Acute Ischemic Stroke Management: A Systematic Review
Caiqing Shao1, Cuilan Li2, Liangchen Hu2, Daojing Li2, Xiaohai Li3, Yongnan Hao4* and Xiang Li5*
1Department of Clinical Medicine, Jining Medical University, Jining, China
2Department of Neurology, Affiliated Hospital of Jining Medical University, Jining, China
3Department of Rehabilitation Therapy, Jining Medical University, Jining, China
4Department of Emergency Stroke, Affiliated Hospital of Jining Medical University, Jining, China
5Department of Rehabilitation, Affiliated Hospital of Jining Medical University, Jining, China
Submission: February 13, 2023; Published: February 28, 2023
*Corresponding author: Xiang Li & Yongnan Hao, Department of Rehabilitation, Affiliated Hospital of Jining Medical University, Jining, China
Department of Emergency Stroke, Affiliated Hospital of Jining Medical University, Jining, China
How to cite this article: Caiqing Shao, Cuilan Li, Liangchen Hu, Daojing Li, Xiaohai Li, Yongnan Hao* and Xiang Li. Acute Ischemic Stroke Management: A Systematic Review. Open Access J Neurol Neurosurg 2023; 18(1): 555977.DOI: 10.19080/OAJNN.2023.18.555977.
Abstract
With the increasing improvement of living standards, stroke also has a very high incidence rate in the world, in which ischemic stroke accounts for 60%-80% [1]. But many people, even medical students, know very little about stroke treatment. Therefore, I intend to write a review of the treatment of Acute Ischemic Stroke (AIS), which will give the reading public a systematic understanding of the treatment of acute ischemic stroke, reduce the public’s fear of stroke, and make patients and their families trust doctors. This article focuses on the treatment options for mild ischemic stroke and heavy ischemic stroke. The treatment options for mild ischemic stroke are thrombolytic therapy or short-term dual antiplatelet therapy [2]; the treatment options for heavy ischemic stroke are bridging therapy or direct endovascular intervention.
Keywords: Ischemic stroke; Thrombolytic drugs; Interventional therapy; Overview
Introduction
With the aging of the population, stroke has become a major killer threatening human life, in which ischemic stroke accounts for 60%-80%, while the mortality of patients with acute ischemic stroke within one year is as high as 10%, and the disability rate is as high as 20% ~ 25% [3]. Among the patients with ischemic stroke, 28%-46% [4] belong to acute severe ischemic stroke. Patients with acute CT-confirmed ischemic stroke were classified as either mild or severe according to National Institutes of Health Stroke Scale (NIHSS) score and clinical symptoms (e.g., presence of binocular gaze, tongue extension to one side, facial paresis, and tetraplegia). Patients with acute mild ischemic stroke with significant disabling deficits (e.g., aphasia or hemiparesis) are treated with thrombolysis, [5] and patients with acute mild ischemic stroke without disabling deficits are treated with short-term dual antiplatelet therapy [6]. Patients with severe acute ischemic stroke are treated with endovascular intervention.
Acute mild ischemic stroke
The definition of acute mild ischemic stroke: [7] It is the sudden focal mild neurological deficits and neurological dysfunction caused by vascular causes are caused by ischemic infarction associated with imaging and clinical symptoms, rather than cerebral hemorrhage detected by imaging. However, specific quantifiers are not standardized and are usually based on the NIHSS, which requires a score ≤1 [8] each or utilizes certain limitations, primarily NIHSS ≤6 [9]. It is difficult to distinguish acute mild ischemic stroke from transient ischemic attack. The fundamental difference between the two definitions is whether there is focal ischemic focus in brain tissue. Early local lesions may not have been found by imaging examination, or the location of local lesions has little impact on the patient’s physical function, so it is sometimes difficult to distinguish the two, but the treatment measures are basically the same.
Two treatments: Currently, there are two controversial treatments for acute mild ischemic stroke. One is intravenous thrombolysis, and a pooled analysis of nine trials showed that in patients with significant disabling impairments such as aphasia or hemiplegia, the chance of good functional outcomes after 3 months of thrombolysis increased by nearly 10% [10,11]. Therefore, current guidelines still recommend thrombolysis only for mild stroke patients with significant disabling symptoms. The other is dual-antibody therapy, [12] IST-3 found evidence that was beneficial for patients presenting within 6 hours of stroke symptoms, but this benefit increased with NIHSS, whereas for patients with mild stroke symptoms patients were less beneficial. Therefore, dual antiplatelet secondary prophylaxis is used for minor stroke and TIA-treated patients without disabling symptoms. However, both treatments have advantages and disadvantages. The PRISMS trial, a randomized controlled trial (RCT), suggested that in patients with low NIHSS and no disabling deficits, rtPA may provide no benefit and may increase the risk of symptomatic intracranial hemorrhage [9]. Because symptoms of the posterior circulation (eg, dizziness, gait imbalance) are underrepresented in the NIHSS, strokes in this region are more likely to be defined as “minor” when using the NIHSS score. If the patient is not treated with thrombolytic therapy, but only dual-antibody therapy may be given to the patient, which may delay the patient’s condition. Advanced imaging techniques may help to better assess the risk-benefit ratio of thrombolytic therapy in mildly symptomatic acute ischemic stroke patients. Further trial results on this topic are expected shortly.
Thrombolytic drugs: First generation thrombolytic drugs: urokinase (UK), streptokinase (SK); Second generation thrombolytic drugs: rt-PA, Pro-UK; Third generation thrombolytic drugs: r-PA, TNK-tPA. The first generation of thrombolytic agents belong to non-specific plasminogen activators, which can induce many inactive plasminogen to change into active plasminogen after injection into blood vessels. A large amount of plasmin can not only degrade fibrin in thrombus, but also fibrinogen and coagulation factors in blood [13]. α2-antiplasmin cannot completely antagonize the large amount of plasmin produced, so the risk of bleeding is high. The second and third generation thrombolytic agents belong to specific plasminogen activators. After being injected into blood vessels, they can only activate plasminogen into plasmin after binding with fibrin in thrombus. So, it can specifically degrade fibrin in thrombus. A small amount of plasmin degrades fibrinogen and coagulation factors in the blood, which will also be α 2-antiplasmin antagonism, so the risk of bleeding is small.
Indications for thrombolytic drugs: 1) The age must be greater than or equal to 18 years. 2) The time from the onset of first symptoms to the administration of the drug, usually within 3-4.5 hours for the application of alteplase, and within 6 hours for the application of urokinase thrombolysis. 3) The patient must be a patient with ischemic stroke with CT exclusion of cerebral hemorrhage. 4) The patient or family must sign an informed consent form.
Complications and treatment measures after thrombolytic drugs
Allergy: [14] usually mild, which can be manifested by skin rash, urticaria, bronchospasm, angiogenic edema, hypotension, shock and other symptoms related to allergic reactions. Treatment methods: 1) Once severe allergy is found, stop thrombolytic drugs immediately and monitor vital signs. 2) Mild cases were treated with steroids and antihistamines; In severe cases, adrenal hormone shall be used and tracheal intubation shall be conducted if necessary, and rescue team shall be started.
Vessel reocclusion: [15] acute ischemic stroke is a common complication of thrombolytic therapy, which is related to the deterioration of clinical symptoms. Early reocclusion predicts poor long-term prognosis. It may be related to the activation and aggregation of exposed platelets of lipid core after thrombolysis or vascular endothelial injury, inadequate use of antiplatelet drugs during perioperative period or resistance to antiplatelet drugs. Treatment methods: the treatment measures for re occlusion of blood vessels within 24 hours include arterial thrombectomy, early antiplatelet, early anticoagulation and combined antithrombotic therapy. The 2014 edition of the Chinese expert consensus on intravascular treatment of acute ischemic stroke points out that thrombolysis combined with antiplatelet therapy may reduce the occurrence of re occlusion; Combined application of GP IIb / IIIa inhibitors can reduce the incidence of reocclusion and treat reocclusion.
Systemic hemorrhage: [16] systemic hemorrhage is another potential complication after thrombolysis. Patients with recent large-scale surgery may have a higher risk of systemic hemorrhage. The common bleeding sites in clinic include gastrointestinal tract, urogenital tract, subcutaneous, retroperitoneal and epistaxis. Treatment methods: 1) Stop rt-PA immediately, detect PT, APTT, fibrinogen, whole blood count and blood matching. 2) Appropriate supportive therapy: monitoring blood pressure, rehydration, blood transfusion and improving circulation. 3) Antifibrinolytic therapy: tranexamic acid, if fibrinogen is too low (<1 g / L), can be given cold cryoprecipitate.
Acute severe ischemic stroke
Discussion on the definition of acute severe ischemic stroke: Acute severe ischemic stroke can be defined according to the comprehensive analysis of clinical symptoms, NIHSS score and auxiliary examination. 1) Severe clinical symptoms. The patient has coma, disappearance of eye reflex to light, double eye gaze, limb muscle strength grade 1-2, etc. Most studies show that the NIHSS score of this kind of ischemic stroke is ≥ 8 ~ 10 [17]. 2) The load of thrombus was large, and the opening rate of intravenous thrombolysis was low. For example, the reopening rate of middle cerebral artery occlusion is 30%, the internal carotid artery terminal is only about 6%, and the basilar artery is 30% [18]. When the thrombus length is ≥ 8mm, the reopening rate of intravenous thrombolysis is less than 1% [19]. 3) The lesion is large in size. The ischemic core area can be seen through CT imaging examination, but the size of the ischemic core area is not specified as severe, which needs to be judged in combination with physical signs. The above three points are the key points and difficulties in the treatment of acute ischemic stroke. Patients with acute ischemic stroke who meet the above three points should be classified as acute severe ischemic stroke.
Endovascular therapy
Arterial thrombolysis: There are several potential advantages of intra-arterial thrombolysis, including angiographic evaluation of thrombus, obstruction and collateral circulation sites, improvement of recanalization, provision of local concentration of higher concentration thrombolytic agents, and prolongation of treatment time. Intra-arterial thrombolysis has great hope [20]. Patients with middle cerebral artery occlusion with NIHSS score of 16 were treated with intra-arterial thrombolysis within 3h. The average time to start treatment was 106 minutes (range: 10-180 minutes). During the follow-up of 1-3 months, 66% of the treated patients had a modified Rankine scale (mRS) score of 2 or less, a recanalization rate of 77%, a symptomatic intracranial hemorrhage rate of 11%, and a mortality rate of 23% [21]. Arterial thrombolysis is still unable to overcome the shortcomings of narrow time window, low recanalization rate and poor prognosis. It is generally not used alone in clinical practice.
Stent-retriever thrombectomy: In 2015, the results of four prospective trials [22-25] showed that endovascular therapy (EVT) was beneficial to some acute severe ischemic stroke patients, and then EVT became the standard treatment. The time window of intravascular interventional therapy < 4.5h, so researchers from the Netherlands conducted a multicenter randomized clinical trial of endovascular treatment of acute ischemic stroke (MR CLEAN) [26]. This study shows that intra-arterial treatment within 6 hours is safe and effective for patients with acute ischemic stroke caused by anterior circulation intracranial artery occlusion. In the 6h treatment window, patients with stent thrombectomy and reperfusion had a high rate, low mortality, and a good prognosis at 90 days. Saver et al. [27] randomly divided the patients with proximal occlusion of intracranial vessels into a control group only receiving intravenous thrombolysis and an intervention group receiving stent thrombectomy within 6 hours of onset. The reperfusion rate in the intervention group was 88%. Both the final control group and the intervention group reduced the disability degree of patients after 90 days, but the mRS score (0[asymptomatic] to 6[death]) of the intervention group was lower than that of the control group. The results showed that patients with intracranial proximal large vessel occlusion who received stent thrombectomy within the 6h treatment window could greatly reduce the mortality and disability degree of patients. Stent thrombectomy has become a class IA indication for patients with acute severe ischemic stroke symptoms [28]. The interventional thrombolytic therapy window is a time set by retrospective analysis of previous thrombolytic patients and comparison of the survival value of patients at various time periods after thrombolytic therapy. However, the situation of each person is different. For adult patients with acute ischemic stroke caused by occlusion of large vessels of the anterior circulation more than 6 hours after onset, mechanical thrombectomy is combined with optimal medical management, is it possible to improve functional outcomes more than medication alone? [29]. The DEFUSE-3 study, and the dawn study conducted clinical trials under their established inclusion criteria. Screening criteria for patients in the DEFUSE-3 study: CTA or MRA at 6-16 hours after onset confirmed that the extracranial or intracranial segment of the carotid artery or the proximal end of the middle cerebral artery was occluded, with small infarct core and large ischemic penumbra (CTP / DWI and MRP assessment: infarct core volume ≥ 70 ml, ischemic tissue / infarct volume ≥ 1.8, and ischemic penumbra volume ≥ 15 ml). Screening criteria for patients in dawn study: CTA or MRA confirmed occlusion of intracranial segment of internal carotid artery or M1 segment of middle cerebral artery or two segments at the same time within 6-24 hours after onset does not match the severity of clinical symptoms and infarct volume (the judgment of infarct volume is based on magnetic resonance DWI or CT perfusion, and the rapid software is used for automatic calculation), age ≥ 18 years, and pre-stroke mRS 0-1 point. The results showed that the onset time of acute ischemic stroke caused by anterior circulation large vessel occlusion was 6-24 hours, and it met the screening criteria of patients in DEFUSE-3 or dawn study. It was recommended to adopt mechanical thrombectomy combined with the best medical management, rather than the best medical management, to improve the functional prognosis (quality of evidence: medium + + +; recommendation level: strong ↑). Nogueira et al. [30] included 206 patients whose clinical symptoms were inconsistent with imaging findings. 107 patients who received stent thrombectomy within 6-24 hours after the onset of the disease constituted the experimental group, and the other 99 patients who received conservative treatment constituted the control group. The 90-day mRS score of the experimental group was 5.5, while that of the control group was 3.4. There was no difference in the rate of intracranial hemorrhage and mortality after 90 days between the two groups. The results showed that the effect of interventional therapy for stroke patients beyond the treatment window was better than that of conservative therapy only. Because stent thrombectomy has the advantages of high vascular recanalization rate, good 90-day prognosis and wide treatment window, it has become the first choice for the treatment of acute severe ischemic stroke.
Contact thrombus aspiration: Contact thrombus aspiration is increasingly accepted in the treatment of intravascular stroke. Gory et al. [31] in a prospective single center series of 20 patients with large vessel occlusive stroke, we retrospectively evaluated the safety and effectiveness of arc intracranial catheter for first line contact aspiration. The median NIHSS score of 20 patients with large vessel occlusion was 15 at admission, and 12 patients (60%) had been treated with intravenous thrombolysis before. Proximal occlusion was in the anterior circulation in 16 patients (80%). In a median time of 26 minutes, primary inhalation was successful in 10 (67%) of 15 patients. In a median time of 31 minutes, 16 patients (80%) achieved overall successful reperfusion. The median discharge score of patients with successful reperfusion was 2.5. Six (35%) of the 17 patients were independent and six (35%) died within 90 days. Symptomatic cerebral hemorrhage occurred in 5% of the patients. Kim et al. [32] retrospectively analyzed 189 stroke patients who underwent contact thrombus aspiration. Among them, doctors used penumbra thrombectomy system to remove thrombi for 90 patients, and Sofia catheter to remove thrombi for 99 patients. The success rate of reperfusion was 94.4% vs 92.9%, p=0.656; Sofia group achieved more first pass effect (20.0% vs 39.4%, p=0.004). The clinical results showed no significant difference in mortality (11.1% vs 6.1%, p=0.213). Thrombus aspiration technology has the advantages of high vascular recanalization rate, low postoperative complications and good 90-day prognosis. BGC catheter can block the proximal blood flow and reduce the risk of distal occlusion caused by small emboli broken during thrombectomy. Kang et al. [33] retrospectively analyzed 429 patients (mean age over 68.4 years; m/f ratio 215:214). BGC was used in 45.2% of patients. Compared with the non BGC group, the BGC group had less distal or different site embolization (0.5% vs. 3.4%). According to many retrospective analyses, contact thrombus aspiration can be used as the first-line treatment for acute intracranial large vessel occlusion. In order to improve its distal navigability and further reduce the risk of mortality and bleeding complications, it is worth conducting a large-scale prospective study.
TRAP technology: TRAP technology is the combination of thrombectomy and aspiration. The principle is that BGC blocks the proximal blood flow, the middle catheter is pumped, and the distal stent is pulled. The therapeutic effect of multiple acute severe ischemic stroke patients has also been affirmed in clinical work. A retrospective analysis of 353 patients, including 215 patients in the stenting group, 32 patients in the thrombus aspiration group and 106 patients in the combined stent aspiration group. There was no significant difference in age and admission rate between groups. The successful recanalization rate was 35.8% in the stent pulling group, 34.4% in the thrombus aspiration group and 55.7% in the stent combined with thrombus aspiration group. Compared with these technologies, the combination of stent and catheter can improve the single successful recanalization rate [34]. In addition to this, there are also high-quality randomized controlled trials of ASTER 2 studies supporting the TRAP technique [35].
Bridge therapy: The treatment was a combination of Mechanical thrombectomy (MT) and intravenous thrombolysis (IVT) [36]. The aim of the THRACE trial was to compare the clinical prognosis of intravenous therapy alone and bridging therapy in 414 patients with moderate to severe stroke due to large artery occlusion enrolled from 26 centers in France and randomly assigned during rt-PA reperfusion therapy. The results showed that the proportion of patients with a good 3-month prognosis (mRS score 0 to 2) was significantly higher in the IVT+MT group than in the IVT alone group, but the incidence of sICH at 24 h and 3 months was not significantly different between the two groups.
Complications and treatment measures after endovascular therapy
Ischemia-reperfusion injury: [37] The substances involved in ischemia/reperfusion injury include white blood cells, Ca2+and free radicals [38]. During ischemia, the blood flow slows down, resulting in white blood cells more easily contacting the blood vessel wall, thus crossing the blood vessel wall and entering the brain tissue, causing immune response [39]. In the ischemic environment of brain cells, the transport rate of sodium and potassium pump on the cell membrane decreases, causing the reverse work of sodium and calcium transporters, thus overloading the intracellular calcium ions. These mechanisms are the main mechanisms of cell injury during ischemia, but the opening of blood vessels can also cause brain cell injury when blood is left in ischemic brain tissue [40]. During blood reperfusion, damaged cells will produce many reactive oxygen free radicals, which will cause damage to brain cells.
Hemorrhagic transformation: [41] When the occluded blood vessel is reopened, the reperfusion blood can exude through the damaged blood vessel wall. For the transformation of symptomatic hemorrhage, antithrombotic therapy and other bleeding drugs should be stopped and CT, Pt and APTT should be performed. The general antithrombotic therapy for asymptomatic bleeding can be continued.
Malignant brain edema: [42] Definition: 1. At least 50% of the blood supply areas of the middle cerebral artery have low density changes with disappearance of the sulcus and gyrus and compression of the lateral ventricle; 2. Midline structures such as septum pellucidum or pineal gland are displaced ≥ 5mm, accompanied by disappearance of basal cistern. Once malignant brain edema occurs, the mortality rate is as high as 80%, so we should strictly grasp the indications for thrombectomy, optimize the stroke treatment process, open blood vessels faster and operate fewer times, which are important preventive measures for malignant brain edema.
Piercing damage: In the process of endovascular mechanical opening, improper operation of catheters, guidewires, balloons, stents and other endovascular treatment devices may lead to vascular perforation; Secondly, intravascular treatment devices are easy to cause vasospasm during intravascular operation, and the occurrence of vasospasm will further increase the risk of vascular perforation. Once vascular perforation occurs, intracranial hemorrhage occurs. The blood pressure of patients should be strictly controlled so as not to be too high, but at the same time, cerebral tissue hypoperfusion should be prevented. Heparin is generally used during the treatment of intravascular opening. Heparin should be stopped immediately after vascular perforation and protamine should be used to neutralize the anticoagulant effect of heparin. For those who use antiplatelet drugs during operation, the transfusion of antiplatelet drugs should be stopped immediately. If necessary, platelet transfusion can help to reverse the antiplatelet effect. For the vascular perforation complications that cannot be effectively stopped by conservative drug treatment, it is also an effective hemostatic method to use balloon low-pressure continuous expansion to compress the bleeding point. However, long-term compression of the perforated blood vessels is likely to lead to the occurrence of distal ischemia. Covered stent implantation can also achieve hemostatic effect by blocking the perforation of the bleeding artery. Stent implantation can easily lead to thrombosis in the stent and occlusion of the perforating artery. For the vascular perforation with small internal diameter that is not suitable for balloon or stent implantation treatment, it is also an important method to quickly occlude the perforated vessels by embolizing the perforated vessels with materials such as spring coils, gel sponges and thrombin (Figure 1 & 2).
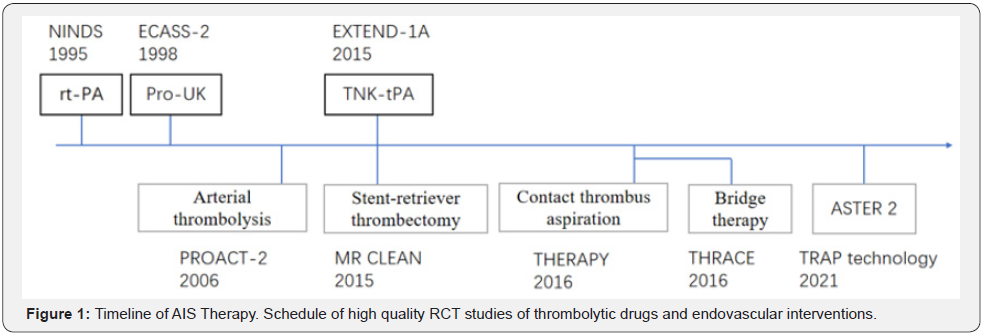
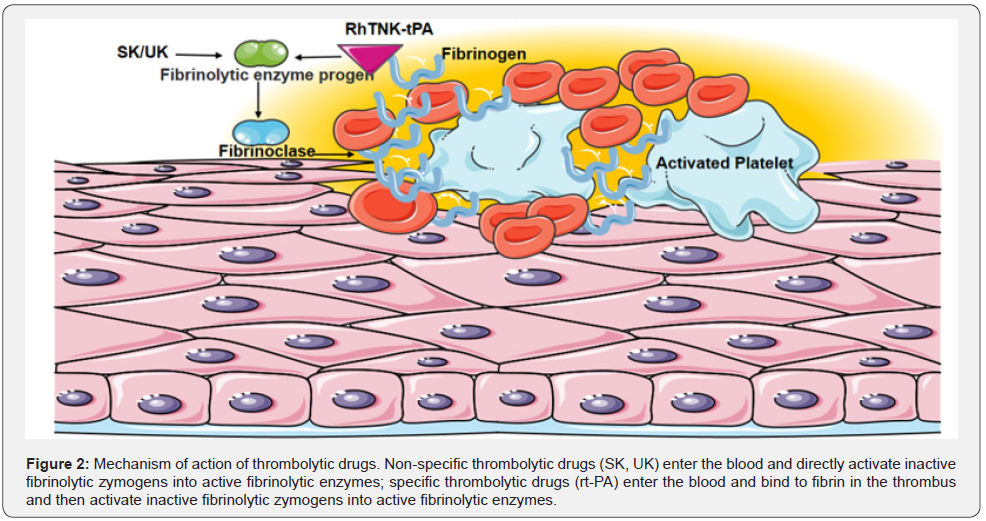
Conclusion
Intravenous thrombolytic drugs are mainly indicated for acute mild ischemic strokes. Intravascular interventional therapies are mainly applicable to large vessel occlusion, such as serial occlusion, end of internal carotid artery occlusion and basilar artery occlusion [43-50]. The treatment of acute ischemic stroke is becoming more available and more effective. However, patients may still have some sequelae, such as hemiparesis, slurred speech and cognitive impairment. Therefore, it is important for physicians to adequately assess patients for compliance with treatment principles and to fully explain possible complications with families and patients before taking therapeutic measures.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding Statement
This work was supported by grants from the National Natural Science Foundation of China (81901228) and the Outstanding Young talents in Medicine and health of Qilu (2021-QLJQ-003).
References
- Song S, Liang L, Fonarow GC, Smith EE, Bhatt DL, et al. (2019) Comparison of. Clinical Care and In-Hospital Outcomes of Asian American and White Patients with Acute Ischemic Stroke. JAMA Neurol 76(4): 430-439.
- Ferrari J, Reynolds A, Knoflach M, Sykora M (2021) Acute Ischemic Stroke with Mild Symptoms-To Thrombolyse or Not to Thrombolyse? Front Neurol 12: 760813.
- Morotti A, Poli L, Costa P (2019) Acute Stroke. Semin Neurol 39(1): 61-72.
- Smith WS, Lev MH, English JD, Camargo EC, Chou M, et al. (2009) Significance of large vessel intracranial occlusion causing acute ischemic stroke and TIA. Stroke 40(12): 3834-3840.
- Campbell B, Mitchell PJ, Churilov L, Yassi N, Kleinig TJ, et al. (2018) Tenecteplase versus Alteplase before Thrombectomy for Ischemic Stroke. N Engl J Med 378(17): 1573-1582.
- Kleindorfer DO, Towfighi A, Chaturvedi S, Cockroft KM, Gutierrez J, et al. (2021) 2021 Guideline for the Prevention of Stroke in Patients with Stroke and Transient Ischemic Attack: A Guideline from the American Heart Association/American Stroke Association. Stroke 52(7): e364-e467.
- Strambo D, Zambon AA, Roveri L, Giacalone G, Di Maggio G, et al. (2015) Defining minor symptoms in acute ischemic stroke. Cerebrovasc Dis 39(3-4): 209-215.
- Fischer U, Baumgartner A, Arnold M, Nedeltchev K, Gralla J, et al. (2010) What is a minor stroke? Stroke 41(4): 661-666.
- Khatri P, Kleindorfer DO, Devlin T, Sawyer RJ, Starr M, et al. (2018) Effect of Alteplase vs Aspirin on Functional Outcome for Patients with Acute Ischemic Stroke and Minor Nondisabling Neurologic Deficits: The PRISMS Randomized Clinical Trial. JAMA 320(2): 156-166.
- Emberson J, Lees KR, Lyden P, Blackwell L, Albers G, et al. (2014) Effect of treatment delay, age, and stroke severity on the effects of intravenous thrombolysis with alteplase for acute ischaemic stroke: a meta-analysis of individual patient data from randomised trials. Lancet 384(9958): 1929-1935.
- Lees KR, Emberson J, Blackwell L, Bluhmki E, Davis SM, et al. (2016) Effects of Alteplase for Acute Stroke on the Distribution of Functional Outcomes: A Pooled Analysis of 9 Trials. Stroke 47(9): 2373-2379.
- Sandercock P, Wardlaw JM, Lindley RI, Dennis M, Cohen G, et al. (2012) The benefits and harms of intravenous thrombolysis with recombinant tissue plasminogen activator within 6 h of acute ischaemic stroke (the third international stroke trial [IST-3]): a randomised controlled trial. Lancet 379(9834): 2352-2363.
- Abdul S, Leebeek FW, Rijken DC, Uitte DWS (2016) Natural heterogeneity of α2-antiplasmin: functional and clinical consequences. Blood 127(5): 538-545.
- Wang YX, Li YQ, Chen Y, Zhang CH, Dong Z, et al. (2018) Analysis of related factors of orolingual angioedema after rt-PA intravenous thrombolytic therapy. Eur Rev Med Pharmacol Sci 22(5): 1478-1484.
- Rapaport E (1991) Thrombolysis, anticoagulation, and reocclusion. Am J Cardiol 68(16): 17E-22E.
- Lo KY, Biby S, Sethi P, Campbell BC (2016) Symptomatic intracerebral and systemic hemorrhage after stroke thrombolysis in patients taking ticagrelor. Int J Stroke 11(6): NP71-NP72.
- Saver JL, Jahan R, Levy EI, Jovin TG, Baxter B, et al. (2012) Solitaire flow restoration device versus the Merci Retriever in patients with acute ischaemic stroke (SWIFT): a randomised, parallel-group, non-inferiority trial. Lancet 380(9849): 1241-1249.
- Alexandrov AV (2010) Current and future recanalization strategies for acute ischemic stroke. J Intern Med 267(2): 209-219.
- Riedel CH, Zimmermann P, Jensen-Kondering U, Stingele R, Deuschl G, et al. (2011) The importance of size: successful recanalization by intravenous thrombolysis in acute anterior stroke depends on thrombus length. Stroke 42(6): 1775-1777.
- Jones MM, Nogajski JH, Faulder K, Harrington T, Ng P, et al. (2005) Intra-arterial thrombolysis in acute ischaemic stroke. Intern Med J 35(5): 300-302.
- Bourekas EC, Slivka A, Shah R, Mohammad Y, Slone HW, et al. (2009) Intra-arterial thrombolysis within three hours of stroke onset in middle cerebral artery strokes. Neurocrit Care 11(2): 217-222.
- Jovin TG, Chamorro A, Cobo E, de Miquel MA, Molina CA, et al. (2015) Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med 372(24): 2296-2306.
- Campbell BC, Mitchell PJ, Kleinig TJ, Dewey HM, Churilov L, et al. (2015) Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med 372(11): 1009-1018.
- Goyal M, Demchuk AM, Menon BK, Eesa M, Rempel JL, et al. (2015) Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med 372(11): 1019-1030.
- Saver JL, Goyal M, Bonafe A, Diener HC, Levy EI, et al. (2015) Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N Engl J Med 372(24): 2285-2295.
- Pirson F, Hinsenveld WH, Goldhoorn RB, Staals J, de Ridder IR, et al. (2021) MR CLEAN-LATE, a multicenter randomized clinical trial of endovascular treatment of acute ischemic stroke in The Netherlands for late arrivals: study protocol for a randomized controlled trial. Trials 22(1): 160.
- Saver JL, Goyal M, Bonafe A, Diener HC, Levy EI, et al. (2015) Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N Engl J Med 372(24): 2285-2295.
- Peisker T, Koznar B, Stetkarova I, Widimsky P (2017) Acute stroke therapy: A review. Trends Cardiovasc Med 27(1): 59-66.
- Albers GW, Lansberg MG, Kemp S, Tsai JP, Lavori P, et al. (2017) A multicenter randomized controlled trial of endovascular therapy following imaging evaluation for ischemic stroke (DEFUSE 3). Int J Stroke 12(8): 896-905.
- Nogueira RG, Jadhav AP, Haussen DC, Bonafe A, Budzik RF (2018) Thrombectomy 6 to 24. Hours after Stroke with a Mismatch between Deficit and Infarct. N Engl J Med 378(1): 11-21.
- Gory B, Turjman F (2018) Contact Aspiration with the New ARC Catheter for Thrombectomy of. Acute Ischemic Stroke: Single-Center Results. World Neurosurg 109: e374-e381.
- Kim YW, Hwang YH, Kim YS, Kang DH (2019) Frontline contact aspiration thrombectomy using SOFIA catheter for acute ischemic stroke: period-to-period comparison with Penumbra catheter. Acta Neurochir (Wien) 161(6): 1197-1204.
- Kang DH, Kim BM, Heo JH, Nam HS, Kim YD, et al. (2018) Effect of balloon guide catheter utilization on contact aspiration thrombectomy. J Neurosurg 1-7.
- Mizokami T, Uwatoko T, Matsumoto K, Ooya Y, Hashimoto G, et al. (2019) Aspiration Catheter Reach to Thrombus (ART) Sign in Combined Technique for Mechanical Thrombectomy: Impact for First-Pass Complete Reperfusion. J Stroke Cerebrovasc Dis 28(10): 104301.
- Lapergue B, Blanc R, Costalat V, Desal H, Saleme S, et al. (2021) Effect of Thrombectomy with Combined Contact Aspiration and Stent Retriever vs Stent Retriever Alone on Revascularization in Patients with Acute Ischemic Stroke and Large Vessel Occlusion: The ASTER2 Randomized Clinical Trial. JAMA 326(12): 1158-1169.
- Bracard S, Ducrocq X, Mas JL, Soudant M, Oppenheim C, et al. (2016) Mechanical thrombectomy after intravenous alteplase versus alteplase alone after stroke (THRACE): a randomised controlled trial. Lancet Neurol 15(11): 1138-1147.
- Kalogeris T, Baines CP, Krenz M, Korthuis RJ (2012) Cell biology of ischemia/reperfusion injury. Int Rev Cell Mol Biol 298: 229-317.
- Iadecola C, Buckwalter MS, Anrather J (2020) Immune responses to stroke: mechanisms, modulation, and therapeutic potential. J Clin Invest 130(6): 2777-2788.
- Kalogeris T, Baines CP, Krenz M, Korthuis RJ (2016) Ischemia/Reperfusion. Compr Physiol 7(1): 113-170.
- Granger DN, Kvietys PR (2015) Reperfusion injury and reactive oxygen species: The evolution of a concept. Redox Biol 6: 524-551.
- Zhu F, Labreuche J, Haussen DC, Piotin M, Steglich-Arnholm H, et al. (2019) Hemorrhagic Transformation After Thrombectomy for Tandem Occlusions. Stroke 50(2): 516-519.
- Fuhrer H, Schönenberger S, Niesen WD, Seide S, Meyne J, et al. (2019) Endovascular stroke treatment's impact on malignant type of edema (ESTIMATE). J Neurol 266(1): 223-231.
- Yang H, Ma N, Zhang S, Huo X, Gao F, et al. (2018) Endovascular revascularisation of acute tandem vertebrobasilar artery occlusion: seven case series with literature reviews. Stroke Vasc Neurol 3(1): 17-21.
- Lescher S, Czeppan K, Porto L, Singer OC, Berkefeld J (2015) Acute stroke and obstruction of the. extracranial carotid artery combined with intracranial tandem occlusion: results of interventional revascularization. Cardiovasc Intervent Radiol 38(2): 304-313.
- Piechowiak EI, Kaesmacher J, Zibold F, Dobrocky T, Mosimann PJ, et al. (2020) Endovascular treatment of tandem occlusions in vertebrobasilar stroke: technical aspects and outcome compared with isolated basilar artery occlusion. J Neurointerv Surg 12(1): 25-29.
- Mizowaki T, Fujita A, Inoue S, Kurihara E (2021) Endovascular Treatment for Vertebrobasilar Artery Tandem Occlusion Beyond 24 Hours from Onset of Stroke-Case Report. Vasc Endovascular Surg 55(1): 77-80.
- Slawski DE, Jumaa MA, Salahuddin H, Shawver J, Humayun MJ, et al. (2018) Emergent carotid endarterectomy versus stenting in acute stroke patients with tandem occlusion. J Vasc Surg 68(4): 1047-1053.
- Dababneh H, Guerrero WR, Khanna A, Hoh BL, Mocco J (2012) Management of tandem occlusion stroke with endovascular therapy. Neurosurg Focus 32(5): E16.
- Egashira Y, Yoshimura S, Enomoto Y, Ishiguro M, Yamada K, et al. (2013) Efficacy and limitations of multimodal endovascular revascularization other than clot retrieval for acute stroke caused by large-vessel occlusion. J Stroke Cerebrovasc Dis 22(6): 851-856.
- Pfefferkorn T, Holtmannspötter M, Schmidt C, Bender A, Pfister HW, et al. (2010) Drip, ship, and retrieve cooperative recanalization therapy in acute basilar artery occlusion. Stroke 41(4): 722-726.






























