Guillain-Barré Syndrome Post Covid-19 Vaccination: An Overview
Maria Alejandra Nieto Salazar1*, Felipe Velasquez Botero2*, Johanna Stefany Canenguez Benitez3, Guillermo Andres Moreno Cortes4, Jose Luis Hernandez Aparicio5, Franklin C Baron1, Laura Sofia Triviño1, María Ostorga6 and Alfredo Juarez Aguilar7
1Juan N Corpas University, Larkin Community Hospital, Miami, Florida
2CES University, Larkin Community Hospital, Miami, Florida
3University of El Salvador, Larkin Community Hospital, Miami, Florida
4Department of Family Medicine, F.U.C.S University, Bogotá, Colombia; Larkin Community Hospital, Miami, Florida
5University of El Salvador, Larkin Community Hospital, Miami, Florida
6Evangelical University of El Salvador, Larkin Community Hospital, Miami, Florida
7Autonomous University of Baja California, Larkin Community Hospital, Miami, Florida
Submission: July 18, 2022; Published: July 28, 2022
*Corresponding author: Maria Alejandra Nieto Salazar, Juan N Corpas University, Larkin Community Hospital, Miami, Florida
Felipe Velasquez Botero, CES University, Larkin Community Hospital, Miami, Florida
How to cite this article: Maria A N S, Felipe V B, Johanna S C B, Guillermo A M C, Jose Luis HA, et al. Guillain-Barré Syndrome Post Covid-19 Vaccination: An Overview. Open Access J Neurol Neurosurg 2022; 17(3): 555962.DOI: 10.19080/OAJNN.2022.17.555962.
Abstract
Guillain-Barré Syndrome (GBS) is a disease of autoimmune origin that causes demyelination of the nerves. It has been reported as a complication following the administration of some vaccines, such as the vaccine against Coronavirus disease (COVID-19). Infection by this virus may also be responsible for this complication. In the United States, it has been observed as a side effect in vaccines manufactured by Pfizer and Johnson & Johnson (Janssen). After the administration of the first dose of Pfizer’s vaccine, some cases of GBS have been reported, with most of the cases being female. Johnson & Johnson, on its part, reported some cases that required hospitalization and one that resulted in a fatality. Concerning Moderna vaccination and the development of GBS, no studies have demonstrated a clear link between the development of this condition. However, an apparent connection exists between the components of each vaccine, their mechanism of action, and each individual’s immunological response. Despite this, there is clear evidence that vaccination against COVID-19 has a low risk of causing GBS.
Keywords: GBS; Guillain-Barré Syndrome; COVID-19 Vaccination; SCoVaG; SARS-CoV-2 vaccination associated with GBS
Abbreviations: GBS: Guillain-Barré Syndrome, COVID-19: Coronavirus disease, SARS-CoV-2: Severe Acute Respiratory Syndrome Coronavirus 2, SCoVaG: SARS-CoV-2 vaccination associated with GBS, RNA: Ribonucleic Acid, mRNA: messenger RNA, CDC: Centers for Disease Control and Prevention, IVIG: intravenous immunoglobulin, COVE: Coronavirus Efficacy Trial, NCS: nerve conduction studies, PEG: polyethylene glycol, FDA: Food and Drug Administration, TTS: thrombocytopenia syndrome, VAERS: Vaccine Adverse Events Reporting System
Introduction
Guillain Barré syndrome (GBS) is a group of acute polyneuropathies of immunological origin mediated by autoantibodies that are usually the result of an immunological reaction induced by a previous infection in the host, which causes cross-linking with specific epitopes of peripheral nerves, a process called molecular mimicry, thus creating an inflammatory self-reaction that can affect any myelinated nerve, whether motor, sensory, cranial, or sympathetic [1]. This inflammatory process leads to demyelination and axonal loss of nerve tissue, producing slow efferent conduction and, consequently, inherent muscle weakness. GBS occurs worldwide with a global incidence of 1 to 2 cases per 100,000 per year. Although all age groups are affected, the incidence increases by approximately 20% with each 10-year increase in age beyond the first decade of life. In addition, the incidence is slightly higher in men than in women [2]. Even though its main trigger is infectious processes (primarily respiratory and gastrointestinal), some cases of GBS have occurred following vaccination.
In the United States, GBS has been reported after administering several vaccines, including meningococcal, recombinant zoster, and influenza. In addition, cases of GBS have been reported after Coronavirus disease (COVID-19) infection or vaccination. However, a possible causal association of COVID-19 with the risk of GBS remains unclear [3].
Coronaviruses are RNA-enveloped, positive-stranded viruses that can affect humans and animals. In late 2019, a new strain called Severe Acute Respiratory Syndrome Coronavirus 2 (SARSCoV- 2) initiated the current COVID-19 pandemic [4]. In efforts to achieve epidemiological control, various vaccines with different immunological mechanisms were created. There are three types of vaccines in use in the United States: Pfizer, Johnson & Johnson (Janssen), and Moderna. Some of them have been linked to GBS, specifically Pfizer and Johnson & Johnson vaccines. By July 24, 2021, 132 preliminary reports of GBS had been received in the United States after approximately 13.2 million doses of the Johnson & Johnson vaccine had been administered [5]. Nevertheless, since then, other cases have been documented attributable to the vaccine manufactured by Pfizer. The primary objective of this review is to compile the most current scientific literature regarding the development of GBS in individuals exposed to COVID-19 vaccination.
Materials and Methods
In order to conduct this review, extensive searches of databases such as PubMed, National Center for Biotechnology Information (NCBI), EBSCO, and UpToDate were performed. Most of the articles included were published in 2019 or later. All of the members of this research review analyzed a total of twenty-two articles, and the most relevant information was taken from each of them. Clinical research papers and observational studies, written and published in English were among the articles examined here.
Discussion
Pfizer-BioNTech COVID-19 Vaccine
Components and mechanism of action
The COVID-19 vaccine is claimed to contain only safe ingredients. COVID-19 vaccines are composed almost entirely of ingredients found in various foods such as fats, sugars, and salts. The Pfizer-BioNTech COVID-19 vaccine contains a harmless piece of Messenger Ribonucleic Acid (mRNA). This mRNA, a nucleosidemodified mRNA encoding the viral spike (S) glycoprotein of SARS-CoV-2, instructs the cells in the body on how to create an immune response against the virus responsible for COVID-19. In producing an immune response, the body discards all of the vaccine ingredients, just as it would discard any substance that is no longer required by cells. This process is a part of normal body functioning [6].
The vaccine contains several lipids which assist the mRNA in entering the cells, as well as salts and sugars such as dibasic sodium phosphate dihydrate, monobasic potassium phosphate, potassium chloride, and sodium chloride and sucrose. Furthermore, these molecules keep the vaccine components stable during manufacturing, freezing, shipping, and storing the vaccine until it is ready to be administered to a vaccine recipient [6].
Common adverse effects
Pfizer COVID-19 vaccine has been associated with pain, swelling, redness, fever, fatigue, headache, chills, vomiting, diarrhea, muscle pain, joint pain, lymphadenopathy, shoulder injury, right axillary lymphadenopathy, paroxysmal ventricular arrhythmia, syncope, and right leg paresthesia (Table 1). Moreover, the Centers for Disease Control and Prevention (CDC) has identified 6 cases of anaphylaxis reaction following the Pfizer- BioNTech vaccine [7].
Cases of Guillain-Barré Post-vaccination
A few cases of GBS have been reported since the Pfizer vaccine was introduced. Finstener J. reported four cases of GBS post-Pfizer vaccination. The first patient was an 86-year-old female diagnosed with GBS one day after receiving the first dose of the mRNA-based SARS-CoV-2 vaccine. After receiving intravenous immunoglobulin (IVIGs) treatment, the patient partially recovered and did not require mechanical ventilation [8]. The second patient with SARSCoV- 2 vaccination associated with GBS (SCoVaG) was an 82-yearold female who developed GBS two weeks after the first dose of the Pfizer-BioNTech vaccine. Since the patient did not undergo nerve conduction studies (NCS), no GBS subtype could be determined. The patient did not require mechanical ventilation, but she was treated with intravenous immunoglobulins, though only a partial recovery was achieved [8]. The third patient was a 77-year-old male diagnosed with GBS after 72 hours of the first dose of the vaccine. Again, IVIGs were administered, and plasma exchange was performed, with a positive outcome [9]. Furthermore finally, the fourth patient was a 52-year-old female diagnosed with GBS three hours after receiving the first dose of a Pfizer vaccine. She was treated with steroids and pregabalin and recovered partially [8].
Kim J. reported five cases of GBS and variations after vaccination, all presenting symptoms soon after receiving the first dose. The first patient, a 32-year-old female, presented with acute cervical-brachial weakness ten days after vaccination. She was treated with intravenous immunoglobulins and slowly recovered. The second patient, a 43-year-old male, showed symptoms eleven days after being vaccinated. This patient was treated with IVIGs and showed satisfactory results. The third patient, a 58-year-old female, presented with symptoms four days after vaccination. After receiving IVIGs, she underwent plasmapheresis. Patients two and three both presented with a classic form of GBS and were still unable to walk two months after the onset of the illness. The fourth patient was an 18-year-old female who presented with symptoms twelve days after vaccination. The patient died from aspiration pneumonia. The fifth patient, a 73-year-old female, presented symptoms 13 days post-vaccination. She was treated with IVIGs and gradually recovered. The fourth and fifth patients both presented with paraparetic GBS [10]. Finally, Laman T. described a case of a 58-year-old female who presented symptoms three days after receiving her first vaccination. Her symptoms improved following IVIG treatment [11]. This possible association with mRNA-based vaccination expands the potential triggers for an autoimmune-based attack on the peripheral nervous system [11].
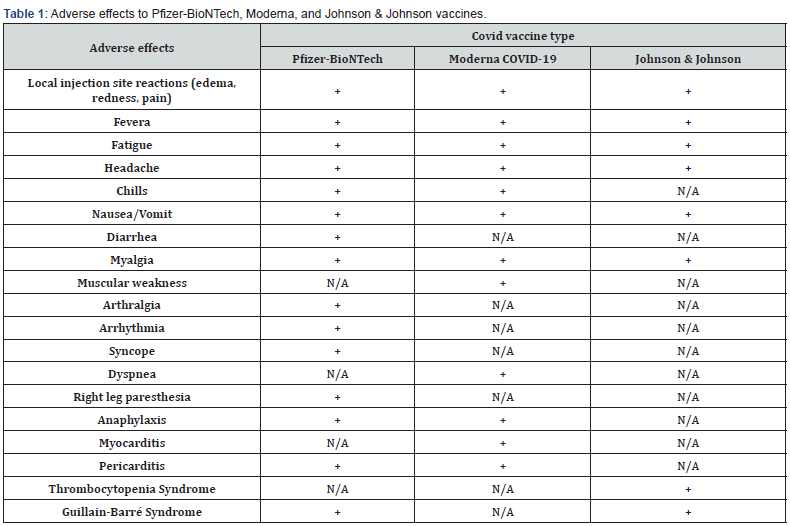
* +: present. N/A: absent.
Moderna COVID-19 Vaccine
Components and mechanism of action
Moderna COVID-19 vaccine is an mRNA-based vaccine. According to the Coronavirus Efficacy Trial (COVE), the mRNA- 1273 vaccine was highly effective at preventing symptomatic SARS-CoV-2 infection in a diverse population of adults [12]. Multiple ingredients are present in the vaccine, including mRNA, lipids, polyethylene glycol, dimyristoyl glycerol, cholesterol, 1.2-distearoyl-sn-glycero-3-phosphocholine, tromethamine, hydrochloride, acetic acid, sodium acetate trihydrate, and sucrose [13]. In addition, the Moderna vaccine contains material that originates from the virus that causes COVID-19, which produces a harmless protein specific to the virus. Once cells copy the protein, the genetic material in the vaccine is destroyed. Upon recognizing the presence of this protein, the immune system creates T cells and B cells that will later be able to neutralize the virus that causes COVID-19 [14].
Common adverse defects
There are a few common adverse effects of the vaccine, including fever, fatigue, nausea, chills, muscular weakness, muscle pain, headache, redness/swelling at the injection site, severe allergic reactions, breathing difficulty, and facial edema. In addition, a few individuals have experienced myocarditis and pericarditis after receiving the vaccine, most of whom were males under 40 years of age (Table 1) [14,15].
Cases of Guillain-Barré Post-vaccination
Currently, there is not enough evidence to suggest a direct link between GBS and Moderna’s COVID-19 vaccine [16,17]. However, it must be acknowledged that many studies are still being conducted with this vaccine, which could result in a finding that Moderna’s SARS-CoV-2 vaccination is associated with GBS.
Johnson & Johnson/Janssen COVID-19 Vaccine
Components and mechanism of action
In the Johnson and Johnson (Janssen) Covid-19 Vaccine, there are different types of constituents, such as sugar, salts (sodium chloride), acid stabilizers (citric acid monohydrate), ethanol (a type of alcohol), Polysorbate-80, 2-hydroxypropyl-β-cyclodextrin, and Trisodium citrate dihydrate. These components provide a mechanism of action that keeps the vaccine molecules stable during their preparation, shipment, and storage until the vaccine is ready for use. The vaccine also includes a protein, which is a replication incompetent recombinant Ad26 (Adenovirus) vector. This is an innocuous version of a virus unrelated to COVID-19 and encodes a stabilized variant of the SARS-CoV-2 Spike (S) protein that causes an immune response that protects the host against infection with COVID-19 [18].
Common adverse effects
Janssen Covid-19 Vaccine has been associated with local side effects such as pain, redness, and swelling at the injection site. Additional side effects might include fever, headache, fatigue, nausea, and muscle aches [19]. In addition, the CDC and the Food and Drug Administration (FDA) recommended a pause in the Janssen COVID-19 vaccine in April 2021 following reports of thrombosis with thrombocytopenia syndrome (TTS). This rare condition affects unusual sites such as the cerebral venous sinus (cerebral venous sinus thrombosis) after receiving the Janssen Vaccine (Table 1) [20].
Cases of Guillain-Barré Post-vaccination
In July 2021, the FDA declared a revision to the Factsheet for the Janssen Covid-19 (Adenovirus-based) vaccine about an increased risk of GBS following administration of the vaccine. The FDA announced about 100 cases of GBS that were reported to the Vaccine Adverse Events Reporting System (VAERS) after the administration of the Janssen Vaccine. In these reports, 95 vaccinated individuals required hospitalization due to GBS, and one of them died. However, the majority of those who developed this condition recovered. Most people with Janssen-related GBS showed symptoms within 42 days following administration of the vaccine.
In most cases, patients complained of difficulty walking, weakness, and tingling sensations in the legs that worsened and spread to the arms and chest, speaking and chewing or swallowing, double vision, difficulty moving the eyes, and problems with bladder or bowel control. Nevertheless, the chances of this adverse effect occurring are minimal. Nevertheless, those who exhibit any of the above symptoms following vaccination with the Janssen Covid-19 vaccine should seek medical attention as soon as possible [21,22].
Conclusion
As the COVID-19 pandemic arose and vaccinations became a day-to-day discussion among healthcare providers, themes such as risks and secondary effects before and after immunization have gained relevance. With the elaboration of this review, we express our concern about a neurological disease post- COVID-19 vaccination exposure. The clinical evidence that suggests a widespread relationship between vaccination and GBS development is low. Our references showed that mRNA-based vaccines, such as Moderna and Pfizer-BioNTech have statistically been less associated with GBS and critical symptomatic hospitalization cases. Moderna vaccination scheme might not represent a risk for the development of the disease or neurological symptoms compatible with GBS since there is not enough and adequate data available.
On the other hand, the Janssen COVID-19 vaccine, based on a different technology, has had several severe cases that required hospitalization linked to GBS symptoms. Overall, we observed a low risk of GBS caused by the induced immune response postvaccination. It is significant to our team to recommend surveillance of future vaccination schemes previously associated with autoimmune-based attacks on the peripheral nervous system, based on the shown evidence and reports (VEARS, COVE trails). Afflicted patients must undergo immediate medical evaluation if noticeable symptoms onset days after being immunized with any COVID-19 vaccine, ensuring an on-time assessment and treatment. Most of the patients who presented vaccination-related GBS have recovered successfully.
Conflict of interest:
The authors did not disclose any conflict of interest during the preparation of this article.
References
- Lu JL, Sheikh KA, Wu HS, Zhang J, Jiang ZF, et al. (2000) Physiologic-pathologic correlation in Guillain-Barré syndrome in children. Neurology 54(1): 33-39.
- Yuki N, Hartung HP (2012) Guillain-Barré N Engl J Med 366(24): 2294-304.
- Baxter R, Lewis N, Bakshi N, Vellozzi C, Klein NP (2012) CISA Network. Recurrent Guillain-Barre syndrome following vaccination. Clin Infect Dis 54(6): 800-804.
- Coronaviridae Study Group of the International Committee on Taxonomy of Viruses. The species Severe acute respiratory syndrome-related coronavirus: classifying 2019-nCoV and naming it SARS-CoV-2. Nat Microbiol 5(4): 536-544.
- Woo EJ, Jonas A, Dimova RB, Alimchandani M, Zinderman CE, et al. (2021) Association of Receipt of the Ad26.COV2.S COVID-19 Vaccine With Presumptive Guillain-Barré Syndrome, 326(16): 1606-1613.
- (2022) Centers for Disease Control and Prevention. Pfizer-BioNTech COVID-19 vaccine overview and Safety. Centers for Disease Control and Prevention.
- Meo SA, Bukhari IA, Akram J, Meo AS, Klonoff DC (2021) COVID-19 vaccines: comparison of biological, pharmacological characteristics and adverse effects of Pfizer/BioNTech and Moderna Vaccines. Eur Rev Med Pharmacol Sci 25(3): 1663-1669.
- Finsterer J, Scorza FA, Scorza CA (2021) Post SARS-CoV-2 vaccination Guillain-Barre syndrome in 19 patients. Clinics (Sao Paulo). 76: e3286.
- Omar MIF, Carazo JMV, Rivas JAP, Rodriguez IV (2021) COVID-19, Guillain-Barré syndrome, and the vaccine. A dangerous combination. Revista clinica espanola 221(9): 555-557.
- Kim JE, Min YG, Shin JY, Kwon YN, Bae JK, et al. (2022) Guillain-Barré Syndrome and Variants Following COVID-19 Vaccination: Report of 13 Cases. Front Neurol 12: 820723.
- Lanman TA, Wu C, Cheung H, Goyal N, Greene M (2022) Guillain-Barré Syndrome with Rapid Onset and Autonomic Dysfunction Following First Dose of Pfizer-BioNTech COVID-19 Vaccine: A Case Report. Neurohospitalist 12(2): 388-390.
- (2021) COVID-19 Vaccine Moderna: EPAR – Product information. Amsterdam: European Medicines Agency.
- World Health Organization (n.d.). (2022) Interim recommendations for use of the moderna mRNA-1273 vaccine against COVID-19. World Health Organization.
- Centers for Disease Control and Prevention (2022) Administration overview for Moderna Covid-19 vaccine. Centers for Disease Control and Prevention.
- Beatty AL, Peyser ND, Butcher XE, Cocohoba JM, Lin F, et al. (2021) Analysis of COVID-19 Vaccine Type and Adverse Effects Following Vaccination. JAMA Netw Open 4(12): e2140364.
- Centers for Disease Control and Prevention (n.d.). (2022) Understanding mrna COVID-19 vaccines. Centers for Disease Control and Prevention.
- Baden LR, El Sahly HM, Essink B, Kotloff K, Frey S, et al. (2021) Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine.published on December 30, 2020, and updated on January 15, 2021,at NEJM.org.N Engl J Med 384: 403-416.
- Centers for Disease Control and Prevention (n.d.). (2022) Johnson & Johnson's Janssen COVID-19 vaccine overview and Safety. Centers for Disease Control and Prevention.
- (2022) Johnson & Johnson COVID-19 Vaccine Authorized by U.S. FDA For Emergency Use - First Single-Shot Vaccine in Fight Against Global Pandemic. Content Lab U.S. (n.d.).
- Oliver SE, Wallace M, See I, Sarah M, Monica G, et al. (2022) Use of the Janssen (Johnson & Johnson) COVID-19 Vaccine: Updated Interim Recommendations from the Advisory Committee on Immunization Practices - United States, December 2021. MMWR Morb Mortal Wkly Rep 71(3): 90-95.
- (2022) Coronavirus (COVID-19) update: July 13, 2021. U.S. Food and Drug Administration.
- (2022) Johnson & Johnson Statement on COVID-19 Vaccine (7/12). Content Lab U.S.






























