Hydrothermal Synthesis of Microemulsion Catalysts for Hydrocracking of Heavy Hydrocarbons
Nikolay M Dobrynkin*, Marina V Batygina and Alexander S Noskov
Department of Catalytic Process Engineering, Boreskov Institute of Catalysis, Russia
Submission: April 02, 2018; Published: April 05, 2018
*Correspondence author: Nikolay M Dobrynkin, Boreskov Institute of Catalysis, pr Akademika Lavrent'eva 5, Novosibirsk, 630090, Russia, Tel: +73833309491; Email: dbn@catalysis.ru/dobrynkin_nik@mail.ru
How to cite this article: Dobrynkin NM, Batygina MV, Noskov AS. Hydrothermal Synthesis of Microemulsion Catalysts for Hydrocracking of Heavy Hydrocarbons. Recent Adv Petrochem Sci. 2018; 4(5): 555648. DOI: 10.19080/RAPSCI.2018.04.555648
Abstract
Hydrothermal method for preparing of microemulsion oxide catalysts based on cobalt and iron nanoparticles for the conversion of heavy hydrocarbons has been tested. The results obtained allowed for concluding that hydrothermal synthesis can be used to create catalytic nanosystems for catalytic upgrading of heavy oils in situ.
Keywords: Heavy oil ;In situ upgrading; Hydrothermal synthesis; Microemulsion oxide catalysts
Introduction
In the last decade, special attention has been paid to the development of nano catalytic systems, including the development of ultrafine catalyst compositions for catalytic recovery and upgrading of heavy and extra-heavy oil under both in situ and ex situ conditions. This approach, which was originally started in preparative purposes to create new promising materials for nano-reactors, is now being practically implemented in several process technologies (CAPRI processes-CAtalytic upgrading PRocess In-situ, Combined EOR-in-reservoir-upgrading method [1-3]. According to existing estimates, the use of nanotechnology allows to increase the oil recovery factor from 50%, which is the upper limit using traditional chemical methods of oil production intensification, up to 70%. In the Russian Federation, the possibilities of using nanoscale and ultra disperse particles of catalysts dispersed in hydrocarbon media for use in slurry and blacking reactors for the hydroconversion of heavy petroleum feedstocks are being actively studied [4].
Our previous studies have shown the possibility of active catalysts synthesis in oxidation-reduction reactions supported on rock matrixes of various types [5-6]. It was noted that the methods developed for the preparation of catalysts and a method for introducing a matrix precursor may be applied in a possible practical use in situ.
In this study, hydrothermal method for preparing microemulsion oxide catalysts based on cobalt and iron nanoparticles for the conversion of heavy hydrocarbons has been tested. The method of preparing the catalyst compositions is based on a combination of double emulsion technologies and hydrothermal hydrolysis of iron nitrates and cobalt nitrates under sub-critical conditions at temperatures of 180-220 °C and a pressure of up to 20atm to obtain nano materials with controlled particle sizes of mixed oxides (Figure 1).
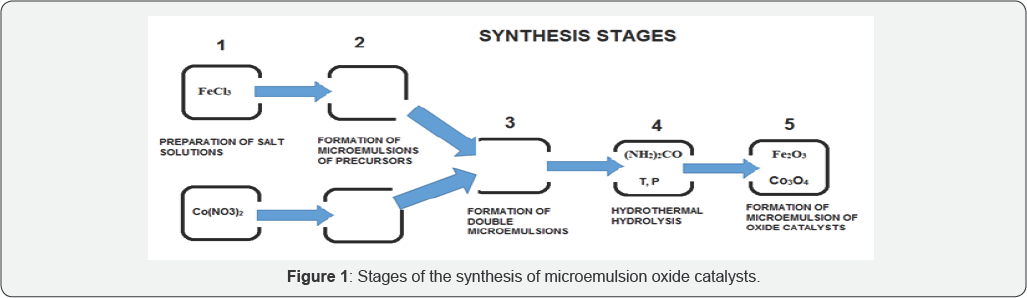
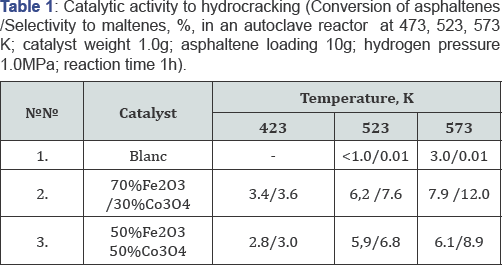
The results of the catalytic activity studies of synthesized catalyst samples in the model reaction of hydrocracking of asphaltenes (the experimental set up and procedure have been reported Dobrynkin [5]) are presented in the Table 1.
Hydrothermal synthesis was carried out in a laboratory high- pressure setup in an inert gas stream using various additives that accelerate hydrolysis. It has been experimentally established that it is optimal to obtain Co3O4 with a particle size of 16.2 to 25nm using Co (NO3)2 * 6H2O cobalt nitrate as a precursor, and CO(NH2)2 urea as the added component at different molar ratios from 4.0 to 0.15 of nitrate to urea. In the joint hydrolysis of cobalt nitrate and ferric chloride in the presence of carbamide, the maximum yield of hematite with a crystallite size of 20nm was observed when equimolar ratios of hydrolysable compounds and carbamide were used.
The sedimentation stability of the synthesized microemulsions was determined by centrifugation at 2600rpm after 24 hours holding at room temperature. Laser correlation spectroscopy methods (emulsion size determination) and high-resolution electron microscopy (TEM) and XRF (to determine the size of metal oxides after heat treatment of emulsions) were used to control the dispersion of emulsion size. It was found that the size of the obtained experimental samples of oxide catalysts microemulsions is 320-360nm with the sizes of metal oxides crystallites of 20-25nm.
Conclusion
The study presents data on hydrothermal synthesis of active catalytic systems for hydrocracking reaction of heavy hydrocarbons. The possibility of regimes choice of nano materials hydrothermal synthesis with controlled particle sizes of mixed cobalt and iron oxides has been determined and optimal conditions (temperature, pressure, duration of synthesis, content of additives accelerating the hydrolysis of metal salts) for synthesis of active catalytic systems for hydrocracking reaction of heavy hydrocarbons. The results obtained allowed for concluding that hydrothermal synthesis can be used to create catalytic nanosystems in situ for catalytic upgrading of heavy oils.
Acknowledgement
This work was conducted within the framework of budget project No. AAAA-A17-117041710077
References
- Almao PP (2012) In situ upgrading of bitumen and heavy oils via nano catalysis. The Canadian J Chem Eng 90(2): 320-329.
- Hamedi SY, Babadagli T (2013) In-SituUpgrading of Heavy Oil/Bitumen During Steam Injection by Use of Metal Nanoparticles: A Study on In- Situ Catalysis and Catalyst Transportation. SPE Reservoir Evaluation and Engineering 16(3): 333-344.
- Abarasi H (2018) How effective is Refinery Regenerated Catalyst for Downhole Catalytic Upgrading of Heavy Crude Oil?. Recent Adv Petrochem Sci 4(2).
- Khadzhiev SN, Kadiev KM, Zhigalina OM, Kadieva MKh, Khmelenin DN, et al. (2015) Structure and properties of molybdenum sulfide nanoparticles synthesized in situ in the hydroconversion process. Petrol Chem 55(8): 655-662.
- Dobrynkin NM, Batygina MV, Noskov AS (2017) Studies of Catalytic Properties of Inorganic Rock Matrices in Redox Reactions. Journal of Sustainable Development of Energy, Water and Environment Systems 5(3): 408-416.
- Dobrynkin NM, Batygina MV, Noskov AS (2017) Catalytic Redox Transformations in Rock Matrices. Kataliz v promyshlennosti 17(6): 436-441.






























