An Atypical Case of Viral Encephalitis
Ishtiaq Ahmad1*, Mohammad Mahade Hassan2 and Mohammad Akram Hossain3
1 Department of Neuromedicine, Imperial Hospital Limited, Bangladesh
2Department of Intensive Care, Imperial Hospital Limited, Bangladesh
3 Department of Microbiology, Imperial Hospital Limited, Bangladesh
Submission: April 29, 2020; Published: June 17, 2020
*Corresponding author: Ishtiaq Ahmad, Department of Neuromedicine, Imperial Hospital Limited, Zakir Hossain Road, Pahartali, Chattogram-4202, Bangladesh
How to cite this article: Ishtiaq A, Mohammad M H, Mohammad A H. An Atypical Case of Viral Encephalitis. Open Access J Neurol Neurosurg. 2020; 13(4): 555869.DOI: 10.19080/OAJNN.2020.13.555869.
Abstract
Introduction: Viral encephalitis is a frequent cause of altered mental status in Bangladesh and all over the world. Here we experience an elderly immunocompetent patient presents with sudden onset of unresponsiveness for last 9 days until she presented to us. She was treated in a remote rural hospital initially following the symptom onset. As patient was declining, patient’s family decided to transfer the patient to our hospital for continued care.
Case presentation: A 90-year-old Bangladeshi female, independent in ADLs prior to the symptom onset, modified Rankin scale 1 developed sudden onset change in mental status and stopped communicating with family members and stopped taking food. She was taken to a local hospital reportedly noted to have normal vitals. NGT feeding was initiated and discharged home with family for continued supportive care. When patient was brought to our hospital after 9 days of symptom onset, on exam she had GCS 5, hypotensive with tachycardia, mild neck rigidity, pupil reactive to light on both sides, planter up going on both sides. There was infected sacral decubiti. Complete blood count showed leukocytosis, mild hyponatremia, high BUN, and CT scan of head was unremarkable. Following admission, patient was started on broad spectrum antibiotic and hydrated well. MRI brain was unremarkable, EEG showed diffuse slowing. CSF showed WBC 5, lymphocytic pleocytosis, high glucose, high protein, culture was negative. CSF MTB-PCR was negative and HSV-PCR (1 and 2) negative. Patient was discharged in 8 days with GCS 10 on oral antibiotic and Acyclovir for 1 weeks.
Conclusion: Elderly patient with altered mental status related to non-HSV encephalitis. Here we discussed primarily viral encephalitis the possible agents we need to keep in mind while managing the patients according the geographical distribution.
Keywords:Encephalitis; Viral encephalitis; Septic shock; Osmotic demyelination syndrome
Introduction
Alteration of mental status is related to functional impairment of brain parenchyma. If that is related to inflammation of brain implicates encephalitis otherwise metabolic and hypoxic abnormalities causes encephalopathy [1-3]. Etiology for brain inflammation are infection and immunologic disorder. Infectious agents to cause encephalitis are viral, bacterial, or parasitic infection etc. Viral encephalitis is very common which carries significant morbidity and mortality specially with herpes simplex virus [4-5]. The work up includes CT brain, MRI brain, EEG, CSF study with the viral-PCR in the CSF.
Case Report
A 90-year-old Bangladeshi female with history of hypertension and type-2 diabetes developed sudden onset change in mental status one evening about 9 days ago prior to be presented to us. She stopped communicating with others and she could not be fed in the morning. She was noted to be normal before bedtime during previous evening. Her family at bed side admits to shortness of breath, but denies any fever, diaphoresis, chest pain, headache, or any other focal neurological deficit. She had left thyroid gland swelling for last 15 years, size remained the same. She was taken to a local rural hospital where Naso-Gastric Tube (NGT) feeding was initiated and Foley catheter was placed. From rural hospital she was discharged after 2 days with plan for supportive care at home. There was no medical record available from the other hospital. As patient was declining progressively, family brought her to our hospital for better understanding of her ailments. History did not reveal any prior episode of seizure, coronary event, or cerebrovascular event. During admission, her GCS score 5 (V1E1M3). Patient was afebrile, dry oral mucosa, BP 90/60, HR 118/min, O2 saturation 99%, mild neck rigidity, pupil equal reactive to light, moved less left side to noxious stimuli, planter is extensor on both sides. There was grade 2 sacral decubiti 5x7 cm which was infected. NGT and Foley catheter were in place. Laboratory exam revealed WBC 18.73 mm9/L, neutrophilic leucocytosis (81.2%), hemoglobin 12.1 gm/dL, hematotrit 37.0%, ESR 93, Hba1c 5.96%, TSH 2.11 microIU/mL, CRP 24 mg/L, Na 132 mM/L, K 5.5 mM/L, Cl 99 mM/L, HCO3 25 mM//L, blood urea nitrogen (BUN) 42 mg/dL, creatinine (Cr) 0.83 mg/dL, glucose 108 mg/dL, ammonia level 29 microM/L (calculated serum osmolarity 285 mOsm). Patient had isotonic hyponatremia associated with severe hypovolemia which could be related to severe dehydration or Cerebral Salt Wasting Syndrome (CSWS). Urinalysis showed WBC 0-2/HPF, specific gravity 1.013, glucose 4+, pH 7.0 (calculated urine osmolarity 390 mOsm). Chest X-ray did not reveal any infiltration. Patient had neutrophilic leukocytosis, as chest X-ray and urinalysis were unremarkable, that led us to consider sacral decubiti or CNS infection as the cause of leukocytosis. CSF study revealed WBC 5/mm3, lymphocytic pleocytosis, no xanthochromia, RBC 18/ mm3, glucose 104 mg/ dL (corresponding blood glucose 92 mg/dL), protein 358 mg/ dL, gram stain was negative for any organism. During lumber puncture, opening pressure was normal. CSF was sent for MTB and HSV PCR. CSF data led towards viral encephalitis rather than bacterial infection. Ultrasound exam of optic nerve did not show feature of elevated ICP (optic nerve diameter was 0.48 cm within normal limit), Inferior Vena Cava (IVC) was noted to be kissing implicating severe dehydration. Internal jugular line revealed Central Venous Pressure (CVP) 5 mm of Hg. EEG revealed diffuse 45 hertz slowing. There was no feature of inter-ictal discharge. MRI did not reveal any acute lesion. There were minimal chronic microvascular ischemic changes in both hemispheres but none in brainstem. Diffusion weighted images did not reveal any acute lesion. She was hydrated with 3 L Normal Saline (NS) with inotropic support. Blood culture was sent prior to administration of antibiotic. For possible systemic infection and encephalitis Linezolid, Meropenem, Acyclovir were initiated IV. Her wound culture for sacaral decubiti revealed growth of coagulase negative Staphylococcus spp which was sensitive to Clindamycin, Gentamycin, Linezolid, Rifampin, Tetracycline, Vancomycin. She was already on Linezolid. Her GCS score improved to 8 (V2E2M4) that evening following hydration. Subsequently her hemoglobin/ hematocrit (H/H) reduced to 9.4/29.0, WBC reduced to 9.3 mm9/L, BUN/Cr reduced to 9.3/0.83. Her echocardiography revealed left ventricular ejection fraction 63% and normal in wall motion. Her cardiac enzyme was negative. Her sacral decubiti improved with twice daily dressing. With this supportive care her BP increased to 150/90 and HR 93/min. She was needed to be on Amlodipine and Atenolol to control hypertension. During discharge after 8 days her GCS score improved to 10 (V2E3M5). Her blood culture was negative for bacterial or fungal organism. CSF Mycobacterium Tuberculosis (MTB) and Herpes Simplex Virus (HSV) PCR were negative. Her family was happy to take her home with antibiotic and Acyclovir orally for 1 week and maintenance of nutritional support as well as hydration by NGT feeding. Her family was educated to maintain twice daily dressing change for sacral decubiti (Table 1).
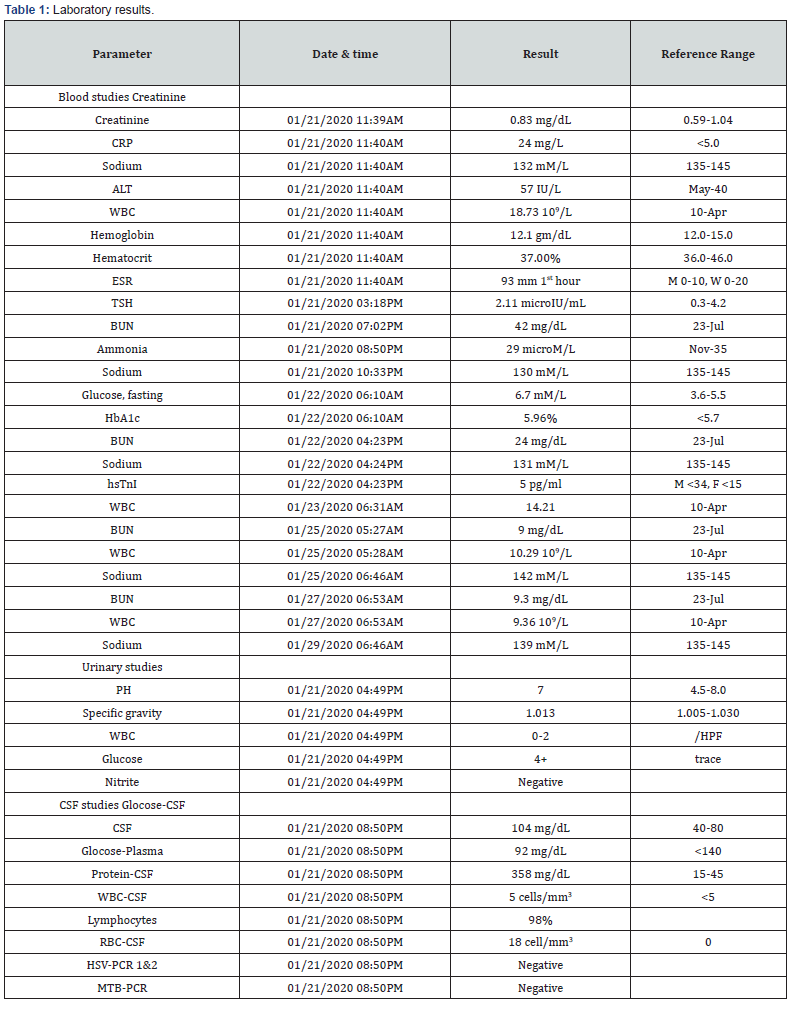
Discussion
Change in mental status in an elderly patient is a medical emergency with wide differential diagnosis related to vascular, infection and metabolic derangement. In our case with history, physical examination along with laboratory work and CT scan of head guided us towards Central Nervous System (CNS) infection with systemic sepsis. CNS vascular event is also a possibility as patient was unconscious with left sided weakness, but it would be a small one as the CT head was unremarkable even after 9 days of symptom onset. CSF study suggested viral encephalitis. EEG did not show any inter-ictal discharge which may explain patient’s unconsciousness. Moreover, EEG did not show Periodic Lateralized Epileptiform Discharge (PLED) which is specific for HSV encephalitis. EEG is showing diffuse theta slowing (Figure1) indicating encephalopathy related to probably dehydration or hyponatremia. MRI brain (Figure 2) did not show any high T2 signal intensity lesion in hippocampus or temporal lobe to implicate HSV encephalitis. Further there was no evidence of acute CNS vascular lesion to explain the reason for patient’s unconsciousness. There are mild microvascular ischemic changes related to patient’s vascular risk factors e.g. hypertension, diabetes and her age. In this clinical presentation the reason for patient’s unresponsiveness at the beginning is probably related to viral encephalitis. Then gradually patient got dehydrated. The laboratory work indicated patient had hyponatremia consistent with isotonic hypovolemic hyponatremia. HSV encephalitis is more commonly associated with hyponatremia than non-HSV encephalitis. Hyponatremia is a poor prognostic sign [6,7]. Usually with encephalitis there are 2 broad kind of hyponatremia, commonly Syndrome of Inappropriate Antidiuretic Hormone (SIADH) which is euvolemic and the other one is CSWS which is hypovolemic [8-15]. The hyponatremia in our patient is consistent with CSWS. The management of these two kinds of hyponatremia is different one is with fluid restriction and other is fluid resuscitation. The contributing factors for our patient’s dehydration are probably reduced fluid intake and CSWS due to viral encephalitis. If the dehydration was purely due to reduced fluid intake than laboratory results will indicate hypernatremia instead of hyponatremia [11]. Our patient responded well to fluid resuscitation.
It is perplexing that our patient did not achieve the baseline cognitive status following treatment but relatively improved with antibiotic, Acyclovir and fluid resuscitation. There could be several reason. First possibility is that the virus which is identified as non-HSV damaged the brain significantly and made it difficult to achieve the baseline cognitive status. The possible endemic viral agents in south Asia and Bangladesh causes residual neurologic deficit following encephalitis are Japanese Encephalitis Virus (JEV) or Nipah Virus (NV) [16-19]. JEV is a flavivirus (single strand RNA) endemic in rural southeast Asia spread by mosquito bite. JEV could invade thalamus, basal ganglia, brain stem, cerebellum, hippocampus and cerebral cortex. Elderly population are with increased risk of mortality of about 50%. During prodromal period patient may complaints of fever, headache, nausea, diarrhea, vomiting and myalgia. Patient may initially present with change in mental status, or confusion and rapidly progress to coma. In late stage patient may present with persistent symptoms e.g. polio-like flaccid paralysis, parkinsonism, seizures, cognitive and language impairment. JEV may have hyponatremia related to SIADH [16,17,18]. Our patient had persistent cognitive impairment, but hyponatremia was related to CSWS not SIADH. JEV-specific IgM Enzyme-Linked Immunoassay (ELISA) on serum or CSF is the diagnostic test which was not done as that will not contribute much in the patient management. Treatment for JEV is symptomatic and no effective antiviral agent exists but prevention is possible through vaccination. NV is a paramyxovirus (enveloped RNA virus) is known since its outbreak in Malaysia during 1998- 99 [20-22]. NV has two different strain identified, the Bangladesh and Malaysia strain with 92% similarity in sequence but different in pathogenicity [23-25]. Fever, headache, dizziness, myalgia are the non-specific prodromal symptoms. The common neurological symptoms are altered mental status, severe weakness, and seizure [26]. In NV encephalitis MRI brain showed multifocal discrete lesions consistent with micro-infarction [27,28]. At the acute stage of the illness NV PCR from throat and nasal swab, CSF and blood is diagnostic. The treatment is supportive, but the use of Ribavirin is being explored [29]. In our patient MRI brain did not reveal any micro-infarction and NV PCR was not done as that will not make any significant change in the management.
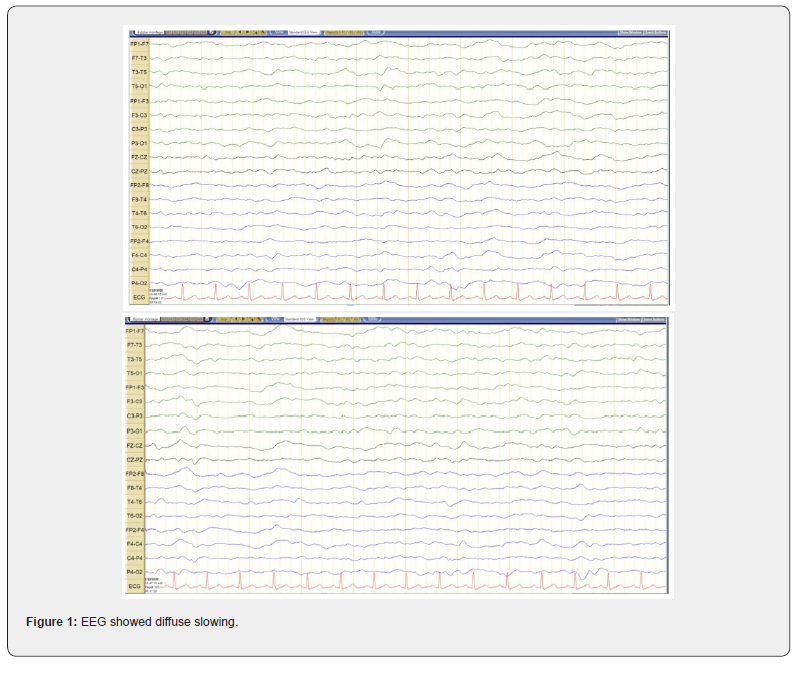
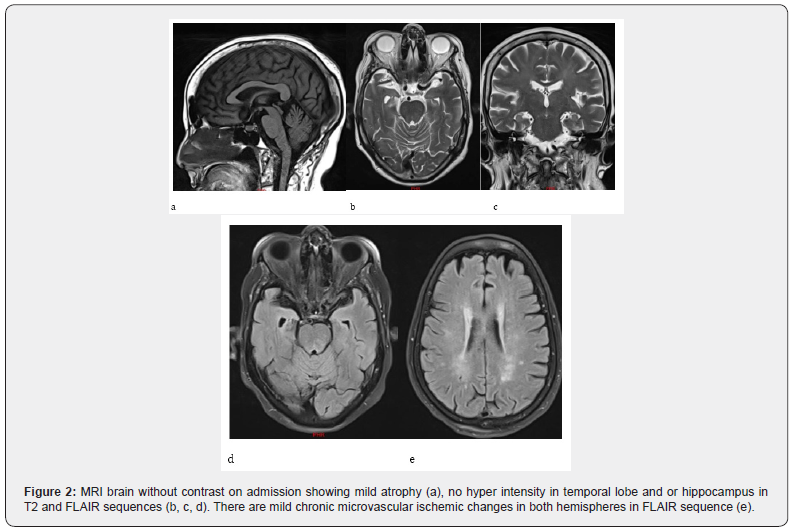
The second possibility for patient’s persistent cognitive deficit is probably related brainstem injury related to prolong hypotension, dehydration, and septic shock. Here we have to keep in mind that our patient remained unmonitored since symptom onset during initial 9-day period. When patient presented to us was noted to be hypotensive and grossly dehydrated but maintaining her O2 saturation. She needed inotropic administration and significant fluid supplement to overcome the hypotension. According to the prior studies, during hypoxic hypotensive events, comparatively hypotension contributes to the brain injury instead of hypoxia [30,31]. Thus, in our patient probably the prolong hypotension could be the significant contributing factor for her persistent cognitive deficit. The third possible cause could be related to correction of hyponatremia. Our patient had isotonic hyponatremia. Sodium level at presentation to us was 130 mM/L, which was treated with IV NS administration as it was a mild hyponatremia. Our patient is consistent with chronic hyponatremia (duration >48 hour) instead of acute hyponatremia (duration <48 hour). Chronic hyponatremia is prone to Osmotic Demyelination Syndrome (ODS) if hyponatremia is corrected rapidly [8]. The goal for correction of hyponatremia is 4-6 mM/L per day to avoid ODS. With IV NS administration after 4 days serum sodium reached up to 142 mM/L. At discharge after 8 days of hospital stay serum sodium was 139 mM/L. Thus, with this gradual correction of hyponatremia we did not expected to have pontine myelinolysis or ODS as cause of patient’s persistent cognitive deficit. Initial MRI brain did not reveal high T2 signal at pons or midbrain. The repeat MRI brain could be further revealing that was beyond our scope in this patient.
Conclusion
Encephalitis is common clinical presentation, especially in a developing country like Bangladesh. Our case is unique as she is elderly and survived unmonitored initial 9-day period. It was further interesting that with optimal treatment she recovered but could not achieved the baseline cognitive status. There are several differential diagnoses we discussed few possible ones related to our case. Thorough diagnostic work could not be entertained as those may not bring significant change in the plan of management which is a limitation in our case. This case teaches us some quick guidance to be hopeful for better outcome in a developing country like Bangladesh although patient look grime at initial presentation.
References
- Ball R, Halsey N, Braun MM, Moulton LH, Gale AD, et al. (2002) Development of case definitions for acute encephalopathy, encephalitis, and multiple sclerosis reports to the vaccine: adverse event reporting system. J Clin Epidemiol 55(8): 819-824.
- Glaser CA, Honarmand S, Anderson LJ, Schnurr DP, Forghani B, et al. (2006) Beyond viruses: clinical profiles and etiologies associated with encephalitis. Clin Infect Dis 43(12): 1565-1577.
- Venkatesan A, Tunkel AR, Bloch KC, Lauring AS, Sejvar J, et al. (2013) International Encephalitis Consortium. Case definitions, diagnostic algorithms, and priorities in encephalitis: consensus statement of the international encephalitis consortium. Clin Infect Dis 57(8): 1114-1128.
- Domingues RB, Lakeman FD, Mayo MS, Whitley RJ (1998) Application of competitive PCS to cerebrospinal fluid samples from patients with herpes simplex encephalitis. J Clin Microbiol 36(8): 2229-2234.
- Sili U, Kaya A, Mert A, HSV Encephalitis Study Group (2014) Herpes simplex virus encephalitis: clinical manifestations, diagnosis and outcome in 106 adult patients. J Clin Virol 60(2): 112-118.
- Basaran S, Yavuz SS, Bali EA, Cagatay A, Oncul O, et al. (2019) Hyponatremia is predictive of HSV-1 encephalitis among patinets with viral encephalitis. Tohoku J Exp Med 247(3): 189-195.
- Riancho J, Delgado-Alvarado M, Sedano MJ, Polo JM, Berciano J (2013) Herpes simplex encephalitis: clinical presentation, neurological sequelae and new prognostic factors. Ten years of experience. Neurol Sci 34(10): 1879-1881.
- Ranger AM, Chaudhary N, Avery M, Fraser D (2012) Central pontine and extrapontine myelinolysis in children: a review of 76 patients. J child Neurol 27(8): 1027-1037.
- Katila J, Singh RK, Misra UK (2017) Cerebral salt wasting is the most common cause of hyponatremia in stroke. J Strke Cerebrovasc Dis 26(5): 1026-1032.
- Moritz ML (2019) Syndrome of inappropriate antidiuresis. Pediatr Clin North Am 66(1): 209-226.
- Bolat F, Oflaz MB, Guyen AS (2013) What is safe approach for neonatal hypernatremic dehydration? A retrospective study from neonatal intensive care unit. Pediatr Emerg Care 29(7): 808-813.
- Cui H, He G, Yang S, Lv Y, Jiang Z, et al. (2019) Inappropriate antidiuretic hormone secretion and cerebral salt-wasting syndromes in neurological patients. Front Neurosci 13: 1170.
- Jain P, Jain A, Kumar A (2014) Epidemiology and etiology of acute encephalitis syndrome in north India. Jpn J Infect Dis 67(3): 197-203.
- Ghosh S, Basu A (2016) Acute encephalitis syndrome in India: the changing scenario. Ann Neurosci 23(3): 131-133.
- Misra UK, Kalita J, Singh RK, Bhoi SK (2019) A Study of Hyponatremia in Acute Encephalitis Syndrome: A Prospective Study from a Tertiary Care Center in India. J Intensive Care Med 34(5): 411-417.
- Soloman T, Ni H, Beasley DW, Ekkelenkamp M, Cardosa MJ, et al. (2003) Origin and evolution of Japanese Encephalitis virus is southeast Asia. J Virol 77(5): 3091-3098.
- Li X, Cui S, Gao X, Wang H, Song M, et al. (2016) The spatio-temporal distribution of Japanese encephalitis cases in different age groups in mainland China, 2004-2014. PLoS Negl Trop Dis 10:1-17.
- Campbell GL, Hills SL, Fischer M, Jacobson JA, Hoke CH, et al. (2011) Estimated Global incidence of Japanese encephalitis: a systemic review. Bull World Health Organ 89(10): 766-774.
- Paul RC, Rahman M, Gurley ES, Hossain MJ, Diorditsa S, et al. (2011). A novel low-cost approach to estimate the incidence of Japanese encephalitis in the catchment area of three hospitals in Bangladesh. Am J Trop Med Hyg 85(2): 379-385.
- Kasiazek TG, Rota PA, Rollin PE (2011) A review of Nipah and Hendra viruses with an historical aside. Virus Res 162(1-2): 173-183.
- Center for disease control and prevention (CDC) (1999) Update: outbreak of Nipah virus - Malaysia and Singapore, 1998-1999. Morb Mortal Wkly Rep 48(16): 335-337.
- Daszak P, Plowright R, Epstein JH, Pullium J, Abdul Rahman D, et al. (2006) The emergence of Nipah and Hendra virus: pathogen dynamics across a wildlife livestock-human continuum. Disease Ecology: Community structure and pathogen dynamics. Oxford Scholarship Online pp186-201.
- Parrish CR, Holmes EC, Morens DM, Park EC, Burke DS, et al. (2008) Cross-Species Virus Transmission and the Emergence of New Epidemic Diseases. Microbiol Mol Biol Rev 72(3): 457–470.
- Hayman DT, Suu-Ire R, Breed AC, McEachern JA, Wang L, et al. (2008) Evidence of hanipavirus infection in West African fruit bats. PloS One 3(7): e2739.
- Banerjee S, Gupta N, Kodan P, Mittal A, Ray Y, et al. (2019) Nipah virus disease: A rare and intractable disease. Intractable Rare Dis Res 8(1): 1-8.
- Sejvar JJ, Hossain J, Saha SK, Gurley ES, Banu S, et al. (2007) Long-term neurological and functional outcome in Nipah virus infection. Ann Neurol 62(3): 235-242.
- Ong KC, Wong KT (2015) Henipavirus encephalitis: recent delopments and advances. Brain Pathol 25(5): 605-613.
- Sarji SA, Abdullah BJ, Goh KJ, Tan CT, Wong KT (2000) MR imageing features of Nipah encephalitis. Am J Roentgenol 175(2): 437-442.
- Chong HT, Kamarulzaman A, Tan CT, Goh KJ, Thayaparan T, et al. (2001) Treatment of acute nipah encephalitis with ribavirin. Ann Neurol 49(6): 810-813.
- Wijdicks EFM, Stevens M (1992) The role of hypotension in septic encephalopathy following surgical procedures. Arch Neurol 49(6): 653-656.
- Adams JH, Brierley JB, Connor RCN, Treip CS (1966) The effects of systemic hypotension upon human brain: clinical and neuropathological observations in 11 cases. Brain 89(2): 235-268.






























