Diffusion Tensor Imaging: The Future of Diagnostics in Amputation Neuromas
Gabriella Glassman1, Isaac Manzanera2, Michael Pridmore2, Alonda Pollins1, Brad Hill1, Leon M Bellan3, Kathleen Weber Montgomery1, Salam Kassis1, Richard Dortch2 and Wesley P Thayer1,3*
1Vanderbilt University Medical Center (VUMC), USA
2Vanderbilt University Institute of Imaging Science (VUIIS), USA
3Vanderbilt University, Departments of Mechanical and Biomedical Engineering, USA
Submission: October 29, 2019;Published: November 18, 2019
*Corresponding author: Wesley P Thayer, Department of Plastic Surgery, Vanderbilt University Medical Center, 1161 21st Ave South, MCN S-2221, Nashville, TN, USA
How to cite this article: Gabriella Glassman, Isaac Manzanera, Michael Pridmore, Alonda Pollins, Brad Hill, Leon M Bellan, Kathleen Weber Montgomery, Salam Kassis, Richard Dortch, Wesley P Thayer. Diffusion Tensor Imaging: The Future of Diagnostics in Amputation Neuromas. Open Access J Neurol Neurosurg. 2019; 12(1): 555830. DOI: 10.19080/OAJNN.2019.12.555830.
Abstract
Each year in the United States, approximately 185,000 people undergo limb amputations. Their recovery can be complex as many will suffer from either phantom pain, infection, ulceration, stump pain, and/or painful neuromas. Painful neuromas complicate rehabilitation and have devastating effects on quality of life. Neuromas are difficult to diagnose with current methods, which include clinical exam, ultrasound and Magnetic Resonance (MR) neurography. Individually, these techniques are often inadequate and cannot reliably distinguish neuromas from complex regional pain syndrome, phantom pain, abscess formation, osteomyelitis or local tumor recurrence. Without a clear diagnosis, amputation neuroma patients are turned away, neglected and /or treated with overly conservative measures. Enhanced detection of neuromas following amputations or traumatic peripheral nerve injuries would allow for earlier surgical intervention and minimized physical disability. Diffusion Tensor Imaging (DTI) is a non-invasive MRI based approach that probes tissue at the microstructural level and has recently demonstrated exciting potential for neuroma diagnosis. Unlike current detection methods, DTI measures water diffusion anisotropy and provides quantitative parameters to describe abnormal nerve growth. Nerve degeneration and/or regeneration in animal models can be monitored with DTI and tractography based on its sensitivity to nerve microstructure. Our lab has further employed DTI for detection of suspected neuromas in animals that failed to recover after sciatic nerve injury and neurorrhaphy. With future advancements in human DTI peripheral nerve protocols, earlier neuroma detection and surgical intervention will be possible.
Keywords: Neuroma MRI DTI Diffusion tensor imaging Fractional anisotropy Amputation
Abbrevations: DTI: Diffusion Tensor Imaging; VUIIS: Vanderbilt University Institute of Imaging Science; SPAIR: Spectral Attenuated Inversion Recovery; FA: Fractional Anisotropy; RD: Radial Diffusivity; AD: Axial Diffusivity; MD: Mean Diffusivity; ADC: Apparent Diffusion Coefficient
Introduction
Neuromas result from a disruption in the normative nerve regeneration process after amputation or following a traumatic nerve laceration, surgery, and/or crush injury [1,2]. These abnormal nerve proliferations are characterized by disorganized axonal growth outside the severed epineural sheath and they typically develop at the distal end of the transected nerve. Each year in the United States approximately 185,000 people undergo limb amputations [3]. It is believed that due to an increased prevalence of vascular disease, diabetes, and trauma the prevalence of limb loss is expected to double by 2050 [4]. Unfortunately, associated postamputation pain can be physically debilitating, making ambulation with prosthesis unbearable [5]. Painful neuromas are often refractory to medicinal therapies, and surgical intervention is the definitive treatment [5,6]. The bulbous neuroma can be resected followed by interventions such as proximal nerve reattachment to a nearby normal nerve or neurorrhaphy, targeted muscle reinnervation, nerve grafting, or epineural closure [6,7]. Nonetheless, roughly 61% of amputation patients report some residual pain. Forty nine percent of amputees’ stump-related pain is presumed to be caused by neuromas [8]; however, neuromas may be overlooked or misdiagnosed as phantom pain, infections, ulceration, cicatrization, or stump pain [9,10]. Enhanced detection of neuromas following amputations or traumatic peripheral injuries would improve surgical management and help minimize physical disability.
Radiographic imaging of neuromas can be difficult and there is no ‘gold standard’ imaging protocol [11]. High resolutionUltrasound (US) and Magnetic Resonance Imaging (MRI) have been implemented to detect nerve damage; [12,13] however, the disruption of normal anatomy and inflammation around an amputation site can interfere with the precision necessary to adequately describe a neuroma. Furthermore, MRI of small caliber nerve trunks in below-the-knee amputations are difficult to visualize [12]. Better diagnostic techniques are needed to help differentiate painful neuroma from complex regional pain syndrome, phantom pain, or local tumor recurrence after a cancer related amputation.
Our experience
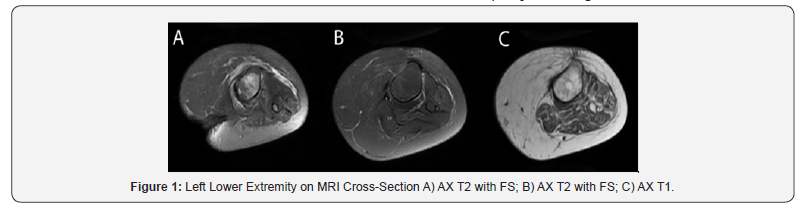
Though current diagnostics methods are inadequate for amputation neuromas, novel imaging protocols have demonstrated promising results. In our experience, a 26-yearold female with a history of polyarteritis nodosa presented with left lower extremity pain approximately 19 months after a below-the-knee amputation. Given her amputation history and physical exam findings, there was a high clinical suspicion for neuroma or nerve entrapment. As part of her neuroma workup a lower extremity non-joint MRI was performed with and without contrast using standard hospital MRI technology. The sequences obtained included axial T1, axial T1 with Fat Suppression (FS), axial T2 with FS, coronal Short Tau Inversion Recovery sequence (STIR), coronal T1, sagittal proton density with FS, axial T1 with FS post contrast and coronal T1 with FS post contrast sequences of the left lower extremity (Figure 1). The radiologist’s impression described a positive fluid collection and surrounding inflammation with signal abnormalities within the adjacent residual tibia. The differential diagnoses included mechanical, inflammatory, and infectious causes. Osteomyelitis was strongly considered given the presumed adjacent abscess formation. The radiologist report concluded there was no evidence of a neuroma. Prior to performing a surgical exploration and possible resection of the suspected neuroma, the Vanderbilt University Institute of Imaging Science (VUIIS) performed an additional scan under an Institutional Review Board approved MRI research study. The VUIIS houses experts of Philips scanners who can perform specialized scans with higher resolution than the scans utilized in the clinical/hospital setting.
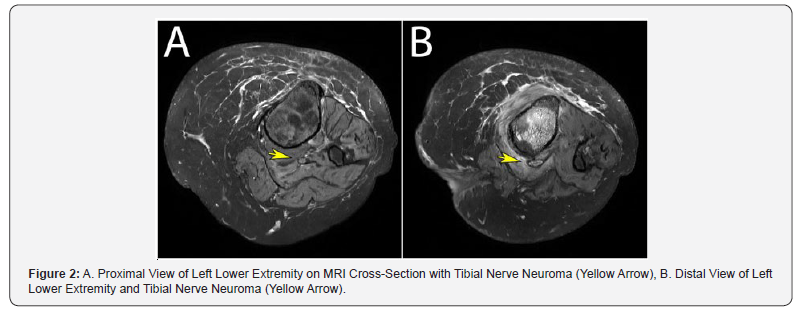
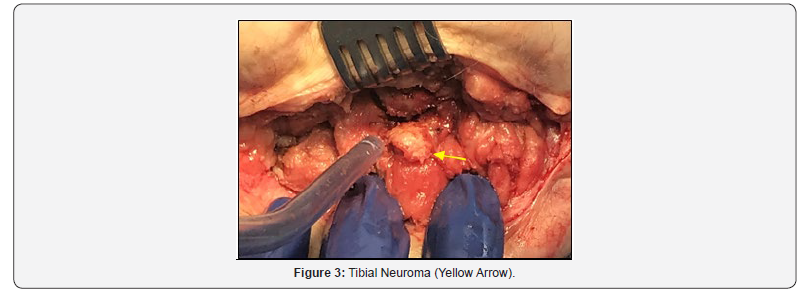
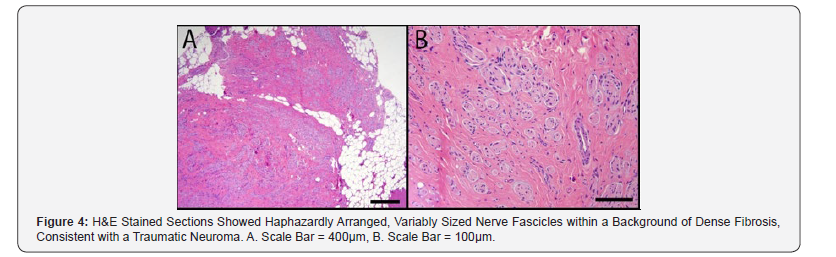
The scan conducted at VUIIS included a proton-densityweighted MRI with Spectral Attenuated Inversion Recovery (SPAIR) and gradient-reversal fat suppression techniques to achieve the more detailed and clearer images seen in Figure 2. Utilizing the Philips SPAIR fat suppression, we achieved an enhanced visualization of irregular thickening of the tibial nerve at the distal end of the stump (Figure 2, yellow arrows). After surgical exploration and resection, the surgical pathology report confirmed our suspicion and agreed with our specialized MRI findings. A traumatic neuroma (Figure 3 & 4) was found adjacent to the nerve with surrounding foreign body giant cell reaction (no osteomyelitis was identified).
Discussion
Improved detection of peripheral neuromas and irregular nerve regeneration could drastically change patient outcomes and improve overall quality of life. Even with technical advancements, current imaging modalities cannot always differentiate neuroma proliferations from peripheral nerveneoplasms, an important distinction in the case of local recurrent cancer [11,14]. This may be due to abnormal scarring, edema, and abscess formation. MRI techniques still require a high level of expertise to enhance the area of interest and eliminate bright signals from fatty atrophy and/ or anatomical abnormalities [12]. When imaging neuromas, some experts suggest using the low signal intensity on T1-weighted images and intermediate to high signal intensity on T2-weighted MR images [15]. Neuroma images typically demonstrate scarring and an absence of a target sign; they can, however, give off a low signal intensity ring on T1 and T2 weighted images [1,15,16]. Existing limitations of standard MRI scans for neuroma patients include problematic issues with metal screening safety and accessibility depending on the state of the patient’s disability, as the subject is required to lay on a table for up to an hour at a time for scanning. Lastly, the abnormal structure of fat, muscle, and bone create a unique challenge to apply MRI scans that were developed for use on healthy subjects.
Diffusion Tensor Imaging (DTI) is a non-invasive MRI based approach that characterizes tissue at the microstructural level. DTI was developed in the 1980s [17,18] and has since then revolutionized MRI detection of human central nervous system pathology including strokes [19], brain tumors [20], multiple sclerosis [21-23] schizophrenia, aging [24], and other cancers [21-26]. DTI measures water diffusion along multiple directions, measuring the effect of tissue barriers that results in diffusion anisotropy. In healthy nerves, the ordered arrangement of axons results in an Apparent Diffusion Coefficient (ADC) that is lower perpendicular to axons than parallel to them. Unlike high resolution ultrasounds and MR neurography, diffusion along multiple directions can be measured with DTI by quantifying indices that describe its diffusion anisotropy. Some of the most important indices are: fractional anisotropy (FA = 0-1, lower values indicate low anisotropy), radial diffusivity (RD, diffusivity perpendicular to the axons), axial diffusivity (AD, diffusivity along the axons), and mean diffusivity (MD, mean value across all directions). The scope of DTI technology has gradually expanded to include the peripheral nervous system; however, image precision remains a challenge due to the small caliber nerves, requiring high spatial resolution balancing signal to noise ratio and time limitations while addressing field inhomogeneity, fat suppression, distortion and motion, [27] was among the first to demonstrate DTI’s use in human peripheral nerves when he accurately scanned and produced DT images of the sciatic nerve in 3 healthy human subjects [28].
In subsequent years, Hiltunen et al. [29] successfully produced DT images, 3D tractography, ADC and FA measurements of the radial, median, ulnar, tibial and peroneal nerves. Hiltunen and coworkers concluded that FA values vary with thickness of the nerve. Meek et al. [30] was among the first to use DTI in a human subject with an isolated lesion of the median nerve. The lesion was repaired 1 month prior to imaging, and therefore Meek et al. [30] exhibited the utility of DTI in differentiationof intact and regenerative nerve fibers in the median nerve. Others further support DTI use in peripheral nerves and are seeking to create standard parameters for detection of axon and myelin sheath integrity of distal nerves of the wrist [31]. DTI functionality in peripheral nerve pathology continues to grow and now encompasses traumatic peripheral nerve injuries [30], carpal and cubital tunnel syndromes [31,32] as well as neoplasms. To date DTI of peripheral neuromas has not been thoroughly studied. Our lab has used DTI parameters to detect suspected neuromas in animals that failed to recover after sciatic nerve injury and repair
Using diffusion MRI techniques and measurements, neuromas that are characterized by dense and incoherent axonal sprouting may be distinguishable from fully regenerated nerves with coherent axonal structure. In addition, virtual fiber tracts (employing DTI measurements to extract the primary direction of diffusion) could provide information regarding the reduced fiber coherence present in neuromas. Nerve degeneration and/ or regeneration in animal models can be monitored with DTI and tractography based on its sensitivity to nerve microstructure. DTI (axonal packing derived diffusivities) and fiber tracking (differences in fiber coherence) techniques have the potential to become an early biomarker in the study and differentiation of neuromas from other regenerating and degenerating nerves after trauma and surgical repair.
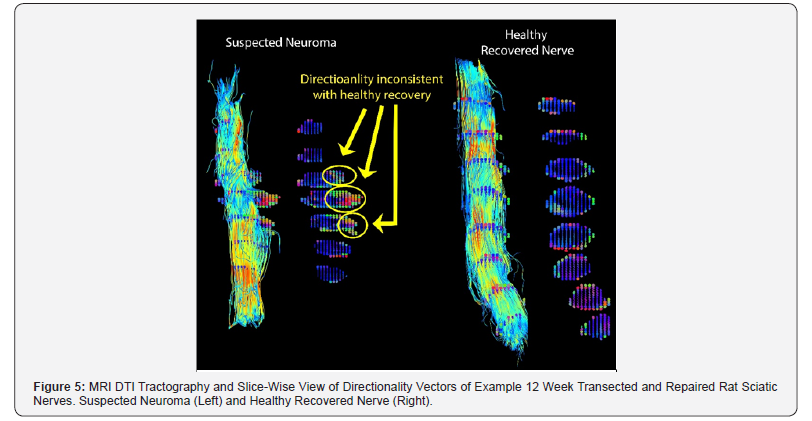
Experiments studying the application of MRI DTI to assess the regeneration of sciatic nerves in a rat model have been conducted in our lab. Post-experimental analysis of complete transection and repair in animals 12 weeks after surgical procedure revealed a possible neuroma. Figure 5 displays the DTI tractography and slice-wise directionality vectors for our suspected neuroma as well as that of a healthy recovered sample. In the slice-wise images of the suspected neuroma, the DTI measurements indicate areas of the nerve where no primary eigenvector can be distinguished. This indicates that the damaged and recovering axons present in this region are dysfunctional and extend in multiple directions, which does not allow for tractography images to be generated in this region. Conversely, in the healthy recovering nerve, the slice-wise DTI measurements consistently indicate axonal extension along the primary eigenvector, producing a tractography image where the entire nerve is fully represented. The preliminary outcomes in animal models are promising and have prompted our research team to employ DTI technology for humans suffering from suspected amputation neuromas.
Conclusion
Amputation patients face adversity adjusting to their new physical limitations, prosthesis rehabilitation, mental exhaustion, and lingering pain. A painful neuroma should be surgically resected shortly after its discovery, but unfortunately current diagnostic methods are inadequate for proper detection and often prolong the repair process. With further advancementsin research and the development of human DTI neuroma protocols, new technology could revolutionize the management of patients suffering from painful neuromas
Funding Acknowledgement
A portion of this work was paid for by NIH R01 NS097821
References
- Singson RD, Feldman F, Slipman CW, Gonzalez E, Rosenberg ZS, et al. (1987) Postamputation neuromas and other symptomatic stump abnormalities: detection with CT. Radiology 162(3): 743-745.
- Rajput K, Reddy S, Shankar H (2012) Painful neuromas. Clin J Pain 28(7): 639-645.
- Owings M, Kozak LJ (1998) Ambulatory and inpatient procedures in the United States, 1996. Vital Health Stat 139: 111-119.
- Ziegler-Graham K, MacKenzie EJ, Ephraim PL, Travison TG, Brookmeyer R (2008) Estimating the prevalence of limb loss in the United States: 2005 to 2050. Arch Phys Med Rehabil 89(3): 422-429.
- Ducic I, Mesbahi AN, Attinger CE, Graw K (2008) The role of peripheral nerve surgery in the treatment of chronic pain associated with amputation stumps. Plast Reconstr Surg 121(3): 908-914.
- Ives GC, Kung TA, Nghiem BT, Ursu DC, Brown DL, et al. (2018) Current State of the Surgical Treatment of Terminal Neuromas. Neurosurgery 83(3): 354-364.
- Dellon AL, Mackinnon SE (1989) Selection of the appropriate parameter to measure neural regeneration. Annals of plastic surgery 23(3): 197-202.
- Buchheit T, Van de Ven T, Hsia HL, McDuffie M, MacLeod DB, et al. (2016) Pain Phenotypes and Associated Clinical Risk Factors Following Traumatic Amputation: Results from Veterans Integrated Pain Evaluation Research (VIPER). Pain med 17(1): 149-161.
- Jensen TS, Krebs B, Nielsen J, Rasmussen P (1985) Immediate and long-term phantom limb pain in amputees: Incidence, clinical characteristics and relationship to pre-amputation limb pain. Pain 21(3): 267-278.
- Jensen TS, Krebs B, Nielsen J, Rasmussen P (1983) Phantom limb, phantom pain and stump pain in amputees during the first 6 months following limb amputation. Pain 17(3): 243-256.
- Ahlawat S, Belzberg AJ, Montgomery EA, Fayad LM (2016) MRI features of peripheral traumatic neuromas. Eur Radiol 26(4): 1204-1212.
- Singson RD, Feldman F, Staron R, Fechtner D, Gonzalez E, et al. (1990) MRI of postamputation neuromas. Skeletal Radiol 19(4): 259-262.
- Cartwright MS, Chloros GD, Walker FO, Wiesler ER, Campbell WW (2007) Diagnostic ultrasound for nerve transection. Muscle Nerve 35(6): 796-799.
- Chhabra A, Williams EH, Wang KC, Dellon AL, Carrino JA (2010) MR neurography of neuromas related to nerve injury and entrapment with surgical correlation. AJNR American journal of neuroradiology 31(8): 1363-1368.
- Henrot P, Stines J, Walter F, Martinet N, Paysant J, et al. (2000) Imaging of the painful lower limb stump. Radiographic No: S219-235.
- Beggs I (1997) Pictorial review: Imaging of peripheral nerve tumours. Clinical Radiology. 52(1): 8-17.
- Le Bihan D BE (1985) Imagerie de diffusion in vivo par résonance magnétique nuclé CR Acad Sci Paris 301: 1109-1112.
- Basser PJ, Mattiello J, LeBihan D (1984) MR diffusion tensor spectroscopy and imaging. Biophys J 66(1): 259-267.
- Chalela JA, Kidwell CS, Nentwich LM, Luby M, Butman JA, et al. (2007) Magnetic resonance imaging and computed tomography in emergency assessment of patients with suspected acute stroke: a prospective comparison. Lancet 369(9558): 293-298.
- Lee WJ, Choi SH, Park CK, Yi KS, Kim TM, et al. (2012) Diffusion-weighted MR imaging for the differentiation of true progression from pseudoprogression following concomitant radiotherapy with temozolomide in patients with newly diagnosed high-grade gliomas. Acad Radiol 19(11): 1353-1361.
- Filippi M, Cercignani M, Inglese M, Horsfield MA, Comi G (2001) Diffusion tensor magnetic resonance imaging in multiple sclerosis. Neurology 56(3): 304-311.
- Werring DJ, Brassat D, Droogan AG, Clark CA, Symms MR, et al. (2000) The pathogenesis of lesions and normal-appearing white matter changes in multiple sclerosis: A serial diffusion MRI study. Brain: A Journal of neurology 123(8): 1667-1676.
- Buchsbaum MS, Tang CY, Peled S, Gudbjartsson H, Lu D, et al. (1998) MRI white matter diffusion anisotropy and PET metabolic rate in schizophrenia. Neuro Report 9(3): 425-430.
- Moseley M (2002) Diffusion tensor imaging and aging - a review. NMR in biomedicine 15(7-8): 553-560.
- Sinha S, Lucas-Quesada FA, Sinha U, De Bruhl N, Bassett LW (2002) In vivo diffusion-weighted MRI of the breast: potential for lesion characterization. J Magn Reson Imaging 15(6): 693-704.
- Conti Nibali M, Rossi M, Sciortino T, Riva M, Gay LG, et al. (2019) Preoperative surgical planning of glioma: limitations and reliability of fMRI and DTI tractography. J Neurosurg Sci 63(2): 127-134.
- Jeon T, Fung MM, Koch KM, Tan ET, Sneag DB (2018) Peripheral nerve diffusion tensor imaging: Overview, pitfalls, and future directions. J Magn Reson Imaging 47(5): 1171-1189.
- Skorpil M, Karlsson M, Nordell A (2004) Peripheral nerve diffusion tensor imaging. Magn Reson Imaging 22(5): 743-745.
- Hiltunen J, Suortti T, Arvela S, Seppä M, Joensuu R, et al. (2005) Diffusion tensor imaging and tractography of distal peripheral nerves at 3 T. Clinical Neurophysiology116(10): 2315-2323.
- Meek MF, Stenekes MW, Hoogduin HM, Nicolai JP (2006) In vivo three-dimensional reconstruction of human median nerves by diffusion tensor imaging. Exp Neurol 198(2): 479-482.
- Heckel A, Weiler M, Xia A, Ruetters M, Pham M, et al. (2015) Peripheral Nerve Diffusion Tensor Imaging: Assessment of Axon and Myelin Sheath Integrity. PLoS One 10(6): e0130833-e0130833.
- Iba K, Wada T, Tamakawa M, Aoki M, Yamashita T (2010) Diffusion-weighted magnetic resonance imaging of the ulnar nerve in cubital tunnel syndrome. Hand surgery: an international journal devoted to hand and upper limb surgery and related research: journal of the Asia-Pacific Federation of Societies for Surgery of the Hand 15(1): 11-15.






























