Congenital Zika Syndrome A Clinical Study of Six Patients
Orlando J Castejón*, Torres Ytalo, Pedro Finol Parra, Patricia Galindez, Maria Salones Castejón and Grundbaum Esmeira
Department of Neuroscience, Zulia University, Venezuela
Submission: December 10, 2018; Published: March 15, 2019
*Corresponding author: Orlando J Castejón, Facultad de Medicina, Instituto de Investigaciones Biológicas, Universidad del Zulia, Apartado 526, Maracaibo, Venezuela
How to cite this article: Orlando J C, Torres Y, Pedro F P, Patricia G, Maria S C,et al. Congenital Zika Syndrome A Clinical Study of Six Patients . Open Access J Neurol Neurosurg. 2019; 10(2): 555782. DOI: 10.19080/OAJNN.2019.10.555782.
Abstract
Six infant patients with microcephaly and neurodevelopmental disorders were studied from the clinical point of view exhibiting mainly reduced cephalic circumference, spastic paralysis of upper and lower extremities, ostetendinous hiperreflexia, bilateral strabismus, dysmorphic faciee and genu valgo bilateral. Transfontanelar ecography showed thin and porous aspect of corpus callosum, and ventricolomegaly. Computerized axial tomography showed lissencephaly and calcifications in basal ganglia and dispersed in the thick and condensed brain parenchyma. Nuclear magnetic resonance neuroimages showed ventriculomegaly, absence of corpus callosum, scarce differentiation of gray and white matter, and vascular and cerebellar hypoplasia. Electroencephalographic recording displayed focal paroxysmal activity and hipsarritmia in some patients. Mothers were infected in the first and second trimester of pregnancy.
Keywords: Microcephaly; Zika virus; Maternal zikv infection; Brain development
Abbrevations: GBS: Guillain-Barré Syndrome; BBB: Blood-Brain Barrier
Introduction
The Zika virus is a flavivirus spread by mosquitoes. Its primary vectors are the Aedes aegypti and the Aedes albopictus [1]. Recent advances in epidemiology, clinical medicine, pathology, and experimental studies have provided a great amount of new information regarding vertical ZIKV transmission and the mechanisms of congenital microcephaly, brain damage, and congenital Zika syndrome in a relatively short time [2]. A spotlight has been focused on the mosquito-borne Zika virus (ZIKV) because of its epidemic outbreak in Brazil and Latin America, as well as the severe neurological manifestations of microcephaly and Guillain-Barré syndrome associated with infection [3].
There is a prevalence of microcephaly in babies born to Zika virus-infected women during pregnancy, since it is known that infection at the first trimester is associated with higher risk to congenital anomalies [4 5]. In addition, Zika virus was a cause of the neurological disorder Guillain-Barré Syndrome (GBS) and other congenital brain abnormalities [6,7]. Multivariate sensitivity analyses indicated that the cumulative incidence of ZIKV infection and risk of microcephaly given maternal infection in the first trimester were the primary drivers of both magnitude and uncertainty in the estimated number of microcephaly cases [8]. A large number of viral particles were found in the central nervous tissue of an electively aborted microcephalic ZIKV-infected fetus [9].
Recent advances in in vitro research show that ZIKV can infect and obliterate cells from the CNS, such as progenitors, neurons, and glial cells. Neural progenitor cells seem to be the main target of the virus, with infection leading to less cell migration, neurogenesis impairment, cell death and, consequently, microcephaly in newborns [10,11]. According to these Authors, ZIKV triggers a strong immune response and disrupts neurovascular development, resulting in postnatal microcephaly with extensive brain damage [12]. Using immunocytochemistry and electron microscopy have shown that ZIKV targets human brain cells, reducing their viability and growth as neurospheres and brain organoids. These results suggest that ZIKV abrogates neurogenesis during human brain development. Significant enhancement of the number of syncytial sprouts was observed in the placentas of women infected during the third trimester, indicating the development of placental abnormalities after ZIKV infection [13].
Brain damage was confirmed through intrauterine ultrasonography and was complemented by magnetic resonance imaging reduction in cerebral volume, ventriculomegaly, cerebellar hypoplasia, lissencephaly with hydrocephalus, and fetal akinesia deformation sequence (ie, arthrogryposis [14,15]. reported ventriculomegaly, brain calcifications and posterior fossa destruction lesions, are also common in this congenital syndrome.
Global gene expression analysis of infected brains reveals upregulation of candidate flavirus entry receptors and dysregulation of genes associated with immune response, apoptosis, and microcephaly [16]. Microglial hyperplasia, reactive gliosis, and myelination delay have been reported in ZIKV-infected microcephalic fetuses. Specifically, ZIKV infection disturbs the proliferation and differentiation of the oligodendrocyte progenitor cells and leads to the abolishment of oligodendrocyte development ZIKV infection leads to cell-cycle arrest, apoptosis, and inhibition of NPC differentiation, resulting in cortical thinning and microcephaly [17].
Anomalies of the corpus callosum and presence of cerebral hyperechogenic ties were significantly more common in the infected group. When the mother was infected during the first or second trimester, there was a greater risk of severe CNS involvement, more signs of infection and intrauterine fetal death than with infection in the third trimester [18]. According to these Authors, a detailed neurosonography on a monthly basis, paying particular attention to the corpus callosum and the presence of cerebral hyperechogenic ties is highly recommended.
Material and Methods
Anomalies of the corpus callosum and presence of cerebral hyperechogenic ties were significantly more common in the infected group. When the mother was infected during the first or second trimester, there was a greater risk of severe CNS involvement, more signs of infection and intrauterine fetal death than with infection in the third trimester [18]. According to these Authors, a detailed neurosonography on a monthly basis, paying particular attention to the corpus callosum and the presence of cerebral hyperechogenic ties is highly recommended.
Clinical Study
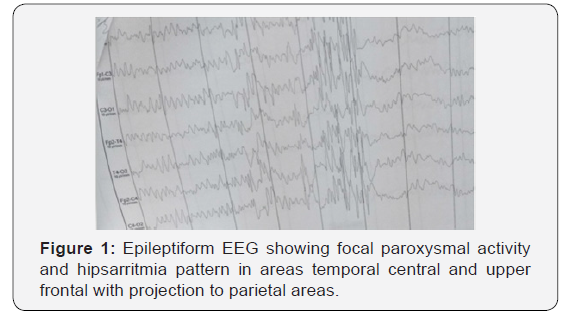
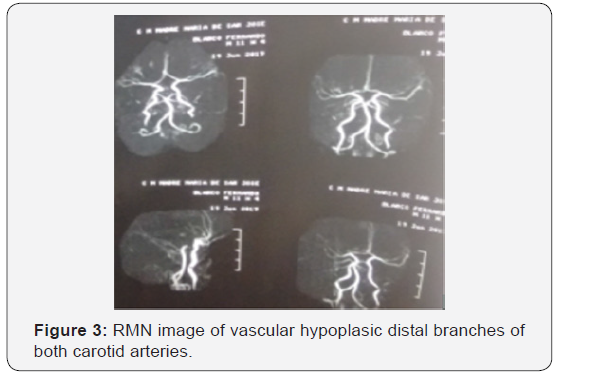
Infant patient, 2 years-old, showing a reduced cephalic circumference featuring postnatal mycrocephaly, spastic paralysis of upper and lower extremities and ostetendinous hiperreflexia (Figure 1). His mother was Zika virus infected during the first trimester of pregnancy. Electroencephalographic recording showed focal paroxistic activity in areas central temporal and superior frontal with projection to parietal areas. Presence of punta, polipunta, slow waves, and light suppression of voltage characteristic of hipsarritmic pattern. Nuclear magnetic resonance neuroimage showed diminution of cranial and brain volume, notably enlargement of lateral ventricles, absence of corpus callosum, scarce differentiation of gray and white matter, and vascular hypoplasia of distal branches of both carotid arteries.
Infant patient one-year-old showing showed reduced cephalic circumference 44cm of cephalic circumference, featuring postnatal microcephaly. Spastic paralysis of upper and lower extremities, ostetendinous hiperreflexia, bilateral strabismus, dysmorphic facie and bilateral genu valgo was observed. His mother was Zika virus infected during the first trimester of pregnancy. MNR exhibited deformities of sulci and gyri of brain cortex and ventriculomegaly (Figure 2). Mother suffered Zika virus infection during first month of pregnancy. The diagnose was early established by transfontanelar ecography (Figure 3).

Ten months-old girl with severe damage during neurodevelopmental process, exhibiting weak neck muscles and difficulties for holding the head, muscle hipotony and spasticity of upper and lower limbs, ostetendinous areflexia, emission of guttural sounds and taquicardy. Mother suffered Zika virus infection during the second trimester of mother pregnancy (Figure 4).

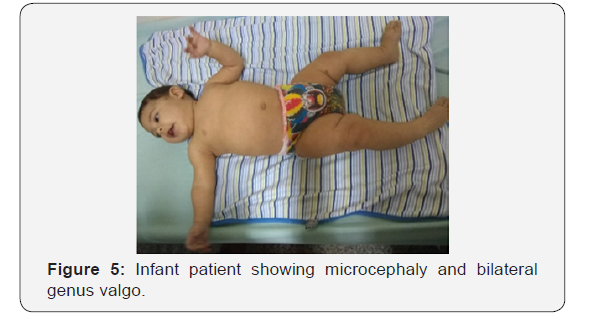
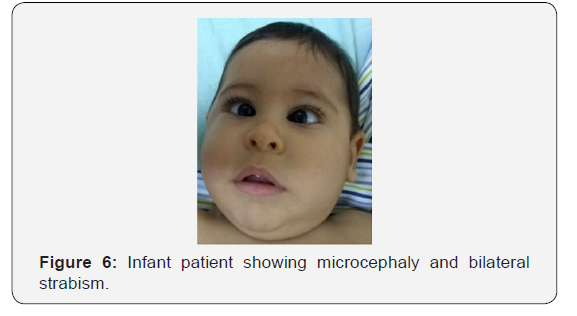
Nine-month-old boy with microcephaly, muscular hipotony, skin café au lait spots, repeated regurgitation of food, depression in the superior part of the skull at temporal and occipital regions. Left eye strabismus, facial dismorphia, hands flexed, sensorineural hearing loss, and upper right leg shorter than the right one. Mother suffered Zika virus infection during first trimester of pregnancy. Computerized axial tomography showed lissencephaly, calcifications in basal ganglia and dispersed in brain parenchyma at temporo-occipital regions (Figure 5).
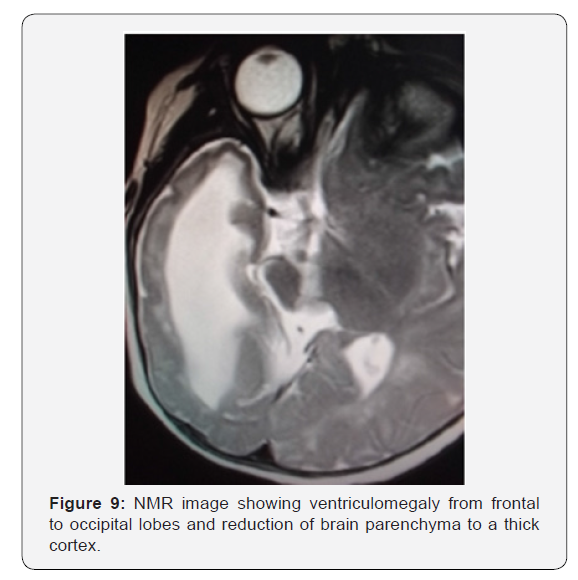
Two years old boy with microcephaly, spasticity of arm, bilateral strabismus, genus valgo bilateral. Mother suffered Zika virus infection during the first trimester of pregnancy. RMN showed ventriculomegaly and brain parenchyma reduced to a compact and thick cortex (Figure 6).
Discussion
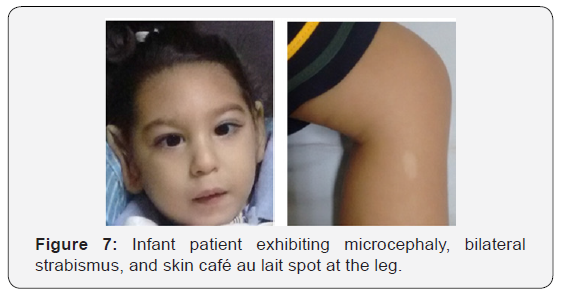
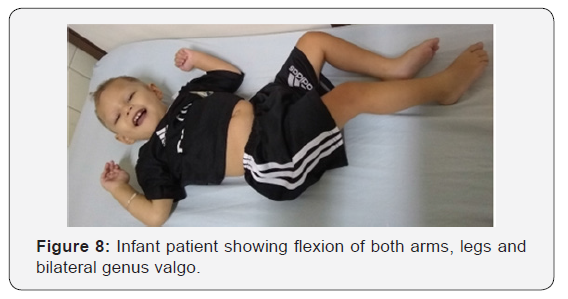
The neuroimage observations in the present paper are similar to those reported by Melo, et al. [14] and Sarno et al. [15] demonstrating the neutropism activity of Zyka virus during brain development. Our study supports the concept postulated by Pokmar, et al. [18] that Zika virus infection during the first trimester of pregnancy evinces a greater risk of severe CNS involvement in the first trimester than infection in third trimester. In case No 1 we observed vascular hypoplasia of distal branches of both carotid arteries. According to Shao, et al. [11]. ZIKV infection leads to abnormal vascular density and diameter in the developing brain, resulting in a leaky Blood-Brain Barrier (BBB), massive neuronal death and BBB leakage indicate brain damage, which is further supported by extensive microglial activation and astrogliosis in virally infected brains. This infant patient also showed absence of corpus callosum apparently related with brain parenchyma reduced blood flow and the hypoxic ischemic encephalopathy, which also explain the bilateral strabismus and genus valgo exhibited by the patient (Figure 7 & 8).
Porous aspect of corpus callosum porous aspect of corpus callosum was observed in case No. 3, which demonstrate the value of suggestion related to carry out a detailed neurosonography on a monthly basis, paying particular attention to the corpus callosum and the presence of cerebral hyperechogenic ties [18]. Brain parenchyma and basal ganglion calcifications as reported in the present paper were also described by [7]. According to de villous immaturity was the main histological finding in the placental tissues, although placentas without alterations were also frequently observed. Significant enhancement of the number of syncytial sprouts was observed in the placentas of women infected during the third trimester, indicating the development of placental abnormalities after ZIKV infection. As above mentioned, large numbers of viral particles were found in the central nervous tissue of an electively aborted microcephalic ZIKV-infected fetus [9] (Figure 9).
As postulated by [10], the mechanisms underlying ZIKV infection in the CNS require further investigations particularly as there are currently no treatments or vaccines against ZIKV infection. New insights into the molecular antigenic structure of Zika virus and flaviviruses in general have provided the foundation for great progress made in developing Zika virus vaccines and antibodies for passive immunization [19].
References
- Varjasi G, Póka R (2017) Zika virus infection in pregnancy. Orv Hetil 158 (15): 563-571.
- Alvarado MG, Schwartz DA (2017) Zika Virus Infection in Pregnancy, Microcephaly, and Maternal and Fetal Health: What We Think, What We Know, and What We Think We Know. Arch Pathol Lab Med 141(1): 26-32.
- Olagnier D, Muscolini M, Coyne CB, Diamond MS, Hiscott J (2016) Mechanisms of Zika Virus Infection and Neuropathogenesis. DNA Cell Biol 35(8): 367-372.
- Tang BL (2016) Zika virus as a causative agent for primary microencephaly: the evidence so far. Arch Microbiol 198(7): 595-601.
- Coelho AVC, Crovella S (2017) Microcephaly Prevalence in Infants Born to Zika Virus-Infected Women: A Systematic Review and Meta-Analysis. Int J Mol Sci 18(8) pii: E1714.
- Krauer F, Riesen M, Reveiz L, Oladapo OT, Martínez-Vega R, et al. (2017) Zika Virus Infection as a Cause of Congenital Brain Abnormalities and Guillain-Barré Syndrome: Systematic Review. PLoS Med 14(1): e1002203.
- Marrs C, Olson G, Saade G, Hankins G, Wen T, et al. (2016) Zika Virus and Pregnancy: A Review of the Literature and Clinical Considerations. Am J Perinatol 33(7): 625-639.
- Ellington SR, Devine O, Bertolli J, Martinez Quiñones A, Shapiro-Mendoza CK, et al. (2016) Estimating the Number of Pregnant Women Infected with Zika Virus and Expected Infants with Microcephaly Following the Zika Virus Outbreak in Puerto Rico. JAMA Pediatr 170(10): 940-945.
- Pawitwar SS, Dhar S, Tiwari S, Ojha CR, Lapierre J, et al. (2017) Overview on the Current Status of Zika Virus Pathogenesis and Animal Related Research. J Neuroimmune Pharmacol 12(3): 371-388.
- Russo FB, Jungmann P, Beltrão-Braga PCB (2017) Zika infection and the development of neurological defects. Cell Microbiol 19(6).
- Shao Q, Herrlinger S, Yang SL, Lai F, Moore JM, et al. (2016) Zika virus infection disrupts neurovascular development and results in postnatal microcephaly with brain damage. Development 143(22): 4127-4136.
- Garcez PP, Loiola EC, Madeiro da Costa R, Higa LM, et al. (2016) Zika virus impairs growth in human neurospheres and brain organoids. Science 352 (6287): 816-818.
- De Noronha L, Zanluca C, Burger M, Suzukawa AA, Azevedo M, et al. (2018) Zika Virus Infection at Different Pregnancy Stages: Anatomopathological Findings, Target Cells and Viral Persistence in Placental Tissues. Front Microbiol 9: 2266.
- Melo AS, Aguiar RS, Amorim MM, Arruda MB, Melo FO, et al. (2016) Congenital Zika Virus Infection: Beyond Neonatal Microcephaly. JAMA Neurol 73(12): 1407-1416.
- Sarno M, Aquino M, Pimentel K, Cabral R, Costa G, et al. (2017) Progressive lesions of central nervous system in microcephalic fetuses with suspected congenital Zika virus syndrome. Ultrasound Obstet Gynecol 50(6): 717-722.
- Li C, Xu D, Ye Q, Hong S, Jiang Y, et al. (2016) Zika Virus Disrupts Neural Progenitor Development and Leads to Microcephaly in Mice. Cell Stem Cell 19(1):120-126.
- Li C, Wang Q, Jiang Y, Ye Q, Xu D, et al. (2018) Disruption of glial cell development by Zika virus contributes to severe microcephalic newborn mice. Cell Discov 4(43).
- Pomar L, Malinger G, Benoist G, Carles G, Ville Y, et al. (2017) Association between Zika virus and fetopathy: a prospective cohort study in French Guiana. Ultrasound Obstet Gynecol 49(6): 729-736.
- Heinz FX, Stiasny K (2017) The Antigenic Structure of Zika Virus and Its Relation to Other Flaviviruses: Implications for Infection and Immunoprophylaxis. Microbiol Mol Biol Rev 81(1) pii: e00055-16.






























