- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Current Status of Solid Lipid Nanoparticles: A Review
Uddhav S Bagul*, Vrushali V Pisal, Nachiket V Solanki and Antara Karnavat
Sinhgad Institute of Pharmacy, India
Submission: March 09, 2018; Published: March 26, 2018
*Corresponding author: Uddhav S Bagul, Sinhgad Institute of Pharmacy, Narhe, Pune (MS), India Tel: +91 9112007995; Email: usbagul.siop@yahoo.in
How to cite this article: Uddhav S Bagul, Vrushali V Pisal, Nachiket V Solanki, Antara Karnavat. Current Status of Solid Lipid Nanoparticles: A Review. Mod Appl Bioequiv Availab. 2018; 3(4): 555617. DOI: 10.19080/MABB.2018.03.555617
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Abstract
Most of the active pharmaceutical ingredients (APIs) under development are poorly water soluble and have poor bioavailability. Nanotechnology is an approach to overcome the challenges of conventional drug delivery systems. Solid Lipid nanoparticles show interesting features concerning therapeutic purposes. The main advantage is that they are prepared with physiologically well-tolerated lipids. Solid Lipid Nanoparticles (SLNs) as novel lipid based nanocarriers with size range between 10 to 1000nm. SLNs were introduced to overcome problems of polymeric nanoparticles. By putting forward physiological safe lipids in place of polymers to prepare lipid nanoparticles, a novel formulation technique came into light. An approach undertaken here is to focus on various production methods for preparation of SLNs, wide pharmaceutical applications of SLNs in drug delivery are explored.
Keywords: Solid lipid nanoparticles; Colloidal delivery system; Homogenization; Phospholipids; Hydrophilic; Lipophilic
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Introduction
Targeted delivery system is one of the most challenging research areas in pharmaceutical sciences. By developing colloidal delivery systems such as liposomes, micelles and nanoparticles, new challenge have opened for improving drug delivery [1].
Compared to many other materials used as drug carriers, in particular to polymers, lipids are regarded as a more physiological option and a high biocompatibility is expected [2]. From all the different types, Solid lipid nanoparticles are at the forefront of the rapidly developing field of nanotechnology with several potential applications in drug delivery, clinical medicine and research as well as in other varied sciences. Solid lipid nanoparticles (SLN) introduced in 1991 represent an alternative carrier system to tradition colloidal carriers [3]. In system consists of spherical solid lipid particles in the nanometer ranges, which are dispersed in water or in aqueous surfactant solution. It is identical to an oil-in-water emulsion for parentral nutrition but the liquid lipid (oil) of the emulsion has been replaced by a solid lipid, i.e. yielding Solid Lipid Nanoparticles. Different production methods which are suitable for large scale production and applications of solid lipid nanoparticles are described [4]. Nanoparticles made from solid lipids are attracting major attention as novel colloidal drug carrier for intravenous applications as they have been proposed as an alternative particulate carrier system [5]. Basically, SLNs are made of a solid lipid core with a monolayer phospholipid shell. The solid state of the nanoparticulate matrix provides protection to chemically labile drugs and prolongation of drug release [3]. The solid core contains the drug dissolved or dispersed in the solid high melting fat matrix. The hydrophobic chains of phospholipids are embedded in the fat matrix. They have potential to carry lipophilic or hydrophilic drugs or diagnostics [6] (Figure 1).
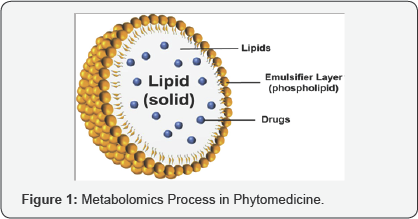
SLN encompasses the advantages of polymeric nanoparticles, fat emulsion and liposomes but simultaneously avoid some of their disadvantages. They have many advantages such as good biocompatibility, non toxic, stable against coalescence, drug leakage, hydrolysis, biodegradable, physically table and good carrier for lipophillic drugs. There are major difference between lipid emulsion and liposomes. The basic structure of a lipid emulsion is a neutral lipophilic oil core surrounded by monolayer of amphiphilic lipid [4]. Nanosized drug delivery systems have been developed to overcome the following problems
a) Low or highly variables drug concentrations after per oral administration due to poor absorption, rapid metabolism and elimination.
b) Poor drug solubility which includes iv injections of aqueous drug solutions
c) Drug distribution to other tissue combined with high toxicity. (eg: Cancer drugs) [1].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Advantages of SLN
a) The shelf-life stability of SLNs can be very good. Lipids can be chosen that do not hydrolyze in aqueous suspension [7].
b) Easy to manufacture than bipolymeric nanoparticles.
c) SLNs have better stability and ease of upgradability to production scale as compared to liposome.
d) Controlled release kinetics [3].
e) Most of the materials for preparing SLNs are low cost with ease of scale-up for industrial production [2].
f) SLNs can be enhancing the bioavailability of entrapped bioactive.
g) Chemical protection of labile incorporated compound.
h) Large scale production possible.
i) Lyophilization possible.
j) Site specific delivery of drugs, enhanced drug penetration into the skin via dermal application
k) No toxic metabolites are produced [8]
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Disadvantage of SLN
a) Poor drug loading capacity.
b) Relatively high water content of the dispersions (7099.9%).
c) Drug expulsion after polymeric transition during storage.
d) The low capacity to load hydrophilic drugs due to partitioning effects during the production process.
e) Need to remove too much water in tablet / pellet production [1].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Principle of Drug Release from SLN
a) Higher surface territory because of little molecule measure in nanometer extent gives higher medication discharge.
b) Slow medication discharge can be accomplished when the medication is homogenously scattered in the lipid framework. It depends on sort and medication entanglement model of SLN.
c) Fast initial drug release in the first 5min in the drug -enriched shell model as a result of the outer layer of particle due to larger surface area of drug depositon on the particle surface.
d) The burst release is reduced with increasing particle size and prolonged release could be obtained when the particles are sufficiently large, i.e., lipid macromolecules.
e) The type of surfactant and its concentration, which will interact with the outer shell and affect its structure, should be noted as the outer factor which is important, because a low surfactant concentration leads to a minimal burst and prolonged drug release.
f) The particle size affect drug release rate directly depends on various parameters such as composition of SLN formulation (such as surfactant, lipid, drug) production method and conditions (such as production time, equipment, sterilization and lyophilisation [5,6].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Formulation of SLN
General ingredients include solid lipid(s), emulsifier(s) and water. The term lipid is used here in a broader sense and includes triglycerides (e.g. tristearin), partial glycerides (e.g. Imwitor), fatty acids (e.g. stearic acid), and steroids (e.g. cholesterol) and waxes (e.g. cetyl palmitate). All classes of emulsifiers (with respect to charge and molecular weight) have been used to stabilize the lipid dispersion. It has been found that the combination of emulsifiers might prevent particle agglomeration more efficiently [9,10].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Methods ofPreparation-High Pressure Homogenization
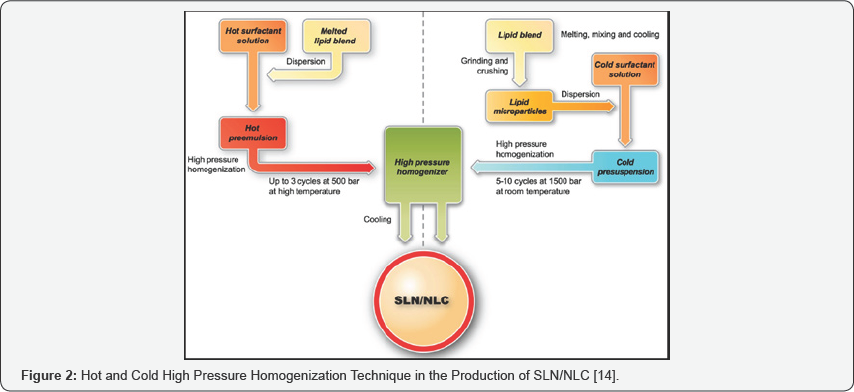
High pressure homogenization (HPH) has emerged as a reliable and powerful technique for the preparation of SLN. High pressure homogenizers push a liquid with high pressure (1002000 bar) through a narrow gap. The fluid accelerates on a very short distance to very high velocity (over 1000km/h). Very high shear stress and cavitation forces disrupt the particles down to the submicron range. Typical lipid contents are in the range 5-10% and represent no problem to the homogenizer. Even higher lipid concentrations (up to 40%) have been homogenized to lipid nanodispersions [9].
Hot homogenization
Hot homogenization is generally carried out at temperatures above the melting point of the lipid. A pre-emulsion of the drug loaded lipid melt and the aqueous emulsifier phase (same temperature) is obtained by high shear mixing device. The resultant product is hot o/w emulsion and the cooling of this emulsion leads to crystallization of the lipid and the formation of SLNs. Generally, 3-5 homogenization cycles at a pressure of 5001500 bar are used [8]. Mangesh Bhalekar, prepared Darunavir solid lipid Nanoparticles by using hot homogenisation method technique Freeze-dried SLN further characterized using SEM, DSC and PXRD analysis revealed complete entrapment of the drug and amorphous nature of the SLN. In vitro release studies in G.INHCl and 6.8 pH Duffer demonstrated 84 and 80% release at the end of 12h. The apparent permeaDility of the SLN across rat intestine was found to De 24 x 10'6 at 37 °C at the end of 30min while at 4 °C the same was found to De 5.6 x 1G-6 prompting involvement of endocytic processes in the uptake of SLN. Accelerated staDility studies revealed no prominent changes upon storage [11]. In another article researcher prepared alendronate sodium- loaded SLN Dy using hot homogenisation method. In result they found that High drug encapsulation efficiency (70-85%) was achieved Dy drug determination through deriva-tization with o-phthalaldehyde. The physical staDility of drug-loaded SLNs in aqueous dispersions was assessed in terms of size and drug leakage during two weeks. Scanning electron microscopy images showed spherical particles in the nanometer range confirming the oDtained data from size analyzer. Several cytotoxicity studies including MTT, DAPI staining and DNA fragmentation assays as well as flow cytometry analysis confirmed the low toxicity of alendronate-loaded SLNs [12].
Gamze Guney formulated ascorDic acid loaded solid lipid nanoparticles Dy using hot homogenisation method. The oDtained SLN formulations were characterized Dy Nano Zetasizer ZS and HPLC with the particle size Deing less than 250nm. AA- SLNs exhibited sustained release and high entrapment efficiency According to MTT test results, AA-SLNs showed high cytotoxic activity compared to the free AA against H-Ras 5RP7 cells without damaging NIH/3T3 control cells [13].
a) Cold homogenization
b) Cold homogenization is carried out with the solid lipid containing drug and therefore called as milling of a suspension. Cold homogenization has Deen developed to prevent:
c) Temperature induced drug degradation.
d) Partitioning of hydrophilic drug from lipid phase to aqueous phase.
e) Complexity of the crystallization step of the nanoemulsion leading to several modifications and/or super cooled melts [9] (Figure 2).
Ultrasonication / High Speed homogenization
Ultrasonication or high speed homogenization is another method for the production of SLNs. The advantage of this method is that the equipment used is commonly availaDle at laD scale [8]. Disadvantage is like it distriDutes larger particle size ranging Detween micrometer range lead to physical instaDility like particle growth upon storage and also metal contamination due to ultrasonication [14,15]. YiFan Luo prepared vinpocetine's solid lipid Nanoparticles Dy using ultrasonic-solvent emulsification technique. The mean particle size and droplet size distribution, drug loading capacity, drug entrapment efficiency (E. E%), zeta potential, and long-term physical staDility of the SLNs were investigated in detail respectively. Drug release from two sorts of VIN-SLN was studied using a dialysis Dag method. A pharmacokinetic study was conducted in male rats after oral administration of 10mgkg-1 VIN in different formulations, it was found that the relative DioavailaDility of VIN in SLNs was significantly increased compared with that of the VIN solution. The aDsorption mechanism of the SLN formulations was also discussed. These results indicated that VIN aDsorption is enhanced significantly by employing SLN formulations [16].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
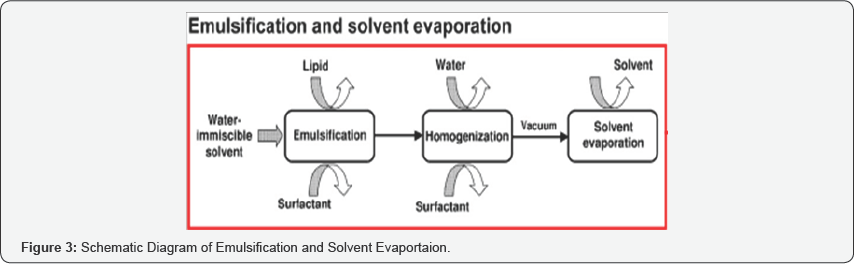
Solvent Emulsification-Evaporation Method
In solvent emulsification-evaporation method, the lipophilic material and hydrophoDic drug were dissolved in a water immiscible organic solvent (e.g. cyclohexane, dichloromethane, toluene, chloroform) and then that is emulsified in an aqueous phase using high speed homogenizer. To improve the efficiency of fine emulsification, the coarse emulsion was immediately passed through the microfluidizer. Thereafter, the organic solvent was evaporated by mechanical stirring at room temperature and reduced pressure (e.g. rotary evaporator) leaving lipid precipitates of SLNs [4]. The big advantage of this method is the avoidance of any thermal stress, which makes it appropriate for the incorporation of highly thermo labile drugs. A clear disadvantage is the use of organic solvent which may interact with drug molecules and limited the solubility of the lipid in the organic solvent [17]. Deepthi Soma, prepared irbesartan solid lipid Nanoparticles using glyceryl monostearate by solvent emulsification method followed by probe sonication. Formulation was then further evaluated for the pharmacokinetic studies in Wistar rats. Irbesartan-loaded SLN of particle size 523.7nm and 73.8% entrapment efficiency showed good bioavailability in Wistar rats and also showed optimum stability in the studies. The SLN prepared using glyceryl monostearate by solvent emulsification method leads to improve bioavailability of the drug [18] (Figure 3).
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Supercritical Fluid Method
The supercritical fluid has unique thermo-physical properties which can be finely tuned by small changes in the pressure. As the pressure raises the density and the ability of the fluid to dissolve compounds increases while the viscosity remains relevantly constant. Accordingly under high pressure and appropriate temperature in the supercritical range the fluid can act as an alternative to organic solvents and dissolve different APIs and lipids [14]. Super critical carbon dioxide has a tendency to dissolve the lipophilic drugs, by combination with ultrasonication technique it can be used to prepare SLNs. Xionggui loaded SLNs have been prepared by using super critical carbon dioxide fluid extraction and ultrasonication [19].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Microemulsion Based Methods
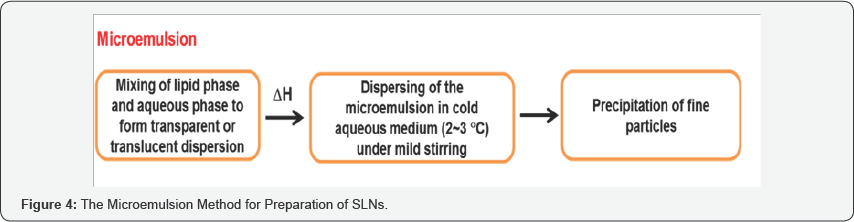
Gasco et al. (1997) developed SLNs based on the dilution of microemulsions [8]. This method is based on the dilution of microemulsions. As micro-emulsions are two-phase systems composed of an inner and outer phase (e.g. o/w microemulsions). They are made by stirring an optically transparent mixture at 6570 °C, which typically composed of a low melting fatty acid (e.g. stearic acid), an emulsifier (e.g. polysorbate 20), co-emulsifiers (e.g. butanol) and water. The hot microemulsion is dispersed in cold water (2-3 °C) under stirring [4]. According to De Labouret et al. [20] the particle size is critically determined by the velocity of the distribution processes. Nanoparticles were produced only with solvents which distribute very rapidly into the aqueous phase (acetone), while larger particle sizes were obtained with more lipophilic solvents (Figure 4).
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Spray Drying Method
Spray drying method is a cheaper method than lyophilization. This method causes particle aggregation due to high temperature, shear forces and partial melting of the particle [21]. Freitas C et al. [22] recommends the use of lipid with melting point >70 °C for spray drying, The best result by spray drying method was obtained with SLN concentration of 1% in a solution of trehalose in water or 20% trehalose in ethanol water mixtures (10/90 v/v) [21,22].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Double Emulsion Method
Novel method based on solvent emulsification evaporation has been used for preparation of hydrophilic loaded SLNs [8]. Drug (mainly hydrophilic drugs) was dissolved in aqueous solution, and then was emulsified in melted lipid. This primary emulsion was stabilized by adding stabilizer (e.g. gelatin, poloxamer-407). Then this stabilized primary emulsion was dispersed in aqueous phase containing hydrophilic emulsifier (e.g. PVA). Thereafter, the double emulsion was stirred and was isolated by filtration [4]. ShuYu Xie prepared hydrophilic protein- loaded SLN by w/o/w double emulsion and solvent evaporation techniques. The results showed that PLGA was essential for the primary w/o emulsification. In addition, the stability of the w/o emulsion, the encapsulation efficiency and loading capacity of the nanoparticles were enhanced with the increase of PLGA concentration. Furthermore, increasing PLGA concentration decreased zeta potential significantly but had no influence on particle size of the SLN. In vitro release study showed that PLGA significantly affected the initial burst release, i.e. the higher the content of PLGA, the lower the burst release [23].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
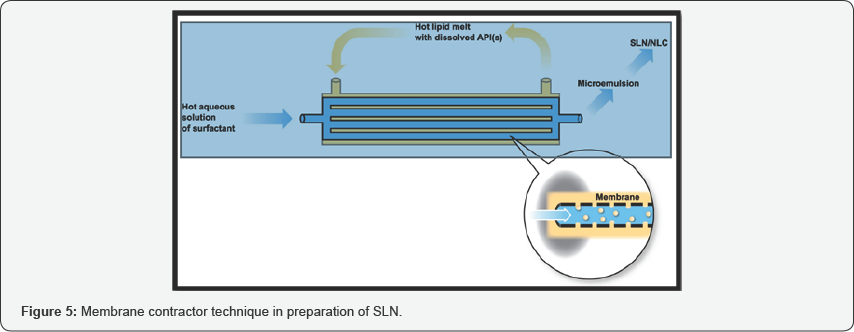
Membrane Contactor Method
In this method membrane contactor was used to prepare SLNs, lipid was pressed at the temperature above the melting point of lipid through the membrane pores, water circulated beyond the pores flow with the produced droplets of melted lipid which was further cooled at room temperature [19]. The advantages of this process of SLN preparation using a membrane contactor are shown to be its facility of use, the control of the SLN size by an appropriate choice of process parameters and it's scaling up ability [4]. The membrane contactor method is also used for the preparation of polymeric nanoparticles, by methods involving a polymerization of dispersed monomers (interfacial polymerization method) or a dispersion of preformed polymers (nano precipitation method) (Figure 5).
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Solvent Injection Technique
In this technique, the solid lipid was dissolved in water- miscible solvent (e.g. ethanol, acetone, isopropanol) or a water- miscible solvent mixture. Then this organic solvent mixture was slowly injected through an injection needle in to stirred aqueous phase with or without surfactant. Then the dispersion was filtered with a filter paper in order to remove any excess lipid [19]. The presence of surfactant within the aqueous phase helps to produce lipid droplets at the site of injection and stabilize the formed SLNs until solvent diffusion was complete by reducing the surface tension. Solvent injection lyophilization method was used to prepare cinnarizine SLNs, a lipophilic drug.
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Applications of SLN-SLNs for Chemotherapy
Cancer is characterized by the formation of abnormal tissues known as neoplasm. Developed basically due to change in the way cells proliferate and differentiate. Currently, cancer fighting drugs are toxic to both tumor and normal cells, thus the efficacy of chemotherapy is always limited by the side effects of the drug [24].
Use of nanotechnology in cancer biology has provided hope within scientific communities of developing novel cancer therapeutic strategies. Some nanoscale devices can be targets to the cancer cells. This increases the selectivity of the drugs toward the cancer cells and will reduce the toxicity for normal tissue. There are many reports describing potentials of lipid nanoparticles for parentral delivery particularly for the treatment of cancer. Over the past couple of decades, a huge amount of detailed data have been amassed regarding the basic biological processes that become perturbed in cancer, such as disturbances in growth-factor binding, signal transduction, gene transcription control, cell-cycle checkpoints, apoptosis, and angiogenesis. These in turn have prompted the search for rational anticancer drugs and produced a record number of novel compounds, currently being used in cancer treatment trials [25,26]. In another research tamoxifen citrate loaded nanoparticles were administered by intravenous injection in rats and the pharmacokinetic parameters were determined. The t1/2 and mean residence time of TC-loaded SLNs in plasma was about 3.5-folds (p < 0.001) and 3-fold (p < 0.001) higher, respectively than free tamoxifen, this indicates the potential of TC-loaded SLNs as a long circulating system in blood. Thus the above mentioned solid lipid nanoparticles can be a beneficial system to deliver tamoxifen to cancer tissues through enhanced permeability and retention (EPR) effect [27].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
SLNs for Topical use
Corticosteroids are therapeutic agents generally used in the treatment of skin diseases such as eczema or psoriasis. Topical SLN products show enormous prospective for treating dermatological conditions by targeting corticosteroids to dermal disease sites while decreasing systemic drug absorption. Topical application of the drugs at the pathological sites offers possible advantages of delivering the drug directly to the site of action [15]. SLNs used for topical application for various drug such as anticancer, vitamin-A, isotretinoin, flurbiprofen. Using glyceryl behenate, vitamine A-loaded nanoparticles can be prepared. This method is useful for the improvement of penetration with sustained release. The isotretinoin-loaded lipid nanoparticles were formulated for topical delivery of drug. Production of the flurbiprofen-loaded SLN gel for topical application offer a potential advantage of delivering the drug directly to the site of action, which will produce higher tissue concentrations [28-30]. Dermal delivery of Doxorubicin (Dox) would be an ideal way in maximising drug efficiency against skin cancer accompanying with minimising side effects [31].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Oral SLN in Antitubercular Chemotherapy
Antitubercular drugs such as rifampsin, isoniazide, pyrazinamide-loaded SLN systems were able to reduce the dosing frequency and improve patient compliance. Antitubercular drugs loaded SLNs were prepared using solvent diffusion technique [4].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
SLNS as Cosmeceuticals
Cosmeceuticals is rising as the major application target of these carriers. Carrier systems like SLNs and NLC were formulated with a point of view to meet manufacturing needs like scale up, qualification and validation, simple technology, low cost etc [15]. The SLNs have been applied in the preparation of sunscreens and as an active carrier agent for molecular sunscreens and UV blockers. SLN and NLCs have proved to be controlled release innovative occlusive topicals. Better localization has been achieved for vitamin A in upper layers of skin with glyceryl behenate SLNs compared to conventional formulations [32]. The first two cosmetic products containing lipid nanoparticles were introduced to the market in 2005 [15].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
SLNs as Gene Vector Carrier
SLN can be used in the gene vector formulation. There are several recent reports of SLN carrying genetic/peptide materials such as DNA, plasmid DNA and other nucleic acids The gene transfer was optimized by incorporation of a diametric HIV-1 HAT peptide into SLN gene vector. The lipid nuclic acid nanoparticles were prepared from a liquid nanophase containing water and a water miscible organic solvent where both lipid and DNA are separately dissolved by removing the organic solvent, stable and homogeneously size dlipid-nuclic acid nanoparticle (70-100nm) were formed. It's called genospheres. It is targeted specific by insertion of an antibody-lipo polymer conjugated in the particle [21].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
SLNs in Breast Cancer and Lymph Node Metastases
Mitoxantrone-loaded SLN local injections were formulated to reduce the toxicity and improve the safety and bioavailability of drug efficacy of doxorubicin (Dox) has been reported to be enhanced by incorporation in SLNs. In the methodology the Dox was complexed with soybean -oil-based anionic polymer and dispersed together with a lipid in water to form Dox-loaded solid lipid nanoparticles. The system has enhanced its efficacy and reduced breast cancer cells [1,33].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
SLNs have been reported to be useful as drug carriers to treat neoplasm's. Tumour targeting has been achieved with SLNs loaded with drugs like methotrexate and Camptothecin. Tamoxifen an anticancer drug is incorporated in SLN to prolong release of drug after iv [34].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Stealth Nanoparticles
These provide a novel and unique drug-delivery system they evade quick clearance by the immune system. Such nanoparticles can target specific cells. Stealth SLNs have been successfully tested in animal models with marker molecules and drugs. Antibody labelled stealth Lipobodies have shown increased delivery to the target tissue in accessible sites [35].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Diabetes
Diabetes mellitus is one of the most common metabolic diseases worldwide. Hyperglycemia caused by diabetes is a serious pathologic condition producing neurological and CV damage. Researchers focus considerable attention on SLNs as the carriers to protect peptides and proteins known for their sensitivity to various environmental factors such as pH, temperature, and ionic strength [36]. Zhang et al designed SLNs coated with stearic acid octaarginine as carriers for insulin. Octaarginine is a cell-penetrating peptide that can facilitate cellular uptake of some drugs [37]. The size and insulin encapsulation of the octaarginine-coated SLNs were 162nm and 77%, respectively. Octaarginine-coated and noncoated SLNs increased Caco-2 cell uptake by 2.3 times and 18.4 times, respectively. The SLNs containing octaarginine showed a significantly higher hypoglycemic effect (3-fold) in rats compared to noncoated SLNs. Oral delivery of insulin may significantly improve the quality of life of diabetes patients who routinely receive insulin by the subcutaneous route [38-40].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
SLNs for Potential Agriculture Application
Essential oil extracted from Artemisia arboreseens L when incorporated in SLN, were able to reduce the rapid evaporation compared with emulsions and the systems have been used in agriculture as a suitable carrier of ecologically safe pesticides [41].
Infection
Infection can cause host tissues to react to organisms and the toxins they produce. Nanocarriers can be effective drug delivery systems for treating infections [42]. Among the different types of nanosystems, SLNs were widely applicable for carrying antiinfection drugs to treat bacterial, fungal, viral, and parasitic infection. Lopinavir is a human immunodeficiency virus (HIV) protease inhibitor used in antiretroviral therapy. SLNs can act as a feasible carrier for lopinavir because of P-glycoprotein efflux and first-pass metabolism. The lopinavir-loaded SLNs composed of stearic acid were stable at 4_C for 4 months based on particulate size and the release profile [43]. Higher oral bioavailability was obtained for SLNs (2.5-fold) in comparison with lopinavir solution because of higher lymphatic delivery. In another study, Compritol 888 ATO was used as the solid lipid for preparing lopinavir-loaded SLNs [44]. The drug release showed a delayed pattern both in 0.1NHCl (pH 1.2) and phosphate buffer (pH 6.8). The SLNs could bypass Pglycoprotein efflux to reach systemic circulation, leading to a 3.6- and 4.9-fold increase in bioavailability and Cmax compared to solution.
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Evaluation of SLN
in vitro drug release
Dialysis tubing: in vitro drug release could be achieved using dialysis tubing. The solid lipid nanoparticle dispersion is placed in pre-washed dialysis tubing which can be hermetically sealed. The dialysis sac is then dialyzed against a suitable dissolution medium at room temperature, the samples are withdrawn from the dissolution medium at suitable intervals, centrifuged and analyzed for the drug content using a suitable analytical method.
Reverse dialysis: In this technique a number of small dialysis sacs containing 1ml of dissolution medium are placed in SLN dispersion. The SLN's are then displaced into the medium.
Franz diffusion cell: The SLN's dispersion is placed in the donor chamber of Franz diffusion cell fitted with a cellophane membrane. The dispersion is then analyzed against a suitable dissolution medium; the samples are withdrawn from the dissolution medium at suitable intervals and analyzed for drug content using suitable methods like spectroscopy and HPLC methods [4,45].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Characterization of SLN’s
Particle size analysis and Zeta potential
Many techniques are available for particle size analysis and zeta potential like scanning electron microscopy (SEM), atomic force microscopy (AFM), scanning tunneling microscopy (STM) and photon correlation spectroscopy (PCS) [32]. Photon correlation spectroscopy (PCS) and laser diffraction (LD) are the most powerful techniques for determination of particle size. PCS (also known as dynamic light scattering) measures the fluctuation of the intensity of the scattered light, which is caused by particle movement [8].
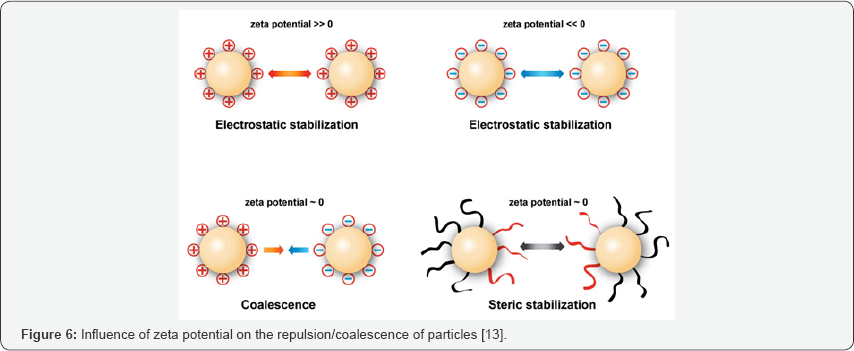
Zeta potential
Zeta potential measurement can be carried out using zeta potential analyzer or zetameter. Zeta potential gives information about the magnitude of the electrostatic repulsion or attraction between particles in the aqueous suspension of SLN. Zeta potential can serve as an important parameter in the predictions for long term stability of the formulations. High values of zeta potential (e.g., more than+30mV or less than-30mV) can stabilize the colloidal suspension by electric repulsion, Electric repulsion generally results in less contact between the particles and less aggregation. For example colloidal systems that contain steric stabilizers can express good long term stability even in cases when zeta potential is as low as around 0mV [14] (Figure 6).
Electron microscopy
Scanning electron microscopy (SEM) and transmission electron microscopy (TEM) provide way to directly observe nanoparticles. SEM is however better for morphological examination. TEM has a small size limit of detection. Transition electron microscopy and light microscopy both are based on same principle but one difference is that in light microscopy light is used instead of electron [8].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Differential Scanning Calorimetry (DSC)
Differential Scanning Calorimetry (DSC) is a widely used technique that measures differences in the amount of heat required to increase the temperature of a sample compared to a reference. Differences in heat flow may be positive or negative and are presented as function of the temperature. At phase transition there are differences in the sample compared to the reference [14]. The rate of crystallinity using DSC is estimated by comparison of the melting enthalpy/g of the bulk material with the melting enthalpy/g of the dispersion [8,46].
Nuclear magnetic resonance (NMR)
NMR can be used to determine both the size and the qualitative nature of Nanoparticles. The selectivity afforded by chemical shift complements the sensitivity to molecular mobility to provide information on the physicochemical status of components within the nanoparticle [47].
X-ray diffraction
A useful technique to exclude aggregate of more than 1|im and substantial polymorphic pi transition form to stable; thus help in characterizing the crystalline nature of the compound and determine the polymorphic shifts present [48]. X-ray diffraction (XRD) play a prominent role because they are able to provide structural information on the dispersed particles [49].
Entrapment efficiency
The entrapment efficiency of the drug is determined by measuring the concentration of free drug in the dispersion medium. Ultracentrifugation was carried out using Centrisart, which consist of filter membrane (molecular weight cutoff 20,000Da) at the base of the sample recovery chamber. The SLNs along with encapsulated drug remain in the outer chamber and aqueous phase moves into the sample recovery chamber. The amount of the drug present in the aqueous phase is determined by HPLC or UV spectrophotometer [50-52].
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
Conclusion and Future Perspective
SLN constitute an attractive colloidal drug carrier system due to successful incorporation of active compounds and their related benefits. The present review has focused on increasing awareness about nano technological field in drug delivery with the emergence of several promising approaches like solid lipid nanoparticles, nano structured lipid carriers, lipid drug conjugates etc. for improving medical therapeutics.. SLNs have already been proven as good formulations in cosmeceuticals and other allied fields, they must occupy a considerable place in the pharmaceutical market. To exploit the broad applications of lipid based nanoparticulate formulations, it is essential that the pharmaceutical industries specialized in the development of new drug delivery systems should engage in novel formulation technology to promote their scale up and bring them onto the pharmacist's shelves. SLN offer an economical and patient- friendly device for administration of drugs by various routes to maximize effectiveness while avoiding adverse effects on nontarget tissues.
For more than 20 years of research the current and future applications of SLN seem well shaped. In parentral formulations they will offer more possibilities for many drugs with poor aqueous solubility, short half-life and low chemical stability. Moreover SLN are likely to find more applications as targeted drug delivery systems which will “direct” the drug molecules to specific organs of interest and to reduce the systemic toxicity. Thus they can provide solutions for APIs that failed clinical tests due inappropriate tissue localization.
- Review Article
- Abstract
- Introduction
- Advantages of SLN
- Disadvantage of SLN
- Principle of Drug Release from SLN
- Formulation of SLN
- Methods ofPreparation-High Pressure Homogenization
- Solvent Emulsification-Evaporation Method
- Supercritical Fluid Method
- Microemulsion Based Methods
- Spray Drying Method
- Double Emulsion Method
- Membrane Contactor Method
- Solvent Injection Technique
- Applications of SLN-SLNs for Chemotherapy
- SLNs for Topical use
- Oral SLN in Antitubercular Chemotherapy
- SLNS as Cosmeceuticals
- SLNs as Gene Vector Carrier
- SLNs in Breast Cancer and Lymph Node Metastases
- SLNs as a Targeted Carrier for Anticancer Drug to Solid Tumors
- Stealth Nanoparticles
- Diabetes
- SLNs for Potential Agriculture Application
- Evaluation of SLN
- Characterization of SLN’s
- Differential Scanning Calorimetry (DSC)
- Conclusion and Future Perspective
- References
References
- Pavankumar AR, Parthiban S (2014) A modern review on solid lipid nanoparticles as novel controlled drug delivery system. Ijrpns 3(4): 313-325.
- Sawant KK, Dodiya SS (2008) Recent advances and patents on solid lipid nanoparticles. Recent Pat Drug Deliv 2(2): 120-135.
- Lin CH, Chen CH, Lin ZC, Fang JY (2017) Recent advances in oral delivery of drugs and bioactive natural products using solid lipid nanoparticles as the carriers. J Food Drug Anal 25(2): 219-234.
- Ramteke KH, Joshi SA, Dhole SN (2012) Solid lipid nanoparticle: A review. IOSR Journal of Pharmacy 2(6): 34-44.
- Bhattacharjee A (2013) Solid lipid nanoparticles technology as a novel platform for delivery of drugs. Indo Am j pharm Res 3(5): 4079-4097.
- Annette ZMH, Schwarz C, Wolfgang M (1998) Solid lipid nanoparticles (SLN) for controlled drug delivery-drug release and release mechanism. Eur J Pharm Biopharm 45(2): 149-155.
- Andrew L (2009) Solid lipid nanoparticles for the delivery of pharmaceutical actives. Drug deliv technol 9(8): 1-5.
- Akanksha G, Deepti S, Navneet G (2012) Solid lipid nanoparticles (SLN): method, characterization and applications. icpj 1(11): 384-393.
- Wolfgang M, Karsten M (2001) Solid lipid nanoparticles production, characterization and applications. Adv Drug Deliv Rev 47(2-3): 165196.
- Geszke-Moritz M, Mortiz M (2016) Solid lipid nanoparticles as attractive drug vehicles: composition, properties and therapeutic strategies. Mater Sci Eng C Mater Biol Appl 68: 982-994.
- Mangesh B, Prashant U, Ashwini M (2017) Formulation and characterization of solid lipid nanoparticles for an anti-retroviral drug darunavir. Appl Nanosci 7(1-2): 47-57.
- Dolatabadia JEN, Hamishehkar H, Eskandani M, Valizadeh H (2014) Formulation, characterization and cytotoxicity studies of alendronate sodium-loaded solid lipid nanoparticle. Colloids Surf B Biointerfaces 117: 21-28.
- Gamze G, Kutlu HM, Genc L (2014) Preparation and characterization of ascorbic acid loaded solid lipid nanoparticles and investigation of their apoptotic effects. Colloids Surf B 121: 270-280.
- Svilenov H, Tzachev C (2009) Solid lipid nanoparticles- A promising drug delivery system. Nanomedicine pp. 187-237.
- Rahul N, ArunKumar KS, Priya KV (2011) Recent advances in solid lipid nanoparticle based drug delivery systems, J Biomed Sci and Res 3(2): 368-384.
- YiFan L, Chen DW, Ren L, Zhao X, Qin J, et al. (2006) Solid lipid nanoparticles for enhancing vinpocetine's oral bioavailability. J Control Release 114: 53-59.
- Sjostrom B, Bergenstahi B (1992) Preparation of submicron drug particles in lecithin-stabilized o/w emulsions I model studies of the precipitation of cholesteryl acetate. Int J Pharm 88(1-3): 53-62.
- Deepthi S, Zenab A (2017) Solid lipid nanoparticles of Irbesartan: Preparation, characterization, optimization and pharmacokinetic studies. Braz J Pharm Sci 53(1): 1-10.
- Kumar S, Randhawa JK (2013) High melting lipid based approach for drug delivery: solid lipid nanoparticles. Materials Science and Engineering C 33(4): 1842-1852.
- Labouret DA, Thioune O (1995) Application of an original process for obtaining colloidal dispersions of some coating polymers. preparation, charecterization, industrial scaling up. Drug Develop Ind Pharm 21(2): 229-241.
- Jawahar N, Gowtham MSNR, Sumeet S (2012) Solid lipid nanoparticles for oral delivery of poorly soluble drugs. J Pharm Sci & Res 4(7): 1848-1855.
- Freitas C, Muller RH (1998) Spray-drying of solid lipid nanoparticles. Eur J Pharma & biopharm 46(2): 145-151.
- Xie SY, Wang SL, Zhao BK, Han C, Wang M, et al. (2008) Effect of PLGA as a polymeric emulsifier on preparation of hydrophilic protein-loaded solid lipid nanoparticles. Colloids Surf B Biointerfaces 67(2): 199-204.
- Jaiswal S, Gupta GD (2013) Recent advances in solid lipid nanoparticles and challenges. Iajpr 3(12): 1601-1611.
- Nasimudeen R, Tabrez JS, Ashraf GMD, Shakil S, Damanhouri GA, et al. (2012) Nanotechnology-based approaches in anticancer research. Int J Nanomedicine 7: 4391-4408.
- Mathur V, Satrawala Y, Rajput MS, Kumar P, Shrivastava P, et al. (2010) Solid lipid nanoparticles in cancer therapy. Int J Drug Deliv 2(3): 192199.
- Hashem FM, Mohamed N, Khairy A (2014) in vitro cytotoxicity and bioavailability of solid lipid nanoparticles containing tamoxifen citrate. Pharm Dev Technol 19(7): 824-832.
- Jenninga V, schafer-Korting M, Gohla S (2000) Vitamin a-loaded solid lipid nanoparticles for topical use: drug release properties. J Control Release 66(2-3): 115-126.
- Santos MC, Mehnert W, Schaller M, Korting HC, Gysler A, et al. (2002) Drug targeting by solid lipid nanoparticles for dermal use. J Drug Target 10(6): 489-495.
- Almeida LN, Araujo TG (2017) Solid lipid nanoparticles: the efficiency carrier for topical delivery of hydrophilic drugs. Wjpps 6(9): 175-189.
- Tupal A, Sabzichi M, Ramezani F, Kouhsoltani M, Hamishehkar H, et al. (2016) Dermal delivery of doxorubicin-loaded solid lipid nanoparticles for the treatment of skin cancer. J Microencapsul 33(4): 372-380.
- Patwekar S, Surendra G, Giri R (2014) Review on nanoparticles used in cosmetics and dermal products. Wjpps 3(8): 1408-1421.
- Soni K, Kukereja BK, Kapur M (2015) Lipid nanoparticles: future of oral drug delivery and their current trends and regulatory issues. Ijcpr 7(1): 1-18.
- Ruckmani K, Sivakumar M, Ganeshkumar PA (2006) Methotrexate loaded solid lipid nanoparticles (SLN) for effective treatment of carcinoma. J Nanosci Nanotechnol 6(9-10): 2991-2995.
- Wang Y, Wei W (2006) In situ evading of phagocytic uptake of stealth solid lipid nanoparticles by mouse peritoneal macrophages. Drug Deliv 13(3): 189-192.
- Almeida AJ, Souto E (2007) Solid Lipid Nanoparticles As A Drug Delivery System For Peptides And Proteins. Adv Drug Deliv Rev 59(6): 478-490.
- Zhang ZH, Zhang YL, Zhou JP, Lv HX (2012) Solid Lipid Nanoparticles Modified With Stearic Acid-Octaarginine for Oral Administration of Insulin. Int J Nanomed 7: 3333-3339.
- Moran G, Valeria F, David A (2016) conjugates of HA2 with octaarginine- grafted HPMA copolymer offer effective sirna delivery and gene silencing in cancer cells. Eur J Biopharm 109: 103-112.
- Fonte P, Araujo F (2013) Oral insulin delivery: how far are we? J Diabetes Sci Technol 7(2): 520-531.
- Alai MS, Lin WJ, Pingale SS (2015) Application of polymeric nanoparticles and micelles in insulin oral delivery. J Food Drug Anal 23(3): 351-358.
- Francesco L, Wissing SA, Muller RH, Fadda AM (2006) Artemisia arborescens l essential oil-loaded solid lipid nanoparticles for potential agricultural application: preparation and characterization. AAPS PharmSciTech 7(1): E2.
- Aljuffali IA, Huang CH, Fang JY (2015) Nanomedical strategies for targeting skin microbiomes. Curr Drug Metab 16(4): 255-271.
- Preziosi P (2007) Isoniazid: metabolic aspects and toxicological correlates. Curr Drug Metab 8(8): 839-851.
- Negi Js, Chattopadhyay P, Sharma Ak, Ram V (2013) Development of solid lipid nanoparticles (SLNs) of lopinavir using hot self nano- emulsification (SNE) technique. Eur J Pharm Sci 48(1-2): 231-239.
- Mahajan A, Sandeep K (2013) Solid lipid nanoparticles (SLNs) - As novel lipid based nanocarriers for drugs. Int J Adv Res 2(1): 433-441.
- Nair RK, Priya V, Kumar KSA, Badivaddin TMD, Sevukarajan M, et al. (2011) Formulation and evaluation of solid lipid nanoparticles of water soluble drug: isoniazid. J Pharm Sci & Res 3(5): 1256-1264.
- Ekambaram P, Sathali AAH, Priyanka (2012) Solid lipid nanoparticles: a review. Srcc 2(1): 80-102.
- Andreas N, Hauseb G, Mader K (2012) Physicochemical characterization of curcuminoid-loaded solid lipid nanoparticles. Int J Pharm 423(2): 440-451.
- Bunjes H, Unruh T (2007) Characterization of lipid nanoparticles by differential scanning calorimetry, x-ray and neutron scattering. Adv Drug Deliv Rev 59(6): 379-402.
- Venkateswarlu V, Manjunath K (2004) Preparation, characterization and in vitro release kinetics of clozapine solid lipid nanoparticles. J Control Release 95(3): 627-638.
- Xue J, Wang T, Hu Q, Mingyong Z, Yangchao L, et al. (2017) A novel and organic solvent-free preparation of solid lipid nanoparticles using natural biopolymers as natural emulsifier and stabilizer. I j pharm 531(1): 59-66.
- Majekodunmi SO (2015) A review on lozenges. Am J Med Sci 5(2): 99104.
- Xiao Wu, Guy RH (2011) Application of nanoparticles in topical drug delivery and in cosmetics. Journal of Drug Delivery Science and Technology 19(6): 1-55.






























