Neurological Dysfunction Associated with Vitamin Deficiencies: A Narrative Review
María Alejandra Nieto-Salazar1, Karla Nicole Aguirre Ordóñez2, Zulma Dorila Sosa Carcamo3, Alisson Cristina Aguirre Ordóñez4, Ericka Alexandra Saldana5, Lidys Lorena Juarez Herrera6, Vilma Maria Turcios Ramos6, Aishwarya Yannamani7, Ernesto Calderon-Martinez8, Ghena Khasawneh9, David Arriaga Escamilla10, Fredy Reynaldo Pavón Enamorado6, Allan Roberto Bueso11 and David Adrian De Leon Garza12
1Juan N Corpas University, Colombia. Larkin Community Hospital, USA
2Pontificia Universidad Católica del Ecuador, Ecuador
3Universidad Evangélica de El Salvador, El Salvador
4Universidad de las Américas, Ecuado
5Universidad Salvadoreña Alberto Masferrer, El Salvador
6Universidad Católica de Honduras, Honduras
7Kasturba Medical College, India
8Universidad Nacional Autónoma de México, México
9Jordan University of Science and Technology, Jordan
10Universidad Justo Sierra, México
11Universidad Tecnológica Centroamericana, Honduras
12Universidad de Monterrey, México
Submission: March 13, 2023; Published: March 28, 2023
*Corresponding author: María Alejandra Nieto-Salazar, Juan N Corpas University, Colombia. Larkin Community Hospital, 6100 City Ave. Philadelphia, PA 19131, USA
How to cite this article: María Alejandra Nieto-Salazar, Karla Nicole Aguirre Ordóñez, Zulma Dorila Sosa Carcamo, Alisson Cristina Aguirre Ordóñez, Ericka Alexandra Saldana, et al. Neurological Dysfunction Associated with Vitamin Deficiencies: A Narrative Review. Open Access J Neurol Neurosurg 2023; 18(1): 555979.DOI: 10.19080/OAJNN.2023.18.555979.
Abstract
Background: Vitamins are essential elements from various food sources that the nervous system requires to function correctly. Vitamin deficiency is a widespread public health problem in some populations particularly susceptible. The deficit of many different types of vitamins is associated with neurologic dysfunction. It commonly results from a complex interplay of factors related to inadequate intake, impaired absorption, increased demand, and/or increased excretion. The prevalence of vitamin deficiency is difficult to estimate precisely as it varies depending on the vitamin in question. In addition, the clinical presentation varies widely based on the type of vitamin deficiency. Specific signs or symptoms accompany the deficiency of each vitamin.
Results: Vitamin B1 leads to Wernicke encephalopathy, Korsakoff syndrome, and/or Beriberi. B2, B3, and B6 lead to neuropathy. Vitamin B12 deficiency leads to megaloblastic anemia and neurological impairment. Folate deficiency increases the risk of neural tube defects in the developing fetus. Vitamin A and C are associated with night blindness and scurvy, respectively.
Conclusion: Vitamins are essential elements that contribute to many functions in the nervous system. This narrative review describes the neurological dysfunctions associated with vitamin deficiencies. Most of them cause peripheral neuropathy as they are implicated in myelin formation, but some particular clinical manifestations might help to get a differential diagnosis. Treatment includes vitamin supplementation, and its posology has been described in-depth. Prospective studies that correlate vitamin levels and neurologic manifestations are still required.
Keywords: Vitamin deficiency; CNS; Central Nervous System effects; Neurologic symptoms; Nutrition and CNS
Abbreviations: CNS: Central nervous system, US: United States, WE: Wernicke encephalopathy, WKS: Wernicke-Korsakoff syndrome, ETK: Erythrocyte transketolase, MRI: Magnetic resonance imaging, CT: Computed tomography, IV: Intravenous, TDD: Thiamine deficiency disorders, NAD: Nicotinamide adenine dinucleotide, NADP: Nicotinamide adenine dinucleotide phosphate, PLP: Pyridoxal-5-phosphate, CDC: Centers for Disease Control and Prevention, ASD: Autism spectrum disorder, DNA: Deoxyribonucleic acid, CBC: Complete blood count, TTP: Tocopherol transfer protein, AVED: Ataxia with vitamin E deficiency, RDA: Recommended Dietary Allowance, FNB: Food and Nutrition Board
Introduction
Vitamins are essential elements obtained from various food sources that the nervous system requires to function correctly. Vitamin deficiency is a widespread public health problem, with pregnant women and children being particularly susceptible [1]. The deficit of many different types of vitamins is associated with neurologic dysfunction, including B-complex vitamins (B1, B3, B6, B9, B12), vitamin A (retinol), C (ascorbic acid), E (tocopherol), among others. Vitamin deficiency commonly results from a complex interplay of factors related to inadequate intake (i.e., poor dietary choices, restrictive diets, or lack of availability of certain foods), impaired absorption (i.e., conditions that affect the gastrointestinal tract, such as inflammatory bowel disease, celiac disease, or surgical removal of part of the intestine), increased demand (during growth, pregnancy, breastfeeding, or recovery from an illness or injury), and/or increased excretion (i.e., chronic kidney disease or alcoholism) [1,2].
In the USA, the prevalence of vitamin deficiency is difficult to estimate precisely as it varies depending on the vitamin in question, population groups, geographical regions, diet, race, comorbidities, and socioeconomic status. Furthermore, the clinical presentation varies widely based on the type of vitamin deficiency. However, the most common symptoms include fatigue, dizziness, pale skin, muscle weakness, poor balance, and neurological or gastrointestinal disturbance [2]. Specific signs or symptoms accompany the deficiency of each vitamin. For example, vitamin B12 deficiency leads to megaloblastic anemia and neurological impairment. Folate deficiency during pregnancy increases the risk of neural tube defects in the developing fetus. Vitamin A and C are associated with night blindness and scurvy, respectively. The diagnosis of vitamin deficiency usually involves a combination of clinical assessment, laboratory testing, and medical history [2,3]. Moreover, treating these deficiencies depends on the specific vitamin and the severity of the deficit. In order to correct the nutritional disorder, a balanced diet, supplements, and treatment of underlying causes should all be taken into consideration [3]. It is important to note that vitamin deficiency is usually severe when most neurological manifestations become evident and must be corrected in the shortest time possible to avoid potentially irreversible sequelae [2]. However, some of these symptoms might be unspecific or poorly understood by many clinicians, generating a challenge for their proper management. The purpose of this narrative review is to provide a comprehensive overview of the neurological manifestations of vitamin deficiency to better understand the overall approach to this condition.
Thiamine (Vitamin B1)
Thiamine, also known as vitamin B1, is an essential nutrient that plays a critical role in energy metabolism and the proper functioning of the nervous system. It is a water-soluble vitamin found in meat, beef, pork, legumes, whole grains, and nuts. Thi amine pyrophosphate, thiamine’s biologically active form, is a coenzyme involved in several biochemical pathways in the brain. The prevalence of vitamin B1 deficiency varies depending on the population and the underlying causes. In developed countries, it is relatively rare and is often associated with alcohol abuse, malabsorption syndromes, and chronic illness. In contrast, in developing countries, thiamine deficiency is more common, especially in populations that rely heavily on a diet consisting mainly of polished rice or other processed grains that have been stripped of their thiamine content [1,2,4].
Vitamin B1 deficiency, also known as beriberi, can result in a wide range of symptoms, depending on the severity and duration of the deficiency. The symptoms can affect various organ systems, including the nervous, cardiovascular, and gastrointestinal systems. Cardiovascular symptoms include cardiomegaly, acute/chronic heart failure, and arrhythmias. These symptoms are more common in severe cases of thiamine deficiency, such as those associated with alcohol abuse. When patients experience heart failure, the condition is known as “wet” beriberi. Thiamine deficiency can also affect the gastrointestinal system, leading to loss of appetite, weight loss, and constipation.
Neurological symptoms are broad, including peripheral numbness, tingling, paresthesias, weakness, loss of reflexes, confusion, memory loss, delirium, seizures, behavioral changes (i.e., apathy, irritability and depression), and visual disturbances (ophthalmoplegia or nystagmus). When the deficiency presents with predominantly symmetrical peripheral neuropathy, it is known as “dry” beriberi and is more prevalent in children. When the deficiency presents with dementia-like symptoms, it is known as Wernicke encephalopathy (WE), which is more common in adults. WE is characterized by the clinical triad of ataxia, confusion, and nystagmus [5]. If left untreated, WE might progress to Korsakoff syndrome or Wernicke-Korsakoff syndrome (WKS). The latter is characterized by the clinical triad of confabulation, altered mental status, and severe memory loss (particularly affecting anterograde memory) [5]. While initial neurologic symptoms are often reversed quickly with prompt treatment, severe cases or delayed treatment lead to irreversible sequelae (i.e., when Korsakoff syndrome is present, the neurological symptoms are permanent) [4,5]. In most severe cases, patients may also experience cardio-respiratory failure, coma, and death.
The diagnosis of thiamine deficiency is laboratory-based and commonly independent of signs and symptoms of the disease. Laboratory tests that may be used include measurement of vitamin B1 metabolite levels in the blood or urine and assessments of erythrocyte transketolase activity (ETK assay), which is a marker of thiamine status. The ETK assay is considered more accurate, as it demonstrates the actual functionality of the vitamin [5,6]. In addition, in selected cases, imaging studies such as magnetic resonance imaging (MRI) or computed tomography (CT) may be performed to evaluate for structural abnormalities or damage to the brain parenchyma.
The treatment of thiamine deficiency involves prompt vitamin supplementation, either orally or intravenously, depending on the severity of the deficit. In mild cases, oral thiamine supplementation is typically sufficient. The recommended dose of oral thiamine is usually between 5 and 30 mg/day, although higher doses may be required in some cases. The frequency of administration may vary but is typically once or twice daily for several weeks or until symptoms resolve. In moderate to severe cases of thiamine deficiency, intravenous (IV) thiamine supplementation is necessary to quickly restore thiamine levels and prevent potential complications. The recommended dose of IV thiamine is usually between 100 and 500 mg per day, administered either as a single dose or divided into multiple doses over the course of a day. The duration of IV therapy may vary but typically lasts for several days or until symptoms resolve. It should be noted that, compared with intravenous doses, experimental and clinical data indicate that orally administered thiamine is less effective at increasing blood thiamine and may not be suitable for treating neurological conditions associated with thiamine deficiency disorders (TDDs) [4,5]. In addition to vitamin supplementation, patients may require supportive care and management of any underlying conditions that may have contributed to the deficiency, such as alcohol use disorder or malnutrition.
Niacin (Vitamin B3)
Niacin is a vitamin that belongs to the vitamin B complex and acts as a coenzyme in multiple metabolic reactions [7]. Its deficiency, which develops in the condition known as pellagra, is most common in countries with low income and high levels of poverty, such as some areas of Africa where it is still a prevalent disease compared to other parts of the world [8]. Due to a diet based mainly on the use of corn in Latin America, China, and India, pellagra is rarely seen. In the United States, pellagra is a rare condition not associated with nutritional deficiencies because of the enrichment of foods like flour. Instead, it occurs due to chronic alcoholism or malabsorption syndrome [9].
Primary niacin deficiency occurs mainly from the lack of its consumption through dietary sources like fish, meats, cereal, bread, and legumes [10]. In the case of secondary pellagra, alcoholism, gastrointestinal diseases, and drug addiction are its most common causes [11]. Characterized by gastrointestinal and dermatological findings, its neurological symptoms and signs include insomnia, anxiety, disorientation, delusions, dementia, encephalopathy, and muscle weakness. When niacin deficiency is suspected, it can be confirmed through laboratory testing of levels of niacin, tryptophan, nicotinamide adenine dinucleotide (NAD), and NAD phosphate (NADP) [12]. The treatment of niacin deficiency relies on the vitamin’s replacement through oral supplementation of nicotinamide, recommended doses of 250 to 500 mg daily. It’s also important to identify the cause of this deficiency, treat it accordingly, and take preventive measures to avoid the development of the disease in the future [13].
Pyridoxine (Vitamin B6)
Pyridoxine, or vitamin B6, is an essential nutrient in numerous biochemical processes. For example, it plays a crucial role in the metabolism of amino acids, synthesizing neurotransmitters, and producing red blood cells. Pyridoxine is found naturally in various foods, including poultry, fish, beans, nuts, and bananas. It is also available as a dietary supplement and is commonly used to treat certain medical conditions, such as morning sickness, carpal tunnel syndrome, and premenstrual syndrome [14,15].
Pyridoxine is necessary for the proper functioning of the nervous system and helps maintain healthy skin, eyes, and liver. It also plays a role in regulating the immune system and hormone levels. The deficit of pyridoxine can result in various symptoms, including anemia, dermatitis, seizures, depression, and confusion. It may also increase the risk of cardiovascular disease and certain types of cancer [15]. Pyridoxine deficiency can be caused by severe malnutrition, alcoholism, liver disease, pregnancy, and treatment with isoniazid [14]. Mild impairments manifest at the neurological level as irritability, confusion, depression, and possibly peripheral neuropathy. Severe deficiency is associated with seizures. In addition, an inborn error of pyridoxine metabolism is responsible for pyridoxine-dependent epilepsy, which presents with refractory neonatal seizures [15].
The diagnosis of pyridoxine deficit involves patient history and laboratory tests. Mean plasma pyridoxal-5-phosphate (PLP) concentrations are commonly measured to detect the condition. PLP levels of 20 - 30 nmol/L (4.9 to 7.4 ng/mL) are generally considered borderline, and levels >30 nmol/L (>4.4 ng/mL) confirm the diagnosis [15]. When vitamin B6 deficiency is caused by malnutrition, the treatment involves the intake of 50 mg of pyridoxine orally daily for several weeks, followed by 2 mg/daily, and then returning to a regular diet. When the deficiency is due to a pyridoxine antagonist drug such as isoniazid, penicillamine, hydralazine, or cycloserine, treatment consists of 50 mg of pyridoxine daily, and when the antagonist is also used [16]. In pyridoxine-dependent epilepsy, standard treatment involves lifetime supplementation of oral pyridoxine (15-30 mg/kg/day divided into 3 doses in infants; up to 200 mg/day in neonates and 500 mg /day in adults) to control seizures, as well as regular clinical monitoring [17].
Folate (Vitamin B9)
Folate is an essential water-soluble vitamin that can be found in fruits, green leafy vegetables, as well as liver. On the other hand, folic acid is a synthetic form of folate that is added to fortified foods and supplements. This form has an increased bioavailability. Folate is essential due to its role in preventing congenital disabilities like neural tube defects. It is used to form purines and pyrimidine synthesis, nucleoproteins, and to aid in erythropoiesis. A study conducted by the CDC showed that women of childbearing age, as well as nonblack Hispanic women, are at high risk of folic acid deficiency due to poor nutrition; it also showed that low-income populations, as well as an institutionalized aging population, have an increased risk for folic acid deficiency because of malnutrition and mental status change. Other causes for folic acid deficiency in adults include celiac disease, gastric bypass, malabsorption syndrome, amyloidosis, other congenital defects of enzymes that are required in folate metabolism like B12 deficiency, therapies with methotrexate, phenytoin, sulfasalazine and trimethoprim, alcoholism, pregnancy, hemolytic anemias, and hemodialysis status.
Currently, most developed countries have mandatory folic acid flour fortification, and they recommend folic acid supplementation before conception and up to the first 3 months of pregnancy [18]. However, high physiological folate concentrations and folate overload may increase the risk of impaired brain development and embryogenesis and can be the cause of precancerous cell formation [19]. The neurologic manifestations of folate deficiency are similar to those of vitamin B-12 deficiency. They include cognitive impairment, combined spinal cord degeneration, dementia, depression, Alzheimer’s disease (with increased homocysteine levels), and even peripheral neuropathy in certain cases. For some patients, folate deficiency can be secondary to psychiatric illness, apathy, or poor diet, aggravating the underlying disorder [20].
Nowadays, more studies are being conducted examining the importance of folic acid in childhood behavioral outcomes, specifically autism spectrum disorder (ASD). Some authors report an increased frequency of serum autoantibodies against folate receptor alpha or polymorphisms in methylenetetrahydrofolate reductase related to elevated ASD risk. Unfortunately, the data is still inconclusive [19,20]. However, given the observational data about the protective effect of folate against neural tube defects, more clinical trials with larger sample sizes and observational periods are necessary.
Diagnosis can be made with vitamin B12 deficiency evaluation, especially in patients with macrocytic anemia. The complete blood count would show an increased mean corpuscular volume greater than 100, a peripheral smear with macrocytic red blood cells (megaloblasts), and hyper segmented neutrophils. Normal serum folate levels range from 2 ng/mL to 4 ng/ mL. Therefore, a folate level of < 2 ng/mL confirms the diagnosis. For patients with borderline results, folate deficiency can be confirmed with the presence of normal levels of vitamin B12 and methylmalonic acid, with raised homocysteine levels. Folate deficiency is treated with supplemental folic acid, typically 1-5 mg oral daily doses. Other routes of administration are used in patients that cannot tolerate oral medications. Therapy duration depends on the deficit’s severity and etiology (i.e., patients with malabsorption syndromes may require long-term treatment). In patients with concomitant vitamin B12 deficiency, this vitamin should also be replenished to avoid the progression of irreversible neurological damage [18].
Cobalamin (Vitamin B12)
Cobalamin, or vitamin B12, is a vital nutritional element for different metabolic processes, such as DNA synthesis and myelin production. It can be found in several animal products, including red meat, dairy, and eggs [21]. Its deficiency is more common in patients around 60 years old, affecting men and women equally. It is more frequent in African and European ascendancy individuals, with a prevalence of 4,3% and 4%, respectively. This condition has an overall incidence of 25/100,00 people [22].
The etiology of cobalamin deficiency is broad and sometimes multifactorial. It may be caused by pernicious anemia, where the immune system attacks the gastric cells that produce intrinsic factors (a protein necessary for vitamin B12 absorption) [21]. Other causes include gastrointestinal disorders such as Crohn’s disease or celiac disease, which can interfere with the absorption of vitamin B12 in the small intestine. Surgery to remove part of the stomach or small intestine can also lead to malabsorption of vitamin B12. Moreover, strict vegetarian or vegan diets, not including animal products, can lead to cobalamin deficiency, as vitamin B12 is found mainly in animal-based foods. Older adults are also at risk of cobalamin deficiency as their ability to absorb vitamin B12 decreases with age. Certain medications, such as metformin used to treat diabetes, can also interfere with vitamin B12 absorption [21].
The clinical presentation of cobalamin deficiency can include megaloblastic anemia, which can lead to fatigue, weakness, and dyspnea. Other symptoms may include skin changes, glossitis, nausea, vomiting, and diarrhea. Moreover, B12 deficiency can cause various neurologic symptoms due to its essential role in maintaining the nervous system. It can lead to demyelination of the nerves, leading to nerve damage and dysfunction. This can result in peripheral neuropathy, which involves numbness, tingling, and weakness in the hands and feet. Other neurologic symptoms may include difficulty with balance and coordination, weakness, spasticity, and impaired fine motor control. If the deficit is severe, it might lead to intense cognitive impairment, dementia, and even psychiatric conditions (i.e., depression, mania, and psychosis) [23]. The neurologic symptoms of cobalamin deficiency are typically irreversible if left untreated for a prolonged period. As such, early recognition and treatment are crucial in preventing the onset of severe neurologic symptoms and irreversible sequelae.
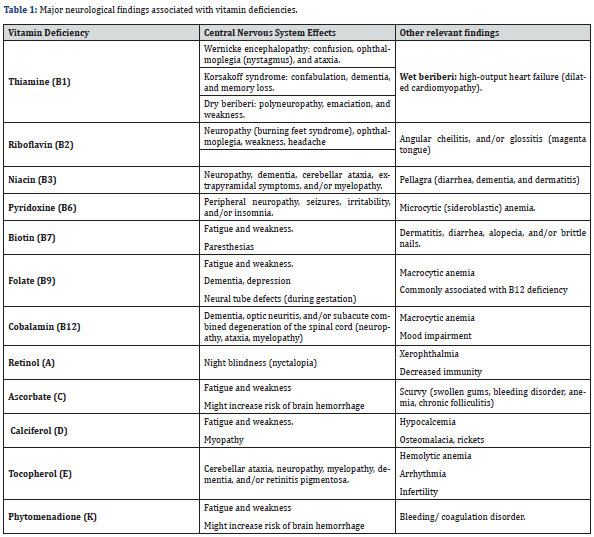
It is important to note that both water-soluble and fat-soluble vitamins can cause similar neurological symptoms, so the diagnostic and therapeutic approach should always combine the patient’s medical history, physical examination, and laboratory findings. Table 1 summarizes the most important neurological and other relevant findings for each vitamin deficiency. The diagnosis of vitamin B12 deficiency includes a complete blood count (CBC), a peripheral blood smear, and serum levels of B12 and folate. Macrocytic anemia should be observed in the CBC, with hyper segmented neutrophils in the blood smear, low serum B12, and normal levels of folate [24]. The treatment of this nutritional deficiency consists of identifying the primary cause of the deficiency and ensuring that the intake is restored. Duration of treatment and supplement doses vary a lot depending on multiple patient factors. While oral supplements might be enough for patients with inadequate diet, long-term parenteral doses are recommended for those with malabsorption syndrome or autoimmunity [21].
Retinol (Vitamin A)
Vitamin A is a fat-soluble compound found in animal products and carotenoids (i.e., fruits and vegetables). There are three active forms of vitamin A: retinol, retinal, and retinoic acid. All of them constitute an essential nutrient needed in small amounts for the normal functioning of the visual system and maintenance of cell function for growth, epithelial integrity, red blood cell production, immunity, and reproduction. However, this vital nutrient cannot be synthesized by the human body and must be provided through diet. When dietary intake is chronically low, insufficient vitamin A will sustain the vision and cellular processes, leading to impaired function [25].
Several circumstances might cause or contribute to vitamin A deficiency. The leading underlying etiology is a diet insufficient in vitamin intake, resulting in an inability to meet physiological needs. When the deficit is chronic or severe, it can result in xerophthalmia, one of the most common causes of preventable childhood blindness [25]. Moreover, infectious diseases have been associated with the depletion of hepatic vitamin A supplies, reduced serum retinol concentrations, and increased excretion of vitamin A in the urine. For example, it is known that measles virus infection precipitates conjunctival and corneal damage, leading to blindness in children with poor vitamin A levels. Furthermore, even children who have a mild deficiency have a higher incidence of respiratory complications and diarrhea, as well as a greater mortality rate from measles infection compared to children who consume enough vitamin A. Vitamin A deficiency and goiter-induced iodine deficiency can coexist in up to 50% of children [26].
There are various clinical manifestations associated with vitamin A deficiency. First, ophthalmological symptoms are among the most common symptoms, including bulging eyeballs, cortical disc swelling, and visual disturbances, especially at night (night blindness). Secondly, dermatological signs are prevalent, including skin discoloration, desquamation, rash, itching, thin/coarse hair, and alopecia of the eyebrows. Finally, neurological symptoms include irritability, severe headache, drowsiness, delirium, coma, increased intracranial pressure (pseudotumor cerebri), bulging fontanelles (in infants), psychiatric changes, and cerebral edema [27]. The diagnosis of vitamin A deficiency consists of a combination between a patient’s medical history, physical examination, and lab tests (particularly serum retinol levels). The normal range of serum retinol is between 28 to 86 mcg/ dL (1 to 3 mmol/L). Levels < 28 mcg/dL confirm the diagnosis. However, it is important to note that these evident reductions in retinol levels only occur in advanced stages as the liver contains large vitamin A reserves. Consequently, the deficiency might be present even with normal vitamin serum levels. Moreover, certain acute conditions (i.e., infection, trauma, etc.) might cause a transient reduction in retinol-binding protein and transthyretin (prealbumin) levels, falsely reducing vitamin A levels. In these challenging situations, a therapeutic vitamin A test can aid in confirming the diagnosis [27,28].
Vitamin A deficiency is treated according to its etiology. Patients with poor dietary intake are treated with vitamin A palmitate oil in doses of 60,000 units orally once daily for 2 days, followed by 4500 units orally once daily. If vomiting, malabsorption, or xerophthalmia is present, a dose of 50,000 - 200,000 units should be given for 2 days, with a third dose 2 weeks later. Prolonged daily administration of higher doses should be avoided, especially in infants, due to the risk of toxicity. In pregnant or breastfeeding women, the therapeutic or prophylactic dose should not exceed 10,000 units/day to avoid possible harm to the fetus or infant [28].
Ascorbate (Vitamin C)
Vitamin C, also known as ascorbic acid or ascorbate, is a key circulating antioxidant with anti-inflammatory and immune- supporting effects. Humans lack L-gulono-1,4-lactone oxidase, the enzyme needed for synthesizing vitamin C. As a result, ascorbate’s presence in the body depends entirely on the amount consumed in the diet. In critically ill patients, vitamin C deficiency develops during these extreme circumstances [29,30]. A national survey in the United States during the years 2003–2004 reported that 7% of individuals had levels of vitamin C below the recommended levels, particularly smokers (who are at a greater risk of this deficiency). There are several pathologies and other situations in which the levels of vitamin C drop in plasma, commonly because these conditions have an enhanced need for vitamin C. Some examples of this include acute surgery, trauma, sepsis, and burns, which might cause a decrease in blood vitamin C. Other conditions, such as acute myocardial infarction and cancer, are associated with a rapid reduction of vitamin C in plasma and tissues [29].
The clinical presentation of vitamin C deficiency is broad, and most symptoms commonly develop when plasma levels are below 0.15 mg/dL. This deficiency is also known as scurvy. Some early manifestations of this condition include lethargy, confusion, and irritability. As the deficit progresses, other symptoms emerge: muscle weakness, swollen and bleeding gums, loss of teeth, petechial hemorrhaging, spontaneous ecchymoses, anemia, impaired wound healing, weakness, myalgia, arthralgia, and weight loss. The most characteristic skin manifestations are purpuric perifollicular hyperkeratotic papules and the presence of kinky hair. Scurvy is potentially fatal, and sudden death can occur as a consequence of cerebral/myocardial hemorrhage or pneumonia. Peripheral neuropathy causing numbness of calves and pain might be related to the epigenetic effect of vitamin C, as it is needed for myelin formation by Schwann cells. Therefore, it is thought that peripheral neuropathy can be due to demethylating impacts, which can affect the epigenetic activation, proliferation, and differentiation of Schwann cells [31,32].
Diagnosing this condition begins with evaluating risk factors and performing a thorough physical examination. Dermoscopy can be used to aid in the diagnosis, confirming follicular purpura and corkscrew hairs with a 4 mm punch biopsy of affected areas showing similar findings by histopathology. Serum testing for low plasma vitamin C (< 0.2 mg/dL) is usually consistent with scurvy. However, recent intake or supplementation may elevate plasma levels and not be reflective of a prior prolonged deficit. The level of vitamin C in leukocytes is more accurate when assessing the sparse vitamin C stores as they are less affected by acute dietary changes. A leukocyte vitamin C level of 0 mg/dL is indicative of latent scurvy. Levels between 0 to 7 mg/dL are consistent with deficiency, and levels > 15 mg/dl are considered adequate. With vitamin C deficiency, imaging studies could reveal fractures, dislocations, subperiosteal elevation, and alveolar bone resorption. Early imaging features of scurvy are observed at the distal ends of the radius, which has fuzziness over the lateral aspect of the cortex. Moreover, progression to osteopenia and osteoporosis is common [33,34].
Direct replacement of vitamin C is the standard treatment for this condition. Doses are up to 300 mg for children and 500 to 1000 mg daily for adults. The intended end point of replacement is one month or upon resolution of clinical symptoms. Alternative treatment regimens for adults include 1 to 2 gr for up to 3 days, followed by 500 mg daily for a week, followed by 100 mg daily for up to 3 months. In addition to immediate supplementation, the patient should be educated on lifestyle modifications to ensure adequate intake and recommend cessation of alcohol and tobacco use. It is essential to address the underlying cause of scurvy as part of management [35].
Tocopherol (Vitamin E)
Vitamin E is one of the most important lipid-soluble antioxidant nutrients. It is found naturally in some foods (i.e., nuts, seeds, and vegetable oils), added to others, and available as a dietary supplement [36]. Vitamin E cannot be synthesized in the human body, so it must be supplied exogenously through diet. The vitamin E family contains eight isomers, four tocopherols (α-, β-, γ-, and δ-tocopherol), and four tocotrienols (α-, β-, γ-, and δ-tocotrienol) [37,38]. After consumption of vitamin E from the diet, all isoforms are absorbed by the small intestine and reach the liver via chylomicrons to be metabolized. Only α-tocopherol preferentially is released into the bloodstream bound to a protein called α-tocopherol transfer protein (α-TTP). Due to its high affinity to α-TTP in the liver, it accumulates in various organs via the bloodstream. On the contrary, the other isoforms of vitamin E are excreted because of their poor affinity to α-TTP. Serum levels of alpha-tocopherol in 0.1% of United States adults over the age of 20 have been found to be deficient. Surveys of the same data set have shown that 89.8% of men and 96.3% of women 19 years of age or older have insufficient alpha-tocopherol intake. Some studies have shown alpha-tocopherol to be lower in pediatric populations and higher in pregnancy [38].
Recent studies have indicated that vitamin E exhibits various potentially beneficial effects on human health, such as anti- allergic anti-atherogenic, anti-cancer, anti-cardiovascular, anti- diabetic, anti-lipidemic, anti-hypertensive, anti-inflammatory, anti-obesity, neuroprotective, and modulation of telomerase activities [38]. Tocochromanols are the most effective group of lipophilic phenolic antioxidants. Researchers theorize that antioxidants protect key cell components by neutralizing free radicals before they can cause lipid oxidation or DNA damage. By reducing free radical attack, antioxidants break the chain reaction of lipid peroxidation (chain-breaking antioxidant) and protect the cell membranes through lipid repair and replacement. Their antioxidant function is attributed to inhibiting membrane lipid peroxidation and scavenging reactive oxygen species. In this way, they may prevent cancer, heart disease, and neurologic disorders. Epidemiological evidence indicates that diet-derived antioxidants (e.g., vitamins A, C, and E) may be necessary for maintaining human and animal health. In addition to its actions as an antioxidant, vitamin E is involved in immune function and, as shown primarily by in vitro studies of cells, cell signaling, regulation of gene expression, and other metabolic processes [39,40].
In addition to dietary intake and oxidative stress conditions, the risk of vitamin E deficiency may be influenced by age, obesity, and sex. In some children and elderly populations, low dietary diversity and poor fruit and vegetable intake may contribute to poor antioxidant vitamin status. The observation that vitamin E deficiency is often accompanied by low circulating levels of other antioxidants, including vitamin C and β-carotene, supports the theory that deficiencies are associated with poor intake and more significant oxidative stress. Among adolescents and adults, obesity and male sex may predispose individuals to vitamin E deficiency. An association between obesity and poor vitamin E status has been demonstrated in multiple studies, possibly due to the sequestration of α-tocopherol in adipose tissue [40]. Severe vitamin E deficiency can profoundly affect the central nervous system due to its vulnerability to the damaging effects of free radicals. Cystic fibrosis, chronic cholestatic liver disease, abetalipoproteinemia, short bowel syndrome, isolated vitamin E deficiency syndrome due to genetic abnormalities in α-TTP, and other malabsorption syndromes may cause varying degrees of neurologic deficits due to related vitamin deficiencies [41].
The classic clinical presentation in vitamin E deficiency progresses from hyporeflexia, ataxia, limitations in upward gaze, and strabismus to long-tract defects, profound muscle weakness, and visual field constriction. Patients with a severe, prolonged deficiency may develop complete blindness (retinitis pigmentosa), infertility, dementia, and cardiac arrhythmias (6). Individuals with autosomal recessive mutations in the gene for α-TTP (ataxia with vitamin E deficiency [AVED]) present with neurodegenerative symptoms, including cerebellar ataxia, loss of deep tendon reflexes, vibratory-sense disturbances, dysarthria, muscle weakness, head tremor, and dystonia. Acanthocytosis, a characteristic alteration of the erythrocyte membrane morphology, is associated with vitamin E deficiency and results in increased erythrocyte hemolysis and anemia. The peripheral neuropathy and anemia seen in vitamin E deficiency are likely caused by excessive free radical damage to the large-caliber axons in sensory neurons and the erythrocyte membrane. Plasma α-tocopherol concentrations < 8 μmol/L are associated with neurologic disease in humans, while concentrations < 12 μmol/L are associated with increased red blood cell fragility in vitro [40].
According to the Food and Nutrition Board of the Institute of Medicine, vitamin E deficiency in normal, healthy adults is defined by a plasma α-tocopherol concentration < 12 μmol/L (0.5 mg/dL). However, studies of vitamin E status in diverse populations have used cutoffs ranging from 2.8 to 24 μmol/L (0.1 to 1.0 mg/dL) to define deficiency and insufficiency [40]. In adults with hyperlipidemia, the rise in lipids may affect vitamin E levels, and serum alpha-tocopherol to lipids level must be lower than 0.8 mg/g to confirm the diagnosis. Pediatric patients with abetalipoproteinemia will have serum alpha-tocopherol levels that are not detectable [38].
Treatment must be tailored to the underlying cause of vitamin E deficiency and may include oral or parenteral vitamin supplementation. The more advanced the deficit, the more limited the response to therapy. Therefore, a good neurologic examination and periodic serum vitamin E levels are essential in patients at risk of vitamin E deficiency [41]. Patients with genetic abnormalities or malabsorptive syndromes require daily pharmacologic vitamin E doses for life to overcome the mechanisms leading to deficiency. Generally, patients with AVED are suggested to consume 1000 mg RRR-α-tocopherol (natural vitamin E) daily in divided doses. Patients with abetalipoproteinemia require 100 mg/kg/day, and those with cystic fibrosis 400 mg/day. It should be noted that patients with fat malabsorption related to impaired biliary secretion generally do not absorb orally administered vitamin E. Consequently, they have to receive other forms of vitamin E, such as α-tocopheryl polyethylene glycol succinate, that spontaneously form micelles and avoid the need for bile acids to get absorbed [42]. In order to prevent vitamin E deficiency from dietary intake, the Recommended Dietary Allowance (RDA) developed by the Food and Nutrition Board (FNB) at the Institute of Medicine of The National Academies is 15 mg daily (or 22 IU) of tocopherol for males and females ages 14 years and older, including women who are pregnant. Lactating women need slightly more at 19 mg (28 IU) daily. Numerous dietary sources can provide vitamin E. Nuts, seeds, and vegetable oils are among the best sources of alpha-tocopherol. Moreover, significant amounts are available in green leafy vegetables and fortified cereals. Most vitamin E sources in the American diet are in the form of gamma-tocopherol, which comes from soybean, canola, corn, and other vegetable oils [38].
Conclusion
Vitamins are essential elements that contribute to many functions in the central nervous system. Their deficiency often originates from malnutrition, intestinal malabsorption, or chronic conditions. This narrative review describes the neurological dysfunctions associated with the most common vitamin deficiencies. Most of them might cause peripheral neuropathy as they are implicated in myelin formation. However, some particular clinical manifestations might aid in obtaining a clue on differential diagnoses, such as the bleeding tendency associated with ascorbate, megaloblastic anemia with cobalamin, pellagra with niacin, cerebellar ataxia with thiamine deficiency, among others. In order to achieve a successful diagnosis, a comprehensive medical history must be obtained, including an entire dietary assessment, physical examination, neurologic evaluation, and laboratory tests. Treatment for most vitamin deficiencies includes nutritional supplementation and the correction of the underlying disorder. Despite the fact that current data is extensive, future large-scale prospective studies that correlate vitamin levels and neurologic manifestations are still required.
References
- Bailey RL, West KP, Black RE (2015) The epidemiology of global micronutrient deficiencies. Ann Nutr Metab 66(2): 22-33.
- (1998) Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate OBV and C. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline.
- National Institutes of Health, Office of Dietary Supplements (2023) Vitamin and Mineral Supplement Fact Sheets.
- Wiley KD, Gupta M (2022) Vitamin B1 Thiamine Deficiency. StatPearls.
- Smith TJ, Johnson CR, Koshy R, et al. (2021) Thiamine deficiency disorders: a clinical perspective. Ann N Y Acad Sci 1498(1): 9-28.
- Whitfield KC, Bourassa MW, Adamolekun B, Bergeron G, Lucien B, et al. (2018) Thiamine deficiency disorders: diagnosis, prevalence, and a roadmap for global control programs. Ann N Y Acad Sci 1430(1): 3-43.
- Oliveira NAC, Magalhaes LA, Matos MRT, Aragao GF, Bachur TPR (2019) The deficiency of thiamine and niacin as a risk factor for neurological disease. Infarma - Ciencias Farmaceuticas 31(2): 80-85.
- Li R, Yu K, Wang Q, Wang L, Mao J, et al. (2016) Pellagra Secondary to Medication and Alcoholism: A Case Report and Review of Literature. Nutr Clin Pract 31(6): 785-789.
- Berdanier CD (2019) Corn ,niacin, and the history of pellagra. Nutr Today 54(6): 283-288.
- Fukuwatari T, Shibata K (2013) Nutritional Aspect of Tryptophan Metabolism. Int J Tryptophan Res 6(Suppl 1): 3-8.
- Badawy AAB (2014) Pellagra and alcoholism: a biochemical perspective. Alcohol Alcohol 49(3): 238-250.
- Gasperi V, Sibilano M, Savini I, Catani MV (2019) Niacin in the Central Nervous System: An Update of Biological Aspects and Clinical Applications. Int J Mol Sci 20(4): 974.
- Hegyi J, Schwartz RA, Hegyi V, (2004) Pellagra: dermatitis, dementia, and diarrhea. Int J Dermatol 43(1): 1-5.
- Baviera MR, Buigues LA, Campins RM, Garces SM, Martinez TI (2022) Refractory status epilepticus due to vitamin B6 deficit in a Parkinson’s disease patient in treatment with levodopa/carbidopa intestinal gel. Neurologia 37(7): 608-609.
- Sassan P, David LB (2023) Overview of water-soluble vitamins - UpToDate.
- Ropper AH (2023) Enfermedades Del Sistema Nervioso a Causa de Deficiencia Nutricional | Adams y Victor. Principios de Neurología, 11e | AccessMedicina | McGraw Hill Medical. McGraw Hill. (Adams & Victor, ed.). McGraw Hill; 2020.
- Wong KL (2022) Pyridoxine-Dependent Epilepsy - ALDH7A1. Epilepsy Case Studies: Pearls for Patient Care.
- Khan KM, Jialal I (2022) Folic Acid Deficiency. StatPearls.
- Shulpekova Y, Nechaev V, Kardasheva S, Sedova A, Kurbatova A, et al. (2021) The Concept of Folic Acid in Health and Disease. Molecules 26(12): 3731.
- Reynolds EH (2014) The neurology of folic acid deficiency. Handb Clin Neurol 120: 927-943.
- Briani C, Torre CD, Citton V, ManaraR, Pompanin S, et al. (2013) Cobalamin Deficiency: Clinical Picture and Radiological Findings. Nutrients 5(11): 4521-4539.
- Forrellat M, Gomis I, Gautier H (1999) Vitamina B12: metabolismo y aspectos clínicos de su deficiencia. Rev Cubana Hematol Inmunol Hemoter 15(3):159-174.
- Senol MG, Sonmez G, Ozdag F, Saracoglu M (2008) Reversible myelopathy with vitamin B12 Singapore Med Journal 49(11): e330-332.
- Antony AC (2009) Megaloblastic Anemias- Hematology: Basic Principles and Practice. 5th ed, Elsevier Churchill Livingstone, UK.
- World Health Organization (WHO) (2023) Vitamin A deficiency.
- Sassan P, David BL (2023) Overview of vitamin A - UpToDate.
- National Institutes of Health (NIH) (2022) Vitamin A and Carotenoids - Health Professional Fact Sheet.
- Larry JE (2023) Vitamin A Deficiency - Nutritional Disorders - MSD Manual Professional Edition.
- Dosedel M, Jirkovsky E, Macakova K, Krcmova LK, Javorska L, et al. (2021) Vitamin C-Sources, Physiological Role, Kinetics, Deficiency, Use, Toxicity, and Determination. Nutrients 13(2): 615.
- Spoelstra de Man AME, Elbers PWG, Oudemans-Van Straaten HM (2018) Vitamin C: should we supplement? Curr Opin Crit Care 24(4): 248-255.
- Young JI, Züchner S, Wang G (2015) Regulation of the Epigenome by Vitamin C. Annu Rev Nutr 35(1): 545-564.
- Valdes F (2006) [Vitamin C]. Actas Dermosifiliogr 97(9): 557-568.
- Ceglie G, Macchiarulo G, Marchili MR, Marchesi A, AufieroLR, et al. (2019) Scurvy: still a threat in the well-fed first world? Arch Dis Child 104(4): 381-383.
- Antonelli M, Burzo ML, Pecorini G, Massi G, Landolfi R, et al. (2018) Scurvy as cause of purpura in the XXI century: a review on this “ancient” disease. Eur Rev Med Pharmacol Sci 22(13): 4355-4358.
- Maxfield L, Crane JS (2022) Vitamin C Deficiency. The Bioarchaeology of Metabolic Bone Disease.
- Colombo ML (2010) An update on vitamin E, tocopherol and tocotrienol-perspectives. Molecules 15(4): 2103-2113.
- Miyazawa T, Burdeos GC, Itaya M, Nakagawa K, Miyazawa T (2019) Vitamin E: Regulatory Redox Interactions. IUBMB Life 71(4): 430-441.
- Kemnic TR, Coleman M (2022) Vitamin E Deficiency. Nutrition and the Eye: A Practical Approach, pp. 121-122.
- National Institutes of Health (NIH) (2023) Vitamin E - Health Professional Fact Sheet.
- Dror DK, Allen LH (2011) Vitamin E deficiency in developing countries. Food Nutr Bull 32(2): 124-143.
- Tanyel MC, Mancano LD (1997) Neurologic findings in vitamin E deficiency. Am Fam Physician 55(1): 197-201.
- Traber MG (2013) Vitamin E Metabolism and Requirements-Caballero B. Encyclopedia of Human Nutrition. 3rd Elsevier.






























