Stroke: Unifying Mechanism Involving Antioxidant Therapy, Reactive Oxygen Species, and Oxidative Stress
Peter Kovacic1* and Wil Weston2
1 Department of Chemistry and Biochemistry, San Diego State University, San Diego, CA, USA
2 Library and Information Access, San Diego State University, San Diego, CA, USA
Submission: December 01, 2018; Published: December 14, 2018
*Corresponding author: Peter Kovacic, Department of Chemistry and Biochemistry, San Diego State University, San Diego, CA, USA
How to cite this article: Peter Kovacic, Wil Weston. Stroke: Unifying Mechanism Involving Antioxidant Therapy, Reactive Oxygen Species, and Oxidative Stress . Nov Appro Drug Des Dev 2018; 4(4): 555641. DOI: 10.19080/NAPDD.2018.04.555641
Abstract
Reactive oxygen species (ROS) and oxidative stress (OS) play roles in stroke, as also in Alzheimer’s (AD), Parkinson’s (PD) disease and Schizophrenia (SCZ). Various sources, including oxidases, serve as generators of ROS-OS, such as mitochondria, NADPH, cytochromes P450, monoamines, ET metal complexes, G72 gene, and microglia. Many novel examples of antioxidants (AOs) exert a positive influence on the harmful effects, namely through a unifying mechanism based on ET-ROS-OS-AO. Drugs for treatment of stroke are discussed in relation to the unifying theme including phenolic and phenolic ethers.
Keywords: Stroke; Ischemia; Radicals; Oxidative Stress; Reactive Oxygen Species; Antioxidants
Abbrevations: ET: Electron Transfer; ROS: Reactive Oxygen Species; OS: Oxidative Stress; AO: Antioxidant.
Introduction
Symptoms
Stroke is a leading cause of death and disability [1] and ischemic stroke is the second leading cause of death worldwide [2]. A stroke is a brain injury that takes place due to disruption of blood supply for various reasons [3]. This event is classified into these categories, hemorrhage (bleeding in diverse parts of the brain, and thrombotic (clot formation in the artery due to atherosclerosis, the most common type.), embolic (a blood clot or other debris traveling to the brain causing harm). Symptoms, which can occur in different areas of the brain, are as follows: dizziness or confusion, numbness, visual disturbance or loss, difficulty walking, slurred speech, seizures, stupor, coma, and irregular breathing [4]. Stroke may be preceded by transient, ischemic attacks. Risk factors include smoking, diabetes, high blood pressure, heart disease, and genetics.
Unifying Mechanism
Stroke fits into the unifying mechanism which has been widely applied, previously in an article involving electron transfer (ET), reactive oxygen species (ROS) and oxidative stress (OS) [5]. This unifying mechanism argues that the preponderance of bioactive substances, usually as the metabolites, incorporate ET functionalities. We believe these ET-metabolites play an important role in physiological responses. The main group includes quinones (or phenolic precursors), metal complexes (or complexes), aromatic nitro compounds (or reduced hydroxylamine and nitroso derivatives), and conjugated imines (or iminium species). Resultant redox cycling is illustrated in Figure 1. In vivo redox cycling with oxygen can occur, giving rise to OS through generation of ROS, such as hydrogen peroxide, hydroperoxides, alkyl peroxides, and diverse radicals (hydroxyl, alkoxyl, hydroperoxyl, and superoxide) (Figure 1). Cellular and mitochondrial enzymes can also perform catalytically in the reduction of O2.
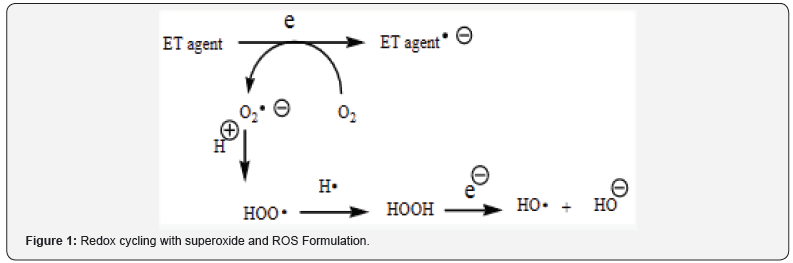
In some cases, ET results in involvement with normal electrical effects (e.g. neurochemistry). Generally, active entities possessing ET groups display reduction potentials in the physiologically responsive range. Hence, ET in vivo can occur resulting in production of ROS, which can be beneficial in cell signaling at low concentrations but produce toxic results at high levels. Electron donors consist of phenols, N-heterocycles or disulfides in proteins, which produce relatively stable radical cations. ET, ROS and OS have been increasingly implicated in the mode of action of drugs and toxins, e.g. anticancer drugs [6], carcinogens [7], cardiovascular toxins [8], toxins [9], ototoxins [10] and various other categories [11].
In addition to the above, there is a plethora of experimental evidence supporting the theoretical framework. This evidence includes generation of the common ROS, lipid peroxidation, degradation products of oxidation, depletion of AOs, effect of exogenous AOs, and DNA oxidation and cleavage products, as well as electrochemical data [5]. This comprehensive, unifying mechanism is consistent with the frequent observation that many ET substances display a variety of activities (e.g. multiple-drug properties), as well as toxic effects. It is important to recognize that mode of action in the biodomain is often involved with many physiological actions and is multifaceted. In addition to ET-ROSOS in relation to mechanism, much attention in the literature is paid to AO action entailing physiological effects.
ROS-OS
ROS can be beneficial, but at high levels toxic effects often predominate. There are various sources for these species [5]. NAPDH oxidase is an important producer of the ROS in various organs. The G72 gene increased radical generation in cells. The gene acts as an activator of oxidase. ROS generated by NO synthase have been implicated in an array of harmful behaviors. Mitochondria provide another source of ROS-OS which appears to contribute to aging. Leakage of electrons occurs in the ET chain which react with oxygen to produce superoxide, a precursor of another ROS. Other examples of ROS producers are cytochrome P450, metal complexes, monoamine oxidase and microglia
There is literature for specific sources of ROS-OS in stroke, which is rare in brain illness. A study found systemic oxidative damage to lipids and proteins at baseline in stroke [12]. Malondialdehyde, an OS marker, concentrations correlated with stroke severity and was associated with hemorrhage complications. ROS generated from reperfusion injury could be a cause of brain injury. Canola oil shortens the life span of strokeprone rats [13]. The oil reduced the AO activities of SOD, GSH peroxides and catalase and produced a change in oxidative status.
Tetrahydrocannabinol (THC) increases OS and induces cerebral mitochondrial dysfunction in cannabis-related stroke [14]. THC increased H2O2 production and mitochondrial free radical leaks, a source of ROS. A relevant report on THC can be found in the recent literature [15]. This stroke article is part of a series on brain illnesses, including Alzheimer’s disease (AD) [16], Parkinson’s disease (PD) [17], Structure-activity relationship (SAR) for AD and PD [18] Schizophrenia [5], Depression [19], Multiple Sclerosis and Amyotrophic Lateral Sclerosis [20], Dementia [21], and Huntington’s disease [22].
Therapy
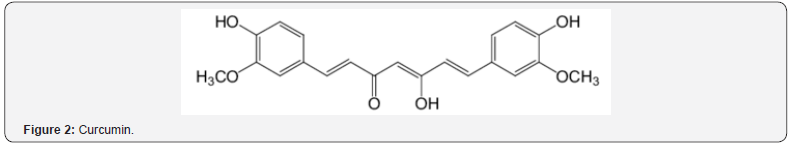
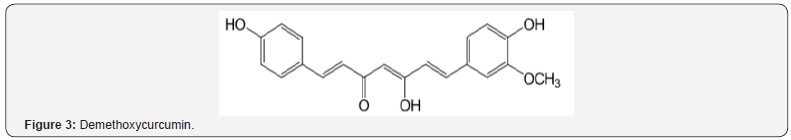
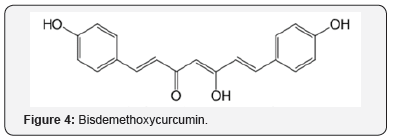
Curcumin (Figure 2) and derivatives of curcumin (Figures 3 & 4) were investigated in experimental stroke [23]. Phenolic ethers (see Figure 2) can undergo cleavage to phenols. Beneficial AO action was observed in all cases with the methoxy types being significantly better. Prior work deals with demethylation to AO phenolics [17]. Another article reports the neuroprotective effect of curcumin in a stroke model [24]. There was protection against ischemia via AO activity and neuronal apoptosis. Other studies of brain illnesses deal with phenolics and phenolic ethers [16,17].
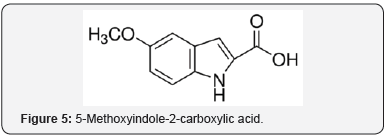
5-Methoxyindole-2-carboxylic acid (MICA) (Figure 5) provides neuroprotection against stroke [25]. There was a decrease in OS in MICA treated rats based on decrease in H2O2 and lipid peroxidation. The mechanism likely involves AO protection, attenuation of OS, and maintenance of mitochondrial function. The mitochondrial aspect is related to another report [9]. Nobiletin (Figure 6) elicits protection against ischemic stroke [26]. There are accompanying AO and anti-inflammatory responses.

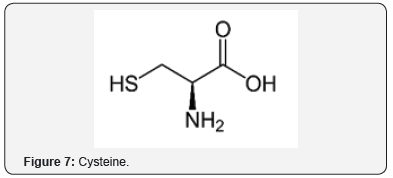

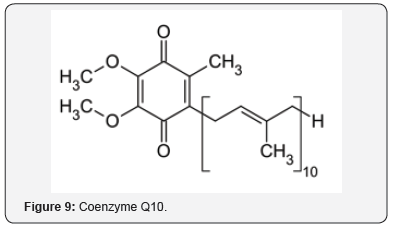
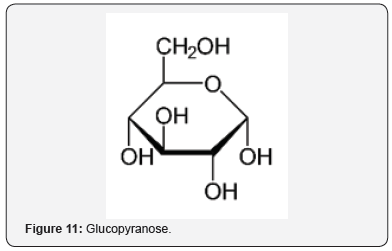
The neuroprotective effects were studied of an AO mixture, Twendee X, composed of ingredients, such as cysteine (Figure 7), ascorbic acid (Figure 8), and coenzyme Q10 (Figure 9) [27]. OS and inflammation are important factors in ischemic stroke. The neuroprotective effects were demonstrated in AO and anti-inflammatory pathways [28]. Ginsenoside Rd (Figure 10), an ingredient in ginseng, can improve stroke outcome [29]. Ginsenoside Rd also attenuates redox imbalance, along with enhancing AO activities. There are other examples of polyols acting as AOs, such as sugars like glucopyranose (Figure 11), which possess significant AO capacity [22].

Astaxanthin (Figures 1 & 2), a natural AO carotenoid, reduces cerebral injury in stroke [30]. Neuroprotection is provided via suppression of ROS and activation of AO defenses. Recovery was increased through promotion of AO defenses. There is inhibition of apoptosis and promotion of neural regeneration. Multiple mechanisms are involved. A related AO is amphotericin B [31].

Mitochondria damage appears to be involved in brain stroke [32]. Diphenyl diselenide (Figures 12 & 13) reduced mitochondrial damage in a stroke model. The neuroprotective action may be due to the maintenance of redox balance. The initial injury is attributed to an increase in ROS. There is a related article by Mancini, et al. (2014), in which the compound, diphenyl diselenide was determined to mimic endogenous antioxidant enzymes or be metabolized by thioredoxin reductase to form selenol intermediate, which can copy the function of the antioxidant selenoenzymes [33].

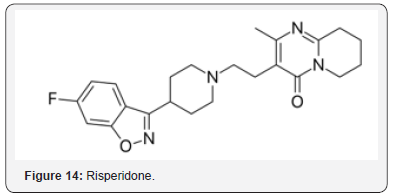
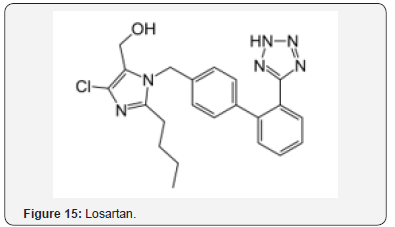
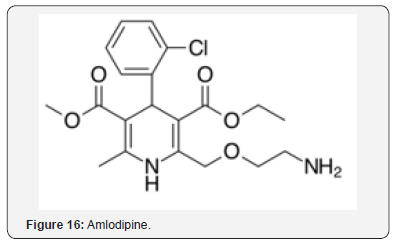
Risperidone (Figure 14), an antipsychotic drug, displays neuroprotective effects in ischemic stroke [34]. Significant protection was observed against neuronal death. The neuroprotective effect is attributed in part to maintenance of AOs. Losartan (Figure 15) and amlodipine (Figure 16) were studied for beneficial effects on stroke prone rats [35]. The two agents upregulated expression of superoxide dismutase (SOD) and decreased apoptosis. Amlodipine was more effective in decreasing apoptosis, which may be related to the AO properties of the agent in an OS environment.
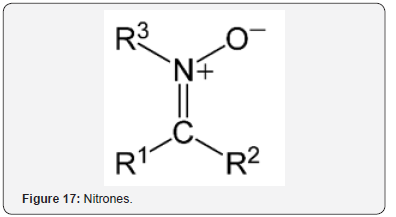
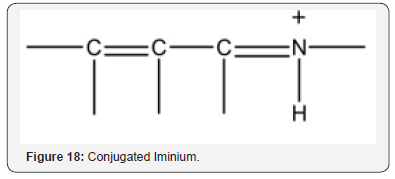
Nitrones (Figure 17) are potent agents for stroke treatment based on AO, anti-inflammatory, and neuroprotective properties [36]. In vitro evaluation of brain blood barrier (BBB) penetration of select nitrones showed that all of them crossed. Nitrones are electrochemically analogous to iminium (Figure 18) as noted in the introduction. Manganese superoxide dismutase (MnSOD) is an important AO enzyme in the central nervous system [37]. MnSOD is an important therapeutic agent in ischemic stroke by alleviating OS and apoptosis Other preventative effects involve AO.
The protein Ubiquilin-1 protects mice from OS and ischemic stroke [38]. The stroke caused neuronal injury which was alleviated. The protein incorporates AO features. OS was appreciably weakened. Evidence points to nicotinamide adenine dinucleotide phosphate (NADPH) as a major source based on generation of OS. Neuroprotection could be provided through stress therapy based on AO action [1]. NADPH oxidase type 4 (NOX4) is a major source of OS in acute stroke [2]. Application of VAS2870, an inhibitor of the oxidase, was remarkably productive. NOX4 represents a novel class of drug target for stroke therapy.
The following is a brief collection of summaries of other articles that speak to AO, ROS, OS in association with stroke. AO and antiapoptotic approaches have been examined in neuroprotection of stroke. In 2013, Rodrigo et. al noted that ROS has been implicated in stroke and suggests novel AO approaches for treatment of ischemic stroke involving OS and pathophysiology [39]. A 2018 report discusses reactive oxygen species – sensitive NO synthase inhibitor, an agent for stroke treatment, involving AO/NO donor [40]. In 2017, a report suggested that AO enzyme therapy would be useful for ischemic stroke [41]. Transglutaminase, a calcium dependent enzyme, was also involved as a therapeutic target for OS and excitotoxicity in stroke [42]. In another study, AO therapy was used on neurotrophins after stroke [43]. Earlier stroke research, in 2011, was performed involving AO therapy and thrombolysis, which suggested that co-administration of AO drugs could augment the value of thrombolytic therapy [44]. A study by Yi et. al demonstrated AO protection by mitochondrial HMG-CoA synthase contributed to healing in stroke prone rats [45]. In a 2010 study, AO therapy involving vascular targeting was carried out with stroke patients [46]. Later, in 2012, Brea et al. found OS markers are linked to vascular recurrence [47].
A 2013 report deals with the effects of OS on vascular reactivity of stroke prone rats [48]. A 2010 study reported on the effects of inflammatory processes on the brain of stroke rats, finding that they could significantly increase survival through the AO and anti-inflammatory effects of their treatment [49]. A receptor reduces ischemic stroke through reduction of OS inflammation [50]. Reception agonist treatment ameliorates OS and neuroinflammation in ischemic stroke [51]. Traditional medicines have also been examined. In a 2016 study, Korean traditional medicine provided neuroprotection for stroke through AO/apoptotic pathways [52]. Additionally, green tea prevents OS in stroke models and may have a beneficial impact on cognitive function after stroke [53]. A 2016 study found that intervention with AOs may have protecting effects in severe heat stroke [54]. In 2011 another study examined the relationship between OS, autoimmunity, and heart risk in Africans with HIV/AIDS [55]. They found that these clustered factors along with OS may explain the high risk of stroke in HIV/AIDS patients.
References
- Radermacher KA, Wingler K, Langhauser F, Altenhöfer S, Kleikers P, et al. (2013) Neuroprotection after stroke by targeting NOX4 as a source of oxidative stress. Antioxid Redox Signal 18(12): 1418-1427.
- Kleinschnitz C, Grund H, Wingler K, Armitage ME, Jones E, et al. (2010) Post-stroke inhibition of induced NADPH oxidase type 4 prevents oxidative stress and neurodegeneration. PLoS Biol 8(9)
- Lewine HE (2017) Stroke overview. In Harvard Medical School (edn.) Health reference series: Harvard Medical School health topics A-Z. Harvard Health Publications: MA, Boston.
- Robinson R, Odle TG, Frey RJ, Alic M (2015) Stroke. In Gale (edn), The Gale encyclopedia of senior health: a guide for seniors and their caregivers (2nd edn). Farmington, MI: Gale.
- Kovacic P, Weston W (2017) Cause and treatment of schizophrenia: Electron transfer, reactive oxygen species, oxidative stress, antioxidants, and unifying mechanism. Chron of Pharm Sci 1(6): 332-340.
- Kovacic P, Osuna JA (2000) Mechanisms of anticancer agents: Emphasis on oxidative stress and electron transfer. Curr Pharmaceut Des 6: 277- 309.
- Kovacic P Jacintho JD (2001) Reproductive toxins. Pervasive theme of oxidative stress and electron transfer. Curr Med Chem 8: 863-892.
- Kovacic P, Thurn LA (2005) Cardiovascular toxicity from the perspective of oxidative stress, electron transfer, and prevention by antioxidants. Curr Vasc Pharmacol 3: 107-117.
- Kovacic P, Pozos RS, Somanathan R, Shangari N, O’Brien PJ (2005) Mechanism of mitochondrial uncouplers, inhibitors, and toxins: Focus on electron transfer, free radicals, and structure-activity relationships. Curr Med Chem 12(22): 2601-2623.
- Kovacic P, Somanathan R (2008) Ototoxicity and noise trauma: Electron transfer, reactive oxygen species, cell signaling, electrical effects, and protection by antioxidants: Practical medical aspects Med Hypotheses 70: 914-923.
- Halliwell B, Gutteridge J (1999) Free Radicals in Biology and Medicine (3rd edn) Oxford University Press, Clarendon Press: New York, USA.
- Domínguez C, Delgado P, Vilches A, Martín Gallán P, Ribó M, et al. (2010) Oxidative stress after thrombolysis-induced reperfusion in human stroke. Stroke 41(4): 653-660.
- Papazzo A, Conlan XA, Lexis L, Lewandowski PA (2011) Differential effects of dietary canola and soybean oil intake on oxidative stress in stroke-prone spontaneously hypertensive rats. Lipids Health Dis 10: 98.
- Wolff V, Schlagowski AI, Rouyer O, Charles AL, Singh F, et al. (2015) Tetrahydrocannabinol induces brain mitochondrial respiratory chain dysfunction and increases oxidative stress: a potential mechanism involved in cannabis-related stroke. Biomed Res Int. 2015; 2015: 323706.
- Kovacic P, Somanathan R (2014) Cannabinoids (CBD, CBDHQ and THC): Metabolism, physiological effects, electron transfer, reactive oxygen species and medical use. Nat Prod J 4(1): 47-53.
- Kovacic P, Weston W (2017). Phenolic antioxidants as drugs for Alzheimer’s disease: Oxidative stress and selectivity. NAPDD 2(5).
- Kovacic P, Weston W (2017) Treatment of Parkinson’s disease with phenolic antioxidant drugs: Oxidative stress, reactive oxygen species and selectivity. Chron Pharm Sci 1(4): 193-198.
- Kovacic P, Weston W (2017) Novel structure-activity relationship and quantitative data for phenolic drugs involving Alzheimer’s and Parkinson’s disease: Antioxidants, oxidative stress, and selectivity. Chron Pharm Sci 1(5): 299-306.
- Kovacic P, Weston W (2018) Brain depression – Unifying mechanism involving antioxidant therapy: Reactive oxygen species, oxidative stress, and phenolics. Chron Pharm Sci 2(3): 545-553.
- Kovacic P, Weston W (2018) Unifying mechanism for Multiple Sclerosis and Amyotrophic Lateral Sclerosis: Reactive oxygen species, oxidative stress, and antioxidants. J Biopharm and Therap Chall 1(1): 1-8.
- Kovacic P, Weston W (2018) Dementia – Unifying mechanism involving antioxidant therapy: Reactive oxygen species, oxidative stress, and Phenolics. Chron Pharm Sci 2(6): 710-717.
- Kovacic P, Weston W (2018) Huntington’s disease-Unifying mechanism involving antioxidant therapy: Reactive oxygen species, oxidative stress, and Phenolics. Clin Biotech and Microbio 2(5): 485-494.
- Ahmad N, Umar S, Ashafaq M, Akhtar M, Iqbal Z, et al. (2013) A comparative study of PNIPAM nanoparticles of curcumin, demethoxycurcumin, and bisdemethoxycurcumin and their effects on oxidative stress markers in experimental stroke. Protoplasma 250(6): 1327-1338.
- Altinay S, Cabalar M, Isler C, Yildirim F, Celik DS, et al. (2017) Is chronic curcumin supplementation neuroprotective against ischemia for antioxidant activity, neurological deficit, or neuronal apoptosis in an experimental stroke model? Turk Neurosurg 27(4): 537-545.
- Wu J, Li R, Li W, Ren M, Thangthaeng N, et al. (2017) Administration of 5-methoxyindole-2-carboxylic acid that potentially targets mitochondrial dihydrolipoamide dehydrogenase confers cerebral preconditioning against ischemic stroke injury. Free Radic Biol Med 113: 244-254.
- Zhang L, Zhang X, Zhang C, Bai X, Zhang J, et al. (2016) Nobiletin promotes antioxidant and anti-inflammatory responses and elicits protection against ischemic stroke in vivo. Brain Res 1636: 130-141.
- Kusaki M, Ohta Y, Inufusa H, Yamashita T, Morihara R, et al. (2017) Neuroprotective effects of a novel antioxidant mixture Twendee X in mouse stroke model. J Stroke Cerebrovasc Dis.; 26(6): 1191-1196.
- Kovacic P, Somanathan R (2014) Inflammation and Anti-Inflammatory Agents – Reactive Oxygen Species and Toxicity. In: Laher I (edn) Systems Biology of Free Radicals and Antioxidants. Springer, Berlin, Heidelberg.
- Ye R, Kong X, Yang Q, Zhang Y, Han J, et al. (2011) Ginsenoside Rd attenuates redox imbalance and improves stroke outcome after focal cerebral ischemia in aged mice. Neuropharmacology 61(4): 815-824.
- Pan L, Zhou Y, Li XF, Wan QJ, Yu LH (2017) Preventive treatment of astaxanthin provides neuroprotection through suppression of reactive oxygen species and activation of antioxidant defense pathway after stroke in rats. Brain Res Bull 130: 211-220.
- Kovacic P, Cooksy A (2012) Novel, unifying mechanism for amphotericin B and other polyene drugs: electron affinity, radicals, electron transfer, autoxidation, toxicity, and antifungal action. Med Chem Comm 3: 274- 280.
- Dobrachinski F, da Silva MH, Tassi CL, de Carvalho NR, Dias GR (2014) Neuroprotective effect of diphenyl diselenide in an experimental stroke model: maintenance of redox system in mitochondria of brain regions. Neurotox Res 26(4): 317-330
- Mancini G, Raniel Straliotto M, da Rocha JB, de Bem AF (2014) Diphenyl diselenide improves the antioxidant response via activation of the Nrf- 2 pathway in macrophage cells. Free Radic Biol Med 75 Suppl 1: S40.
- Yan BC, Park JH, Ahn JH, Kim IH, Park OK, et al. (2014) Neuroprotection of posttreatment with risperidone, an atypical antipsychotic drug, in rat and gerbil models of ischemic stroke and the maintenance of antioxidants in a gerbil model of ischemic stroke. J Neurosci Res.; 92(6): 795-807.
- He DH, Zhang LM, Ning RB, Wang HJ, Xu CS, et al. (2013) Differential effects of antihypertensive treatments on apoptosis, oxidative stress, and expression of angiotensin receptors in the cerebral cortex from the onset of prehypertension and hypertension in stroke-prone spontaneous hypertensive rats. Neuroreport 24(16): 911-917.
- Chioua M, Sucunza D, Soriano E, Hadjipavlou Litina D, Alcázar A, Ayuso I, et al. (2012) Α-aryl-N-alkyl nitrones, as potential agents for stroke treatment: synthesis, theoretical calculations, antioxidant, antiinflammatory, neuroprotective, and brain-blood barrier permeability properties. J Med Chem 55(1): 153-168.
- Huang HF, Guo F, Cao YZ, Shi W, Xia Q (2012) Neuroprotection by manganese superoxide dismutase (MnSOD) mimics: antioxidant effect and oxidative stress regulation in acute experimental stroke. CNS Neurosci Ther 18(10): 811-818.
- Liu Y, Lü L, Hettinger CL, Dong G, Zhang D, et al. (2014) Ubiquilin-1 protects cells from oxidative stress and ischemic stroke caused tissue injury in mice. J Neurosci 34(8): 2813-2821.
- Rodrigo R, Fernández Gajardo R, Gutiérrez R, Matamala JM, Carrasco R, et al. (2013) Oxidative stress and pathophysiology of ischemic stroke: novel therapeutic opportunities. CNS Neurol Disord Drug Targets 12(5): 698-714.
- Nash KM, Schiefer IT, Shah ZA (2018) Development of a reactive oxygen species-sensitive nitric oxide synthase inhibitor for the treatment of ischemic stroke. Free Radic Biol Med 115: 395-404.
- Davis SM, Pennypacker KR (2017) Targeting antioxidant enzyme expression as a therapeutic strategy for ischemic stroke. Neurochem Int 107: 23-32.
- Basso M, Ratan RR (2013) Transglutaminase is a therapeutic target for oxidative stress, excitotoxicity and stroke: a new epigenetic kid on the CNS block. J Cereb Blood Flow Metab 33(6): 809-818.
- Karakulova YV, Selyanina NV, Zhelnin AV, Filimonova TA, Cepilov SV (2016) Effect of antioxidant therapy on neurotrophins and processes of rehabilitation after stroke. Zh Nevrol Psikhiatr Im S S Korsakova.; 116(8): 36-39.
- Amaro S, Chamorro Á (2011) Translational stroke research of the combination of thrombolysis and antioxidant therapy. Stroke 42(5): 1495-1499.
- Yi W, Fu P, Fan Z, Aso H, Tian C, et al. (2010) Mitochondrial HMG-CoA synthase partially contributes to antioxidant protection in the kidney of stroke-prone spontaneously hypertensive rats. Nutrition 26(11-12): 1176-1180.
- Greig JA, Shirley R, Graham D, Denby L, Dominiczak AF, et al. (2010) Vascular-targeting antioxidant therapy in a model of hypertension and stroke. J Cardiovasc Pharmacol 56(6): 642-650.
- Brea D, Roquer J, Serena J, Segura T, Castillo J (2012) Oxidative stress markers are associated to vascular recurrence in non-cardioembolic stroke patients non-treated with statins. BMC Neurol 12: 65.
- Takemori K, Tahara A, Murakami T, Kometani T (2013) Effects of oxidative stress on vascular reactivity in the offspring of proteinrestricted stroke-prone spontaneously hypertensive rats. Biosci Biotechnol Biochem 77(8): 1689-1693.
- Gelosa P, Banfi C, Gianella A, Brioschi M, Pignieri A, et al. (2010) Peroxisome proliferator-activated receptor {alpha} agonism prevents renal damage and the oxidative stress and inflammatory processes affecting the brains of stroke-prone rats. J Pharmacol Exp Ther 335(2): 324-331.
- Han Z, Shen F, He Y, Degos V, Camus M, et al. (2014) Activation of α-7 nicotinic acetylcholine receptor reduces ischemic stroke injury through reduction of pro-inflammatory macrophages and oxidative stress. PLoS One 9(8): e105711.
- Han Z, Li L, Wang L, Degos V, Maze M, et al. (2014) Alpha-7 nicotinic acetylcholine receptor agonist treatment reduces neuroinflammation, oxidative stress, and brain injury in mice with ischemic stroke and bone fracture. J Neurochem 131(4): 498-508.
- Kim SH, Chung DK, Lee YJ, Song CH, Ku SK (2016) Neuroprotective effects of Danggui-Jakyak-San on rat stroke model through antioxidant/ antiapoptotic pathway. J Ethnopharmacol 188: 123-133.
- Altermann CDC, Souza MA, Schimidt HL, Izaguirry AP, Martins A, et al. (2017) Short-term green tea supplementation prevents recognition memory deficits and ameliorates hippocampal oxidative stress induced by different stroke models in rats. Brain Res Bull 131: 78-84.
- Jin H, Li Z, Guo X, Tong H, Liu Z, et al. (2016) Microcirculatory disorders and protective role of antioxidant in severe heat stroke: A Rat Study. Shock 46(6): 688-695.
- Longo Mbenza B, Longokolo Mashi M, Lelo Tshikwela M, Mokondjimobe E, Gombet T, et al. (2011) Relationship between younger age, autoimmunity, cardio metabolic risk, oxidative stress, HAART, and ischemic stroke in Africans with HIV/AIDS. ISRN Cardiology 2011: 897908.






























