Intracranial Extradural IgG4-Related Disease Extended from the Infratemporal Fossa
Kais Maamri1*, Mohamed Amine Hadj Taieb1, Nesrine Nessib1, Ghassen El Kahla1, Maher Hadhri1, Atef Ben Ncir1, Rym Hadhri2 and Mehdi Darmoul1
1Department Neurosurgery, Faculty of Medicine of Monastir, Fatouma Bourguiba Hospital, Tunisia
2Department of Pathology, Faculty of Medicine of Monastir, Fatouma Bourguiba Hospital, Tunisia
Submission: October 8, 2020; Published: December 10, 2020
*Corresponding author: Kais Maamri, Department Neurosurgery, Fatouma Bourguiba Hospital, Faculty of Medicine of Monastir, Monastir, Tunisia
How to cite this article: Case R. Intracranial Extradural IgG4-Related Disease Extended from the Infratemporal Fossa. Open Access J Neurol Neurosurg 2020; 14(4): 555894. DOI: 10.19080/OAJNN.2020.14.555894.
Abstract
IgG4-related disease (IgG4-RD) is a recently recognized chronic inflammatory condition, characterized by tissue infiltration with lymphocytes and IgG4-secreting plasma cells, which can involve any organ or system [1,2]. It is a relapsing–remitting disease associated with a tendency to form tissue-destructive lesions. It can damage several sites especially pancreas, bile ducts, lacrimal and salivary glands. Head and brain involvement is rare [1,2]. The clinical expression of this disease varies depending on the organ involved. Generally, patients with IgG4-RD have a subacute clinical presentation. Principal neurological manifestations result from pachymeningitis, pituitary gland and stalk involvement [3]. In this article, we are presenting our experience with a rare case of an intracranial IgG4 related disease extended from the infratemporal fossa. The purpose of this report is to supply further information helpful in distinguishing this disease from other extra-axial tumors.
Observation
We report the case of a 37-year-old man with a 6-month history of headaches and blurred vision. Our patient had been followed by an otorhinolaryngologist during two years for a cervical lymphadenopathy and a right submandibular swelling. The cervical lymphadenopathy biopsy was non-diagnostic twice, showing a non-specific inflammatory disease. He had no other medical background and no personal or familiar history of an autoimmune disease.
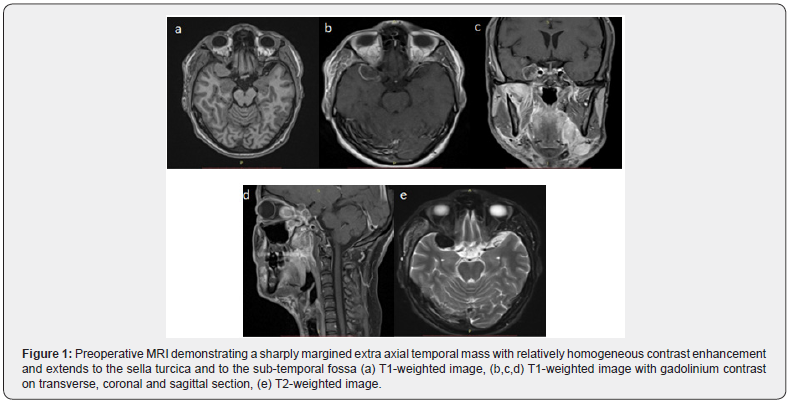
On examination, he had significant swelling of the right hemi face and the neck with a trismus and a decrease in the visual acuity of the right eye. The dilated fundus examination showed a right papillary paleness. Peripheral blood markers of inflammation were elevated. Screening for immunodeficiency and mycobacterial infections was negative. Cerebral MRI showed a pseudo-tumoral lesion developing in the right pterygo-palatin fossa spreading to the orbital and the intracranial cavity through the superior orbital fissure. The intracranial portion forms a temporal extra-axial mass mimicking a meningioma that infiltrates the lateral wall of the cavernous sinus. The lesion was strongly enhanced after injection of gadolinium (Figure1) CT scans of chest, abdomen and pelvis were normal.
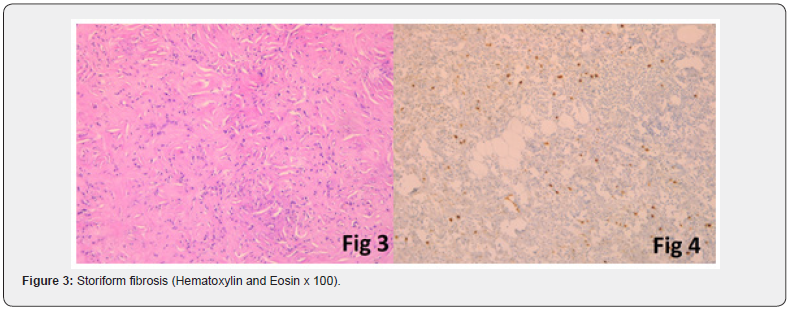
The patient was operated via a pterional approach. Our first strategy was a gross total resection of the intracranial portion of the tumor. Regarding to its very firm consistency, we opted for a large biopsy of the extra-axial lesion. The tumor was solid, well-delineated and strongly adherent to the temporal lobe. Histological examination showed dense lympho-plasmocytic infiltrate with storiform fibrosis (Figure 2, Figure 3). Immunohistochemical staining revealed an increased number of IgG4-positive plasma cells (Figure 4). The inflammation is often focal, predominantly in a perivascular location. Our patient received high doses of corticosteroids (0.6 mg/kg/day) followed by a progressive tapering. His neurological manifestations gradually improved and resolved after two months. A cerebral MRI was done 1 month after a well-conducted treatment and showed a reduction of the tumor’s size.


Discussion
IgG4-RD was firstly recognized as being associated with sclerosing diseases, in 2001, by Hamano et al. after finding elevated serum levels of IgG4 in patients having autoimmune pancreatitis compared to patients with other causes of chronic pancreatitis [4]. Nowadays, IgG4-RD is known as a systemic, chronic inflammatory process whose origin remains poorly understood, with some theories suggesting an autoimmune or allergic mechanism. It is characterized by increasing IgG4-positive plasma cells and lymphocytes that infiltrate one or multiple organs such as lacrimal gland, salivary gland, pancreas, bile duct, liver, kidney, and retro-peritoneum [1,5,6]. IgG4-RD occur predominantly in men and are more common in the fifth to sixth decade [7]. When it comes to the central nervous system, its involvement was only documented in less than 2% of IgG4-RD and it commonly involves pachymeninges forming hypertrophic pachymeningitis. In rare instance, it may form inflammatory pseudo-tumoral masses resembling meningiomas. To our knowledge, only two cases of intracranial spread of IgG4-RD via skull base foramina have been reported in literature [8].
The clinical presentations of IgG4 neuropathy are various. Chronic headache is the most frequent symptom.
Patients with IgG4-RD usually have increased serum IgG4 level. However, this elevation is not constant which makes it a nonspecific index of this pathology [2,6]. Imaging studies in IgG4- RD are useful both for diagnostic and monitoring purposes of these intracranial pseudotumoral masses. On Computed Tomography (CT) scans, lesions are hypo dense and demonstrate strong contrast enhancement. On T2-weighted-MRI images, lesions are typically hypo intense. T1-weighted images usually demonstrate homogenous and gradual gadolinium enhancement [2,9-11]. Despite the increasing frequency of IgG4-related neuropathy, there is no consensus guidelines on its treatment. Commonly, Glucocorticoids, usually methylprednisolone or prednisolone, are the first-line treatment with an excellent response [12,13]. A dose of 0,6 mg/kg/day is given for at least two to four weeks for response, followed by tapering schedule over a period of 3–6 months down to a maintenance dose (2.5–5 mg/day) maintained for up to 3 years [14]. In case of steroid resistance, other immunosuppressants like cyclophosphamide and methotrexate were used with success in refractory case [15-18]. When it comes to intracranial pseudo-tumors, it is still unclear whether biopsy or par-tial/subtotal resection is better. Surgical decompression may be required only in the presence of compressive symptoms [18]. Finally, the present case suggests that IgG4- RD can cause intracranial pseudo-tumoral masses, meaning that serologic and histologic examinations for IgG4- positive plasma cells must be performed to diagnose the etiology of unusual intracranial masses. However, at present, reports of patients with IgG4-related pseudo-tumors are still rare. Further reports will be useful to establish this disease concept and guidelines on its treatment.
Conclusion
The IgG4-RD is one of the central nervous system tumor differential diagnoses. Early recognition of IgG4-RD and appropriate establishment of its long-term treatment may avoid unnecessary investigations and morbidity.
References
- Arkadiusz Nowak, Tomasz Dziedzic, Tomasz Czernicki, PrzemysławKunert, Andrzej Marchel (2014) Surgical treatment of parasagittal and falcine meningiomas invading the superior sagittal sinus. NeurologiaiNeurochirurgiaPolska 48(3): 174-180.
- Chae-Yong Kim, Hee Won Jung (2009) Falcine Meningiomas. In: Lee JH(eds.), Meningiomas. Springer, London, pp.319-325.
- Fu-Xing Zuo, Jing-Hai Wan, Xue-Ji Li, Hai-Peng Qian, Xiao-Li Meng (2012) A proposed scheme for the classification and surgical planning of falcine meningioma treatment. Journal of Clinical Neuroscience 19(12): 1679-1683.
- Jan J Heimans, Jaap C Reijneveld (2012) Factors affecting the cerebral network in brain tumor patients. J Neurooncol108(2):231-237.
- Cecilia Casali, Massimiliano Del Bene, Francesco DiMeco (2020) Falcine meningiomas. Handb Clin Neurol 170:101-106.
- Evangelia Liouta, Christos Koutsarnakis, FaidonLiakos, George Stranjalis (2015) Effects of intracranial meningioma location, size, and surgery on neurocognitive functions: a 3-year prospective study. J Neurosurg 124(6):1578-1584.
- Sophie J M Rijnen, Ikram Meskal, Marjan Bakker, Wouter De Baene, Geert-Jan M Rutten, et al. (2019) Cognitive outcomes in meningioma patients undergoing surgery: individual changes over time and predictors of late cognitive functioning. Neuro Oncol21(7):911-922.
- Ikram Meskal, Karin Gehring, Geert-Jan MRutten, Margriet MSitskoorn (2016) Cognitive functioning in meningioma patients: a systematic review. J Neurooncol128(2): 195-205.
- Ikram Meskal, Karin Gehring, Sophie D van der Linden, Geert-Jan M Rutten, Margriet M Sitskoorn (2015) Cognitive improvement in meningioma patients after surgery: clinical relevance of computerized testing. J Neurooncol121(3):617-625.
- Hiroyasu Koizumi, Makoto Ideguchi, Hideyuki Iwanaga, Satoshi Shirao, HirokazuSadahiro, et al. (2014) Cognitive dysfunction might be improved in association with recovered neuronal viability after intracranial meningioma resection. Brain Res 1574:50-59.
- Wolfgang Krupp, Christoph Klein, Ronald Koschny, Heidrun Holland, Volker Seifert, et al. (2009) Assessment of neuropsychological parameters and quality of life to evaluate outcome in patients with surgically treated supratentorial meningiomas. Neurosurgery 64(1):40-47.
- Yoshihiko Yoshii, Daisuke Tominaga, Kouichi Sugimoto, Yukihiro Tsuchida, Akio Hyodo, et al. (2008) Cognitive function of patients with brain tumor in pre- and postoperative stage. Surg Neurol69(1):51-61.
- Oliver Tucha, Christian Smely, Michael Preier, Georg Becker, Geraldine M Paul, et al. (2003) Preoperative and postoperative cognitive functioning in patients with frontal meningiomas. J Neurosurg98(1):21-31.
- Alissa M Butts, Stephen Weigand, Paul D Brown, Ronald C Petersen, Clifford R Jack Jr, et al. (2017) Neurocognition in individuals with incidentally-identified meningioma. J Neurooncol 134(1):125-132.
- DainaKashiwazaki, Akiko Takaiwa, Shoichi Nagai, Naoki Akioka, Kunikazu Kurosaki, et al. (2014) Reversal of cognitive dysfunction by total removal of a large lateral ventricle meningioma: a case report with neuropsychological assessments. Case Rep Neurol 6(1): 44-49.
- Yoshihiko Yoshii, Daisuke Tominaga, Kouichi Sugimoto, Yukihiro Tsuchida, Akio Hyodo, et al. (2008) Cognitive function of patients with brain tumor in pre-and postoperative stage. Surg Neurol69(1):51-61.
- Pearl JC van Lonkhuizen, Sophie JM Rijnen, Sophie D van der Linden, Geert Jan MRutten, Karin Gehring, et al. (2019) Subjective cognitive functioning in patients with a meningioma: Its course and association with objective cognitive functioning and psychological symptoms. Psycho‐Oncology 28(8):1654-1662.
- Sanne van der Vossen, Vera P M Schepers, Jan Willem Berkelbach van der Sprenkel, Johanna M A Visser-Meily, Marcel W M Post (2014) Cognitive and emotional problems in patients after cerebral meningioma surgery. J Rehabil Med. 46(5):430-437.
- John M Tew Jr, Harry R van Loveren, Jeffrey T Keller (2001) Atlas of Operative Microneurosurgery. Philadelphia: Saunders, p. 8-26.
- Oyama Hirofumi, Kito Akira, Maki Hideki, Hattori Kenichi, Noda Tomoyuki (2012) Surgical results of parasagittal and falx meningioma. Nagoya J Med Sci 74(1-2):211-216.






























