Effect of Bed Porosity on CO2 and CH4 Adsorption inside Packed Bed Column for CO2-CH4 Gas Mixture
Bhajan Lal*, Ali Qasim, Azmi Mohd Shariff and Hilmi Mukhtar
Department of Chemical Engineering, Universiti Teknologi Petronas, Malaysia
Submission: November 17, 2017 ; Published: December 14, 2017
*Correspondence author: Bhajan Lal, Department of Chemical Engineering, Universiti Teknologi Petronas, Bandar Seri Iskandar, Perak, 32610 Malaysia, Email: bhajan.lal@utp.edu.my
How to cite this article: Bhajan L, Ali Q, Azmi M S, Hilmi M. Effect of Bed Porosity on CO2 and CH4 Adsorption inside Packed Bed Column for CO2-CH4 Gas Mixture. Recent Adv Petrochem Sci. 2017;4(1): 555629. DOI: 10.19080/RAPSCI.2017.04.555629
Abstract
The mass transfer phenomenon is observed inside the fixed-bed column by doing the kinetic study of the process. This work studies the effect of bed porosity on CO2 adsorption for CH4/CO2 gas mixture inside packed-bed nanoporous media through Computational Fluid Dynamics (CFD) approach. The adsorbent used in the process is nonoporous activated carbon. Simulations have been done in ANSYS FLUENT® 14. The effect of bed porosity on CO2 and CH4 concentration factors is investigated and it is found out that higher bed porosity reduces the CO2 adsorption uptake and bed performs efficiently at lower values of porosity.
Introduction
The increase in atmospheric concentration of carbon dioxide is proving to be detrimental for the environment and will ultimately result in global climate change [1]. It is estimated that since the time of industrial revolution to 2015, atmospheric CO2 concentration has risen to a value of 401ppm [2]. This trend will continue to rise if anthropogenic sources of emission are not regulated properly. Different techniques have been proposed in order to capture CO2 from flue gases on large scale. These techniques include cryogenic distillation, membrane purification, and absorption using liquids and adsorption by employing solid adsorbents [3]. The use of adsorption is advantageous as solvent is not required for the process and also the adsorbent can be regenerated.
The agricultural waste which is mostly solid is found extensively in Malaysia and thus it can be effectively utilized. For this work, activated carbons are used as an adsorbent to study the adsorption process. These activated carbons are derived from palm mesocarp fibers. Activated carbons can prove to be useful as adsorbents as they have both microporous and mesoporous surfaces [4].
Several different mass transfer models have been proposed to model and simulate adsorption inside fixed-bed column which include linear driving force, quadratic driving force or pore diffusion model [5]. In this study, Computational Fluid Dynamics (CFD) approach has been used to determine the mass transfer phenomenon of CO2/CH4 gas mixture inside the high pressure volumetric analyzer (HPVA) packed bed filled with the adsorbent.
Methodology
Integrated CFD approach has been used to model adsorption dynamics in the fixed bed column. The model is based on convective-diffusive mass transfer. Inlet feed concentrations of CO2/CH4 are varied on the basis of natural gas treatment system. The column operation is considered to be isothermal due to the small length-scale of HPVA column. Mass transfer is represented by Linear Driving Force (LDF) model. The mass transfer coefficient takes into account the external fluid film resistance and macropore diffusion.
Bed porosity is calculated by using the following expression [6]: ^ ^
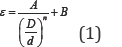
A, B and n are the constants dependent on the shape of particles while 'D' and 'd' are vessel diameter and particle diameter respectively
The general mass balance equation for the column is expressed
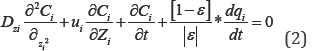
The rate of adsorption based on linear driving force (LDF) model is:
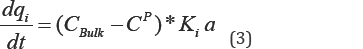
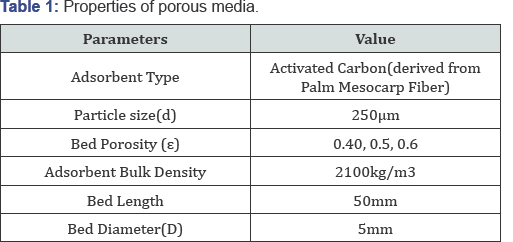
The properties of porous media and the specifications regarding adsorbent are summarized in Table 1.
Result and Discussion
Effect of bed porosity on CO2 and CH4 bed concentration factor (r)
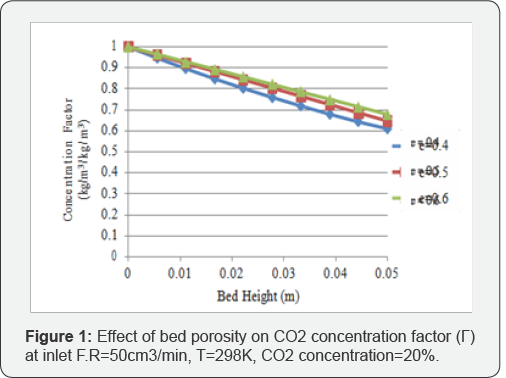
The effect of bed porosity on CO2 and CH4 concentration factors is investigated by considering the bed porosity (e) values at 0.4, 0.5 and 0.6. The selected porosity values are in line with the past researches as done by Cavenati Delgado [7,8]. Concentration factor is ratio of outlet gas concentration to inlet gas concentration. The inlet feed flow rate is constant at 50cm3/min. Carbon dioxide and methane concentration factor profiles along the column for different bed porosities are shown in Figure 1 & 2 respectively. It can be seen from the Figure 1 that concentration factor increases with the increase in porosity from 0.4 to 0.6 as higher bed porosity produces high CO2 concentration in gas phase. This trend is due to the reason that smaller bed porosity increases mass transfer and adsorption rate. This phenomenon shows that the effect of mass transfer is dominant at lower porosity values as low bed porosity values.
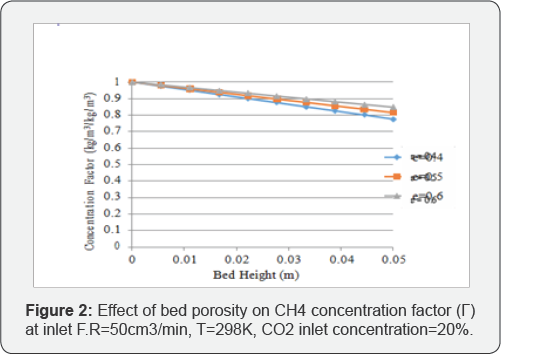
The change in concentration factor values for methane with respect to bed porosity is shown in Figure 2. It can be observed from the trend that lower bed porosity results in higher concentration factor values for the gas. In case of lower bed porosity, an increased amount of methane can be adsorbed inside the system as surface area of the bed is increased to accommodate the gas molecules.
Conclusion
The adsorption phenomenon for CO2-CH4 gas mixture inside the high pressure volumetric analyzer (HPVA) chamber is simulated through CFD approach. The convective-diffusive mass transfer model is based on linear driving force model. For bed porosity, it has been concluded that the bed performs efficiently at lower bed porosity values as more amount of gas molecules can attach to the surface of the adsorbent when it is compact and thus increasing the mass transfer rate.
References
- Guerrero SR, Sayari A (2010) Modeling adsorption of CO2 on amine- functionalized mesoporous silica. 2: Kinetics and breakthrough curves. Chemical Engineering Journal 161(1-2): 182-190.
- Adam G, Aslan F, Portenkirchner E, Stadler P, Scharber MC, et al. (2015) Electrocatalytic reduction of carbon dioxide using sol-gel processed copper indium sulfide (cis) immobilized on ito-coated glass electrode. Electrocatalysis 6(4): 405-413.
- Khalil SH, Aroua MK, Daud AW (2011) Impregnation of commercial palm shell activated carbon with monoethanolamine for adsorbing Co2 from gas mixture. 2011 International conference on biology, Environment and Chemistry 24(2011): 438-443.
- Choi S, Drese JH, Jones CW (2009) Adsorbent materials for carbon dioxide capture from large anthropogenic point sources. Chem Sus Chem 2(9): 796-854.
- Ruthven DM, Farooq S, Knaebel KS (1993) Pressure swing adsorption. wiley-VCH1994. p. 376.
- Pushnov A (2006) Calculation of average bed porosity. Chemical and Petroleum Engineering 42(1-2): 14-17.?
- Cavenati S, Grande CA, Rodrigues AE (2006) Separation of CH4/CO2/N2 mixtures by layered pressure swing adsorption for upgrade of natural gas. Chemical engineering science 61(12): 3893-3906.
- Delgado JA, Uguina MA, Sotelo JL, Ruiz B (2006) Fixed-bed adsorption of carbon dioxide-helium, nitrogen-helium and carbon dioxidenitrogen mixtures onto silicalite pellets. Separation and Purification Technology 49(1): 91-100.






























