Use of Intravenous Fluorescein for Intra-Operative Localization of an Intramedullary Spinal Cord Tumour; A Technical Note
John Adams, Murray Hong, Sean D Christie and Sean P Barry*
Department of Surgery (Neurosurgery), Dalhousie University, Canada
Submission: February 02, 2018; Published: March 19, 2018
*Corresponding author: Sean P Barry, Department of Surgery (Neurosurgery), Dalhousie University, Halifax, Nova Scotia, 1796 Summer Street, B3H 3A7, Canada, Email: SPBARRY@dal.ca
How to cite this article: John A, Murray H, Sean D , Sean P B. Use of Intravenous Fluorescein for Intra-Operative Localization of an Intramedullary Spinal Cord Tumour; A Technical Note. Open Access J Neurol Neurosurg. 2018; 7(3): 555711. DOI: 10.19080/OAJNN.2018.07.555711
Abstract
Background: Localization of intramedullary spine tumors can be difficult. Various intraoperative aids have previously been described, but have limited use due to expense, complexity, and time. Intravenous fluorescein is an inexpensive and safe drug that may be useful in the localization of such tumors. We describe a technical description of the intra-operative use of fluorescein sodium as an aid in the localization of a high grade intramedullary spine tumour.
Clinical presentation: We present a case of a 56 year old man presenting with a intramedullary tumor at the level of C5-6. Intra-operatively, intravenous Fluorescein was administered and the Pentero microscope BLUE™ 400 feature was used to accurately identify the lesion. Multiple biopsies of the fluorescent tissue were taken.
Conclusion: Final pathology confirmed this tissue was consistent witha high-grade glioma. The use of intravenous fluorescein was a valuable aid in localizing the lesion and minimizing the size of our myelotomy. The use of intravenous fluorescein to localize high grade intramedullary spinal cord tumours appears to be safe, accurate, and inexpensive.
Keywords: Fluorescein; Localization; Glioma; Intramedullary tumour
Background and Importance
Surgical resection of intramedullary spine tumors can be associated with significant morbidity. Therefore the management of such tumors often begins with biopsy; then, depending on the pathology, may or may not be followed by surgical resection. Intra-operatively, the lesion of interest may not be seen by direct visualization. After opening dura it is common to see what appears to be normal spinal cord. This can make it difficult in deciding where to perform the necessary myelotomy. In the case of astrocytomas, it can also be difficult to find a plane of dissection between normal and abnormal tissue. Various imaging modalities have been described to aid in the intraoperative localization of these tumors. Intra-operative MRI may be useful but is expensive and time consuming [1]. Ultrasound is inexpensive but can be technically difficult to interpret [2-4]. An accurate and inexpensive adjuvant to localize spine tumors could prove to be very useful in planning a myelotomy and deciding on the ideal sites for biopsy.
Recently,variousstudieshavelookedattheuseof5-Aminolevulinic acid (ALA) to help localize and aid in the resection of high grade gliomas in the brain. 5-ALA is metabolized into fluorescent porphyrin's, which can be visualized under blue light [5]. Zeiss has modified their Pentero 900 microscope to include a Blue light function. Using this filter, the accumulation of 5-ALA in high grade gliomas has been demonstrated in a phase III trial [6]. Similar techniques have not yet been described for intrinsic spinal cord tumours.
Currently 5-ALA is only available in Europe. However, sodium fluorescein is a fluorescent hydrocarbon that is approved for use in Ophthalmology within North America in both intravenous and topical forms. Similar to 5-ALA, fluorescein emits a yellow- green fluorescence when excited by 490nm light. It too has been used in neurosurgery to help localize brain tumors. However, previous studies focused on its ability to stain the color of pathologic tissue yellowish-green under white light. They did not utilize the fluorescent properties seen under the ideal filter, and therefore had to administer high doses to see the desired results. Glioblastoma appears yellow-green under white light after the intravenous administration of fluorescein [7-9]. With this in mind, we planned to utilize the fluorescent properties of fluorescein as well as the blue 400 function of the Pentero microscope, to test a novel technique to aid in the localization of intramedullary spine tumors.
Clinical Presentation
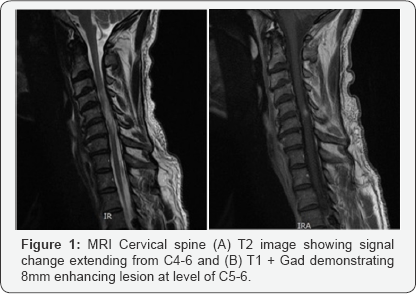
A 56 year old man presents with a 4 month history of progressive functional decline. He has severe myelopathy affecting both upper and lower extremities. A MRI of the spine demonstrated a 8mm enhancing lesion at C5-6 with surrounding edema extending from C4 to C6 (Figure 1). We were concerned about a malignant primary intramedullary tumor and therefore arranged for biopsy for tissue diagnosis.
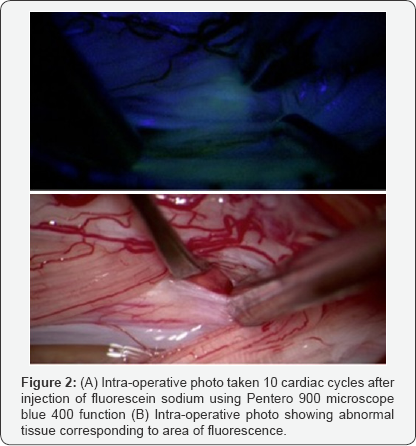
A standard posterior approach with C5-7 laminectomies and a midline dural opening was performed. Normal spinal cord was visualized at the level of the suspected lesion. The operative microscope (PENTERO 900, Carl Zeiss Meditec, Germany) was brought into place. Anesthesia administered a 20mg/kg dose of Intravenous fluorescein (Fluorescite®, Alcon, Canada) while the surgical team observed the surgical field using the Blue 400 function of the Pentero microscope. Almost immediately we began to observe the uptake of fluorescent green in the cerebrospinal fluid (CSF) space, followed by the exposed nerve root sleeves. After 10 cardiac cycles the fluorescent coloring was isolated to what we identified as the intramedullary lesion; this lasted for the time of the procedure (Figure 2). A myelotomy was made just lateral to the dorsal root entry zone adjacent to the suspected lesion. Abnormal tissue was confirmed via intraoperative pathological analysis in the area consistent with the fluorescence observed under the Blue light function. Three separate biopsies were taken, all of which were consistent with a Grade 3 astrocytoma on final pathology.
Discussion
Using the described technique we were able to accurately localize the lesion. Following our myelotomy it was clear that the observed fluorescence corresponded to the area of pathology. Because of this we were able to minimize our myelotomy while still obtaining positive biopsies. Similar techniques have been described in the brain to help guide the extent of resection of high grade gliomas. To our knowledge, this is the first time fluorescein has been used to facilitate localization of an intramedullary spine tumor.
There are several advantages to this technique. The use of a 5cc vial of fluorescein is inexpensive (US$5.99) compared to other drugs, such as 5-ALA which would cost about US$150 in a similar setting. Unlike MRI and intraoperative ultrasound it is also technically simple and easy to interpret, and therefore would not require any additional training of surgeons and/or staff. Compared to 5-ALA it can be given intravenously seconds before the time of interest, as opposed to 3 hours before surgery. Based on its use in ophthalmology we know it is safe to use intravenously with minimal adverse affects [10,11]. Most common side effects are nausea (0.7%), vomiting (0.4%), and dizziness (0.3%). Patients may experience transient skin and urine discoloration, which resolves in 6-12 hours. Less than 0.01% of patients have a severe reaction, such as anaphylaxis [12]. Finally, based on our experience, which is limited to this single case, it is an accurate tool in localizing high grade gliomas of the spine.
Despite the advantages noted above, the intra-operative use of fluorescein to localize spine tumors does have some potential limitations. The uptake of fluorescein into tumors seems to be dependent on the breakdown of the blood brain barrier (BBB). Diaz et al demonstrated that fluorescein accumulated in the extracellular space as opposed to glioma cells in a malignant glioma mouse model. They also demonstrated that fluorescein correlated with areas of gadolinium enhancement. This suggests that the uptake of fluorescein relies on the breakdown of the BBB, a characteristic seen only in higher-grade tumors. Therefore, its utility may be limited to tumors such as high grade gliomas and intramedullary spinal metastases.
The Blue 400 function was developed for use with 5-ALA. The filters correspond to the fluorescent properties of 5-ALA, with excitation in the 400-410nm wavelength range and display in the 620-710nm wavelength range. This differs from fluorescein, which has a peak excitation at 490 nm and emission between 500-550nm. Therefore, although it delineated the tumour in our particular case, a filter more representative of the properties of fluorescein may be more accurate than using the Blue 400 function. Zeiss has developed a yellow 560 function, which visualizes fluorescent dyes in the wavelength range from 540 to 690nm. This has been used to help resect cerebral metastases in a small case series [13], and may be a better alternative to the blue 400 function in future cases. Further study will be required to determine it's utility.
Conclusion
The intra-operative use of fluorescein sodium to localize high grade intramedullary tumors is accurate, inexpensive, and safe.
References
- Woodard EJ, Leon SP, Moriarty TM, Quinones A, Zamani AA, et al. (2001) Initial experience with intraoperative magnetic resonance imaging in spine surgery. Spine 26(4): 410-417.
- Regelsberger J, Fritzsche E, Langer N, Westphal M (2005) Intraoperative sonography of intra and extramedullary tumors. Ultrasound Med Biol 31(5): 593-598.
- Sosna J, Barth M, Kruskal J, Kane R (2005) Intraoperative sonography for neurosurgery. Journal of Ultrasound Medicine 24(12): 1671-1682.
- Vetrano I, Prada F, Nataloni I, Bene M, Dimeco F, et al. (2015) Discrete or diffuse intramedullary tumor? Contrast-enhanced intraoperative ultrasound in a case of intramedullary cervicothoracic hemangioblastomas mimicking a diffuse infiltrative glioma: technical note and case report. Neurosurgical Focus 39(2): 1-6.
- Stummer W, Stocker S, Wagner S, Stepp H, Fritsch C, et al. (1998) Intraoperative detection of malignant gliomas by 5-aminolevulinic acid-induced porphyrin fluorescence. Neurosurg 42(3): 518-526.
- Stummer W, Novotny A, Stepp H, Goetz C, Bise K, et al. (2000) Fluorescence-guided resection of glioblastoma multiforme by using 5-aminolevulinic acid-induced porphyrins: a prospective study in 52 consecutive patients. Journal of Neurosurg 93(6): 1003-1013.
- Shinoda J, Yano H, Yoshimura S, Okumura A, Kaku Y, et al. (2003) Fluorescence-guided resection of glioblastoma multiforme by using high-dose fluorescein sodium. J Neurosurg 99(3): 597-603.
- Diaz R, Dios R, Hattab E, Burrell K, Rakopoulos P, et al. (2015) A Study of the biodistribution of fluorescein in glioma-infiltrated mouse brain and histopathological correlation of intraoperative findings in high- grade gliomas resected under fluorescein fluorescence guidance. J Neurosurg 122(6): 1360-1369.
- Koc K, Anik I, Cabuk B, Ceylan S (2008) Fluorescein sodium-guided surgery in glioblastoma multiforme: a prospective evaluation. Br J Neurosurg. 22(1): 99-103.
- Lipson B, Yannuzzi L (1989) Complications of intravenous fluorescein injections. Int Ophthalmol Clin 29(3): 200-205.
- Kwan A, Barry C, McAllister I, Constable I (2006) Fluorescein angiography and adverse drug reactions revisited: the Lions Eye experience. Clinical Exp Ophthalmol 34(1): 33-38.
- Fineschi V, Monasterolo G, Rosi R, Turillazzi E (1999) Fatal anaphylactic shock during a fluorescein angiography. Forensic Sci Int 100(1-2): 137142.
- Schebesch K, Hoehne J, Hohenberger C, Proescholdt M, Riemenschneider MJ, et al. (2015) Fluorescein sodium-guided resection of cerebral metastases-experience with the first 30 patients. Acta Neurochirurgica 157(6): 899-904.






























