Formulation and Evaluation of Floating Gastro Retentive Glipizide Tablets
Uddhav Bagul S1*, Yogesh Shirsath A1and Kishore Gujar N2
1Sinhgad Institute of Pharmacy, Narhe, India
2Sinhgad College of Pharmacy, Vadgoan, India
Submission: July 01, 2017; Published: August 04, 2017
*Corresponding author: Uddhav S Bagul, Sinhgad Institute of Pharmacy, Narhe, Pune (MS) India, Tel: +91 9552595800; Email: usbagul.siop@yahoo.in
How to cite this article: Uddhav B S, Yogesh S A, Kishore G N. Formulation and Evaluation of Floating Gastro Retentive Glipizide Tablets. Mod Appl Bioequiv Availab. 2017; 1(5): 555573.
Abstract
Glipizide, a BCS class II drug commonly prescribed for the type II diabetes, as an oral hypoglycaemic agent. But its insolubility in water leads to low oral bioavailability due to limiting dissolution rate. Therefore, the solubility of glipizide was increased by solid dispersion method followed by formulation of floating tablets using 32 full factorial designs. Solid dispersion of PEG 4000 and 6000 with glipizide at different ratio was prepared by fusion method. The floating tablets were prepared by direct compression method, using HPMC K4M, HPMC K15M and sodium bicarbonate was used to maintain buoyancy. The floating tablets were evaluated for various physiochemical properties and in vitro drug release studies.
The saturated solubility of pure glipizide was 7.9^g/ml which was enhanced to 204.3^g/ml, after preparation of solid dispersion, in 1:6 ratios with PEG 6000. The glipizide-PEG complex was confirmed by FT-IR spectroscopy and DSC thermo gram. All the formulations showed floating lag time 73-145 seconds, floating duration more than 24 hours and drug content was found in the range of 95.41 to 99.02%. Batch number F7 showed 61.48% of in vitro drug release in 8 hours hence, batch F7 was compared with marketed Glynase XL and showed 51.58% similarity factor. In vitro release kinetics of batch F7 followed the zero order release and super class II transport diffusion
Keywords: Glipizide; Solid dispersion; Factorial design; Floating tablets; Buoyancy; Floating time
Introduction
From past few decades' greater attention have been focused on development of sustained release (SRJ or controlled release (CRJ drug delivery systems due to Complications and expense involved in marketing of new drug entities [1]. The real challenge in the development of an oral controlled-release drug delivery system is not just to sustain the drug release but also to prolong the presence of the dosage form within the gastrointestinal tract (GIT) until all the drug is completely released at the desired period of time which over comes problem of gastric retention as in case of conventional oral delivery. Indeed, gastric drug retention is receiving significant interest now a day [2]. Gastro retentive drug delivery systems are the systems which are retained in the stomach for a longer period of time and thereby improve the bioavailability of drugs that are preferentially absorbed from upper GIT [3]. Various approaches have been pursued to increase the retention of an oral dosage form in the stomach, including floating systems [4,5] and expanding systems bio adhesive systems, modified-shape system and high-density systems [6]. The floating drug delivery systems, designed on the basis of delayed gastric emptying and buoyancy principles, appear to be an effective and rational approach to the modulation of controlled oral drug delivery. These systems were useful for those drugs that act locally in the proximal part of gastrointestinal tract or are poorly absorbed in the intestine. These dosage forms have a bulk density lower than that of the gastric fluid. After oral administration, they can remain in the stomach and deliver drugs in a sustained release manner [7].
Solid dispersion technique was selected as it was utilized in limited number of researches to increase the solubility of glipizide. It has been widely used to improve the dissolution rate, solubility and oral absorption of poorly water-soluble drugs [8,9]. In solid dispersions, the particle size of the drugs was reduced, thewettability and the dispersibility were enhanced; therefore, drug dissolution was improved markedly [10]. Glipizide is an oral hypoglycemic agent, which is a commonly prescribed drug for the treatment of patients with type II diabetes. Glipizide is a weak acid (pKa=5.9) practically insoluble in water and acidic environment and highly permeable (class II) drugs according to the Biopharmaceutical Classification System (BCS) [11]. The oral absorption is uniform, rapid and complete with a bioavailability of nearly 100% and an elimination half-life of 2-4 hours. Glipizide is reported to have a short biological half-life (3.4±0.7 hours) requiring it to be administered in 2 to 3 doses of 2.5 to 10mg per day. SR formulations that would maintain plasma levels of drug for 8 to 12 hours might be sufficient for once a day dosing for glipizide. SR products are needed for glipizide to prolong its duration of action and to improve patient compliance [12].
Materials and Methods
Materials
Glipizide was generously gifted by USV, Ltd. Mumbai, India. HPMC (K4M, K15M) and microcrystalline cellulose were obtained from Signet, Pune, India. Sodium bicarbonate, magnesium stearate, PEG 4000, 6000 and talc were purchased from Thomas Baker, Mumbai.
Method
Preparation of the glipizide PEG complex: Solid dispersions (SDs) at various weight ratios1:1, 1:2, 1:3, 1:4, 1:5, and 1:6 were prepared by melting method. Glipizide was added to the molten base comprising of PEG4000 and PEG 6000 respectively. The blend was heated 10 °C above the melting point of each carrier for 5min with continuous stirring. The systems were placed 24 hours for drying. The mass was crushed, ground gently with a mortar and pestle and passed through sieve# 40.
Evaluation of Glipizide PEG complex Solubility measurements: Saturated solutions were prepared by adding the glipizide to 0.1 NHcl. It was performed by adding gradually amount of glipizide to the solution until undisclosed glipizide was present after 24h of stirring on a magnetic stirrer. The solutions were filtered using a cellulose acetate membrane (0.45|im). The concentrations of the glipizide were determined spectroscopic ally with UV/Vis Spectrophotometer at wavelength 276nm [13].
Glipizide-PEG complex study
The infrared spectra of pure drug (glipizideJ and drug-PEG complex (1:1) were recorded between 400 and 4000cm-1 by FT- IR spectrometer (Jasco 4100 series) using KBr pellet technique. Similarly DSC thermo gram of above combination was recorded and interpreted [14].
Preparation of Tablets
All the tablets were prepared by direct compression method. All the ingredients (Table 1) were passed through sieveno. 40# and blend in an octagonal blender for 10min. Magnesium stearate was used to lubricate the blend. The lubricated blend was then compressed on 12 stations rotator tablets machine (CIPS machinery India) using single 8mm flat punches.
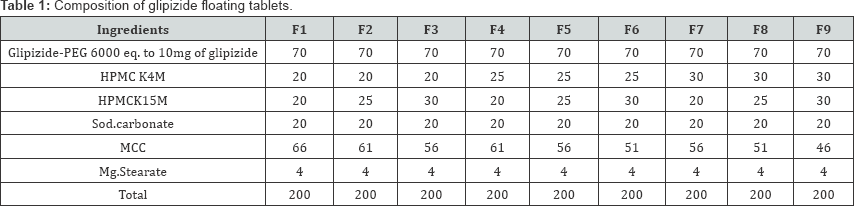
Evaluation of Tablets
Floating lag time and buoyancy time: The tablets were placed in a 100ml beaker containing 100ml 0.1 NHcl. The time required for the tablet to rise to the surface and float was taken as the floating lag time. The time for which tablets kept floating was termed as 'buoyancy time' of the tablets which was determined for all the formulations [3].
Percentage of drug content: Twenty Tablets were weighed individually and the drug was extracted in 0.1 NHcl followed by filtration through 0.45|im. The solution was analysed by using spectrophotometer at 276mm.
Swelling study: Water uptake study of the dosage form is conducted by using USP dissolution apparatus-II in 900ml of distilled water which is maintained at 37±0.5 °C, rotated at 100rpm. At selected regular intervals, the tablet is withdrawn and weighed. Percentage swelling of the tablet is expressed as percentage water uptake (%WU) or % Swelling index [2].
% Swelling index= Wt - Wo* 100
Wt
Where,
Wt - weight of the swollen tablet,
Wo - initial weight of the tablet
In vitroRelease Studies: Drug release studies of the prepared floating tablets as well as the commercially available Glynase XL 10mg (USV Ltd) tablets were performed, in triplicate, in a USP dissolution tester apparatus, type- II (Paddle method) at 37 °C ± 0.5 °C and 100 rpm. The tablets were placed into 900ml of 0.1NHcl solutions (pH 1.2). The drug content was determined spectrophotometrically at a wavelength of 276mm.
Kinetic modelling of drug release profiles: The drug release kinetics was studied by plotting the data obtained from the in vitro drug release in various kinetic models like zero order, first order, Higuchi, and Hixson-Crowell model. The model with the highest correlation coefficient was considered to be the best fitting one.
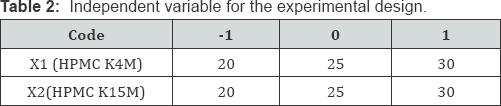
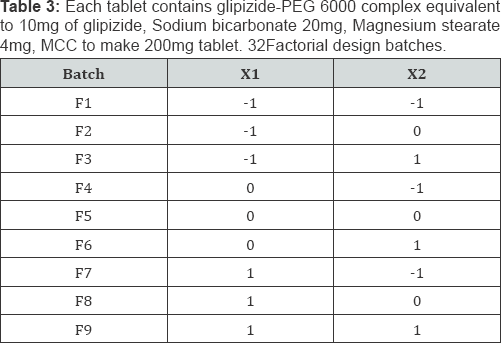
Factorial design: A 32 full factorial design was constructed to study the effect of the amount of HPMC K4M(X1) and the amount of HPMC K15M(X2) on the drug release from gastro retentive glipizide tablets (Table 2 & 3). The dependent variables chosen were % drug release and floating lag time (FLT). A statistical model incorporating interactive and polynomial terms was utilized to evaluate the response.
Y = b0 + b1X1 + b2X2 + b12X1X2 + b11X12 + b22X22
Where Y is the dependent variable, b0 is the arithmetic mean response of the 9 runs, and bi is the estimated coefficient for the factor Xi. The main effects (X1 and X2) represent the average result of changing one factor at a time from its low to high value. The interaction terms (X1X2) show how the response changes when 2 factors are changed simultaneously. The polynomial terms (X12 and X22) are included to investigate nonlinearity [15-17].
Results and Discussion
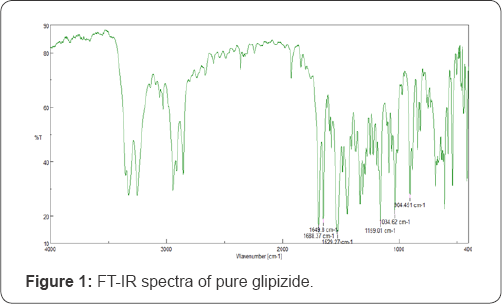
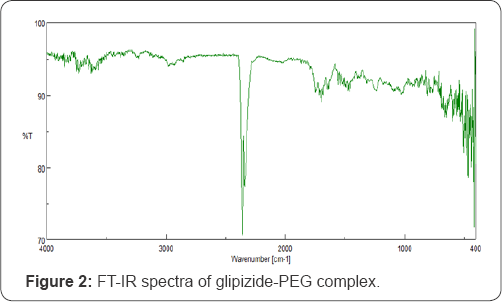
The solubility of glipizide was increased as a linear function of carrier concentration. Solid dispersion prepared by PEG 6000 (1:6 ratio), showed 204.3 ppm solubility which is 2 5 times more than solubility of pure glipizide. The FT-IR spectrum of glipizide showed principle functional groups wave number at 1688.37, 1649.8, 1529.27, 1159.01, 1034.62 and 904.45cm'1 (Figure 1). In the glipizide-PEG 6000 complex spectra principle wave number peak was diminished which indicates the formation of glipizide- PEG 6000 complex (Figure 2). DSC of pure glipizide showed the peak at the 218.0 Celsius while only one peak at 62.3 Celsius of excipients observed for glipizide-PEG complex which confirms the formation of glipizide-PEG complex(Figure 3).

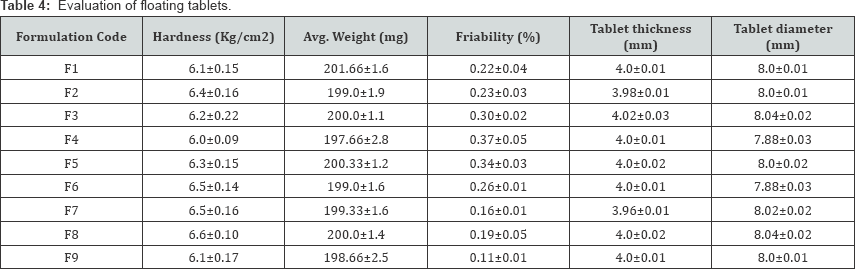
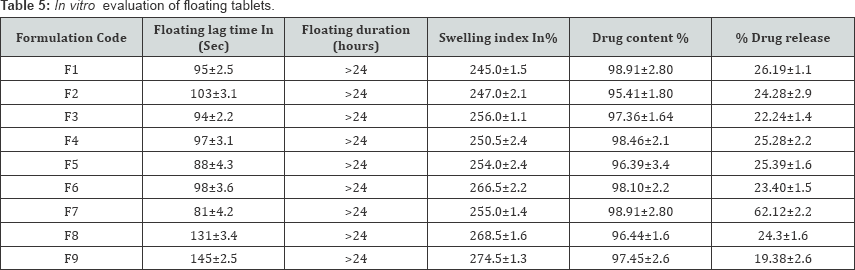
The in vitro testing revealed the ability of all the tablets to maintain buoyant more than 24h (Table 4). This indicates that the gel layers, formed by the HPMC, enabled efficient entrapment of the generated gas bubbles. The possible increase in tablet porosity made it float on the test medium (0.1NHcl) for this extended period of time. The formulations with HPMC K4M and HPMC K15M showed significant swelling and good tablet integrity. The formulations with HPMC K15M showed higher swelling compared to formulations with K4M. Drug uniformity results were found to be good among all batches; the percentage of drug content ranged from 95.41% to 99.78% (Table 5).
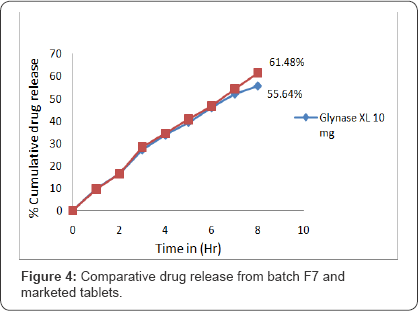
Optimized batch F7 and marketed preparation (Glynase XL) were selected for the similarity factor study. The percentage of glipizide released from batch F7 and Glynase XL in 8 hours found to be 61.48% and 55.64 % respectively (Figure 4). Similarity factor was calculated using formula 50+log [{1+ (Rt- Tt)*1/n}-0.5]. Similarity factor was found to be 51.58%, more than 50% value is acceptable.
In the release kinetics study, data obtained from the dissolution studies was fitted in different models like zero order, first order, Higuchi, and Hixson-Crowell model. The model with the highest correlation coefficient was considered to be the best fitting one [18-20]. The release mechanism was super case II transport as n value was 1.153. The best fitting model for F7 batch was zero order kinetics. This relationship can be used to describe the drug dissolution of several types of modified release dosage forms, as in the case of some transdermal systems, as well as matrix tablets with low soluble drugs.
Conclusion
From this study, it may be concluded that floating tablets of glipizide can be formulated as a sustained released formulation. This approach can increase the gastric residence time and thereby improve its bioavailability. Reproducibility in formulation indicates easy scale up of formulation at large scale.
Acknowledgement
We are thankful to the Sinhgad Institute of Pharmacy, Principal and all the faculty members.
References
- Radhika P, Pala TK, Sivakumar T (2009) Formulation and evaluation of sustained release matrix tablets of glipizideIranian. J Pharm Sci 5(4):
- Tadros MI (2010) Controlled-release effervescent floating matrix tablets of ciprofloxacin hydrochloride: development, optimization and in vitro-in vivo evaluation in healthy human volunteers. Eur J Pharm Biopharm 74(2): 332-339.
- Ddhav B, Ramakant P, Niranjan P, Kishor G (2012) Trilayer Mucoadhesive Gastro Retentive Tablets: Formulation and in vitro Evaluation, Journal of Pharmacy Research 5(5): 2463-2466.
- Singh BN, Kim KH (2000) Floating drug delivery systems: an approach to oral controlled drug delivery via gastric retention. J Control Release 63(3): 235-259.
- Arza RA, Gonugunta CK, Veerareddy PR (2009) Formulation and evaluation of swellable and floating gastro retentive ciprofloxacin hydrochloride tablets. AAPS Pharm Sci Tech 10(1): 220-226.
- Sauzet C, Bruno MC, Nicolas M, Kister J, Piccerelle P, et al. (2009) An innovative floating gastro retentive dosage system: formulation and in vitro evaluation. Int J Pharm 378(1-2): 23-29.
- Liandong H, Li L, Yang X, Liu W, Yang J, et al. (2011) Floating matrix dosage form for Dextromethorphan hydro bromide based on gas forming technique: In vitro and in vivo evaluation in healthy volunteers. Eur J Pharm Biopharm 42(1-2): 99-105.
- El-Badry M, Fetih G, Fathy M (2009) Improvement of solubility and dissolution rate of indomethacin by solid dispersions in Gelucire 50/13 and PEG4000. Saudi Pharm J 17(3): 217-225.
- El-Badry M, Fathy M (2005) Properties of solid dispersions of Piroxicam in pluronic F-98. J Drug Del Sci Technol 14: 199-205.
- Craig DQ (2002) The mechanisms of drug release from solid dispersions in water-soluble polymers. Int J Pharm 231(2): 131-144.
- Patel JK, Patel RP, Amin AF (2005) Formulation and evaluation of Mucoadhesive glipizide microspheres. AAPS Pharm Sci Tech 6(1): E49- 55.
- Chowdary KP, Roa YS (2003) Design and in Vitro and in vivo evaluation of Mucoadhesive microcapsules of glipizide for oral controlled release: a technical note. AAPS Pharm Sci Tech 4(3): E39.
- Unga J, Tajarobi F , Norder O (2009) Relating solubility data of parabens in liquid PEG 400 to the behaviour of PEG 4000-parabens solid dispersions. Eur J Pharm Biopharm 73(2): 260-268.
- Verma RK, Garg S (2005) Selection of excipients for extended release formulations of glipizide thoursough drug-excipient compatibility testing. J Pharm Biomedical Analysis 38(4): 633-644.
- Dave BS, Amin AF, Patel MM (2004) Gastro retentive drug delivery system of ranitidine hydrochloride: formulation and in vitro evaluation. AAPS Pharm Sci Tech 5(2): e34.
- Shinde AJ, Patil MS, More HN (2010) Formulation and evaluation of an oral floating tablet of cephalexin. Indian J Pharm Educ Res 44: 3.
- Gambhire MN, Ambade KW, Kurmi SD (2007) Development and in vitro evaluation of an oral floating matrix tablet formulation of diltiazem hydrochloride. AAPS Pharm Sci Tech 8(3): E73.
- Suvakanta D, Padala NM, Nath L (2010) Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol Pharm 67(3): 217-223.
- Meka L, Kesavan B, Chinnala MK, Vobalaboina V, Yamsani MR, et al (2008) Preparation of a matrix type multiple-unit gastro retentive floating drug delivery system for captopril based on gas formation technique: in vitro evaluation. AAPS Pharm Sci Tech 9(2): 612-619.
- Bomma R, NaiduRA, Yamsani MR (2009) Development and evaluation of gastro retentive norfloxacin floating tablets. Acta Pharm 59(2): 211221.






























