Status Dystonicus- Case Report and Intensive Care Management
Patrick Britell1, 2 and Julio A Chalela2,3*
1Department of Anesthesia, Medical University of South Carolina, USA
2Department of Neurosurgery, Medical University of South Carolina, USA
3Department of Neurology, Medical University of South Carolina, USA
Submission: September 19, 2019;Published: October 09, 2019
*Corresponding author: Julio A Chalela, Department of Neurosurgery & Neurology, Medical University of South Carolina, USA
How to cite this article: Patrick Britell, Julio A Chalela. Status Dystonicus- Case Report and Intensive Care Management. Open Access J Neurol Neurosurg. 2019; 11(4): 555820. DOI: 10.19080/OAJNN.2019.11.555820.
Abstract
Background: Status dystonicus, a rare complication of dystonia occurring almost exclusively in children is defined as a sudden and severe worsening of dystonic movements leading to bulbar weakness, airway compromise, respiratory failure and metabolic derangements progressing to pain and exhaustion
Objectives: to describe a case of status dystonicus and provide an overview of its critical care management.
Methods: Case report study and literature review
Results: A 23-year-old female with chronic cervico-brachial dystonia presented to the hospital with severe, painful, sustained dystonic posture involving the neck, arm, and hand. The dystonia prevented her from eating, drinking, or sleeping. She was well controlled with a deep brain stimulator and physical therapy. On admission she was dehydrated, had elevated creatine kinase and mild pre-renal azotemia. Intravenous benzodiazepines failed to improve her symptoms. She was transferred to the intensive care unit where she was hydrated with intravenous crystalloids and started on continuous infusions of dexmedetomidine and ketamine as well as PRN intravenous opiates and benzodiazepines reserving intubation, sedation with propofol, and possible neuromuscular blockade for lack of improvement. The patient responded to escalating doses of dexmedetomidine/ketamine and intubation was foregone. Her dystonic posture improved, and she was able to resume oral intake and transfer to the ward.
Conclusion: Status dystonicus is a rare neurologic emergency that may require intensive care unit admission. We successfully treated a patient with status dystonicus with simultaneous infusions of an alpha-2 agonist and an NMDA-antagonist avoiding the need for intubation, mechanical ventilation, sedation, or neuromuscular blockade. Status Dystonicus (SD) is defined as a sudden and severe worsening of dystonic movements leading to bulbar weakness, airway compromise, respiratory failure and metabolic derangements progressing to pain and exhaustion [1,2]. It is a more severe and persistent presentation of the baseline dystonia that can cause systemic complications and require Neurosciences Intensive Care Unit (NSICU) admission. Occurring in patients with primary or secondary dystonia, SD is a rare disorder with only 100 reported cases mostly in children [1,2]. We hereby describe a case of de novo SD occurring in a young adult. We review the clinical syndrome and propose a treatment algorithm for patients with SD in the NSICU
Keywords: Dystonia Status Intensive care Status dystonicus Neurosciences intensive Care unit Deep brain stimulator Sleep Lorazepam Dystonic posture Aggressive interventions Neurologic emergencies Aspiration pneumonia Neurogenic hyperventilation Jaw dystonia
Abbrevations: SD: Status Dystonicus; NSICU: Neurosciences Intensive Care Unit; DBS: Deep Brain Stimulator
Case Report
A 23-year old female with secondary focal dystonia was admitted to the hospital with persistent, painful dystonic movements for one week. She had a long-standing history of dystonia affecting the left neck (laterochollis) and the left wrist/hand and was being treated only with a Deep Brain Stimulator (DBS) and physical therapy. She was able to perform all normal activities. One week prior she developed sustained, painful, left laterochollis and sustained painful left wrist flexion and hand contraction. She was unable to eat, drink, or sleep due to bizarre neck positioning and pain. She had a recent upper respiratory infection but no other triggers. Due to lack of response to intravenous lorazepam she was transferred to the NSICU.
The patient was awake and alert and in moderate distress. She was afebrile with a blood pressure of 90/55 mmHg and a heart rate 106. Her mentation was normal, and her speech was clear. She had marked laterochollis to the left, inward rotation of the left shoulder, flexion of the left arm, forced flexion of the left wrist, forced supination of the wrist, and forced contracture of the left hand (akin to writer’s cramp). Sensation and coordination were normal in non-affected limbs. Reflexes were brisk and symmetric. No myoclonic or choreiform movements were appreciated. No involvement of the respiratory muscles was evident, but she complained of dysphagia and difficulty coughing due to her dystonic posture. She was deemed to be a grade 4 in the Dystonia Severity Action Plan Scale (DSAPS) [3].
Laboratory studies showed blood urea nitrogen of 34, Cr of 0.9, and Creatine Kinase (CK) of 800. Chest X-ray showed left lower lobe atelectasis. She was vigorously hydrated with isotonic crystalloids. Her DBS battery was changed without symptom relief. Supplemental oxygen was administered anticipating the need for intravenous sedatives/hypnotics. Concomitant infusions of Ketamine (1mcg/kg/min to 10 mcg/kg/min) and dexmedetomidine (0.25 mcg/kg/hour to 1.0 mcg/kg/hr) were initiated for analgesia, sedation, and anxiolysis. Intermittent PRN hydromorphone and Diazepam 5 mg IV every 6 hours were used for analgesia and muscle relaxation respectively. The above treatment plan was continued for 24 hours, with the understanding that if it was unsuccessful, endotracheal intubation and deep sedation would be performed. The patient was monitored for response and exhaustion signs.
24 hours later, after titration of ketamine to 4 mcg/kg/min and dexmedetomidine to 1.0 mcg/kg/min, the patient improved to a grade 2 DSAPS. The current therapy was continued, and intubation foregone. The patient was able to attain 15 degrees elbow extension, able to bring her chin close to midline, and able to halfway open her hand. After additional 48 hours of ketamine/ dextemedetomidine infusion she improved to a grade 1 DSAPS and was able to ingest fluids. Renal function and CK normalized and she was transferred to the ward.
Management of status dystonicus
In 1982 Jankovic reported a child with dystonia muscularis deformans who developed severe painful torticollis evolving into generalized dystonia with fever, intense pain and teacolored urine [4]. He required intubation and neuromuscular blockade and subsequently developed rhabdomyolysisinduced renal failure. Treatment with dopamine blockers, anticholinergic agents, sedatives, neuromuscular blockers and anticonvulsants was ineffective and he required a ventrolateral right thalamotomy. The cardinal findings of SD are bulbar dysfunction with potential airway compromise, progressive impairment of respiratory function, exhaustion and pain, and metabolic derangements [1,2,5]. Infections, new medications, or cessation of medications can trigger SD. Untreated SD canlead to rhabdomyolysis, metabolic disturbances, aspiration, and respiratory failure. Fatality is 10% and more common among males with tonic dystonia [6]. Involvement of the diaphragm or severe opisthotonos can lead to severe respiratory failure [5]. Superimposed hyperkinetic movements and bizarre body positions may make NSICU management challenging and predispose to rhabdomyolysis and exhaustion. Most important, SD causes extreme discomfort to the patient and anxiety to the family. Traditional dystonia therapies often fail in SD and admission to the NSICU may be needed for more aggressive interventions. To our knowledge this is the first report of SD in the neurocritical care literature.
As in most neurologic emergencies airway, breathing, and circulation take priority in managing SD. Intubation, sedation and neuromuscular blockade are needed in approximately 30- 40% of patients [6]. Respiratory compromise can result from bulbar dysfunction, aspiration pneumonia, atelectasis, or from abnormal chest wall compliance (from opisthostonos, severe trunk flexion, or diaphragmatic dystonia). Abdominal wall dystonia may exert restrictive effect on the lungs. Laryngeal muscle involvement may complicate management and hinder extubation. Bag mask ventilation prior to anesthesia induction may be challenging in patients with neck or jaw dystonia. Venous thromboembolism due to immobility can also cause respiratory failure. Neurogenic hyperventilation is common and may require sedation and/or neuromuscular blockade. In rare circumstances in which dystonia interferes with ventilation mechanical ventilation may be required for months [5].
After a patent airway has been established all efforts should be made to ensure adequate hydration. Volume depletion stems from poor oral intake and increased insensible losses. Muscle induced exothermic causes increased temperature and dehydration. Fractures resulting from violent dyskinetic movements and/or falls can be a source of bleeding and hypovolemia. Temperature can rise to 107 degrees Fahrenheit increasing insensible losses [4]. Hydration with isotonic crystalloids is recommended while monitoring for signs of rhabdomyolysis and acute kidney injury. Levels of CK are typically 5 times normal. Rhabdomyolysis is a common complication of SD and if untreated, can lead to renal failure [1]. Elevations in CK may lag behind other clinical findings [1].
Systemic complications are common in patients with SD and need to be addressed promptly. Pneumonia, venous thromboembolism, metabolic acidosis, sepsis, acute renal failure, ileus, acute malnutrition, rhadomyolysis, soft tissue injuries, trauma from violent movements, and toxic drug reactions are common [7]. Antipyretics, cooling blankets, or endovascular devices may be required to control hyperthermia. Complications may also arise from the natural morbidity associated with deep sedation and neuromuscular blockade. Nursing care in the NSICU can be challenging as the dystonic movements can displace medical devices, interfere with monitoring and evenbreak orthopedic casts [5]. The psychological impact on the patient and family is immense and requires a multidisciplinary approach. Early tracheostomy and gastrostomy placement are often needed given the prolonged need for mechanical ventilation and the ubiquitous occurrence of dysphagia and malnutrition.
Specific treatment for SD is tailored to the severity; generally, grades 4-5 will require NSICU admission. A management algorithm is proposed in figure 1. Agents used prior to NSICU admission can be attempted at higher doses but most cases require therapy escalation. Obviously, there is more than one dystonic syndrome and it is not clear if merely achieving muscle relaxation or resting the dystonic brain is adequate treatment for all. Treatment goals are controlling the dystoniato achieve synchronous ventilation, preventing aspiration, preventing exothermic heat loss, alleviating pain, preventing rhabdomyolysis, providing relief from exhaustion, and ensuring adequate nutrition and hydration. Levo-dopa trials are warranted in early-onset cases to exclude dopa-responsive dystonia [5]. Intravenous anticholinergic medications (trihexyphenidyl) can also be attempted. Tetrabenazine can be used to deplete all three monoamine neurotransmitters (dopamine, noradrenaline, serotonin) [5]. Post-synaptic blockade of dopamine receptors can be achieved with pimozide, haloperidol, or other phenothiazines. Intravenous benzodiazepines (midazolam, clonazepam, diazepam), oral baclofen, and oral gabapentin have been used with variable results [1]. Clonidine may be used in non-hypotensive patients.
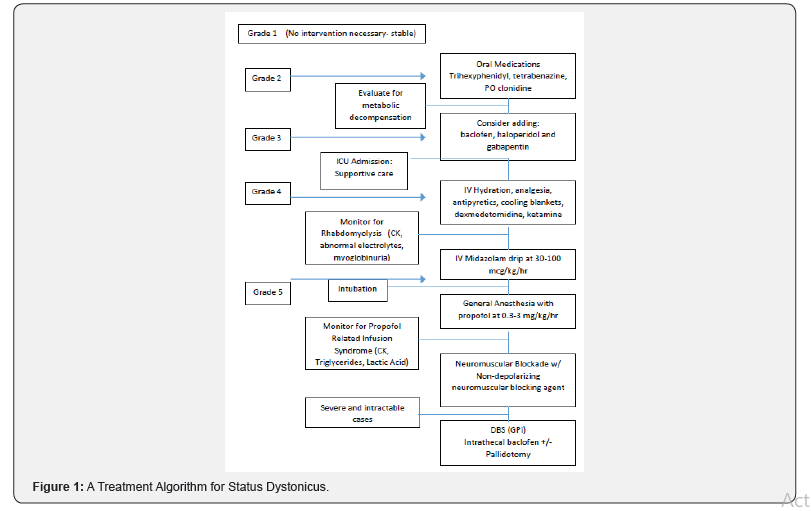
Patients who do not respond to the above-mentioned interventions will require intubation, mechanical ventilation, sedation, and possibly neuromuscular blockade. Intubating the patient with cervical dystonia can be challenging as the dystonic posture may persist after induction doses of benzodiazepines, fentanyl, and propofol.8 Furthermore, acute airway obstruction can complicate their administration and require airway adjuncts use [7,8]. A careful airway exam should note Mallampati Score, thyromental distance, and ability to prognath. Neck mobility may be very limited. Oromandibular dystonia resulting in tongue biting with resulting edema and bleeding can hinder laryngoscopy [7]. In some situations, a laryngeal mask airway can be used to ventilate the patient with continued neck ante flexion after induction [8]. However, obtaining a seal and ventilating with a laryngeal mask airway can also be complicated due to tracheal displacement from cervical dystonia.
Neuromuscular blocking drugs administered with intubation may be helpful in reducing the degree of spasm associated with SD. However, depending on symptom duration, some degree of abnormal head posture may remain and may complicate intubation and ventilation. Neuromuscular blocking drugsshould be administered with caution due to the potential of acan’t ventilate, can’t intubate scenario [8].
After intubation, temporizing measures such as sedation and spasm relief can proceed in a stepwise fashion. Dexmedetomidine; an alpha 2 agonist with a similar mechanism of action to clonidine, has been used in the placement of DBS in patients with SD [9]. It can be a useful adjunct in accomplishing analgesia, sleep and light sedation. Ketamine addition may potentiate analgesia. Midazolam, a short acting benzodiazepine, in doses from 30-100 mcg/kg/hr can be used help to alleviate the spasms associated with SD. Tolerance to Midazolam can develop quickly and this drug may promote delirium in mechanically ventilated patients [10].
General anesthesia should be undertaken with propofol if the above measures fail. Propofol is rapidly titratable to induce dystonia relief. However, propofol related infusion syndrome can occur during prolonged infusion (> 48hrs) and coexisting rhabdomyolysis and acidosis may complicate its detection as they may occur concomitantly in SD. Triglycerides should be monitored, and propofol discontinued in the presence of hypertriglyceridemia associated with acidosis or elevated CK [11].
Persistent SD after anesthesia implementation requires neuromuscular blockade with a non-depolarizing muscle relaxant. Some non-depolarizing muscle blockers are renally excreted and should be used with caution if rhabdomyolysisinduced acute renal failure coexists. The exception to this is cisatracurium, which undergoes Hoffman elimination (pH and temperature dependent) and does not rely on liver metabolism and renal excretion. Severe hyperthermia may interfere with the action of cisatracurium. If after intubation, sedation, anesthesia and paralysis, the patient remains in refractory SD then more invasive therapies are warranted. Treatment options for superrefractory SD include, intrathecal baclofen, DBS placement in the globus pallidus and pallidotomy [1]. There is limited data on the implementation of intrathecal baclofen, in patients with refractory SD with most data derived from case reports.1 Intrathecal baclofen used may be limited by technical failures and/or tolerance. This can be combined with unilateral or bilateral pallidotomy to achieve symptom relief. However, these therapies carry with them a high eventual mortality and pallidotomy has largely been replaced by DBS placement.
Some authors advocate for early DBs placement in the globus pallidus in patients with refractory SD. This has been found to be highly effective with a low incidence of recurrence [1,2,12] Placement of a DBS may prevent some of the complications associated with mechanical ventilation, anesthesia, and neuromuscular blockade, and reduce length ofstay. The benefits of DBS use may be appreciated immediately or make take weeks/months to occur. Placement in children may be complicated by electrode displacement with subsequent growth [2]. Neurointensivists should consult with movement disorder specialists and functional neurosurgeons to determine if surgical options are feasible.
Conclusion
This case outlines the management of stage 4 DSAPS with good resolution. It illustrates the NSICU management priorities in status dystonicus. It demonstrates the need to be cognizant of temperature management, pain control, and fluid resuscitation. Fortunately, it was not necessary to progress in the algorithm to intubation and general anesthesia. The symptoms gradually abated, and she was able to be discharged to the general ward and then home.
References
- Allen NM, Lin JP, Lynch T, Kind MD (2014) Status dystonicus: a practice guide. Dev Med Child Neurol 56(2):105-112.
- Sobstyl MR, Sławek JW, Ząbek M (2014) The neurosurgical treatment of patients in dystonicstate - Overview of the literature. Neurologiai Neurochirurgia Polska 48(64): 63-70.
- Lumsden DE, Lundy C, Fairhurst C, Lin JP (2013) Dystonia Severity Action Plan: a simple grading system for medical severity of status dystonicus and life-threatening dystonia. Dev Med Child Neurol 55(7): 671-672.
- Jankovic J, Penn AS (1982) Severe dystonia and myoglobinuria. Neurology 32(10): 1195-1197.
- Manji H, Howard RS, Miller DH, Hirsch P, Carr L, et al. (1998) Status dystonicus: the syndrome and its management. Brain 121(121): 243-252.
- Fasano A, Ricciardi L, Bentivoglio AR, Canavese C, Zorzi G, et al. (2012) Status dystonicus: predictors of outcome and progression patterns of underlying disease. Mov Disord 27(6): 783-788.
- Marras CE, Rizzi M, Cantonetti L, Rebessi E, De Benedictis A, et al. (2014) Pallidotomy for medically refractory status dystonicus in childhood. Dev Med Child Neurol 56(7): 649-656.
- Byrappa V, Redhu S, Varadarajan B (2016) Anesthetic management of a patient with severe neck dystonia during MRI. J Anaesthesiol Clin Pharmacol 32(1): 115-116.
- Maurtua MA, Cata JP, Martirena M, Deogaonkar M, Rezai A, et al. (2009) Dexmedetomidine for deep brain stimulator placement in a child with primary generalized dystonia. J Clin Anesth 21(3): 213-216.
- Riker R, Shehabi Y, Bokesch P (2009) Dexmedetomidine vs Midazolam for sedation of critically ill patients: a randomized trial. JAMA 301(5): 489-499.
- Mirrakhimov A, Voore P, Halytskyy O, Khan M (2015) Propofol Infusion Syndrome in Adults: A Clinical Update. Critical Care Research and Practice 2015:1-10.
- Maurtua MA, Cata JP, Martirena M, Deogaonkar M, Rezai A, et al. (2009) Dexmedetomidine for deep brain stimulator placement in a child with primary generalized dystonia. J Clin Anesth 21(3): 213-216.






























