Radiopharmaceuticals Regulations on Bioavailability and Bioequivalence: Present Status and Future Requirements
Sandeep Sharma1, Ashish Baldi2 and Rakesh Kumar Sharma3*
1Research Scholar IK Gujral Punjab Technical University, India
2Maharaja Ranjit Singh Punjab Technical University, India
3Defence Food Research Laboratory, India
Submission: June 07, 2017; Published: June 27, 2017
*Corresponding author: Rakesh Kumar Sharma, Defence Food Research Laboratory, Siddartha Nagar, Mysuru, India, Tel: 0821-2473783; Email:director@dfrl.drdo.in
How to cite this article: Sandeep S, Ashish B, Rakesh K S. Radiopharmaceuticals Regulations on Bioavailability and Bioequivalence: Present Status and Future Requirements. Mod Appl Bioequiv Availab. 2017; 1(4): 555567.
Abstract
Recent era has seen an inordinate rise in use of radiopharmaceuticals owing to their use both as diagnostic and therapeutic agent. Major factors propelling their growth are its non invasive nature, growing cancer patient population and alpha radio-immunotherapy based targeted cancer treatment with high accuracy and precision. Indian market which has always been a hotspot amongst the innovators for carrying research owing to widespread climatic conditions and genetic variability has also undergone great development in the field of Molecular imaging and Nuclear medicine. However the Drugs & Cosmetic Act 1940 and the rules frame there under governing pharmaceutical industry in India has exempted radiopharmaceuticals from certain provisions and as such radiopharmaceuticals do not enjoy the full status of drug. This review aims to explore existing literature surrounding radiopharmaceuticals regulations in India and the need to intensify efforts for bringing out harmonized regulatory guidelines for conducting bioavailability and bioequivalence studies of radiopharmaceuticals which has global acceptance and will bring out ease in marketing authorization.
Keywords: Radiopharmaceuticals; Guidelines; Bioavailability; Bioequivalence
Introduction
Radiopharmaceuticals are the agents which are used for diagnostic and therapeutic purposes. They consist of two functional components, one of which is radioactive and another which is non-radioactive in nature. In diagnosis, the radioactive component which is nothing but a radio-nuclide with appropriate physical properties enables the detection of the product and is the active agent of the radiopharmaceutical during treatment. The non-radioactive component is a molecule or biological tracer with appropriate pharmacokinetics and shows organ specificity and metabolism. As radiopharmaceuticals consist of two components-pharmaceutical part and radioactive part, strict requirement exists to fulfill the quality specifications for both pharmaceutical part (pH, organoleptic properties, sterility, apyrogenicity, chemical purity, dosage related properties) and the radioactive part (radionuclide purity, radiochemical purity, radio assay, etc.) [1]. These quality control tests are necessary for finished radiopharmaceutical products before the licensing of the finished product can be obtained. Radiopharmaceuticals are now a crucial part of the healthcare industry due to their ability to identify various disease processes much earlier than other diagnostic tests [2]. The increasing acceptance of disease targeted treatment and rise in cancer patients coupled with high demand specifically from emerging countries due to its non invasive nature and presence of potential radioisotopes in the pipeline have opened new frontiers for the ever increasing radiopharmaceutical use.
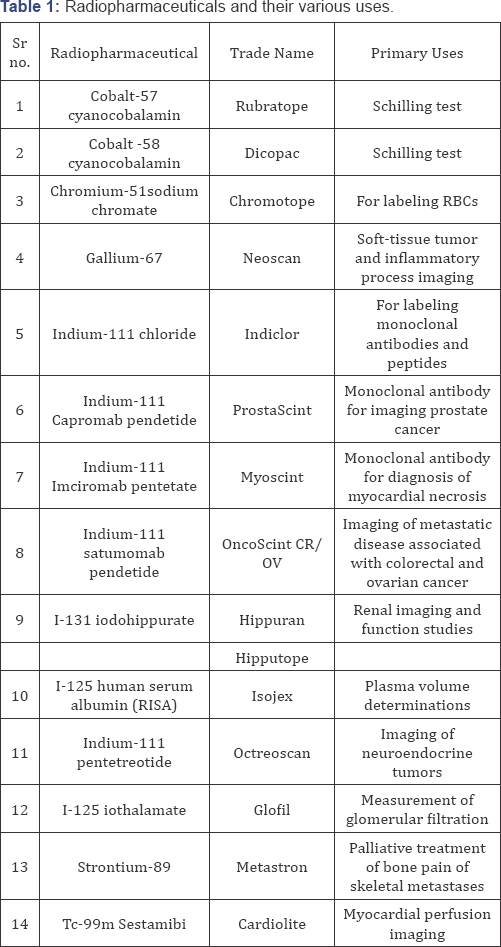
The development of nuclear medicine over the past several years highlights the strong link between advances in chemistry and the development of radionuclide and radio labeled compounds [3]. Radiopharmaceuticals are extensively used under different trade name for various purposes as summarized in Table 1.
The global radiopharmaceuticals market is driven mostly by an aging population and cardiovascular, oncological and neurological disorders. Moreover radiopharmaceuticals use for pediatric population has now also been regulated [4]. Although developed countries largely dominate the share of radiopharmaceutical market, the Asian-Pacific countries too have attained a sizeable share during the twenty first century India too is emerging a strong player for the radiopharmaceutical market and research due to its diverse climatic conditions and genetic diversity. However the regulatory setup in India for radiopharmaceutical is still in its infancy stage and no strict and detailed guideline exist. Although the Indian Pharmacopoeia 2014 have added 20 radiopharmaceuticals still they are not considered as drug and are exempted from certain provisions of Drug and Cosmetic Act 1940. There is a lack of adequate regulatory setup for preclinical studies, clinical trials and various bioavailability and bioequivalence studies for radiopharmaceuticals in India. This article attempts to highlight the present scenario of radiopharmaceutical setup in India and present a way forward so that high quality radiopharmaceuticals are delivered to the patients in the most cost effective manner.
Radiopharmaceuticals as Special Class
Radiopharmaceuticals are special class of pharmaceuticals and utmost care should be taken for their handling, storage, dispensing and use. The characteristic which sets them apart from pharmaceuticals includes their short half life, inherent hazardous nature of radioisotope, issue of maintaining sterility with radiation safety simultaneously, storage, transport and waste disposal issues and the fact that minute change in dose may cause faulty diagnosis or even over exposure. Therefore the guidelines applicable to pharmaceuticals are not relevant for radiopharmaceutical and calls for separate regulatory setup for radiopharmaceuticals.
Regulatory requirement for pre clinical studies of radiopharmaceuticals
Presently the regulatory setup for conducting preclinical studies using radiopharmaceuticals is rather weak even at international level. Even the most sought after guideline from ICH have no supporting literature available for pre clinical studies of radiopharmaceuticals [5]. These guidelines are pertinent as they help in establishing the risk-to-benefit ratio which further helps in justifying the rationale for an effective and safe use of radiopharmaceutical. The parameters to focus must include the bio distribution profile, toxicological evaluation and in vivo stability of the radiopharmaceutical. Radiopharmaceutical can be used for diagnostic as well as therapeutic purpose. The dose and frequency required for latter case is generally multiple and larger and therefore invites toxicological and safety issues [6]. This necessitates the need for separate guidelines for preclinical studies of diagnostic and therapeutic radiopharmaceuticals.
Regulatory requirement for clinical studies of radiopharmaceuticals
Till date no guideline is available in India for carrying out the clinical trials of radiopharmaceuticals which are used both for diagnostic and therapeutic purpose. The radiopharmaceuticals employed for diagnostic purpose generally have a low radiation dose as compared to therapeutic radiopharmaceuticals [7,8]. Moreover different half life exists for different radiopharmaceuticals. For eg. C11 used for cancer study have half life of 20.4 minutes while Iridium192 which is used for cancer treatment has a half life of almost 74 days [9]. Therefore clinical studies for them must separately address the safety and quality concerns before they are finally administered to the subjects. This is because very short half life radiopharmaceuticals have to be immediately administered to the subject after their manufacture and therefore must be released from the laboratory before all the quality parameters have been fulfilled. In this case a robust quality assurance setup and elaborated SOPs have to be in place. Moreover toxicological profile, genotoxicity profile, reproductive toxicity and dosimetry profile also need to be established. Therefore an elaborated guideline detailing all such intricacies should be drafted and implemented.
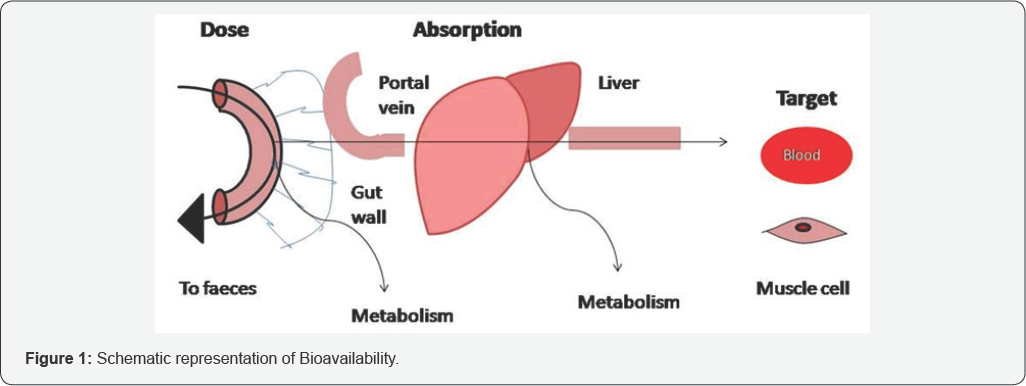
As both bioavailability and bioequivalence focus on the release of drug substance from its dosage and its corresponding absorption into systemic circulation, similar approaches for measuring bioavailability should be sought in demonstrating bioequivalence. Figure 1 depicts the schematic representation of bioavailability.
Regulatory requirement for Bioavailability and Bioequivalence studies
The bioavailability [F] of the drug may be determined by comparison of the area under curve (AUC) between serum concentration and time for the particular dosage form against the intravenous form and is given by the following formula:

Bioequivalence and bioavailability studies are important criteria to ensure the therapeutic equivalence between a test product and a reference product. Both in vitro and in vivo approaches can be followed for assessing the product quality. In vivo approaches include a comparative clinical trial or comparative clinical pharmacodynamics study. Sometimes cases exists when a new drug is to be administered parent rally having same concentration of active substance with comparable excipients concentration or do not have excipients affecting gastro-intestinal transit etc. Such cases do not require bioequivalence studies. In India Central Drug Standards Control Organization (CDSCO) under Ministry of Health and Family Welfare issued Guideline for Bioavailability and Bioequivalence studies in 2005 [10]. The guidelines explicitly details the design and conduct of studies including study design, study population and condition, bio analytical methodologies involved and the facilities for conducting BA/BE studies of pharmaceuticals. However no sufficient information is available for the special case of radiopharmaceutical which has a very short half life and poses handling and storage problems. Bioequivalence studies helps in comparison of two products having same active substance thus providing an objective means of assessing the possibility of use of any of them. An explicit regulatory document detailing design and conduct of studies including pharmacokinetic studies, study population, selection of number of subjects, study condition, bio analytical methodology and statistical evaluation for radiopharmaceuticals is need of the hour. The systemic exposure study obtained during clinical trial in the early drug development can serve as a benchmark for subsequent BE studies.
Recent Regulatory Advances
CDSCO is actively participating and issuing various office order From time to time for regulating radiopharmaceuticals. Recently the issue for import of various radiopharmaceuticals was taken up and office orders came on 29th June, 2016 in which it was conceived that all importers shall have to comply with regulations of Drug and Cosmetic Act, 1940 as amended for importing radiopharmaceuticals [11]. Simultaneously on 20th Feb, 2017, matter of import of radiopharmaceuticals for in vitro and in vivo use was taken up and it was notified that on import the sample from first five imported consignment shall be sent to Radiation Medicine Centre of Bhabha Atomic Research Centre (BARC) or to Institute of Nuclear Medicine and Allied Sciences (INMAS), Delhi for testing [12]. Importer shall be required to communicate the testing result to CDSCO which will then release the product into market after evaluation of test results. However it was further observed that importers were aggrieved since Radiation Medicine Centre of BARC and Institute of Nuclear Medicine and Allied Sciences had not accepted the samples for testing. After series of meeting with importers and CDSCO it was finally conceived by office order that importer shall voluntarily make the sample available for testing at the test centre stated within 10 working days of office orders. Furthermore it was also notified that in vivo radiopharmaceutical having no indigenous manufacturer shall be granted license in Form 10 by central government and those having indigenous manufacturer shall be required to have import registration certificate as well as license in Form 10 [13].
Discussion and Conclusion
The ever increasing demand of radiopharmaceuticals has highlighted the need for strong regulatory setup for their expedited entry from bench side to bedside. The present regulatory setup has many lacunae which make it confusing and therefore the manufacturer and investigators abstain from investing in radiopharmaceutical domain. The guidelines overseeing radiopharmaceuticals in India lacks clear cut information for commencing preclinical studies, clinical trials and various bioavailability and bioequivalence studies. In India radiopharmaceuticals fall under Schedule K of Drug and Cosmetic Act 1940 in which rule 123 clearly states that Schedule K drugs shall be exempted from provision of chapter IV of this act [14]. Thus radiopharmaceuticals enjoy immunity from the ambit of Drug and Cosmetic Act 1940. The same has been highlighted by Atomic Energy Regulatory Board which has mentioned in one of its document that radiopharmaceuticals are exempted from chapter IV of Drug and Cosmetic Act 1940 due to its "low toxicity”. However this statement is ambiguous and self conflicting as radiopharmaceuticals used for therapeutic interventions have multiple dosing protocols and the chances of overexposure leading to toxicity cannot be overruled. Therefore it is pertinent to give radiopharmaceuticals the full status of drug so that India Pharmacopoeia 2014 as amended which has included 20 radiopharmaceuticals can be appropriately implemented and radiopharmaceuticals enjoy full status of drug. Radiopharmaceuticals need to be regulated appropriately and completely. Non fulfillment of such a situation may cause administration of non-sterile formulation leading to systemic problems, faulty diagnosis due to change in dose, fatal results on overdose and problem of radiation safety and associated risks. Explicit evidence-based guidelines will help to improve the process and outcomes of health care and helps in creating improved methodologies based on most rigorous science available leading to patient and personnel health and safety.
References
- Santos-Oliveira R, Benevides CA, Kohlman-Rabbani ER, Ricarte-Freitas IMA (2008) Radiopharmaceuticals Industry Based on the Brazilian Regulations. FABAD J Pharm Sci 33(4): 205-209.
- Venugopal V (2011) Societal Applications of Nuclear Technology in Health Care, Industry and Water Resource Management in India. Asian Nuclear Prospects 2010, Energy Procedia 7: 553-559.
- Gutfilen B, Valentini G (2014) Radiopharmaceuticals in Nuclear Medicine: Recent Developments for SPECT and PET Studies. Biomed Res Int 2014(2014): 426892.
- Gelfand MJ, Parisi MT, Treves ST (2011) Pediatric Radiopharmaceutical Administered Doses: 2010 North American Consensus Guidelines. J Nucl Med 52(2): 318-322.
- Baum R (2014) Therapeutic Nuclear Medicine. (1st edn), Springer Verlag, Berlin, Germany, p. 951.
- Spence AM, Muzi M, Graham MM, O'Sullivan F, Link JM, et al. (2002) 2-[18F]Fluoro-2-deoxyglucose and Glucose Uptake in Malignant Gliomas before and after Radiotherapy. Clin Cancer Res 8: 971-979.
- Edvin Hansson (2012) The Internal Radiation Dosimetry of Diagnostic Radiopharmaceuticals across Different Asian Populations.
- Eberlein U, Bröer JH, Vandevoorde C, Santos P, Bardiès M, et al. (2011) Biokinetics, and dosimetry of commonly used radiopharmaceuticals in diagnostic nuclear medicine-a review. Eur J Nucl Med Mol Imaging 38(12) : 2269-2281.
- Kastner J (1951) The Half-Life of Iridium 192. Canadian Journal of Physics 29(6): 480-481.
- (2005) Guidelines for Bioavailability and Bioequivalence Studies, Central Drug Standard Control Organization, Ministry of Health and Family Welfare, Government of India, New Delhi, India.
- F. No.29/Misc./4/2016-DC(101) Government of India, Director General of Health Services, Central Drug Standard Organization, New Delhi, India.
- File No. DCG (I)/Misc/2017 (11), Directorate General of Health Services, Office of Drug Controller General, India.
- F.No. DCG(I)/Misc/2017 (11) Directorate General of Health Services, Office of Drug Controller General, India (Import and Registration).
- The Drugs & Cosmetics Act 1940 and Rules 1945, 1945. Delhi (India): Ministry of Health and Family Welfare (Govt. of India); 1945.






























