Overview on the Mechanism of Action of Two Well-known Antibacterial Drugs Based on Enzyme Inhibition
Santarupa Thakurta1* and Bikram Ghosh2
1Assistant Professor, Department of Chemistry, Prabhu Jagatbandhu College, India
2Ex-student, Department of Chemistry, Prabhu Jagatbandhu College, India
Submission: August 28, 2021; Published: September 09, 2021
*Corresponding author: Santarupa Thakurta, Department of Chemistry, Prabhu Jagatbandhu College, India.
How to cite this article: Santarupa T, Bikram G. Overview on the Mechanism of Action of Two Well-known Antibacterial Drugs Based on Enzyme Inhibition. Nov Appro Drug Des Dev. 2021; 6(2): 555682. DOI: 10.19080/NAPDD.2021.06.555682
Abstract
Most of the drugs achieve their therapeutic effects by interacting with specific target enzymes of the humans or infectants. The action of such drugs involves enzyme inhibition which can be reversible or irreversible. In this review, the discovery, structure, mechanisms of action of two important drugs based on enzyme inhibition are discussed. Among them, Penicillins are irreversible inhibitors whereas Sulphonamides are reversible inhibitors.
Keywords: Enzyme inhibitors; Penicillin; Sulphonamides; Drugs
Abbreviations: NAM: N-acetylmuramic acid; NAG: N- acetylglucosamine; SAR: Structure-Activity-Relationship
Introduction
During this pandemic, chemistry shines the brightest light on our path in the form of medicines. Drug is a chemical substance which has a physiological effect for treating diseases when ingested or administered into the body. From the times of Paul Ehrlich, father of modern chemotherapy, understanding the mechanism of action of drugs has been one of the key interests of scientific community. The journey of drug development starts with identifying lead compound, which may act on specific genes or proteins involved in a disease. The analysis of Structure-Activity-Relationship (SAR) enables the determination of the chemical group responsible for evoking a target biological effect in the organism. A drug achieves its therapeutic effects by interacting with specific target. These targets are the important biochemical components of the humans or infectants. Among these drug targets, enzymes are the very basic ones. Enzymes catalyze chemical reactions in the biological mediums. Binding of the substrates to its active site with the help of nonbonding interactions, is the main feature of an enzyme catalyzed reaction [1]. These interactions are balanced so that they can hold the substrates and allow products to leave the binding site, more over an enzyme stabilizes transition state of the reaction by binding it most strongly [2]. Selective inhibition of these enzymes leads to the desired effects of a drug. Inhibition of an enzyme can be achieved in many different ways. Types of inhibitors which play a significant role in drug discovery processes are discussed below [3].
Reversible inhibitors
This type of inhibitors bind to the enzymes by non-covalent interaction, such as H-bonding, salt-bridge, van der Waals interaction and the enzyme inhibition can be reversed if the inhibitor is removed.
Drugs which are designed to be similar to the natural substrate, can fit to the active site more strongly than the substrate itself. It may not undergo any reaction when it is in the active site, but as long as it stays there it blocks access to the natural substrate and prevents the enzymatic reaction. This is known as Competitive inhibition. Uncompetitive inhibitor binds exclusively with enzyme-substrate complex. Non-competitive inhibitor binds at an allosteric site and distorts the active site. As long as the non-competitive inhibitor is bound, the enzyme remains catalytically inactive.
Irreversible inhibitors
Irreversible enzyme inhibitors can form a covalent bond to a key amino acid in the active site and permanently block the action of the target enzyme. These cannot be affected by substrate concentration. Sometimes, the target enzyme commits suicide by activating the drug, so they are also known as suicidal substances.
Based on enzyme inhibition a vast number of drugs have been developed. Therapeutic uses of some drugs based on enzyme inhibition are listed in Table 1.
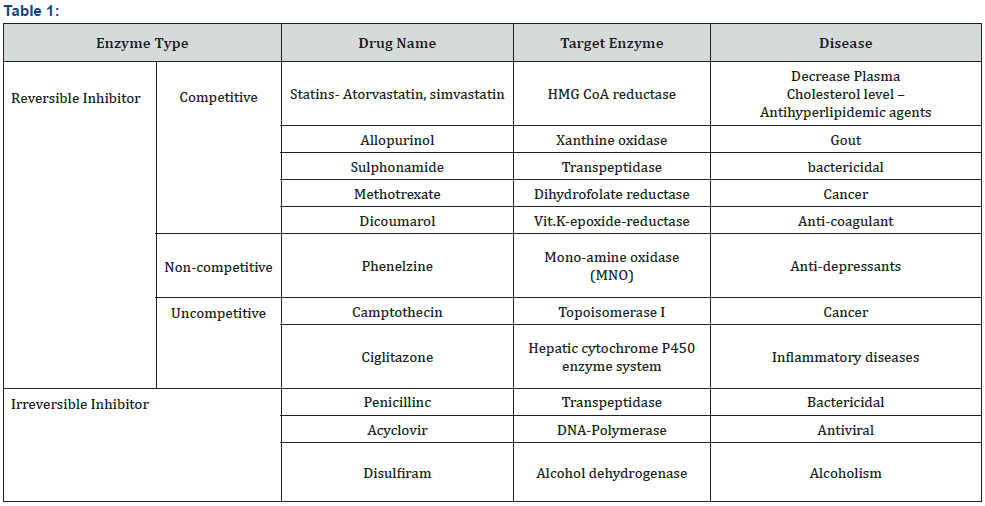
Among this huge variety of drugs, in this article, we are presenting a brief review on two representative drugs: Penicillin as irreversible inhibitor and Sulphonamide as reversible inhibitor. Our objective is to study their structure, function of the target enzyme and mechanism of drug action on that enzyme.
Penicillin
Background
In 1928 Scottish Scientist Alexander Fleming discovered Penicillin accidently [4]. He observed that a blue-green mold of fungus had developed on a staphylococcus culture plate and the fungal colonies killed nearby bacterial colonies. He identified the fungus as a rare species of penicillium notetum [5]. But he could not separate the active compound and concluded that it is too unstable to isolate. In 1938, this problem was solved by Howard Florey and Ernst Chain. They developed the technology for largescale production of the active component produced by the mold. Bio-technological production of Penicillin revolutionized the medicinal field in the 20th century. Penicillin was available in the market as an antimicrobial drug saving the lives of many soldiers during Second World War. The Nobel Prize in Medicine in 1945 was awarded to Alexander Fleming along with Howard Florey and Ernst Chain for their contribution in creating the first massproduced antibiotic.
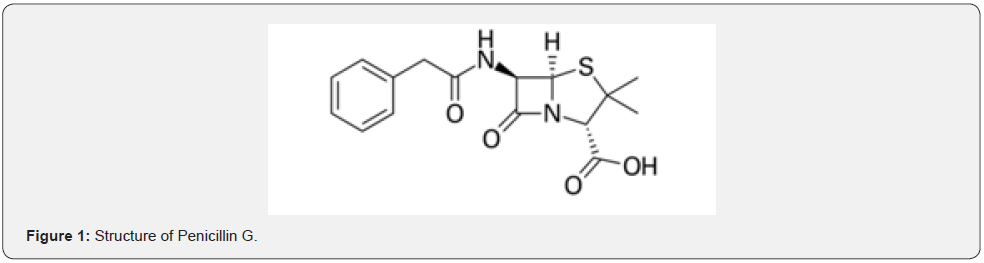
In 1945, Dorothy Hodgkin established the structure of Penicillin using X-ray Crystallography (Figure 1). Penicillins are part of a broader class of antibiotics known as beta-lactam antibiotics. Its structure consists of a fused thiazolidine ring and a β lactum ring (cyclic amide). Penicillins are used to treat certain infections caused by bacteria such as pneumonia and other respiratory tract infections, scarlet fever, and ear, skin, gum, mouth, and throat infections.
Function of target enzyme
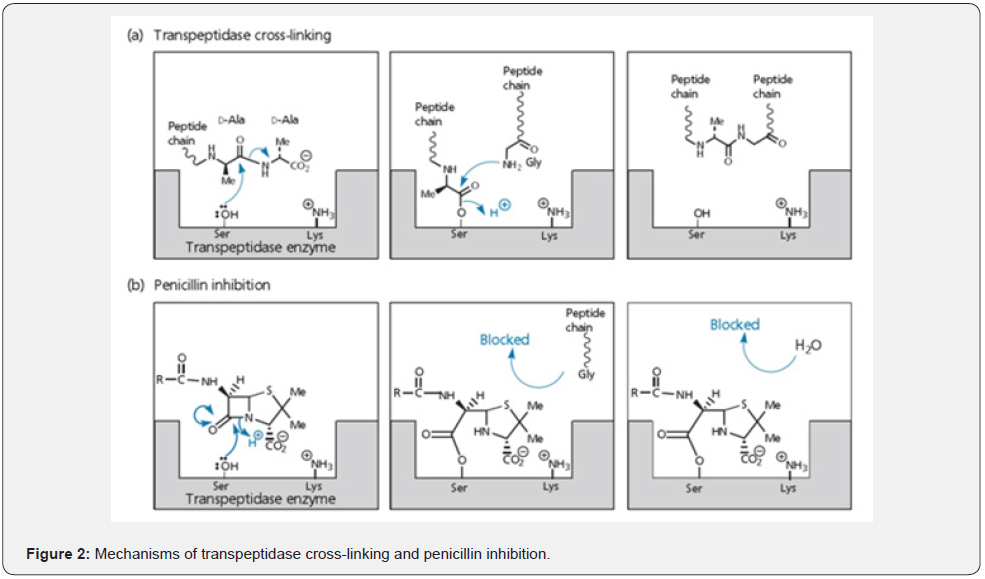
Bacteria have cell wall in order to survive a range of environmental conditions. Without cell wall, water enters to the bacterial cell, lysis takes place. The wall is a peptidoglycan structure, made up of peptide and sugar units. The structure of the wall consists of a parallel series of sugar backbones containing two types of sugar, namely N-acetylmuramic acid (NAM) and Nacetylglucosamine (NAG). Peptide chains are bound to the NAM sugars and both D & L- amino acid present in this chain. In the final stage, the peptide chains are linked together by the displacement of d-alanine from one chain by glycine in another. This crosslinking reaction is catalysed by transpeptidase enzyme [6].
Transpeptidase has serine residue at the active site, which catalyzes the hydrolysis of peptide bonds (Fig. 2). Serine acts as a nucleophile to split the peptide bond between the two d-alanine units on a peptide chain. The terminal alanine departs the active site, leaving the peptide chain bound. The pentaglycyl moiety of another peptide chain now enters the active site and the terminal glycine forms a peptide bond to the alanine group, displacing it from serine and linking the two chains together [7].
Mechanism of drug action
Penicillin works by inhibiting transpeptidase enzyme, which is involved in the last step of cell wall synthesis. Penicillin is the structural analogue of the d-Ala-d-Ala moiety. So, it binds to the enzyme. Once bound, penicillin is subjected to nucleophilic attack by serine (Figure 2). The enzyme can attack the β-lactam ring of penicillin and cleave it in the same way as it did with the peptide bond [8]. Penicillin is cyclic so the molecule is not split in two and nothing leaves the active site. Subsequent hydrolysis of the ester group linking the penicillin to the active site does not take place either, as the penicillin structure blocks access to the pentaglycine chain or water. In this way Penicillin binds with transpeptidase enzyme and inhibits it irreversibly. Since Penicillin is able to kill the bacterial cell, it is classed as bactericidal. Human cell remains unaffected by penicillin because human cells do not contain peptidoglycan.
Penicillin resistance
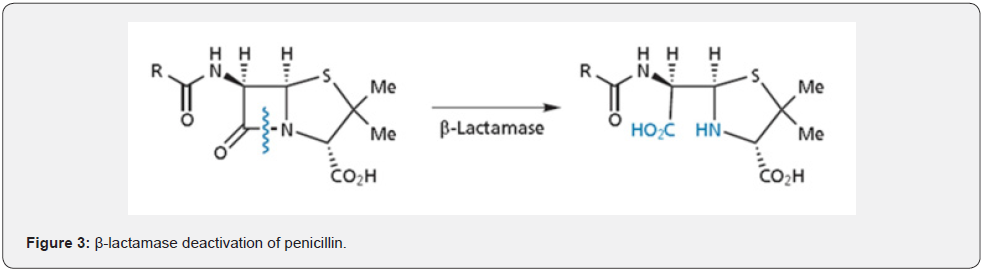
The overuse and misuse of Penicillin have led to a major problem of antibiotic resistance. Certain Bacteria gain penicillin resistance by the presence of β-lactamase enzymes which are the mutated form of transpeptidases (Figure 3). They can hydrolyse β-lactam ring of penicillin, thus making it inactive [9]. Also, some gram-negative bacteria produce excess amount of transpeptidase enzyme and Penicillin is incapable of inactivating all the enzyme molecules present.
Successful derivatives & broad spectrum penicillins
Those penicillins having useful activity against both Grampositive and Gram-negative bacteria are known as broad-spectrum antibiotics. Examples are Ampicillin, Amoxicillin, Carbenicillin, Ureidopenicillins. These are the first line of defence of any bacterial infection [10]. Many successful derivatives of penicillin are developed by changing the ‘R’-group (shown in Figure 3). Some of them are listed in Table 2.
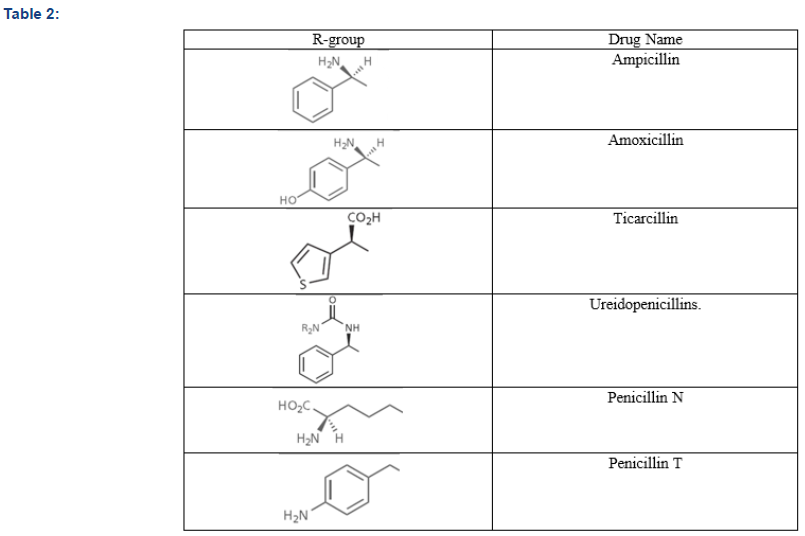
Sulphonamide
Background
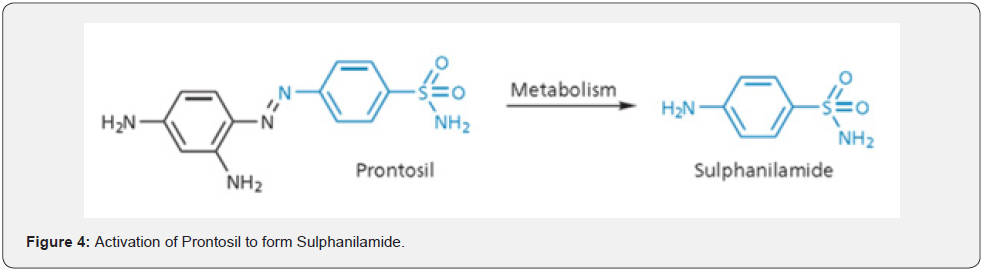
In 1935, German bacteriologist and pathologist Gerhard Domagk discovered that a red dye Prontosil had in vivo antibacterial properties against fatal streptococci infections in humans. But strangely no antibacterial effect was observed in vitro [11]. This remained a mystery until French scientist Ernest Fourneau discovered that prontosil was metabolized by bacteria present in the small intestine of the test animal to give a product called Sulphanilamide (Figure 4). It was this compound which was the true antibacterial agent [12]. Domagk got Nobel Prize in 1939 for development of sulfonamides such as Prontosil. Sulphonamides are a group of synthetic medicines that contain the sulphonamide chemical group (Figure 5). They may also be called sulpha drugs. They are used to treat urinary tract infections, infections of mucous membranes, gut infections, etc.
Function of target enzyme
Bacteria require dihydropteroate for the synthesis of tetrahydrofolate (Coenzyme-F), which is necessary for DNA synthesis of bacteria (Figure 6). Para-aminobenzoic acid (PABA) first binds with the enzyme dihydropteroate synthetase and then enzyme-substrate complex reacts with 6-hydroxymethylpterin diphosphate to produce dihydropteroate, which reacts with Lglutamic acid to form dihydrofolate. Dihydrofolate again binds with dihydrofolate reductase enzyme to produce tetrahydrofolate (Coenzyme F). Now coenzyme-F is used for DNA synthesis.
Mechanism of drug action
Sulphonamides are the example of competitive enzyme inhibitors and structural analogues of para-aminobenzoic acid (PABA) [13]. Sulphonamide binds with dihydropteroate enzyme instead of PABA and inhibits it reversibly (Figure 6). So, the resulting enzyme-substrate complex does not undergo further enzymatic reaction. In this way, Sulphonamide blocks the biosynthesis of tetrahydrofolate which is required for DNA synthesis of bacteria. Now, bacteria cannot grow, and our human immunity system gets sufficient time to kill the bacteria. Sulphonamide does not actively kill the bacteria, only prevent the cell growing and multiplying, hence it is classed as bacteriostatic.
These drugs are further classified as antimetabolites since they prevent the cell metabolism of bacterial cell.
Selectivity of sulphonamide drug
Tetrahydrofolate is also necessary for human cell. But sulphonamide is not toxic for human body. This is because human cell can synthesis tetrahydrofolate from folic acid - a totally different pathway. This folic acid is obtained from the diet as a vitamin and then transport to human cell by transport protein. Bacterial cell is unable to follow the same mechanism of human cell due to lack of transport protein. The success of sulphonamides is based on the utilization of metabolic differences between mammalian and bacterial cells.
Sequential blocking
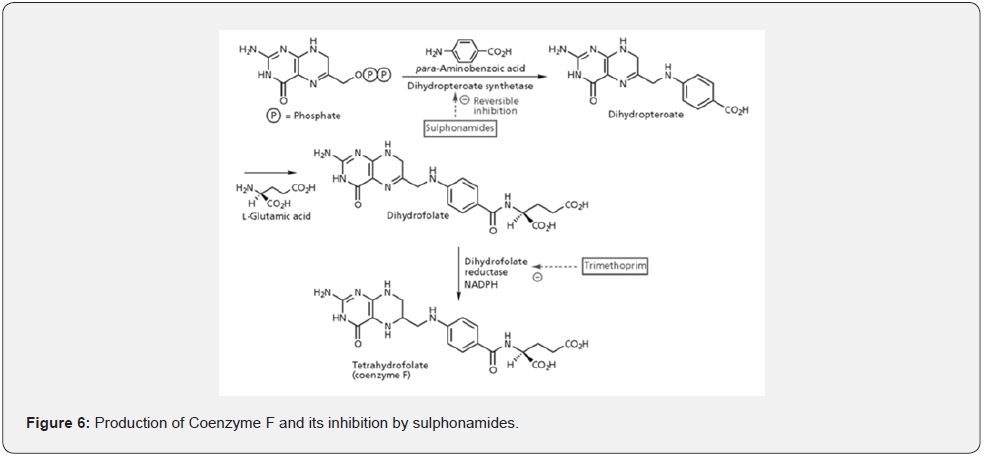
As sulphonamides are reversible inhibitors, their efficiency depends on the substrate concentration but administration of large amounts these drugs produced toxic effects on the patients. This drawback is solved by sequential blocking of two enzymes required in a biosynthetic pathway [14]. For example trimethoprim which blocks Dihydrofolate reductase, is used with sulphonamides to produce antibiotic effect of higher strength (Figure 6).
Successful derivatives
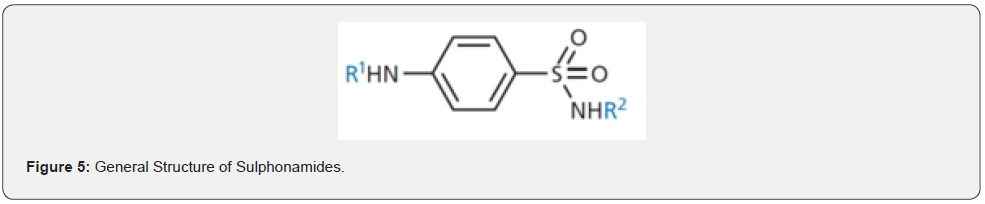
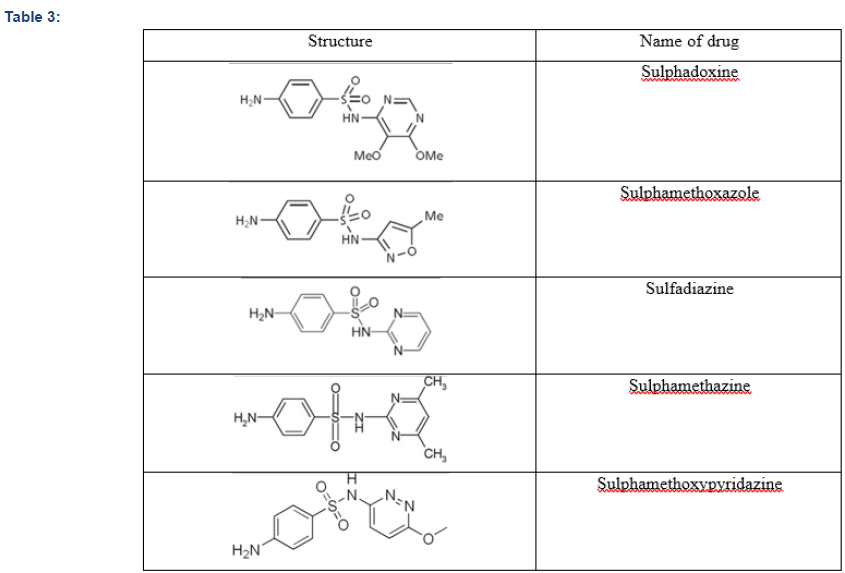
Sulfanilamide itself has been superseded in medicines by its derivatives. Structure-activity relationships reveal that the fruitful derivatives have heterocyclic ring as substituent in place of one hydrogen of sulphonamide group (-SO2NHR2, Figure 5). Some of them are listed in Table 3.
Conclusion
Enzymes remain prime focus for current drug design because altering enzyme activity has immediate and defined effects. In this review, we have discussed discovery, structure, mechanisms of action of two important drugs based on enzyme inhibition. Penicillin which is irreversible inhibitor of transpeptidase enzyme can kill the bacterial cell. On the other hand Sulphonamides which are reversible inhibitors prevent the cell metabolism of bacterial cell. Profound understanding of the target enzyme function and course of an enzyme-catalysed reaction can help to conceptualize different types of inhibitors. Currently, 47% of total drugs in the world are based on enzyme inhibition. With the help of computer modelling and spectroscopic tools the drug design and development will reach new heights at near future.
References
- D Nelson (2013) Lehninger principles of biochemistry, New York: W.H. Freeman and Company.
- TL Amyes, JP Richard (2013) Specificity in Transition State Binding: The Pauling Model Revisited. Biochemistry 52: 2021-2035.
- J Berg (2002) Biochemistry, New York: W.H. Freeman.
- Fleming (1929) On the Antibacterial Action of Cultures of a Penicillium, with Special Reference to Their Use in the Isolation of B. influenzae. British Journal of Experimental Pathology 10: 226-236
- JW Bennett, KT Chung (2001) Alexander Fleming and the discovery of penicillin. Advances in Applied Microbiology 49: 163-184.
- MJ Osborn (1969) Structure and Biosynthesis of the Bacterial Cell Wall. Annual Review of Biochemistry 38: 501-538.
- GPatrick (2018) An introduction to medicinal chemistry, Oxford, United: Oxford University Press.
- RR Yocum, JR Rasmussen, JL Strominger (1980) The mechanism of action of penicillin. Penicillin acylates the active site of Bacillus stearothermophilus D-alanine carboxypeptidase. Journal of Biological Chemistry 255: 3977-3986.
- JDavies, D Davies (2010) Origins and Evolution of Antibiotic Resistance. Microbiology and Molecular Biology Reviews 74(3): 417-433.
- EL Miller (2002) The Penicillins: A Review and Update. Journal of Midwifery & Women Health 47: 426-434.
- G Domagk (1935) Ein Beitrag zur Chemotherapie der bakteriellen Infektionen. DMW - Deutsche Medizinische Wochenschrift 61: 250-253.
- The First Miracle Drugs: How the Sulfa Drugs Transformed Medicine, OXFORD UNIV PR, 2006.
- RJ Henry (1943) The Mode of Action of Sulfonamides. bacteriology Reviews 7: 175-262.
- SR Bushby (1975) Synergy of trimethoprim-sulfamethoxazole. Can Med Assoc J 14: 63-66.






























