Neuro-Imaging of Phacomatoses: about 20 Cases
Aichouni N*, Nasri S, Kamaoui I, Skiker I
Radiology department, Oujda University Hospital, Morocco
Submission: May 27, 2020; Published: July 07, 2020
*Corresponding author: Narjisse Aichouni, Radiology department, Oujda University Hospital, Morocco
How to cite this article: Aichouni N, Nasri S, Kamaoui I, Skiker I. Neuro-Imaging of Phacomatoses: about 20 Cases. Open Access J Neurol Neurosurg. 2020; 13(5): 555872.DOI: 10.19080/OAJNN.2020.13.555872.
Summary
Phacomatoses are a group of congenital diseases that are very different genetically, clinical-ly and radiologically. They are responsible for the formation of hamartomatous or tumor lesions, the preferential impairment of which is the central nervous system and the skin. Neurological manifestations are often a major component of the prognosis. Imaging, including MRI, allows diagnosis, patient monitoring and screening in family members. The most common diseases reported in our article are neurofibromatosis type 1 and 2, Bournevilletubepink sclé,von Hippel-Lindau disease and Sturge-Weber syndrome.
Keywords:Phacomatoses; Imaging; MRI; Tumor
Work Objectives
To write the different radiological aspects on the brain and spinal floor of the main phacomatoses:
Neurofibromatosis TYPE I
Neurofibromatosis TYPE II
Bourneville Tuber Sclerose
Sturg Weber’s Disease
From Hippel Lindau’s Disease
Define the place of imaging in the diagnosis and management of patients with the disease
Materials and Methods
This is a descriptive respective study conducted over a 6-year period (2014-2019) at the Medi-Cal Imaging Department at the
Mohammed VI University Hospital in Oujda, Morocco, involving 20 cases of phacomatoses with recognized diagnostic criteria: Neurofibromatosis type 1 (10cas-es); cas); Neurofibromatosis type 2 (1cas); Sclérose tub Bourneville tuberous pink Scl (7cas-es); Sturge weber disease (1 case); Von Hippel Lindau disease (1 case).
(TDM) cere19 patients were explored by magnetic resonance imaging (MRI) on a GE signa HDXT 1.5 Telsa 16-channel device and 1 patient received a CT scan. brale.
Results
Age
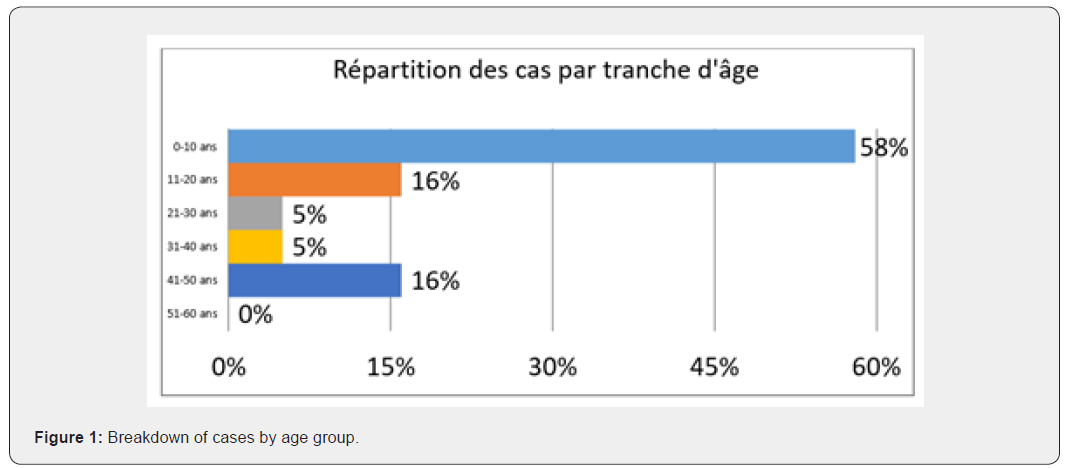
The age of patients in our series varies between 2 years and 49 years (with an av-erage age of 25.5 years), the majority of patients with phacomatosis had an age between 0 and 10 years or 65%(Figure1).
Sex
In our series, there is a slight predominance of the male sex; 10♂/9♀ with a sex ratio of 1.1 (Figure 2).
Reason for consultation
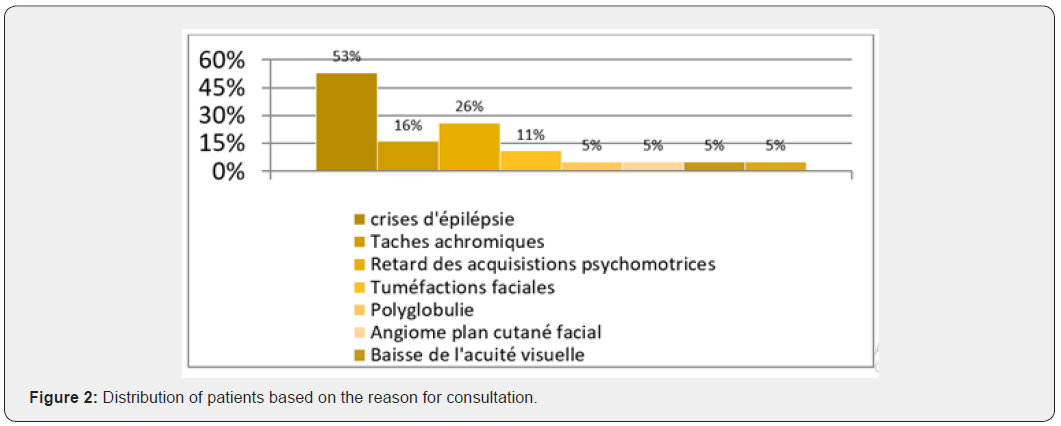
In our series we note the following distribution according to the reason for consultation: Seizures (no.10) or 53%; a chromic spots (no.3) or 16%; Delay in psychomotor acquisi-tions (no.5) or 26%; Facial swelling (no.2) or 11%; Polyglobulia (No.1) or 5%; Angioma plan cutanfacial (no.1) or 5%; decrease in visual acuity (no.1) or 5%; headache and balance disorder (no.1) or 5%.
Imaging Data
Observation 1: Neurofibromatosis type 1
a 4-year-old child is admitted for delaying psychomotor acquisitions, arable cereatic MRI has been performed (Figure 3).
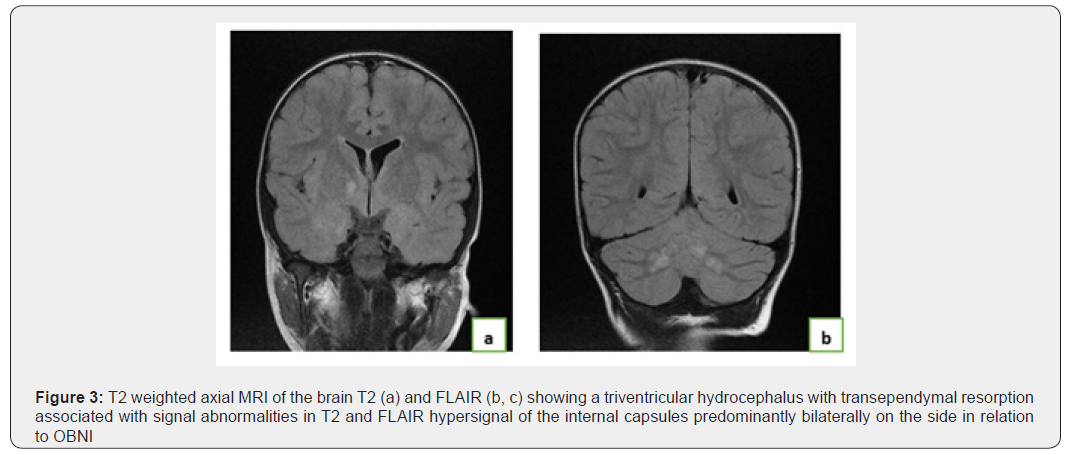
Observation°2: Neurofibromatosis type 1
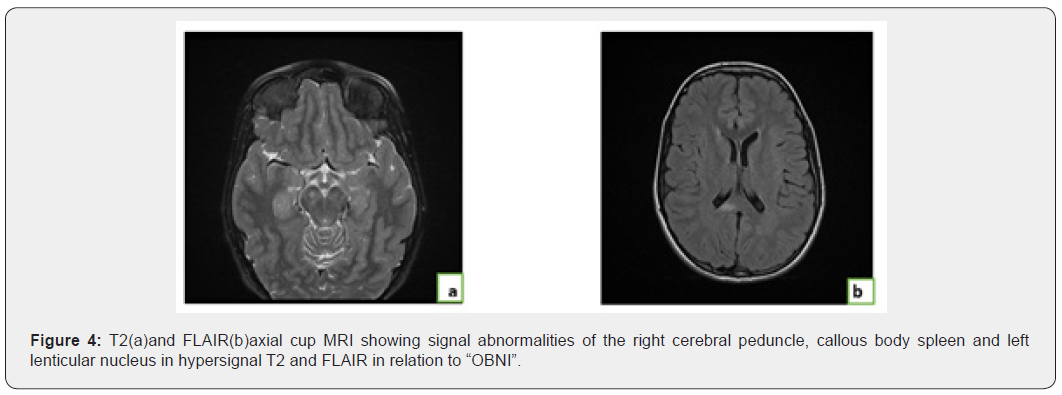
A 10-year-old child who senses mental retardation associated with a chromic stain and epi-leptic seizures. The a Bennyficie of an MRI CéréDude (Figure 4).
Observation°3: Neurofibromatosis type 1
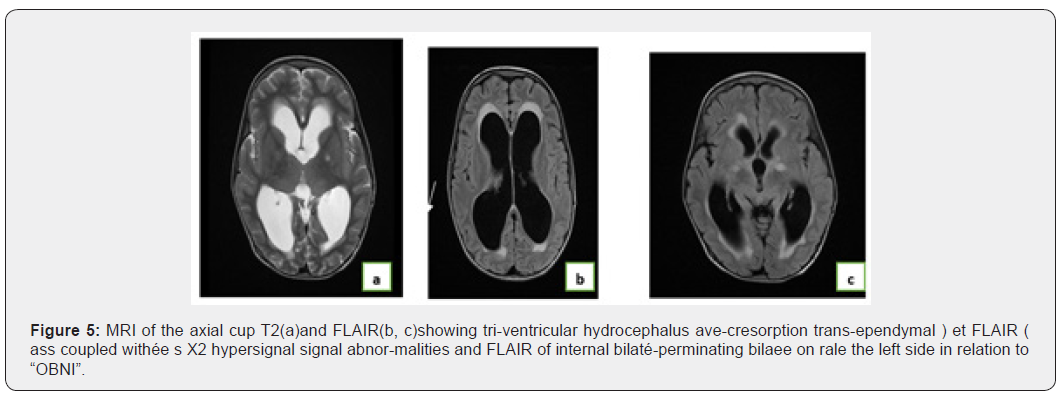
A 7-year-old child is admitted for seizures, followed by psychomotor delay, clinical examination reveals the presence of a chromic spots. Hereceiveda cerebraleMRI (Figure 5).
Observation°4: Neurofibromatosis type 1
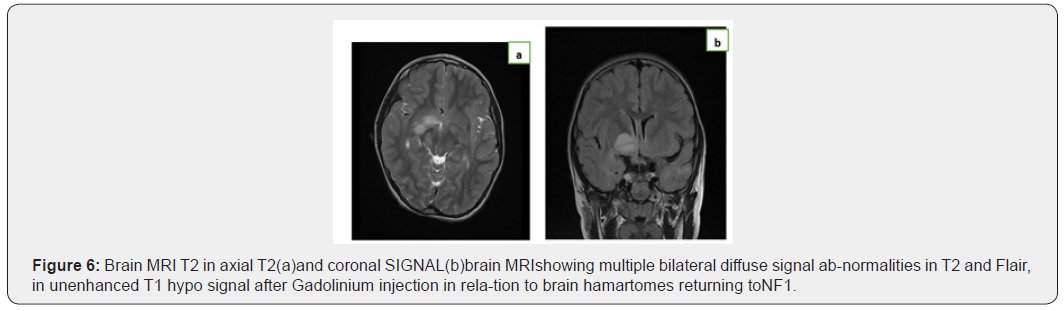
A 7-year-old girl is admitted for a delay of psychomotor acquisitions, anencephalic I RM has been performed (Figure 6).
Observation°5: Neurofibromatosis type 1
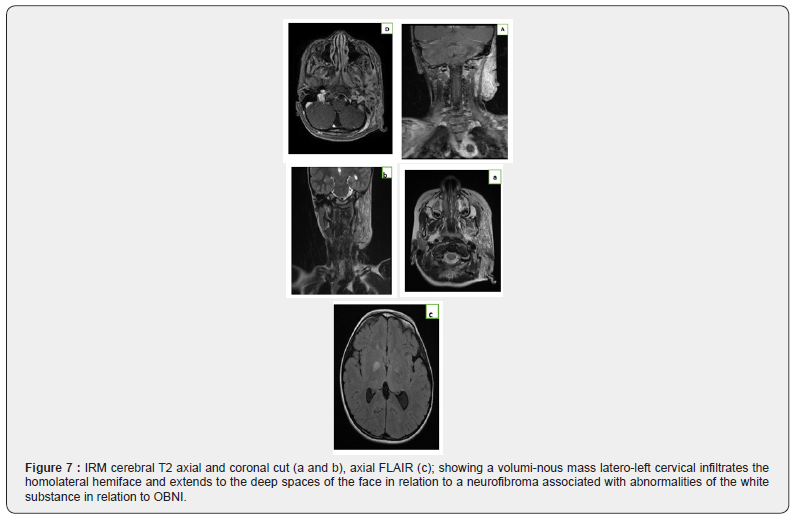
A Child Aged 9-year-old follow-up for neurofibromatosis type I and which has a left cervical swelling in rétroauriculaire soft consistency associated with stains coffee at Milk. Udoes MRI created Dude was achieved (Figure 7).
Observation°6: Neurofibromatosis type 1
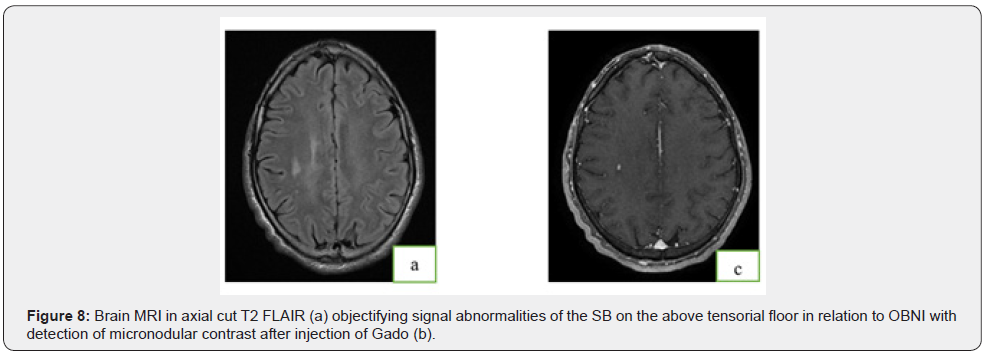
patient 9 years of age followed for neurofibromatosis type 1 with epileptic seizures associat-ed with achromic spots. A Brain MRI with injection of contrast product it has been carried out (Figure 8).
Observation°7: Neurofibromatosis type 1
P49-year-old Years follow-up for neurofibromatosis type 1. A Brain MRI was achieved in the Patient (Figure 9).
Observation°8: Neurofibromatosis type 1
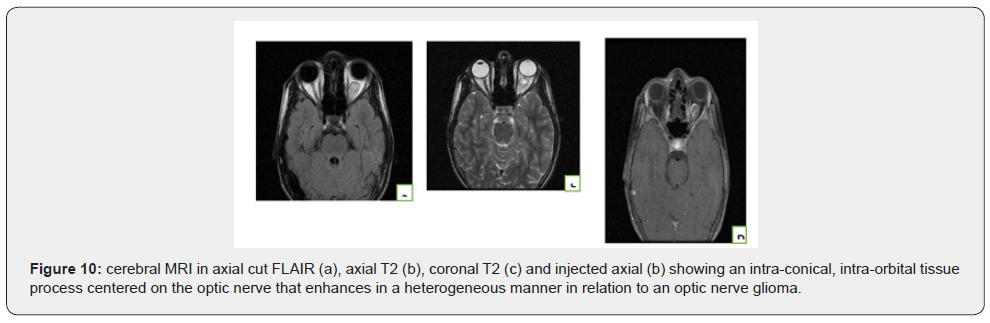
An elderly patient 12-year-old follow-up for neurofibromatosis type 1 admitted for de-creased acuity visual left. A Orbital MRI was performed (Figure 10).
Observation°9: Neurofibromatosis type 1
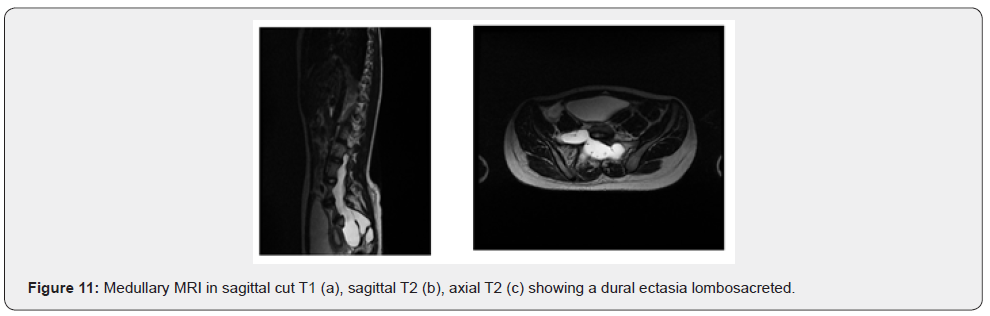
A e 19-year-old girl who was being followed for NF1 and who had been exhibiting bilateral lumbosciatic pain low back pain for the past bilatéyear, accentuated by walking. rale accentu A spinal mri was per-formed (Figure 11).

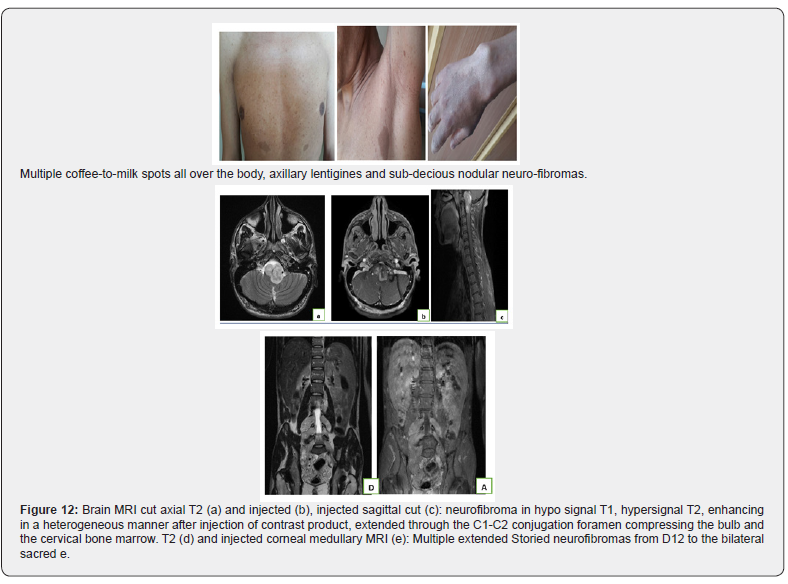
Observation 10: Neurofibromatosis type 1:
Patient of 26 followed for a functional impotence of the right upper limb, with a reduction of the progressive installation walking perimeter, without notion of sphincteric disorders (Figure 12).
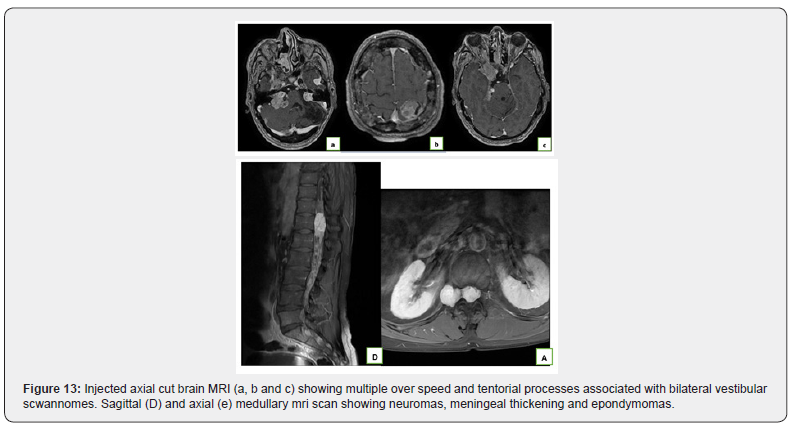
Observation11: 1 Neurofibromatosis type 2:
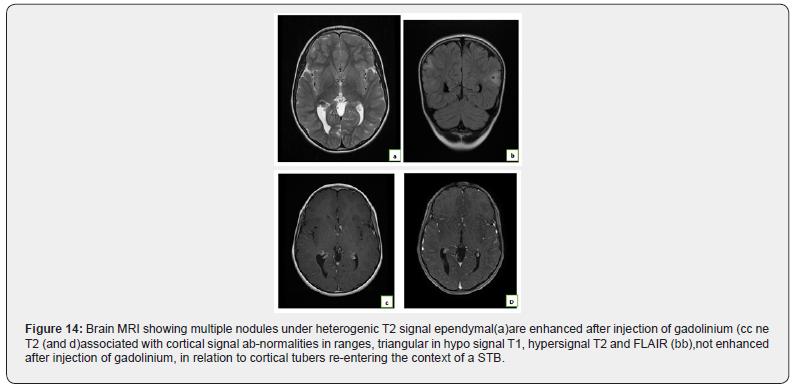
Patient aged 46 years, has BAV, right ptosis, bilateral tinnitus and deafness as well as low back pain. Acerebro-spinal MRI was performed (Figure 14).
Observation No. 12: Bourneville-Pringle disease
A 6-year-old child followed for repeated epileptic seizures, and as part of an etiologi-calcheck-uphe received an encephalic MRI;
Observation No. 13: Bourneville-Pringle disease
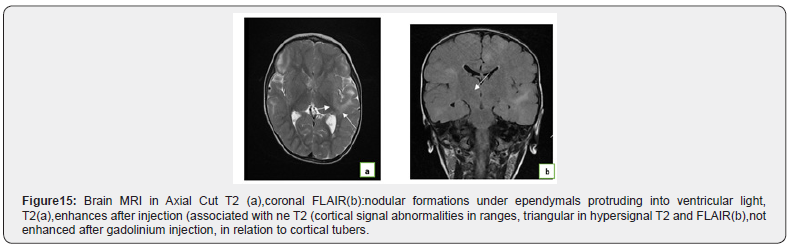
A2-year-old girl is admitted for psychomotor delay and seizures with diffuse achromic spots. Theepileastial ENc MRIinsagittal cut T1, axial T2 and diffusion, coronal T2 FLAIR showed: (Figure 5) (Figure 15)
Observation No. 14: Bourneville-Pringle disease
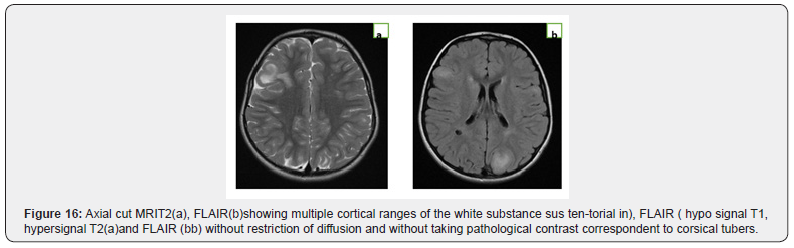
A 4-year-old girl is admitted for epileptic seizures without a found cause, a T1 sagittal cut brain MRI, axial T2 and diffusion, coronal T2 FLAIR was done Figure 16.
Observation No. 15: Bourneville-Pringle disease
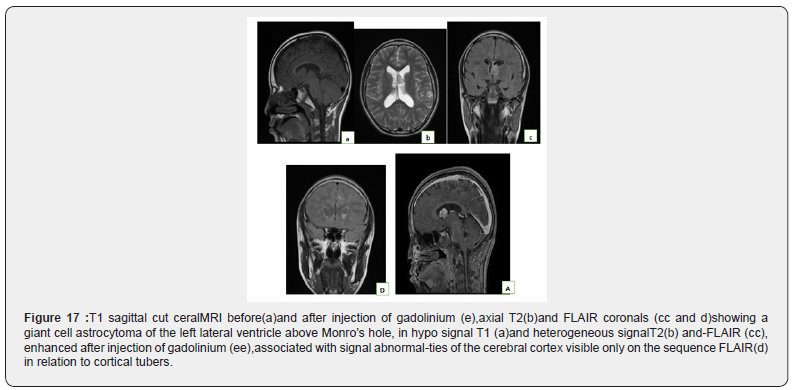
A 14-year-old child followed for seizures, a brain MRI was performed (Figure 17)
Observation No. 16: Bourneville-Pringle disease
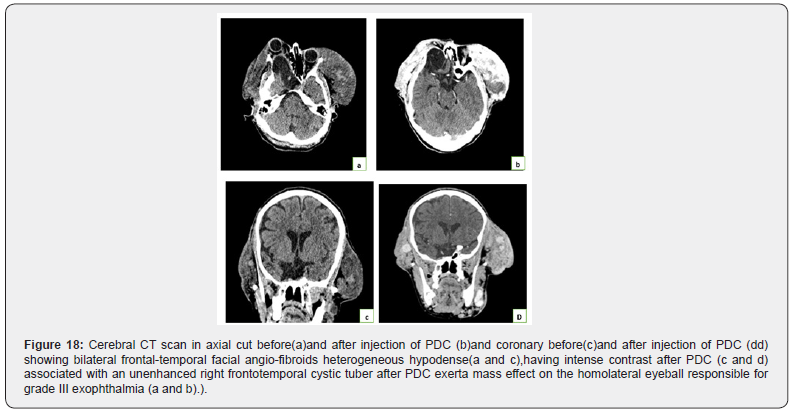
A 35-year-old woman followed for STB, who has bilateral frontotemporal facial swell-ings with right exophthalmia. A brain scan in axial cuts of 1 to 3 mmthick without and with injection of PDC with multi-planar reconstructions was carried out objectively: (Figure 8) (Figure 18)
Observation No. 17: Bourneville-Pringle disease
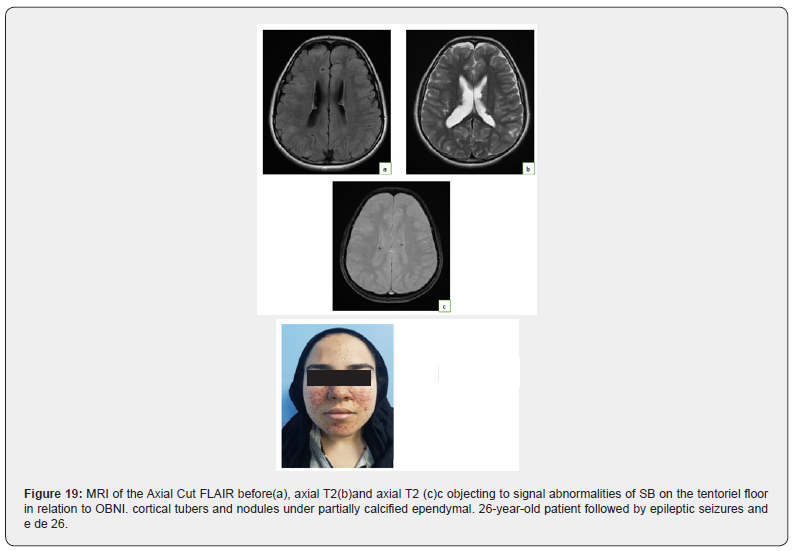
E nfant aged 9 years, followed for epileptic seizures. a brain MRI was performed (Figure 19) and hide the patient’s eyes.
Observation No. 18: Bourneville-Pringle disease
26-year-old patient, followed by epileptic seizures and e de 26 (Figure 20)
Observation n° 19: Syndrome de Sturge Weber
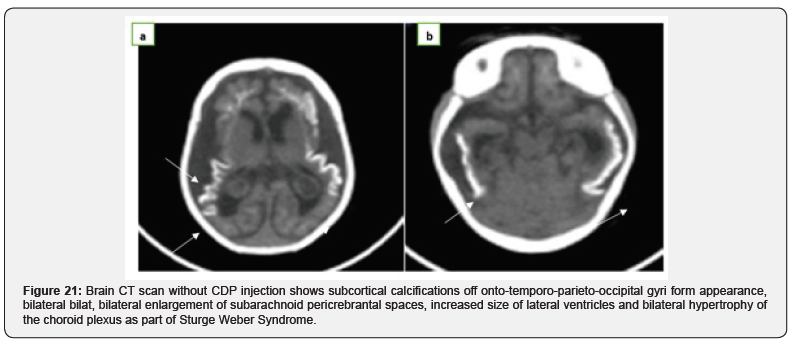
A three-year-old girl with no particular pathological history is admitted for simple partial seizures. Clinical examination reveals the presence of a cut a facial plan angioma median extending over the upper eyelids. A brain CT scan without injection of the contrast product is performed (Figure 9) :) (Figure 21). CT scans show subcortical calcifications off rondo-temporo-parieto-occipital gyri form appearance, bilateral bilat, bilateral enlargement of subarachnoid peri cerebral spaces, in-creased size of lateral ventricles, and bilateral hypertrophy of the choroid plexus. Faced with these CT scans and the presence of cutan angioma in V1 territory, the diagnosis of Weber Sturg-Weber syndrome / Von Hippel-Lindau Disease. was made.
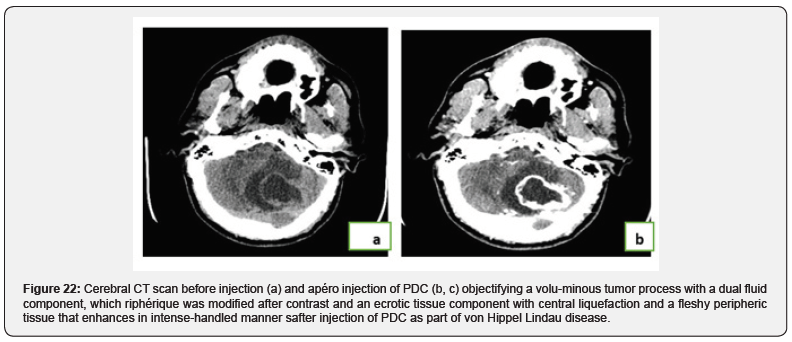
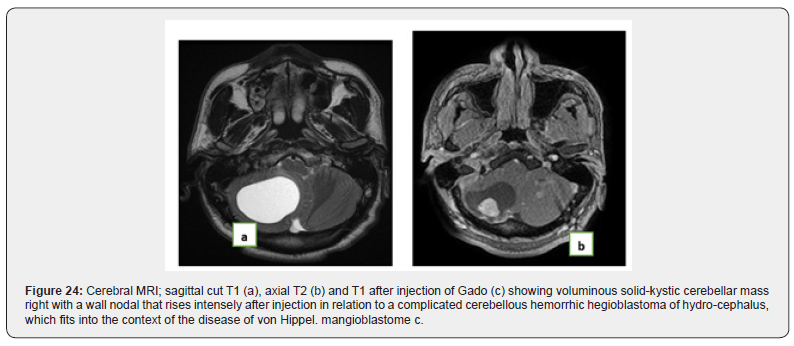
Observation No. 20: Von Hippel Lindau Disease
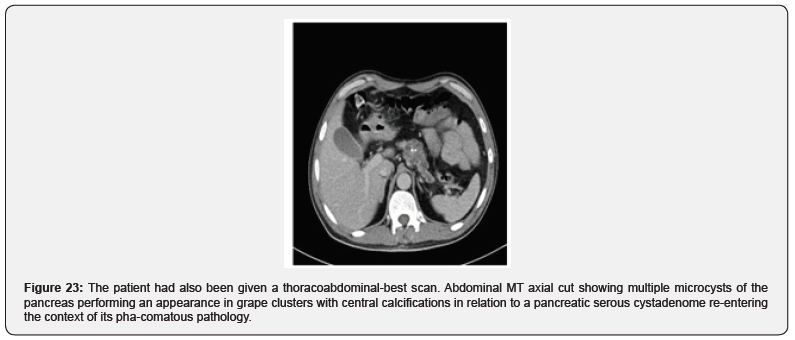
A 48-year-old hypertensive diabetic man being followed for polyglobulia, he received brain scan. cérébral. The patient had also been given a thoracoabdomino-best scan (Figures 22,23).

Discussion
We will limit our analysis to the four most frequently encountered groups of conditions. Namely: neurofibromatosis type 1 and 2, Bournevilletubepink sclé,von Hippel-Lindau disease and Sturge-Weber syndrome.
Neurofibromatosis type 1 (NF1):
Neurofibromatosis type 1 (NF 1) or von Recklinghausen disease is the most common of genetic diseases, its frequency is about 1/3000 in the general population [1]. It is a dominant autosomal transmission genetic disease. The abnormal gene is located on the long arm of chromosome 17 (17q11.2). The protein coded by this gene, neurofibromin, intervenes in the control of cell differentiation and proliferation. The phenotypic expression is verysleea-ble [1,2]. It is characterized by a progressive and polymorphic impairment that can interest most organs with a predisposition for benign tumors and mylines [1]. The diagnosis of NF1 is essentially clinical and is based on well-defined criteria established at the 1987National Institute of Health Consensus Conference. Skin manifestations, particularly latte stains, epihbleds and skin neurofibromas, are at the forefront and often indicative of the disease. Neurological manifestations are also common, mainly represented by behavioral disorders, learning difficulties, seizures and symptoms related to tumor impairment of the optic pathways or even the hypothalamo-hypophysic axis [3,4]. Its management is multidisciplinary and imaging, and especially MRI, plays a prominent role in the initial assessment and monitoring of malignant-potential lesions [5,6].
NF1 Diagnostic Criteria
Despite the devability of the gene responsible for the disease, the diagnosis remains clinical and based on criteria defined by the 1987National Institute of Health (NIH)consensus.
The diagnosis of NF1 is based on the presence of at least two of the following criteria:
a. at least 6 latte stains larger than 5 mm in diameter in pre-pubescent individuals and more than 15 mm in pubescent individuals.
b. two or more neurofibromas of any type or plexiform neurofibroma.
c. axillary or inguinal ephelics;
d. a glioma of the optical pathways.
e. two or more Lisch nodules;
f. a characteristic bone lesion, such as sphenoidal dysplasia, thinning of the cortical long bones with or without pseud arthritis;
g. a first-degree relative with NF1 according to the previous criteria.
Neurofibromatosis type 2
Neurofibromatosis type 2 (NF2) is a genetic disease with dominant autosomal transmission characterized by the presence of bilateral vestibular schwannomes. The incidence of NF2 was estimated at 1 per 25,000 births in a study conducted in the northwest region of Eng-land with a population of 4.1 million. In the same study, the prevalence of NF2 was 1 in 100,000 inhabitants, and the annual incidence was 1 new case diagnosed in 1,312,000 inhab-itants per year [7]. The NF2 gene is the only known gene responsible for neurofibromatosis type 2. This gene encodes a protein, called merlin or schwannomine, inhibiting the growth of certain tumors and composed of 595amins.s. Clinical symptoms appear around the age of 20-30, later than NF1. Revealing symptoms are often due to the absence of acoustic neuromas with deafness and dizzi-ness. Root pain, due to one or more lumbar neuromas, is more rarely a mode of entry into the diagnosis of NF2.
Two types of clinical forms have been described:
A severe form (Wishart type) characterized by an injury before the age of 25, the development of more than 3 tumors, repeated surgeries, and few survivors after the age of 40; a moderate form (Gardner type) characterized by a later onset age, fewer and slow-growing tumors, and many survivors after age 50. About 10% of NF2s are symptomatic before the age of 10. Skin signs are rare (about 30% of cases) compared toNF1, which is why the disease is often overlooked at first. Eye lesions (juvenile cataract) are frequent (50 to80% of cases). The diagnostic criteria for NF2 are strict, the main criterion being represented by a neuroma of bilatéral acoustics. However, this neurofibromatosis is characterized by multiple tumor damage to the central nervous system (meningiomas,e’pendymmes). Papillary pseudo-edema has also been reported to a gliotic pre-hair membrane [8].The following diagnostic criteria, called Manchester Criteras are necessary to establish the diagnosis early [8] bilateral vestibular schwannomes 2A first-degree parent with AND unilateral vestibular schwannome OR two ab-normalities: meningioma, schwannome, neurofibroma, glioma, cataract Unilateral vestibular schwannoma and two abnormalities: meningiome, schwannome, neurofibroma, glioma, cataract Multiples meningiomas AND unilateral vesti bular schwiomeOR two following abnormalities: schwannome, neurofibroma, glioma, cataract
Bourneville Tuberous Sclerosis (STB):
Bourneville tube-pink (STB) is the most frequent phacomatosisafterneurofibromatosis type 1, the incidence approaching 1/5800érose tub.
STB is a dominant hereditary disease of autosomal transmission, but 65-75% of cases are sporadic, corresponding to a mutation of novo [8]. There is genetic heterogeneity and genetic studies have currently identified 2 major genes, TSC1 and TSC2 [9,10].
This disease is characterized by dysplasias and/or neoplasias in the organs derived from the ectodermic leaf (skin, central nervous system Tre intandpe-ericic, eyes), structures derived from mesoderma (blood vessels, bones and cartilage) and endoderm (intestinal epithelium) may also be of interest. Central nervous system impairment is responsible for the most common clinical manifestations including epilepsy (West syndrome) and mental retardation.
Maladie de Sturge Weber (SSW)
Sturge-Weber syndrome (SSW), or angiomatosis encephalxeotrigor meningo-facial com-bines a plane angioma of a hemiface, in the territory of the tri-twin nerve (its upper branchV1), with a homolaterallepto-meningangioma and frequently a choroidal angioma. This condition is rare, its incidence is estimated at 1 /40,000. Pathogenesis is not known. This disease has no racial or ethnic predominance [11], it is con-genital, sporadic in most cases but family cases have been reported. It affects both sexes in the same way.
The angioma lepto-mening éing éingéing is constant, homolateral to the facial angioma, locatise at the level of the magpie mother most often at the occipital level but also at the parietal or even temporal lobe. The frontal lobe is usually spared. The cerebral cortex in front of it has a secondary atrophy that extends beyond the boundaries of the angioma, with ne-crosis and calcifications. Consequences of chronic ischemia, they are pericap and localized at the level of the 3rd and 4th atrophied cortical layers, and also at the level of the underly-ing white substance, neurons and vascular walls. There are outbreaks of cortical gliosis and white matter, is chic leons éand demyelination [12] (Table 1).

Maladie de Von Hippel Lindau (VHL):
Von Hippel Lindau disease (VHL) is a rare phacomatosis, it is a dominant autosomal genetic condition with variable expression, affecting1/36,000 to 1/40,000 people [13]. The Von Hippel Lindau disease gene is located on the short arm of chromosome 3 (3p25- 26). Like other phacomatoses, there are no skin manifestations in VHL disease.
The he mangioblastoma of the central nervous system represents the most typical and consistent is ion of this condition [14]. The relative frequency of major lesions of Von Hippel Lindau’s dis-ease is as follows:
a. Hemangioblastoma of the central nervous system: 60- 80%
b. Retinal Hemangioblastoma: 50-60%
c. Pancreatic disease: 30-65%
d. Cancers or kidney cysts: 30-60%
e. Phenochromocytome: 11-19%
VHL’S Diagnostic Criteria
The clinical diagnosis [15] is based on:
a. in the absence of family history, the presence of two hemangioblastomas or heman-gioblastoma and a visceral lesion (pheochromocytoma, multiple renal cysts, kidney cancer, cysts or pancreatic tumors, cystadenoma of the epididym);
b. with a family history, the presence of a hemangioblastoma or a visceral lesion.
Conclusion
Phacomatoses, or neurocutaneous syndrome, include a large group of genetically-cause conditions responsible for the formation of hamartomata and/or tumor lesions mainly affecting structures of ectodermal origin and mainly resulting in skin and central nervous system damage. Neurological manifestations are often a major component of the prognosis. The most common diseases are type 1 and 2 neurofibromatosis, rose tub Bourneville tuberous pink sclerus, von Hippel-Lindau disease and Sturge- Weber syndrome. Neu-roimaging, including MRI, has an essential role in positive diagnosis, prognostic evaluation, re-ferral of therapeutic management, evolutionary follow-up of lesions and family screening.
References
- Wimmer K (2005) Neurofibromatosis: the most frequent hereditary tumor predisposition syndrome. Wien Med Woch-enschr 155(11-12): 273-80.
- Viskochil D (2002) Genetics of neurofibromatosis 1 and the NF1gene. J Child Neurol 17(8): 562-570.
- (1988) Neurofibromatosis Conference statement. National Institutes of Health Consensus Development Conference. Arch Neurol 45(5): 575-588.
- Tonsgard JH (2006) Clinical manifestations and management of neurofibromatosis type 1. Semin Pediatr Neurol 13(1): 2-7.
- Aidara CM, Diop D, Ndiaye OK, Diop AD, Ndiaye MA, et al. (2015) Contribution of imaging in neurofibroma-tosis type 1. Radiol Leaf 55: 60-65.
- Van Es S, North KN, McHugh K, De Silva M (1996) MRI findings in children with neurofibromatosis type 1: a prospective study. Pediatr Radiol 26(7): 478-487.
- Antinheimo J, Sankila R, Carpen O, Pukkala E, Sainio M, et al. (2000) Population-based analysis of sporadic and type 2 neurofibromatosis-associated meningiomas and schwannomas. Neurology 54(1): 71-76.
- N Girard (2005) Imaging of Neurofibromatosis Type 2. J Neuroradiol 32: 198-203.
- MacCollin M, Kwiatkowski D (2001) Molecular genetic aspects of the phacomatoses: tuberous sclerosis complex and neurofibromatosis 1. Curr Opin Neurol 14(2): 163-169.
- Roach ES, Gomez MR, Northrup H (1998) Tuberous sclerosis complex consensus conference: revised clinical diag-nostic criteria. J Child Neurol 13(12): 624-628.
- (2004) Baselga E Sturge-Weber syndrome, Semin Cutan Med Surg 23(22): 87-98.
- Comi AM (2003) pathophysiology of Sturge-Weber Syndrome. J Child Neurol 18(8): 509-516.
- Chateil JF, Brun M, Le Manh C, Diard F, Labrèze C Phacomatoses in children. Encycl Méd Chir, Radiodiag-nostic - Neuro-radiology-Musculoskeletal device 31-625-A-10, 2000, 23 pp.
- Caron S, Soto-Ares G, Vinchon M, Dhellemmes P, Pruvo JP (2004) Neuroimaging of phacomatoses. Feuill Radiol 44: 241-63.
- LF EL AmranI (2011) Interest of cerebral CT in the systematic assessment of neurofibromatosis type 1. Thesis No. 81/11. CHU Fez.






























