Mini Review on Synthesis of Pyrimidinthione, Pyrimidinedione Derivatives and Their Biological Activity
Nadia Ali Ahmed Elkanzi1,2* and Hajer Hrichi1,3
1Chemistry Department, Jouf University, Saudi Arabia
2Chemistry Department, Aswan University, Egypt
3National Institute of Applied Sciences and Technology, Carthage University, Tunisia
Submission: September 20, 2018; Published: September 27, 2018
*Corresponding author: Nadia Ali Ahmed Elkanzi, Chemistry Department, Jouf University, Saudi Arabia and Aswan University, Egypt.
How to cite this article: Nadia A A E, Hajer H. Mini Review on Synthesis of Pyrimidinthione, Pyrimidinedione Derivatives and Their Biological Activity. Recent Adv Petrochem Sci. 2018; 6(1): 555676. DOI: 10.19080/RAPSCI.2018.06.555676
Abstract
The chemistry of heterocycles has received amongst the chemicals prominent attention in recent years owing to its importance in the pharmaceutical sector. Organic compounds carrying pyrimidinthione, pyrimidinedione, pyridazine rings have been reported to demonstrate a wide range of pharmacological activities, which includes antibacterial, antimicrobial, antioxidant, anti-HIV and anticancer activity. These observations have been guiding for the synthesis of various derivatives of these compounds enclosing biologically active nuclei and study their pharmacological activities.
Keywords: Chalcone; Pyrimidinthione; Pyrimidinedione; Biological activities
Introduction
Over the past decade the evolution of organisms resistant to nearly all the class of antimicrobial agents has become a severe public health concern [1,2]. Heterocyclic compounds have received considerable attention owing to their synthetic and biological importance in the enhancement of the quality of human life. Among numerous heterocycles compounds that have been synthesized and evaluated for their pharmacological activities, chalcone, pyrimidinone and pyrimidinethione have played a crucial role in medicinal chemistry. It was demonstrated that the presence of reactive α, β-unsaturated keto function in chalcones was responsible for their antibacterial and antifungal activities [3].
The pyrimidinones compounds have gained interest in recent years due to their wide-ranging biological activity. These compounds displayed therapeutic applications, as anticancer [4,5], antihypertensive [6], hypoglycaemic [7], antiviral [8], anticonvulsive [9], anti-inflammatory and analgesic [10] drugs.
On the other hand, literature surveys revealed that pyrimidinethione derivatives are an important class of heterocyclic molecules possessing a wide variety of biological properties. In fact, different studies demonstrated that various compounds possessing pyrimidinethione nucleus exhibited broad range of biological activities such as antimicrobial [11-13], antioxidant and antitumor activities [14], antitubercular [15] and hypoglycemic activity [16]. The current mini review aims to focus on some synthetic procedures of pyrimidinones and pyrimidinethione derivatives to facilitate the development of new heterocyclic compounds with more efficient and promising pharmacological activities.
Result and Discussion
Pyrimidinedione derivatives were synthesized from various arylmethylene acetophenone derivatives [17]. The chalcone derivatives were prepared using substituted ketone and distinct substituted benzaldehyde through condensation reaction. Likewise, pyrimidinedione derivatives (4) were synthesized via reaction between aryl methylene derivatives and/ or chalcones with thiourea and KOH in ethanol (50ml) in microwave oven for sufficient time and under an appropriate temperature. The detailed steps used in the synthesis of the pyrimidinedione derivatives were described in the literature [18].
The pyrimidine-2-thione derivatives (4) were screened in-vitro against gram positive and gram-negative strains and were found to be less active against the gram-positive bacteria in comparison with the minimal required dose for the action against the gram-negative bacteria in most cases [17] (Figure 1) (Table 1).
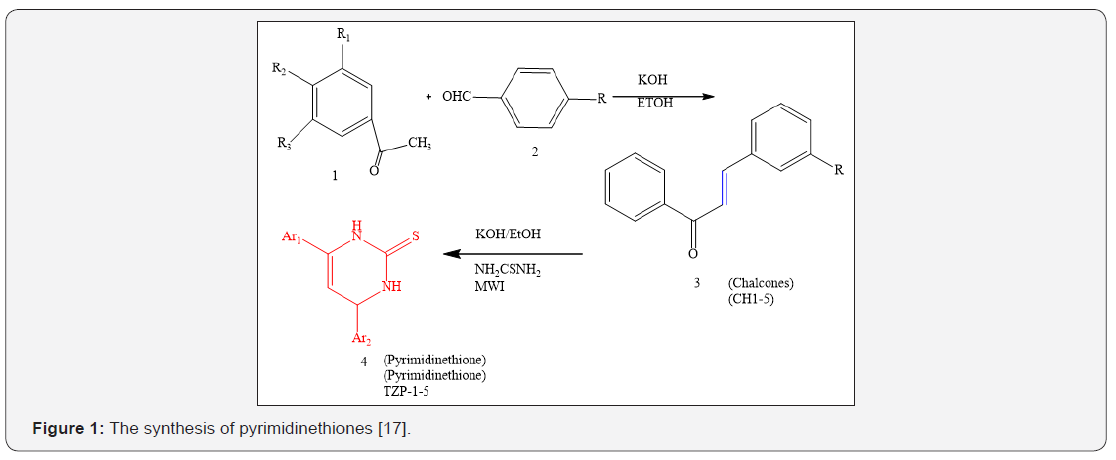
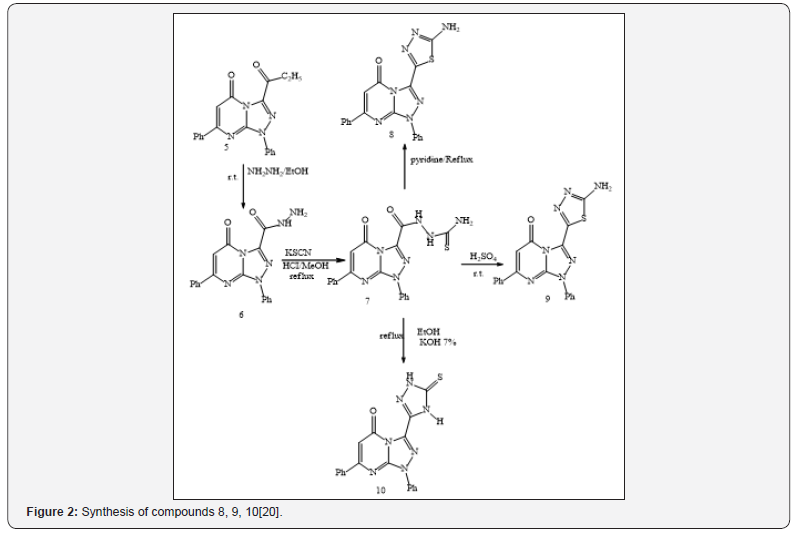
Ethyl 1,5-dihydro-5-oxo-1,7-diphenyl-1,2,4-triazolo [4,3-a] pyrimidine-3-carboxylate (5) was treated with hydrazine hydrate, in refluxing ethanol to give the corresponding acid hydrazide (6) in good yield [19,20]. Acid hydrazide (6) was treated with potassium thiocyanate in refluxing methanol, in the presence of hydrochloric acid to produce1-(1,5-dihydro-5-oxo-1,7-diphenyl- 1,2,4-triazolo[4,3-a] pyrimidine-3-carbonyl) thiosemicarbazide (7). Compound (10) 3-(5-mercapto-4H-1,2,4-triazol-3-yl)-1,7- diphenyl-1,2,4-triazolo [4,3-a] pyrimidin-5-(1H)-one (10) was prepared by oxidative cyclization of compound (7) in basic medium (7% KOH) under reflux with subsequent acidification. It was found that further reflux of compound (7) with dry pyridine gave a product identified as 3- (5-amino-1,3,4-oxadiazol-2-yl)-1,7-diphenyl- 1,2,4-triazolo [4,3-a]pyrimidin-5-(1H)-one (8) (Figure 2).
On the other hand, the reaction involving the synthesis of 1,7-diphenyl-3-(5-amino-1,3,4-thiadiazol-2-yl)-1,2,4-triazolo [4,3-a] pyrimidin-5-(1H)-one (9) took place through a dehydrative cyclization by reacting compound (3) with conc. sulfuric acid. The resulting product (9) was separated as green solid, and was slightly soluble in most organic solvents (Figure 2). Acid hydrazide (6) reacted with phenyl isothiocyanate in refluxing ethanol to produce 1-(1,5-dihydro-5-oxo-1,7-diphenyl-1,2,4-triazolo[4,3-a] pyrimidine-3-carbonyl) phenylthiol semicarbazide (11).
Besides, acid hydrazide (6) was treated with phenyl isothiocyanate in refluxing ethanol to give compound (11)1-(1,5-dihydro- 5-oxo-1,7-diphenyl-1,2,4-triazolo [4,3-a] pyrimidine 3 carbonyl) phenyl thiosemicarbazide as shown in Figure 3. The latter product was subjected to intramolecular cyclization when treated with sulfuric acid, dry pyridine and KOH (5%) demonstrating a new synthetic route to produce compounds 12, 13 and 14, respectively. Subsequent treatment of compound (14) with methyl iodide in the presence of sodium ethoxide solution, yielded 3-(5-methyl thio-4-phenyl-4H-1,2,4-triazol-3-yl)-1,7-diphenyl- 1,2,4-triazolo[4,3-a]pyrimidin-5-(1H)-one (15). It is worth noting that the treatment of compound (14) or (15) with hydrazine hydrate under reflux condition produced the same product, identified as 3- (5-hydrazino-4-phenyl-4H-1,2,4-triazol-3-yl)-1,7-Diphenyl- 1,2,4-triazolo[4,3-a]pyrimidin-5(1H)-one (16) (Figure 3).
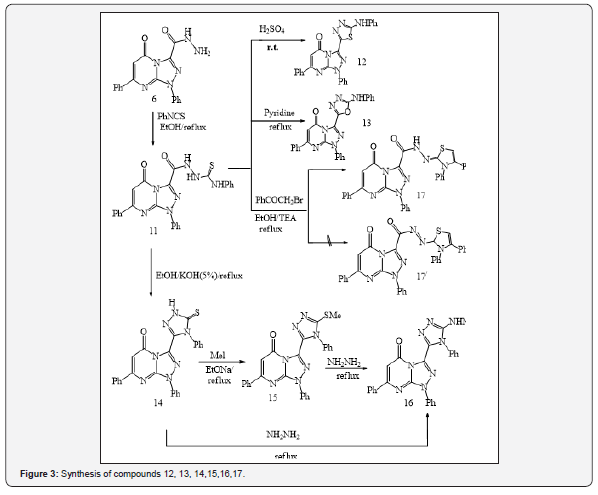
Besides, the treatment of compound (11) with phenacyl bromide in refluxing ethanol, in the presence of triethylamine, yielded 1,5-dihydro-5-oxo-1,7-dipheny-lN-(3,4-diphenyl-3H-thiazol- (2E)-ylidene-1,2,4-triazolo[4,3-a] pyrimidine-3-carbohydrazide (17) as shown in Scheme 3. Treatment of acid hydrazide (6) with carbon disulfide in ethanol, in the presence of potassium hydroxide at room temperature, resulted in the formation of potassium salt (18). The latter product reacted with ethanolic potassium hydroxide to afford 3-(5-mercapto-1,3,4-oxadiazole-2-yl)-1,7-diphenyl- 1,2,4-triazolo[4,3-a]pyrimidin-5(1H)-one (19) (Figure 3). The mechanism of formation of the corresponding ox diazole (19) was studied and discussed in previous studies [21-23]. Moreover, potassium salt (18) was subjected to treatment with hydrazine hydrate, in refluxing ethanol, the reaction yielded as 3-(5-mercapto- 4-amino-4H-1,2,4-triazol-3-yl)-1,7-diphenyl-1,2,4-triazolo[4,3-a] pyrimidin-5(1H)-one (17) (Figure 3).
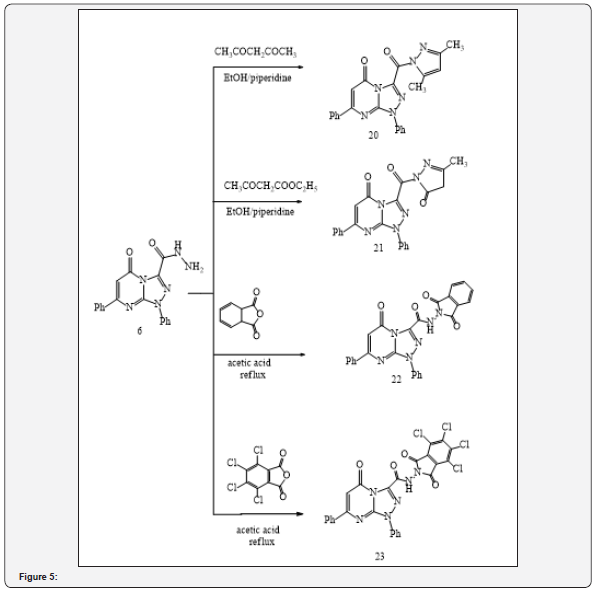
Additionally, it was reported that the reaction involving the synthesis of 4-amino-1, 2, 4-triazole can took place by a simple conversion of 1, 3, 4-oxadiazole under the action of hydrazine hydrate [24,25]. Likewise, the corresponding dicarbonyl compounds including acetylacetone and ethyl acetoacetate were subjected to condensation with acid hydrazide (6) in the presence of an appropriate amount of piperidine as catalyst to give the substituted pyrazole derivatives 3-(3,5-dimethylpyrazole-1-carbonyl)-1,7-diphenyl- 1,2,4-triazolo[4,3-a]pyrimidin-5(1H)-one (20) and 3-(3-methylpyrazole-5-oxo-1-carbonyl)-1,7-diphenyl-1,2,4-triazolo[ 4,3-a]pyrimidin-5(1H)-one (21), respectively (Figure 4).
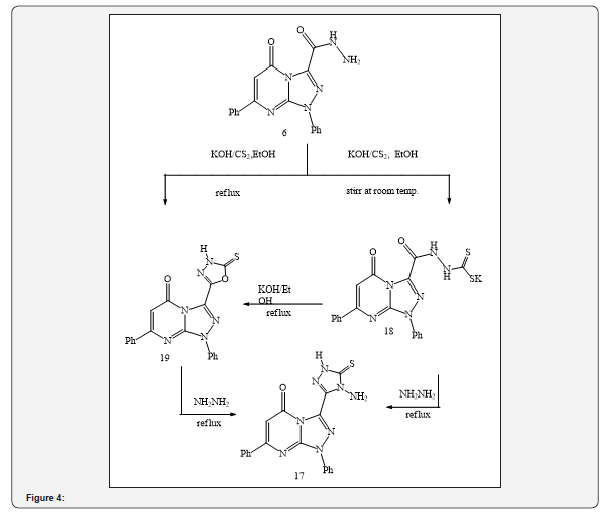
The structure of these compounds was established based on spectral and elemental analysis reported in the related literature [26-28]. Finally, condensation of acid hydrazide (6) with acid anhydrides, namely phthalic anhydride and 2, 3, 4, 5-tetrachlorophthalic anhydride, in refluxing glacial acetic acid, produced the corresponding imides (22) and (23), respectively (Figure 4). Biological studies demonstrated that these compounds exhibited antihypertensive activity [20] (Figure 5).
Conclusion
In this mini-review, we report on the efficient procedures for the synthesis of pyrimidinedione and pyrimidinethione derivatives. The experimental results showed that the prepared product displayed outstanding pharmacological activities when screened In-vitro against gram positive and gram-negative strains and could be further exploited in medicinal chemistry.
References
- Gold HS, Moellering RC, Engl N (1996) Antimicrobial-drug resistance. Journal of Medicinal Chemistry 335: 1445-1453.
- Vanden H, Bossche P, Marichal FCO (1994) Molecular mechanisms of drug resistance in fungi. Trends in Microbiology 2(10): 393-400.
- Solankee A, Patel J (2004) Synthesis of chalcones, pyrazolines, amino pyrimidines and pyrimidinethiones as antibacterial agents. Indian Journal of Chemistry-B 43B: 1580-1584.
- Rashad AE, Shamroukh AH, Yousif NM, Salama MA, Ali HS, et al. (2012) New pyrimidinone and fused pyrimidinone derivatives as potential anticancer chemotherapeutics. Archiv der Pharmazie 345(9): 729- 738.
- Holla BS, Rao BS, Sarojini BK, Akberali PM (2004) One pot synthesis of thiazolodihydropyrimidinones and evaluation of their anticancer activity. Eur J Med Chem 39(9): 777-783.
- Bernhart CA, Haudricourt FB, Assens JL, Gougat J, Lacour C, et al. (1994) Cyclopentanespiro-3H-dihydro-pyrimidinones as Angiotensin II AT1 receptor antagonists. Bioorganic & Medicinal Chemistry Letters 4(1): 157-162.
- Yamaguchi M, Wakasugi K, Saito R, Adachi Y, Yoshikawa Y, et al. (2006) Syntheses of vanadyl and zinc(II) complexes of 1-hydroxy-4,5,6- substituted 2(1H)-pyrimidinones and their insulin-mimetic activities. Journal of Inorganic Biochemistry 100(2): 260-269.
- Rashad AE, Shamroukh AH, Randa EAM, Mostafa A, Elshesheny R, et al. (2010) Synthesis and screening of some novel fused thiophene and thienopyrimidine derivatives for anti‐avian influenza virus (H5N1) activity. Eur J Med Chem 45(11): 5251-5257.
- White DC, Greenwood TD, Downey AL, Bloomquist JR, Wolfe JF, et al. (2004) Synthesis and anticonvulsant evaluation of some new 2-substituted-3-arylpyrido[2,3-d] pyrimidinones. Bioorg Med Chem 12(21): 5711-5717.
- Ranise A, Bruno O, Schenone S, Bondavalli F, Falcone G, et al. (1997) Synthesis of 6-thiosubstituted 5-ethoxycarbonyl-1,3-diphenyl-2- thioxo-2,3-dihydropyrimidin-4(1H)-ones, 6-substituted 5-hydroxy- 1,3-diphenyl-2,3-dihydrothieno[2,3-d]pyrimidin-4(1H)-ones and their esters with local anesthetic, antiarrhythmic, antiinflammatory and analgesic activities. Farmaco 52(8-9): 547-555.
- Bansode TN, Ansari RM, Gawale YK (2011) Synthesis and antibacterial and antifungal activity of some new pyrimidin-2(5H)-one/thione and 4H-chromen-4-one derivatives. Pharmaceutical Chemistry Journal 45: 366-368.
- Cocco MT, Congiu C, Onnis V, Piras R (2001) Synthesis and antitumor evaluation of 6-thioxo-, 6-oxo- and 2,4-dioxopyrimidine derivatives. Farmaco 56(10): 741-748.
- Cocco MT, Congiu C, Onnis V, Schivo ML, Logu DeA, et al, (1995) New thioxopyrimidines. Synthesis and evaluation for antimicrobial activity. Farmaco 50(1): 73-76.
- Salem MA, Marzouk MI, Mahmoud NF (2014) Synthesis of various fused pyrimidine rings and their pharmacological and antimicrobial evaluation. Journal of the Serbian Chemical Society 79: 1059-1073.
- Paghdar DJ, Akabari JD, Tala SD, Dhaduk MF, Joshi HS, et al. (2007) Synthesis of some new thiopyrimidine and oxopyrimidine heterocycles bearing 4-(methylsulfonyl)phenyl nucleus as potent antitubercular and antimicrobial agents. Indian journal of heterocyclic chemistry 17: 113-116.
- Baucer V, Dalalian H, Safir S (1968) 4-(4-Pyrimidinyl)pyridinium Salts. Analogs of the Hypoglycemic 4-Pyrazolylpyridinium Salts. Journal of Medicinal Chemistry 11(6): 1263-1264.
- Jaiswal M (2017) Microwave-assisted synthesis of pyrimedinethione derivatives. Journal of Pharmacology and Biomedicine 1: 115-118.
- Salama MA, El-Essa SA (2003) Synthesis and reactions of some new 2,3-dihydro-5H-5,7-diarylthiazolo-[3,2-a]pyrimidine-3-one derivatives and their antibacterial and fungicidal activity. Indian journal of chemistry 42B: 173-179.
- Shawali AS, Abdallah MA, Mosselhi MA, Faraghaly TA (2002) A facile one‐pot regioselective synthesis of [1,2,4]triazolo[4,3‐a]5(1H)‐ pyrimidinones via tandem Japp-Klingemann, Smiles rearrangement, and cyclization reactions. Heteroatom Chem 13(2): 136-140.
- Korany AA, Eman AR, Thoray AF, Mohamed MA (2011) Synthesis of new functionalized 3 subsitituted [1, 2, 4] triazolo [4, 3-a] pyrimidine derivatives: Potential antihypertensive agents. Acta Pol Pharm 68(2): 237-247.
- Kau ST, Keddie JR, Andrews D (1984) A method for screening diuretic agents in the rat. J Pharmacol Methods 11(1): 67-75.
- Bunag RD (1973) Validation in awake rats of a tail-cuff method for measuring systolic pressure. J Appl Physiol 34(2): 279-282.
- Austen KF, Brocklehurst WE (1961) Anaphylaxis in chopped guinea pig lung. I. Effect of peptidase substrates and inhibitors. J Exp Med 113: 521-539.
- Ram VJ, Goel A, Sarkhel S, Maulik PR (2002) A convenient synthesis and hepatoprotective activity of imidazo [1,2-c] pyrimido [5,4-e] pyrimidine, tetraazaacenaphthene and tetraazaphenalene from cyclic ketene aminals through tandem addition-cyclization reactions. Bioorganic & Medicinal Chemistry 10(5): 1275-1280.
- Tenser RB, Gaydos A, Hay KA (2001) Inhibition of Herpes Simplex Virus Reactivation by Dipyridamole. Antimicrobial Agents and Chemotherapy 45: 3657-3659.
- Cruz DLJP, Carrasco T, Ortega G, Sanchez DLCF (1992) Inhibition of ferrous‐induced lipid peroxidation by pyrimido‐pyrimidine derivatives in human liver membranes. Lipids 27(3): 192-194.
- Sharma P, Rane N, Gurram VK (2004) Synthesis and QSAR studies of pyrimido[4,5-d]pyrimidine-2,5-dione derivatives as potential antimicrobial agents. Bioorg Med Chem Lett 14(16): 4185-4190.
- Cieplik J, Pluta J, Gubrynowicz O (2003) Synthesis and antibacterial activity of 1,3-diarylpyrimido[4,5-d]pyrimidines. Bollettino chimico farmaceutico 142(4):146-150.






























