Spinal Metastasis of Hemangiopericytoma
Suffian Wee Ahmad*
Department of surgery, John Hunter Hospital, Australia
Submission: December 17, 2018; Published: April 16, 2018
*Corresponding author: Suffian Wee Ahmad, Department of surgery, John Hunter Hospital, New South Wales, Australia, Email: ahmad.shafiq@hotmail.com
How to cite this article: Suffian W A. Spinal Metastasis of Hemangiopericytoma. Open Access J Neurol Neurosurg. 2018; 7(3): 555715. DOI: 10.19080/OAJNN.2018.07.555715.
Abstract
A 42 year old female presented with a 12 months' history of worsening lower back pain due to a hemangiopericytoma in the thoracic region. She was subsequently managed aggressively with combined modality approach (pre-operative embolization, radical tumor resection and radiotherapy). Patient exhibited promising improvement of function post treatment. Histological findings of the tumor was characteristic of hemagiopericytoma.
Introduction
Hemangiopericytoma has classically been labeled as a variant of meningioma. However that classification has now been modified to malignant mesenchymal non meningeal tumor [1]. It is composed mainly of pericytes, which are the contractile pericapillary cells that surround blood capillaries. It is well known for its aggressive nature, with high propensity towards local recurrences and late extraneural metastasis mainly to bone, eyes, liver, and lungs [2-4]. Multiple treatment modalities have been tested, but macroscopic total surgical resection with adjuvant post-op radiotherapy remains the most acceptable and common form of treatment; chemotherapy remains inefficacious as a form of treatment.
Case Presentation
We present the case of a 42 year old Caucasian lady who was referred from a rural general surgeon for further investigation and management of an incidental finding of a spinal lesion at the T8 level on computed tomography (CT) chest. The CT was initially performed to investigate back pain post cholecystectomy. The patient's background history was significant for previous parietal lobe hemangiopericytoma which had been surgically extirpated and radiated ten years prior to her current presentation, as well hypothyroidism and lower back injury 3 decades prior.
The patient reported intermittent localized back pain without any exacerbating or alleviating factors. She denied any sensorimotor symptoms, and did not present with any loss of bowel or bladder function. There was no history of unexplained weight loss and no features of infection. Physical examination was unremarkable, with normal sensorimotor function, and negative Hoffman and Babinski signs. On admission, a CT scan of the thorax, abdomen and pelvis showed two lytic lesions involving the T8 vertebral body and left acetabulum posteriorly, suggesting metastatic disease, as well as canal narrowing at T8 level due to soft tissue mass replacement of the vertebral body.
Magnetic resonance Imaging (MRI) of the brain was unremarkable; however, spine MRI demonstrated avid enhancement within the T8 vertebral body, extending into the right pedicle of T8 as well as the right transverse process. It also extended superiorly into the T7 vertebral body. The lesion also spread inferiorly into the anterior epidural space at the level of T9. There was an epidural component to the lesion at the level of T8 and T9, with extrusion into the central canal and right exit foramina, thereby causing moderate to severe canal stenosis at the T8 level, with subsequent posterolateral displacement of the cord. There was also mild loss of the T8 vertebral body height, thoracic curvature was however preserved (Figure 1-3).
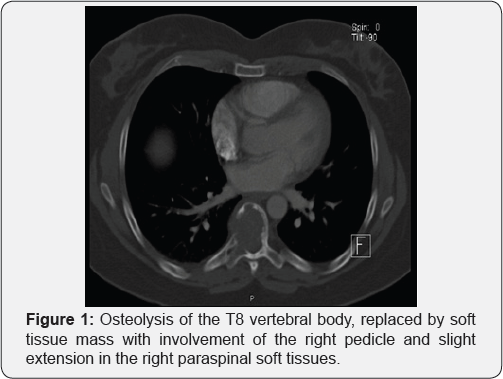
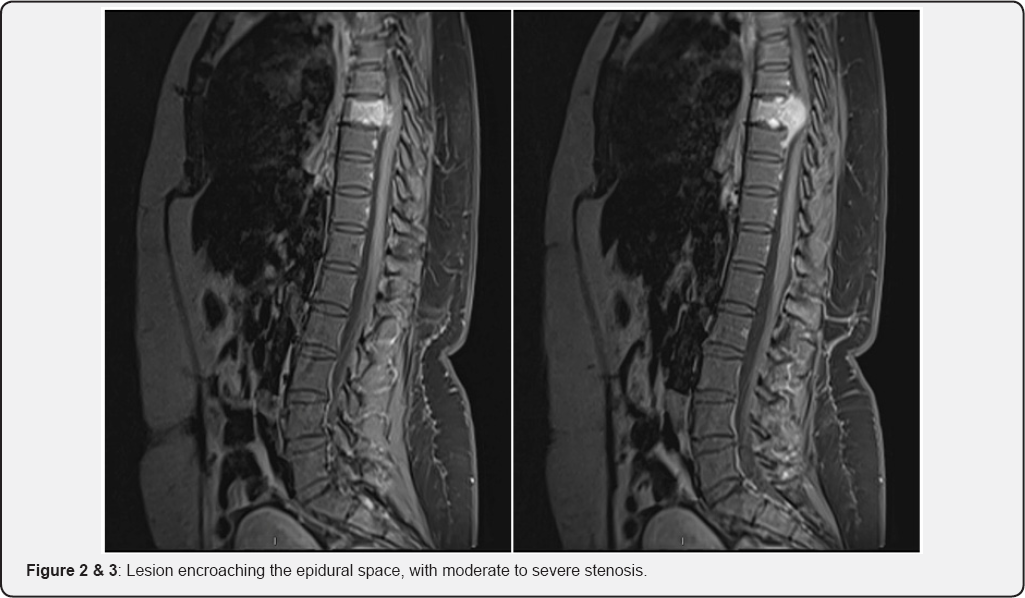
Histopathological analysis of the biopsied left acetabular lesion was consistent with metastatic hemangiop, with features of haphazard short spindle cells in a background of collagen and abundant thin-walled vessels. Immunohistochemical stains were significantly positive for CD34 and STAT6. The Ki67 proliferative index was <5%. The patient subsequently underwent a pre-operative embolization in view of T8 vertebrectomy the following day. Distal embolization of the vertebral body tumor was attempted, but was however abandoned due to irregular walls and perforated the distal branch. The procedure was terminated after the set of T8 segmental arteries were found to have been occluded.
The patient underwent T8 verterbrectomy, with concurrent T6-T10 pedicle screw stabilization and cage reconstruction. The T8 vertebra was sent for histopathology, which confirmed hemangioperictyoma. The morphology and the immunoprofile of the lesion were similar to the acetabular biopsy. The overall appearance was consistent with hemangiopericytoma. Her recovery was uneventful, with no neurological deficit postprocedure. She underwent 2 weeks of rehabilitation, followed by radiotherapy. She was reviewed 3 months later and was back to baseline function, having resumed work.
Discussion
Hemangiopericytoma is a rare hyper vascular soft tissue tumor, and is thought to arise from pericytes [5,6]. Three types of spinal hemangiopericytoma have been described, namely, intramural hemangiopericytoma, intraspinal extradural meningeal hemangiopericytoma and secondary metastatic osseous hemangiopericytoma [6]. In relation to the case report, our patient had secondary metastatic osseous hemangiopericytoma, which presented as a recurrence from her previous intracranial hemangiopericytoma.
Current evidence suggests that surgery (with or without pre-op embolization) followed by radiotherapy is the mainstay of treatment. The role of adjuvant chemotherapy remains unestablished, as there has been no concrete evidence of mortality or morbidity benefits with current chemotherapeutic agents [3].
Surgery provides the benefits of tissue diagnosis and gross total resection which increases the overall survival and recurrence-free interval [5,7]. In our case, cage reconstruction and screw stabilization were performed in order to restore and maintain optimum kyphosis alignment post thoracic vertebrectomy.
Surgery has been proven to be the most effective way in treating hemangiopericytoma; however, it comes with high risk of intraoperative bleeding, mainly due to the marked dilatation of the vascular bed in the vicinity of the neoplasm [5,7]. Other possible complications associated with surgical managementinclude neurological deficit, CSF leakage, rebleeding, infection, cage dislocation, construct failure and bronchial perforation.
Endovascular surgery has been widely used for treatment of hypervascular tumor in head, neck and spinal tumour [8]. The aim of preoperative embolization ranges from complete tumor devascularisation and necrosis to a degree of ischemia and hypovascularity, with the aim to achieve decreased overall intraoperative blood loss [9]. It helps to improve visualization at surgery and to occlude deep, inaccessible arterial feeders to the tumor. Having said that, the risk of bleeding does not diminish post embolization as there could be collateralization or angiographically occult vascularization after embolization. In the case of our patient, even though the surgery was uneventful, she still needed blood transfusion post operatively.
Ashour [8] has suggested that the general indications for preoperative embolization are related to feasibility in terms of anatomic, physiologic and technical standpoint. Benefits of embolization would include decreased procedural time, decreased anesthesia time anda reduced need for blood transfusions. All these factors potentially lead to a reduction in the total cost of treatment [10,11]. Disadvantages of embolization are an increased risk of triggering angio-related toxicity and pulmonary oedema, as well as the potential to create sparks during if contacted by monopolar cautery [8]. Possible complications associated with embolization are the inherent risks of anesthesia, allergic reaction to contrast, groin hematoma or pseudoaneurysm, retroperitoneal hematoma, embolization related ischemia, hemorrhage, and last but not least, peritumoraloedema which may subsequently cause neurological deficitson the relevant spinal level and below [8,11].
In intracranial hemangiopericytoma patients, adjuvant radiotherapy or radiosurgery has been proven to play a definite role in disease management [12]. However, evidence for and against radiotherapy is lacking for intraspinal hemangiopericytoma patients per se. Recent evidence describes the efficacy of post-op radiotherapy for positive surgical margins and high grade histology in hemangiopericytoma of the head and neck, and for reducing the risk of local recurrence in meningeal hemangiopericytoma [1,3,13,14]. On the other hand, radiotherapy can also be used preoperatively in recurrent cases where it shrinks the tumor, thereby making gross total surgical resection possible [9]. Many authors have suggested that chemotherapy is futile for therapy and its role is purely palliative [6]. Nevertheless, there is an increasing body of evidence suggesting benefits associated with the usage of antiangiogenic agents such as sunitini b and sorafeni b for the treatment of hemangiopericytoma [2].
Take Home Messages
A. New onset pain, without neurological deficit in patient with history of hemangiopericytoma might indicate a new hemangiopericytoma lesion, however differentials of other soft tissues pathology (malignant fibrous histiocytoma, liposarcoma, and synovial sarcoma) should not be dismissed until proven otherwise.
B. Given that Hemangiopericytoma is a tumor that is known to have high recurrence rate, patient should be followed up in outpatient clinic with at least annual serial MRI, given that 71% can still have progressive disease post radical resection and radiotherapy [2].
C. Role of chemotherapy might be useful in combination of angiogram
D. MRI is the best imaging to visualize the disease extension.
References
- Galanis E, Buckner JC, Scheithauer BW, Kimmel DW, Schomberg PJ, et al. (1998) Management of recurrent meningeal hemangiopericytoma. Cancer 82(10): 1915-1920.
- Ecker RD, Marsh WR, Pollock BE, Kurtkaya-Yapicier O, McClelland R, et al. (2003) Hemangioperictyoma in the central nervous system: treatment, pathological features, and long term follow up in 38 patients. J Neurosurg 98(6): 1182-1187.
- Espat NJ, Lewis JJ, Leung D, Woodruff JM, Antonescu CR, et al. (2002) Conventional hemangiopericytoma: Modern analysis of outcome. Cancer 95(8): 1746-1751.
- K Fathie (1970) Hemangiopericytoma of the thoracic spine; Case report. J Neurosurg 32(3): 371-374.
- Enzinger F, Smith BH (1976) Hemangiopericytoma: An analysis of 106 cases. Hum Pathol 7(1): 61-82.
- Nonaka M, Kohmura E, Hirata M, Hayakawa T (1997) Case Report: Metastatic meningeal hemangiopericytoma of the thoracic spine. Clin Neurol Neurosurg 100(3): 228-300.
- Woitzik J, Sommer C, Krauss JK (2003) Delayed manifestation of spinal metastasis: a special feature of hemangiopericytoma. Clin Neurol Neurosurg 105(3): 159-166.
- Ashour R, Aziz-Sultan A (2014) Preoperative Tumor Embolization. Neurosurgery Clinics of North America 25(3): 601-617.
- Torigoe T, Higure A, Hirata K, Nagata N, Itoh H (2003) Malignant hemangiopericytoma in the pelvic cavity successfully treated by combined-modality therapy: Report of a Case. Surg Today 33(6): 479-482.
- Kriss FC, Khan RD, Schneider RC (1968) Value of angiography in intraspinal mediastinal hemangioperictyoma. J Neurosurg 29(5): 535539.
- Mavrogenis AF, Rossi G, Rimondi E, Papagelopoulos PJ, Ruggieri P (2011) Embolization of bone tumors. Orthopedics 34(4): 303-310.
- Chang SD, Sakamoto G (2003) The role of radio surgery for hemangiopericytomas. Neurosurg Focus 14(5): e14.
- Mira JG, Chu FC, Fortner JG (1977) The role of radiotherapy in the management of malignant hemagiopericytoma: Report of eleven new cases and review of the literature. Cancer 39(3): 1254-1259.
- Staples JJ, Robinson RA, Wen BC, Hussey DH (1999) Hemangiopericytoma-The role of radiotherapy. Int J Radiat Oncol Biol Phys 19(2): 445-451.






























