Anti-Inflammatory Effects of Orally Administered Dipeptide JBP 485 in Rats and Mice
Xiaobo Yang1, Taiichi Kaku2 and Kexin Liu1*
1Department of Clinical Pharmacology, College of Pharmacy, Dalian Medical University, China
2Japan Bioproducts Co. Ltd, Japan
Submission: August 15, 2018; Published: September 04, 2018
*Corresponding author: Kexin Liu, Department of Clinical Pharmacology, College of Pharmacy, Dalian Medical University, 9 West Section, Lvshun South Road, Lvshunkou District, Dalian, 116044, China; Fax: +86-411-86110407; Tel: +86-411-86110406; Email: kexinliu@dlmedu.edu.cn
How to cite this article: Xiaobo Y, Taiichi K, Kexin L. Anti-Inflammatory Effects of Orally Administered Dipeptide JBP 485 in Rats and Mice. Mod Appl Bioequiv Availab. 2018; 4(1): 555627.
Abstract
The goal of this study was to evaluate the anti-inflammatory properties of JBP 485 isolated from the human placenta hydrolyzate in rats and in mice. JBP 485 markedly inhibited paw swelling in anti-CII mAbs (monoclonal antibodies to type II collagen) plus LPS (lipopolysaccharide)-induced mice paw edema at a dose of 25mg/kg after oral administration. Additionally, 100mg/kg dose of JBP 485 significantly suppressed colonic inflammation induced by DNBS (2,4-dinotrobenzene sulfonic acid) after oral administration and apparently repaired tissue injury, such as diarrhea, colonic ulcer and adhesions between the small intestine and inflamed colon in this rat model of inflammatory bowel disease. Furthermore, the application of 1000M JBP 485 significantly inhibited the releases of pro-inflammatory cytokines such as TNF-α (tumour necrosis factor alpha), IL-1β (interleukin-1β), IL-6 (interleukin-6), IL-2 (interleukin-2) and INF-γ (interferon-gamma) from human peripheral blood mononuclear leukocytes (PBMNL) in a concentration dependent manner. Safety of JBP 485 was verified by negligible cytotoxicity in PBMNL. These results suggest that JBP 485 displays considerable anti-inflammatory effects and may be useful for clinical applications.
Keywords: JBP 485; Concanavalin A; Lipopolysaccharide; Anti-inflammatory; Inflammatory bowel disease
Abbreviations: PBMNL: Peripheral Blood Mononuclear Leukocytes; TNF-α: Tumour Necrosis Factor Alpha; INF-γ: Interferon-Gamma; ILs: Interleukins; ELISA: Enzyme Linked Immune Sorbent Assay; MIF: Migration Inhibitory Factor; LPS: Lipopolysaccharide
Introduction
The inflammatory reaction delivering the appropriate defense against harmful stimuli is involved in various pathological and physiological processes. Beneficial inflammatory responses which promote local coagulation to confine tissue damage and infection can be triggered by the controlled production of tumour necrosis factor alpha (TNF-α) and proinflammatory cytokines, such as interleukins (ILs) [1]. However, uncontrolled inflammation can produce many cytokines, resulting in extensive organ dysfunction and tissue damage and chronic inflammatory diseases, such as autoimmune disease, cardiovascular disease and Alzheimer’s disease [2,3]. In face of the fact that the clinical application of the commonly used anti-inflammatory drugs clinically, including non-steroidal and steroidal anti-inflammatory drugs, is limited due to their side effects, new potential drugs with low toxicity and good efficacy for inflammation treatment is urgent needed.
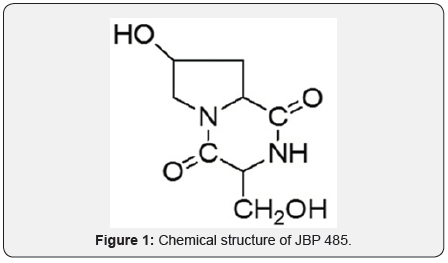
JBP 485 (Cyclo-trans-4-L-hydroxyprolyl-L-serine, Figure 1), a dipeptide, was first isolated from Laennec® (a trade name for hydrolysate of human placenta) which has been used clinically to treat chronic hepatic injury for over five decades [4,5]. Our previous studies showed that JBP 485 had no obvious toxicity and exhibited excellent gastrointestinal absorption [5,6] as well as had clear anti-hepatitis effect after oral administration, probably due to its apoptosis-inhibiting and antioxidant properties, which have been demonstrated in animal experiments [6-8]. These results indicate JBP 485 has a potency as an anti-inflammatory agents. However, scientific reports describing the anti-inflammatory effects of JBP 485 by oral administration are lacking. Therefore, in the present work, we focused on the investigation of the anti-inflammatory effects of JBP 485 on two experimental animal models in rats and in mice.
Materials and Methods
Reagents
JBP 485 was obtained from Japan Bioproducts Industry Co. Ltd (Tokyo, Japan). Concanavalin A was provided by Panlabs, Co. Ltd (Taipei, Taiwan). DNBS (2,4-dinotrobenzene sulfonic acid) was from Panlabs, Co. Ltd (Taipei, Taiwan). Anti-CII mAbs mixture and lipopolysaccharide were provided by Panlabs, Co. Ltd (Taipei, Taiwan). Murine TNF-α, IL-1β, IL-6, IL-2 and IFN-γ ELISA kits were from Panlabs, Co. Ltd (Taipei, Taiwan). DNBS (2,4-dinotrobenzene sulfonic acid) was from Panlabs, Co. Ltd (Taipei, Taiwan). Human peripheral blood mononuclear leukocytes were obtained from Panlabs, Co. Ltd (Taipei, Taiwan). Other reagents will be further specified.
Animals
Male BALB/cByJ mice (20±2g) and Wistar derived male rats (20020g) (Panlabs, Co. Ltd (Taipei, Taiwan)) were housed at 23±2 ℃ under a 12h light-dark cycle, at 50±5% relative humidity throughout the whole experimental period. All mice/rats were allowed free access to water and chow diet. All experimental protocols were approved by the Institutional Animal Ethics Committee of Panlabs, Co. Ltd, which follows the standards of the Committee for the Purpose of Control and Supervision of Experimentation on Animals, Government of Taiwan.
Anti inflammatory tests
Monoclonal antibody to collagen plus lipopolysaccharide induced arthritis in mice: Arthritis was induced in BALB/ cByJ mice using the method previously established by Terato et al. [9,10]. A combination of 4 different monoclonal antibodies (mAbs: D8, F10, DI-2G, A2) to type II collagen (anti-CII mAbs) was administered by intravenous administration (4mg/mouse) on day 1 and challenged 3 days later (day 4) with intravenous administration of lipopolysaccharide (LPS, 25g/mouse). From day 4, beginning one hour post LPS, JBP 485 (25mg/kg) were administered by oral administration once daily for 3 consecutive days. Indomethacin (3mg/kg) (Panlabs, Co. Ltd, Taipei, Taiwan) as a positive control and vehicle (distilled water) as negative control were similarly used with JBP 485. For each animal, increase in volume of both hind paws was measured using a plethysmometer (Ugo Basile Cat. #7150) with a water cell (12 mm diameter) on days 5, 7, 10, 14 and 17. The inhibition percent of increased volume was calculated by following formula:
Inhibition (%) = [1-(Tn-T0)/(Cn-C0)] × 100
Where C0 (Cn) expresses the swollen volume of day 0 (day n) in vehicle control and T0 (Tn) expresses the swollen volume of day 0 (day n) in JBP 485 or indomethacin treated group, respectively. The reduction of edema in the hind paws by 30% or more (≥30) was considered significant.
DNBS (2,4-dinotrobenzene sulfonic acid)-induced bowel disease in rats: The experimental animal model of DNBS-induced colitis is well recorded previously [11,12]. Wistar derived male rats weighing 20020g and fasted for 24 hours were used. Distal colitis was induced by intra-colonic instillation of DNBS (30mg in 0.5ml 30% ethanol). In briefly, a rubber cannula (outside diameter 2.3mm) was inserted intrarectally into a metofaneanesthetized rat so that the tip was 8cm proximal to the anus. After instilling the DNBS-ethanol solution, the cannula was gently removed. Another ruber cannula with the same outside diameter was inserted intrarectally into the rat so that the tip was 2cm proximal to the anus. Then, air (2ml) was gently injected through the cannula to ensure that the DNBS-ethanol solution stayed in the colon. JBP 485 (6.25, 25 and 100mg/ kg) dissolved in a vehicle of 2% Tween 80 was administered by oral administration 24 and 2 hours before DNBS instillation and then daily for 5 consecutive days. Blank control group was similarly treated with vehicle alone while negative control group was treated with vehicle plus DNBS. Sulfasalazine (300mg/kg) (Panlabs, Co. Ltd, Taipei, Taiwan), a drug well known for antiinflammatory bowel disease as positive control was similarly used with JBP 485 groups. The animals were sacrificed 24 hours after the final dose administration (day 7) and each colon was removed and weighed. During the experiment, the presence of diarrhea was recorded daily. Further, when the abdominal cavity was opened before removal of the colon, adhesions between the colon and other organs were also recorded. Colonic ulceration was noted after removal and weighing of each colon. Colon-tobody weight ratio was then calculated for each animal according to the formula as follows:
Colon-to-body weight ratio (%) = Colon (g)/body weight (g) × 100
The “Net” increase in ration of negative control group relative to blank control group is used as a base for comparison with test substance treated groups and expressed as “Decrease (%)” (percent decrease). A 30% or more (≥30%) reduction in colon-to-body weight ratio was considered significant.
Cytotoxicity of JBP 485 in PBMNL: JBP 485 (1-1000μM) was incubated with a suspension of PBMNL (1 × 106/well) in the presence of Con A (10g/ml) or LPS (25ng/ml) for 16 hours in RPMI 1640 medium (pH 7.4) containing 10% FBS with 50 unit/ml of penicillin and 50μg/ml of streptomycin at 37 °C in 5% CO2. Alamar Blue reagent (Sigma-Aldrich) was added and the cells were incubated at 37 °C for another 16 hours. Living cells will take up Alamar Blue and emit fluorescence. Fluorescence intensity is measured using a Spectro Fluor Plus plate reader (Shimadzu, Kyoto, Japan) with excitation at 485nm and emission at 590nm. Decrease of 50 percent or more (≥50%) in fluorescence intensity relative to vehicle treated controls indicates significant cytotoxicity.
Measurement of inflammatory mediator release from human leukocytes: For the interleukin-2 (IL-2) and interferongamma (IFN-γ) release, JBP 485 (1-1000μM), vehicle (0.4% DMSO) and dexamethason (0.0001-1μM, positive control) (Panlabs, Co. Ltd, Taipei, Taiwan) were incubated with a suspension of human peripheral blood mononuclear leukocytes (PBMNL, 1 × 106/well) in the presence of concanavalin A (Con A, 10 µg/ml). For the interleukin-1β (IL-1β), interleukin-6 (IL-6) and TNF-α release, the same concentrations of JBP 485, vehicle and dexamethason were incubated with the same PBMNL in the presence of lipopolysaccharide (LPS, 25ng/ml). The levels of cytokines in cell supernatants were quantitated using sandwich ELISA kits after incubation for 24h [13].
Results
Effect of JBP 485 on arthritis induced by monoclonal antibody to collagen plus LPS in mice
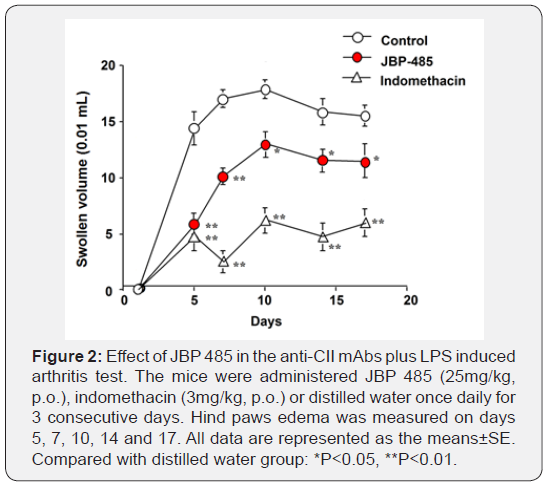
In order to confirm the anti-inflammatory activity of JBP 485 in acute-phase inflammation in vivo, an anti-CII mAbs plus LPSinduced mice paw edema experiment was conducted. As shown in Figure 2, the edema in hind paws appeared markedly after intravenous administration of anti-CII mAbs plus LPS in control group. The peak of edema was at the day 10 after administration of anti-CII mAbs. Compared with control, when 25mg/kg of JBP 485 was administered by oral administration for 3 consecutive days the edema in hind paws after intravenous administration of anti-CII mAbs was significantly inhibited. The difference in the effect from that of the control group was greatest at 5 d after anti-CII mAbs injection, and the inhibition percentage value of JBP 485 was 59.26%. Indomethacin (3mg/kg, p.o.), as a positive control, obviously suppressed the edema (P< 0.01), with a largest inhibition percentage of 85.31% at 7d after mAbs injection.
Effect of JBP 485 on DNBS-induced inflammatory bowel disease in rats

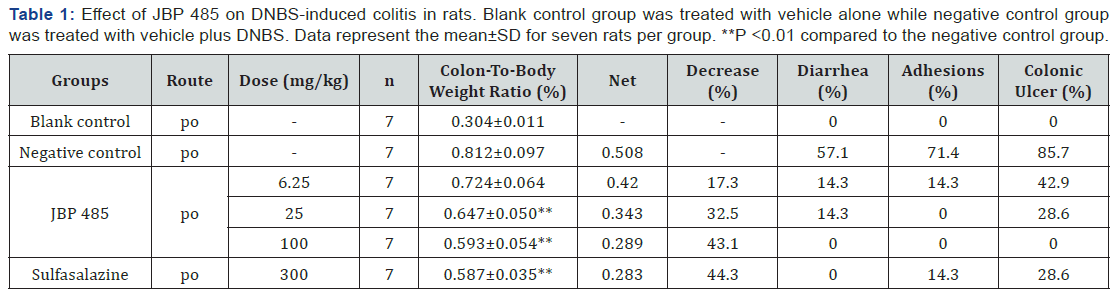
To reveal the effect of JBP 485 on inflammatory bowel disease, a DNBS-colitis rat model was established. As shown in Figure 3 and Table 1, JBP 485 (100 and 25mg/kg, p.o.) significantly decreased the colon-to-body weight ratio (P< 0.01). The values were 0.65% and 0.59%, respectively. The lowest dose of JBP 485 (6.25mg/kg, p.o.) did not affect the value of the colon-to-body weight ratio (P>0.05). Sulfasalazine (300mg/kg, p.o.), as a positive control, markedly suppressed the colon-tobody weight ratio (P< 0.01), with a value of 0.59%. The effect of 100mg/kg JBP 485 was similar with that of sulfasalazine. On day 7, the presence of diarrhea, adhesions between small intestine and the inflamed colon as well as colonic ulcer was noted. For the negative control group, diarrhea was noted in 4 out of 7 (57.1%) rats, adhesions were noted in 5 out of 7 (71.4%) rats and colonic ulcer was noted in 6 out of 7 (85.7%) rats killed 6 days after induction of colitis. The highest dose of JBP 485 (100mg/kg, p.o.) completely decreased the incidence of diarrhea (0 of 7 rats or 0%), adhesions (0 of 7 rats or 0%) and colonic ulcer (0 of 7 rats or 0%). Rats dosed with 25mg/kg and 6.25mg/kg of JBP 485 experienced a much lower incidence of diarrhea (1 of 7 rats or 14.3%, 1 of 7 rats or 14.3%, respectively), adhesions (0 of 7 rats or 0%, 1 of 7 rats or 14.3%, respectively) and colonic ulcer (2 of 7 rats or 28.6%, 3 of 7 rats or 42.9%, respectively) compared to vehicle-treated inflamed controls. Following treatment of rats with 300mg/kg of sulfasalazine, diarrhea, adhesions and colonic ulcer were observed in 0 of 7 rats (0%), 1 of 7 rats (14.3%) and 2 of 7 rats (28.6%), respectively (Figure 3 & Table 1).
Cytotoxicity of JBP 485 in PBMNL
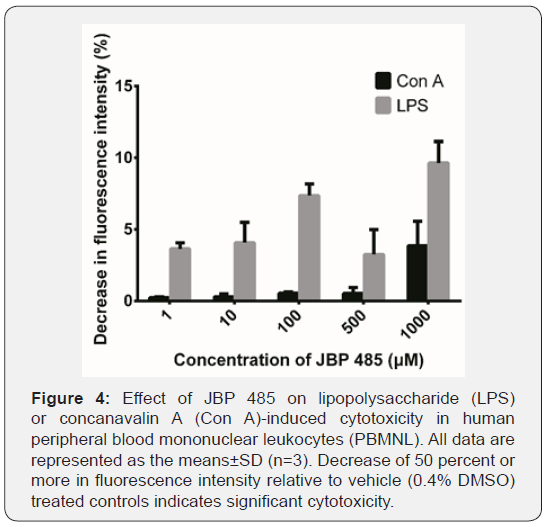
To reveal the effect of JBP 485 on Con A or LPS-induced cytotoxicity in PBMNL, we examined the cytotoxicity of JBP 485 by a fluorometric assay method [14]. As a result, JBP 485 showed negligible cytotoxicity up to a concentration of 1000μM (Figure 4).
Effect of JBP 485 on inflammatory mediator release induced by concanavalin A or lipopolysaccharide from human leukocytes
To examine whether JBP 485 inhibits the release of inflammatory mediator, the effects of JBP 485 on IL-1β, IL-6 and TNF-α releases induced by lipopolysaccharide as well as on INF-γ and IL-2 releases induced by concanavalin A from human leukocytes were observed. As shown in Figure 5, in the range of 1-1000 M, JBP 485 with negligible cytotoxicity (Figure 4) inhibited the releases of IL-1β, IL-6, IL-2, TNF-α and INF-γ in a concentration dependent manner and the inhibition effects for all inflammatory mediators were significantly different from control at 1000M (P< 0.05). Dexamethason (0.01-1μM) treatment of DNBS-colitis rats as well resulted in a significant inhibition on the production of the cytokines mentioned above compared to DNBS-colitis control rats (P< 0 .05).
Discussion
Our previous studies proved that JBP 485 can repair liver function after both oral and i.v. administrations. It can also exhibit an anti-hepatitis effect directly on cultured hepatocytes [5,6]. But the investigation of the anti-inflammatory effects of JBP 485 on experimental animal models of inflammation is not recorded previously. Firstly, we examined the effect of JBP 485 on arthritis induced by anti-CII mAbs plus LPS in mice in comparison with indomethacin, a COX-2 inhibitor. Anti-CII mAbs plus LPS-induced arthritis has many similarities with human arthritis [15], so we used this model to assess the anti-inflammatory effects of JBP 485. In this model of arthritis, the increase in volume of both hind paws of each mouse was measured and the inhibition percent of increased volume was calculated. Our data showed that JBP 485 significantly suppressed the increase of paw volume at a dose of 25mg/kg after oral administration. 25mg/kg/day of JBP 485 and 3mg/kg/day of indomethacin for 3 consecutive days resulted in the greatest inhibition of paw volume at 5d and 7d after anti-CII mAbs injection, respectively, compared with the vehicle treated group (Figure 2). Although human arthritis might not develop in a way identical to rodent arthritis, it is certain that TNF-α plays an important role in inflammation, cartilage damage and bone destruction in anti-CII mAbs plus LPS-induced arthritis [16,17]. TNF α as an important pro-inflammatory cytokine produced by lymphocytes and macrophages during acute inflammatory responses, acted a pivotal part in activation of inflammatory transcriptional factors [18]. Thornton et al. demonstrated that TNF-α mRNA was increased both in acute and in late phases, while IL-1β and IL-6 mRNA expression peaked at the acute stage of conventional collagen-induced arthritis [19]. They claimed that TNF-α had a role in promoting both the early and chronic stages of arthritis and IL-1β and IL-6 would be involved in the acute stage of the disease. Furthermore, for the response to anti-CII mAbs plus LPS, Shinohe et al. suggested that elevation of TNF-α mRNA occurred early in local sites of inflammation. Comparatively later, the expression of IL-1β and IL-6 mRNA were upregulated, leading to the progression of arthritis at the chronic stage [20]. Therefore, the effect of JBP 485 on inflammatory mediator release such as IL-1β, IL-6 and TNF-α should be determined hereafter. In the present study, the application of 1-1000µM JBP 485 inhibited the releases of pro-inflammatory cytokines such as TNF-α, IL-1β and IL-6 from PBMNL in a concentration dependent manner and the inhibition effects for all inflammatory mediators were significantly different from control at 1000µM (Figure 5).
Next, we studied the effect of JBP 485 on DNBS-induced bowel disease in rats compared with sulfasalazine, a drug well known for anti-inflammatory bowel disease. The DNBS caused injury is more closely similar to human colitis in inflammation, histological and macroscopic damage score [21]. Our results demonstrated that the colon-to-body weight ratio was significant decreased after oral administration of JBP 485 compared to the negative control group (Figure 3 & Table 1). Surprisingly, 100mg/kg dose of JBP 485 showed almost comparable colon-to-body weight ratio reduction than 300mg/kg dose of sulfasalazine. Moreover, JBP 485 in any dose (6.25, 25 and 100mg/kg) significantly repaired the diarrhea, adhesions between the small intestine and inflamed colon as well as colonic ulcer in this experimental model of inflammatory bowel disease after oral administration (Figure 3 & Table 1).

Cytokines take a central part in the pathogenesis of inflammatory bowel disease. It has been reported that TNF-α is necessary for the promoting of DNBS colitis, as TNF-α-deficient mice revealed resistance to DNBS, and its over expression is related to more severe colitis in mice [22]. INF-γ is a vital proinflammatory cytokine with pleiotropic functions varying between augmentation of crucial histocompatibility complex expression, antiviral activity and stimulation of natural T and killer cells [23]. Khairy et al. [11] found that the levels of TNF-α and INF-γ were significantly higher in colitis rats compared with those of control rats [11]. It has been reported that pro-inflammatory cytokine IL-2 is involved in a number of inflammatory processes [24-26]. Any successful attempt to block this cytokine would prove to be therapeutically useful [27]. Previously, we demonstrated that immune-mediated liver damage could be prevented by JBP485, and that may be mainly due to the immunomodulatory effects of JBP 485 on T cells and adhesion molecules [7]. In this study, 1000µM JBP 485 significantly inhibited the releases of cytokines such as TNF-α, IL-2 and INF-γ from PBMNL compared to DNBS-colitis control group (P< 0 .05) (Figure 5). The detailed mechanism remains to be further elucidated.
Conclusion
In conclusion, we firstly demonstrated that JBP 485 significantly inhibited the arthritis induced by anti-CII mAbs plus LPS after oral administration in mice. JBP 485 also suppressed colonic inflammation and apparently reduced tissue injury after oral administration in a rat model of DNBS-induced inflammatory bowel disease. Safety of JBP 485 was also verified by negligible cytotoxicity in PBMNL. Further this study, the mechanism of anti-inflammatory activity of JBP 485 should be investigated to further ascertain its applicability to clinical stages.
Conflict of Interest
The authors confirm that this article content has no conflict of interest.
Acknowledgement
This work was supported by the National Natural Science Foundation of China under Grant No. 81473280 and No. 81502992.
References
- Ulloa L, Tracey KJ (2005) The “cytokine profile”: a code for sepsis. Trends Mol Med 11(2): 56-63.
- Heppner FL, Ransohoff RM, Becher B (2015) Immune attack: the role of inflammation in Alzheimer disease. Nat Rev Neurosci 16(6): 358-372.
- Viola J, Soehnlein O (2015) Atherosclerosis - A matter of unresolved inflammation. Seminars in Immunology 27(3): 184-193.
- Liu KX, Kato Y, Kaku T, Sugiyama Y (1998) Human placental extract stimulates liver regeneration in rats. Biol Pharm Bull 21(1): 44-49.
- Liu KX, Kato Y, Kaku TI, Santa T, Imai K, et al. (2000) Hydroxyprolylserine derivatives JBP923 and JBP485 exhibit the antihepatitis activities after gastrointestinal absorption in rats. J Pharmacol Exp Ther 294(2): 510- 515.
- Wu J, Wang C, Liu Q, Yang T, Zhang Q, et al. (2008) Protective effect of JBP485 on concanavalin A-induced hepatocyte toxicity in primary cultured rat hepatocytes. Eur J Pharmacol 589(1-3): 299-305.
- Yang T, Wu J, Wang C, Liu Q, Ma X, et al. (2009) Protective effect of JBP485 on concanavalin A-induced liver injury in mice. J Pharm Pharmacol 61(6): 767-774.
- Wang W, Liu Q, Wang C, Meng Q, Kaku T, et al. (2011) Effects of JBP485 on the expression and function of PEPT1 in indomethacin-induced intestinal injury in rats and damage in Caco-2 cells. Peptides 32(5): 946-955.
- Terato K, Harper DS, Griffiths MM, Hasty DL, Ye XJ, et al. (1995) Collagen-induced arthritis in mice: synergistic effect of E. coli lipopolysaccharide bypasses epitope specificity in the induction of arthritis with monoclonal antibodies to type II collagen. Autoimmunity 22(3): 137-147.
- Ichiyama H, Onodera S, Nishihira J, Ishibashi T (2004) Inhibition of joint inflammation and destruction induced by anti-type II collagen antibody/lipopolysaccharide (LPS)-induced arthritis in mice due to deletion of macrophage migration inhibitory factor (MIF). Cytokine 26(5): 187-194.
- Khairy H, Saleh H, Badr AM, Marie MS (2018) Therapeutic efficacy of osthole against dinitrobenzene sulphonic acid induced-colitis in rats. Biomed Pharmacother 100: 42-51.
- Morris GP, Beck PL, Herridge MS, Depew WT, Szewczuk MR, et al. (1989) Hapten-induced model of chronic inflammation and ulceration in the rat colon. Gastroenterology 96(3): 795-803.
- Welker P, Lippert U, Nürnberg W, Krüger-Krasagakes S, Möller A, et al. (1996) Glucocorticoid-induced modulation of cytokine secretion from normal and leukemic human myelomonocytic cells. Int Arch Allergy Immunol 109(2): 110-115.
- Page B, Page M, Noel C (1993) A new fluorometric assay for cytotoxicity measurements in-vitro. Int J Oncol 3(3): 473-476.
- Okumura A, Saito T, Otani I, Kojima K, Yamada Y, et al. (2008) Suppressive role of leukocyte cell-derived chemotaxin 2 in mouse antitype II collagen antibody-induced arthritis. Arthritis Rheum 58(2): 413-421.
- Kagari T, Doi H, Shimozato T (2002) The importance of IL-1 beta and TNF-alpha, and the noninvolvement of IL-6, in the development of monoclonal antibody-induced arthritis. J Immunol 169(3): 1459-1466.
- Rioja I, Bush KA, Buckton JB, Dickson MC, Life PF (2004) Joint cytokine quantification in two rodent arthritis models: kinetics of expression, correlation of mRNA and protein levels and response to prednisolone treatment. Clin Exp Immunol 137(1): 65-73.
- Stucchi A, Reed K, O Brien M, Cerda S, Andrews C, et al. (2006) A new transcription factor that regulates TNF-alpha gene expression, LITAF, is increased in intestinal tissues from patients with CD and UC. Inflamm Bowel Dis 12(7): 581-587.
- Thornton S, Duwel LE, Boivin GP, Ma Y, Hirsch R (1999) Association of the course of collagen-induced arthritis with distinct patterns of cytokine and chemokine messenger RNA expression. Arthritis Rheum 42(6): 1109-1118.
- Rosal-Vela A, García-Rodríguez S, Postigo J, Iglesias M, Longobardo V, et al. (2015) Distinct serum proteome profiles associated with collagen-induced arthritis and complete Freund’s adjuvant-induced inflammation in CD38(-)/(-) mice: The discriminative power of protein species or proteoforms. Proteomics 15(19): 3382-3393.
- Hawkins JV, Emmel EL, Feuer JJ, Nedelman MA, Harvey CJ, et al. (1997) Protease activity in a hapten-induced model of ulcerative colitis in rats. Dig Dis Sci 42(9): 1969-1980.
- Neurath MF, Fuss I, Pasparakis M, Alexopoulou L, Haralambous S, et al. (1997) Predominant pathogenic role of tumor necrosis factor in experimental colitis in mice. Eur J Immunol 27(7): 1743-1750.
- Farrar MA, Schreiber RD (1993) The molecular cell biology of interferon-gamma and its receptor. Annu Rev Immunol 11: 571-611.
- Wong HR, Finder JD, Wasserloos K, Lowenstein CJ, Geller DA, et al. (1996) Transcriptional regulation of iNOS by IL-1 beta in cultured rat pulmonary artery smooth muscle cells. Am J Physiol 271(1 Pt 1): L166-L171.
- Perkins DJ, Kniss DA (1997) Tumor necrosis factor-alpha promotes sustained cyclooxygenase-2 expression: attenuation by dexamethasone and NSAIDs. Prostaglandins 54(4): 727-743.
- Dinarello CA (2002) The IL-1 family and inflammatory diseases. Clin Exp Rheumatol 20(5 Suppl 27): S1-S13.
- Ashalatha K, Venkateswarlu Y, Priya AM, Lalitha P, Krishnaveni M, et al. (2010) Anti-inflammatory potential of Decalepis hamiltonii (Wight and Arn) as evidenced by down regulation of pro inflammatory cytokines- TNF-alpha and IL-2. J Ethnopharmacol 130(1): 167-170.






























