Abstract
Heat stress is a major environmental constraint that negatively affects broiler productivity by impairing growth performance, metabolic efficiency, and vital organ function, particularly in tropical and subtropical regions. This study evaluated the effects of dietary gamma-aminobutyric acid (GABA) supplementation on growth performance, hepatic and renal function, and related biochemical and histopathological parameters in broiler chickens subjected to chronic heat stress. A total of 360 broiler chickens were randomly assigned to six dietary treatments containing 0, 25, 50, 75, 100, or 150mg /kg GABA feed and exposed to heat stress (32–34°C) from day 12 to day 32 of age. Growth performance indices were recorded, while serum liver enzymes (AST, ALT, and ALP), kidney function markers (urea, uric acid, and creatinine), total protein, and albumin concentrations were analysed. Histopathological evaluations of liver and kidney tissues were also performed. Heat stress significantly reduced body weight gain and feed efficiency and increased biochemical indicators of hepatic and renal dysfunction in unsupplemented birds (P<0.01). Dietary GABA supplementation significantly improved body weight gain and feed conversion ratio (P<0.05), with optimal responses observed at 50–75mg/kg feed. Moderate GABA inclusion normalized liver enzyme activities and improved serum protein, albumin, and renal function markers compared with the heat-stressed control group (P<0.05). Histopathological findings supported these results, showing improved liver and kidney architecture at 75mg/kgGABA, whereas excessive supplementation (150mg/kg) did not confer protective effects. In conclusion, dietary GABA supplementation effectively mitigates heat stress-induced physiological and performance impairments in broiler chickens, with 50–75mg/kg identified as the optimal supplementation range under heat stress conditions.
Keywords: Gaba; Heat Stress; Growth Performance; FCR; Liver Function; Kidney Function.
Abbreviations:FCR: feed conversion ratio; GABA: Gamma-aminobutyric acid; NRC: National Research Council; BWG: Body Weight Gain; ALT: Alanine Aminotransferase; AST: Aminotransferase; ALP: Alkaline Phosphatase; H&E: Haematoxylin and Eosin; GABA: Dietary Gamma- Aminobutyric Acid; TP: Total Protein; ROS: Reactive Oxygen Species; RPM: Rotation Per Minute
Introduction
Heat stress is one of the most critical environmental challenges limiting poultry production worldwide, particularly in tropical and subtropical regions. In countries such as Bangladesh, broiler chickens are frequently exposed to ambient temperatures that exceed their thermoneutral zone, resulting in substantial economic losses due to reduced growth performance, compromised health, and increased mortality [1]. Prolonged exposure to high environmental temperatures disrupts physiological homeostasis, impairs metabolic efficiency, and suppresses immune function, thereby negatively affecting overall productivity [2].
Physiologically, heat stress induces panting and peripheral vasodilation as thermoregulatory responses, which markedly increase maintenance energy requirements while reducing nutrient availability for growth and tissue accretion [3]. Additionally, heat stress triggers a complex cascade of metabolic, endocrine, and behavioral alterations, including reduced feed intake, impaired nutrient digestion and absorption, and elevated production of reactive oxygen species (ROS). Excessive ROS accumulation leads to oxidative stress, cellular damage, and disruption of vital organ functions, particularly the liver, which plays a central role in metabolism and detoxification [1,4]. These pathological changes are often accompanied by immune suppression, hormonal imbalance, and altered lipid and protein metabolism, ultimately manifesting as reduced body weight gain and increased feed conversion ratio (FCR) in broiler chickens [5,6]. Multiple strategies have been explored to mitigate the adverse effects of heat stress in broiler production systems. Environmental interventions, including reduced stocking density, open-sided housing, intermittent lighting programs, and enhanced ventilation, have been shown to partially alleviate thermal load and improve bird comfort [7]. Genetic approaches, such as the selection of heat-tolerant lines and the exploitation of major genes associated with improved thermotolerance and productivity, have also been proposed as long-term solutions [8]. However, the practical application of many environmental and genetic strategies is often constrained by high costs, infrastructure requirements, and complex management demands, limiting their feasibility under commercial production conditions. Consequently, nutritional manipulation has emerged as a widely adopted and cost-effective strategy to attenuate the detrimental effects of heat stress in poultry. Dietary interventions incorporating vitamins, minerals, prebiotics, probiotics, essential amino acids, and Phyto biotics have demonstrated considerable potential in improving physiological resilience, metabolic efficiency, and overall performance of broilers exposed to heat stress [9-12]. Gammaaminobutyric acid (GABA), a major inhibitory neurotransmitter in the central nervous system of vertebrates, has recently gained considerable attention as a functional feed additive due to its ability to modulate stress responses and enhance physiological resilience. Beyond its neuroregulatory functions, GABA has been shown to exert antioxidant, anti-inflammatory, and metabolic regulatory effects, thereby improving adaptation to environmental stressors [13,14]. Experimental evidence suggests that dietary GABA supplementation can improve growth performance, immune competence, and intestinal health in broilers subjected to thermal stress by enhancing digestive enzyme secretion, stabilizing intestinal morphology, and attenuating oxidative damage [15,16].
Importantly, emerging evidence indicates that GABA plays a protective role in maintaining hepatic function during thermal stress by modulating endocrine responses, reducing circulating cortisol concentrations, and improving antioxidant defence mechanisms. Given the pivotal role of the liver in regulating nutrient utilization and metabolic homeostasis, preservation of liver health is fundamental to sustaining productive performance in heat-stressed broilers. However, despite increasing interest in GABA as a nutritional intervention, comprehensive investigations elucidating its multi-system protective effects—particularly its role in hepatic protection under chronic heat stress conditions typical of South Asian poultry production systems—remain limited. In Bangladesh, where high ambient temperatures prevail for much of the year, the poultry industry urgently requires sustainable and effective nutritional strategies to mitigate heat stress–induced productivity losses. Therefore, the present study was designed to evaluate whether dietary gamma-aminobutyric acid supplementation can ameliorate heat stress–induced declines in productive performance of broiler chickens through improvement of liver health. Specifically, the study examined growth performance parameters alongside liver and kidney function markers, protein and albumin status under heat stress conditions representative of commercial broiler production in Bangladesh.
Methodology
Experimental Design
One-day-old commercial broiler chicks (ROSS 308) were procured from Kazi Hatchery Ltd. and immediately housed in a well-equipped experimental poultry facility. All chicks were brooded under standard management conditions until day 11 of age. A total of 120 broiler chicks were randomly assigned to six dietary treatment groups receiving 0, 25, 50, 75, 100, 150 mg γ-aminobutyric acid (GABA) per kg of feed. Each treatment consisted of two replicates with 10 birds per replicate, following a completely randomized design. From day 12 to day 32, all birds were subjected to controlled heat stress conditions. Ambient temperature was maintained at 34±2°C for 4 h daily (12:00–16:00 h) and subsequently reduced to 28±2°C for the remaining period. Desired environmental temperatures were achieved using an electrical water boiler and mechanical ventilation system.
Experimental feed and growth management
Throughout the experimental period, chicks were provided ad libitum access to the experimental diets and fresh drinking water. The basal diet was procured from Kazi Agro Feed Ltd. and formulated in accordance with the nutrient requirements recommended by the National Research Council (NRC) for broiler chickens. To ensure uniform incorporation of γ-aminobutyric acid (GABA), the basal feed was finely ground and thoroughly mixed with the predetermined experimental doses of GABA. The diets were subsequently re-pelleted using a poultry pelletizing machine before feeding to the birds.
Growth Performance
A predetermined quantity of feed was offered to the broiler chickens twice daily, and feed refusals were collected and weighed the following morning. Daily feed intake was calculated as the difference between feed offered and feed refused. Based on daily measurements, weekly and cumulative feed intake were determined. Body weight was recorded weekly to calculate weekly body weight gain (BWG), and feed conversion ratio (FCR) was calculated accordingly. At day 32 of the experimental period, final BWG and FCR were calculated and compared among the experimental groups.
Blood Collection
At the end of the experiment, birds from each group were randomly selected. Blood samples were collected from five randomly selected birds from each group via wing vein, under gentle restraint without anaesthesia and kept it without anticoagulant and processed serum for biochemical analyses.
Preparation of serum for biochemical analysis
Following the collection of blood from wing vein, samples were transferred into the Eppendorf tubes without any anticoagulant and allowed to stand at room temperature, inclined at approximately 45◦, for 2 h. Subsequently, the Eppendorf tubes were refrigerated overnight. The resultant serum was carefully separated and transferred into fresh Eppendorf tubes, after which it underwent centrifugation to eliminate excess cells. This centrifugation process was conducted using the Wise Spin R centrifuge machine, Model- DM0506, DLAB Scientific Co., Ltd, USA, employing low-speed settings at 2000 rotation per minute (rpm) for 20 min. Following centrifugation, the serum was transferred to another fresh tube and stored at freezing temperatures until subsequent analysis.
Serum Biochemical tests
Serum concentrations of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), total protein, albumin, creatinine, urea and uric acid were measured using a Semi-automated biochemical analyser (Labo med BAS-150 TS, USA) in the Department of Pharmacology and Toxicology, Sher-e Bangla Agricultural University, Dhaka, Bangladesh according to the manufacturer’s instructions.
Histopathological analysis of Liver and Kidney
For histopathological examination, the liver and kidney from the experimental broilers were meticulously harvested. Each sample were carefully washed in a Petri dish using normal saline and subsequently transferred into an organ tube containing a 15 % formalin solution, with each specimen treated individually. The specimens were allowed to rest in ambient room conditions until the commencement of the histological procedures.
Sample preparation and staining of collected tissue
The liver and kidney tissues were processed in the laboratory following standard histological procedures. Initially, the tissues were separated, cut, and fixed in Bouin’s fluid for 24 h. Subsequent dehydration was performed using ascending concentrations of alcohol (70 %, 80 %, 90 %, 95 %, 100 % I, 100 % II, and 100 % III), each for 2 h. The tissues were then cleared in three changes of xylene, followed by infiltration with paraffin. After embedding, 5-μm thick sections were cut using a sliding microtome, floated on a lukewarm water bath and mounted on slides coated with egg albumin. The slides were dried for 12 h before staining. The sections were stained using Haematoxylin and Eosin (H&E), following the protocol according to our previous study [17]. Finally, the stained sections were examined under an Olympus photomicroscope (CX43) at 10× objective for histological analysis.
Statistical Analysis
The data were presented as mean±SEM. for each group, and statistical analyses were performed using Graph Pad Prism Version 9.3.1 (GraphPad Software, San Diego, CA). A one-way ANOVA followed by Tukey’s multiple comparisons test was employed to assess Body weight, FCR, and biochemical, a twoway ANOVA followed by Tukey’s multiple comparisons test was applied. A significance level of P<0.05 was established as the criterion for statistical significance.
Results
Effects of GABA supplementation on growth performance under heat stress
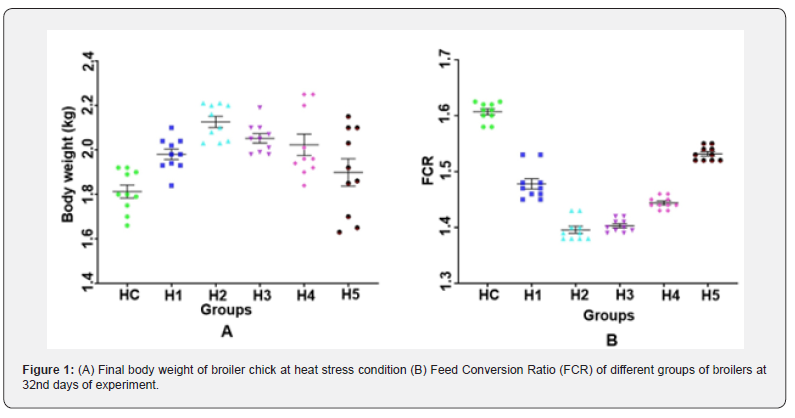
The effects of dietary gamma-aminobutyric acid (GABA) supplementation on body weight gain (BWG), feed intake, and feed conversion ratio (FCR) of broiler chickens under heat stress conditions are presented in Figure 1. Weekly body weight gain was monitored throughout the experimental period under heat stress conditions. During the first two weeks of heat exposure, no statistically significant differences (p>0.05) in BWG were observed between the heat-stress control group and the GABAsupplemented groups. However, by the end of the experimental period (32nd day), heat stress caused a marked reduction in BWG in the control group, whereas birds receiving GABA supplementation exhibited significantly higher BWG (p<0.05) compared to the heat-stress control.
The highest final body weight gain was recorded in the group receiving 50mg/kgGABA (2.10±0.03kg), while the lowest gain was observed in the heat-stress control group (1.82±0.03kg). Heat stress significantly (p<0.05) reduced BWG compared to GABAsupplemented groups. In contrast, dietary supplementation with GABA significantly improved BWG under heat stress conditions. Among the treated groups, the highest final BWG was observed in broilers receiving 50mg/kgGABA (2.10±0.03kg), followed by the 75mg/kg and 100mg/kg supplementation groups. Although BWG tended to decline slightly at higher inclusion levels, all GABA-supplemented groups maintained significantly higher BWG compared to the heat-stress control (p<0.05). These findings suggest that GABA supplementation partially alleviated the growth-suppressive effects of heat stress, with 50mg/kg appearing to be the optimal dietary level for maximizing body weight gain.
Feed conversion ratio (FCR) was significantly influenced by dietary GABA supplementation under heat stress conditions (Figure 1B). At the end of the second week of the experiment (day 25), broilers receiving 75mg/kg GABA exhibited the lowest FCR (1.177 ±0.02), which was significantly lower than that of the heat-stress control group (1.358±0.01; p<0.05). This early improvement in feed efficiency suggests that GABA supplementation enhanced nutrient utilization during the initial phase of thermal challenge. At the end of the experimental period (day 32), FCR values remained significantly improved in GABAsupplemented groups compared to the heat-stress control. The heat-stress control group recorded the highest FCR (1.607±0.08), indicating poor feed efficiency under prolonged heat exposure. In contrast, broilers supplemented with 50mg/kg and 75mg/kg GABA exhibited comparable and significantly lower FCR values (1.396±0.01and1.403±0.01, respectively p<0.05). Although the 100mg/kg group also showed improvement compared to the control, the magnitude of response was less pronounced than that observed at moderate supplementation levels.
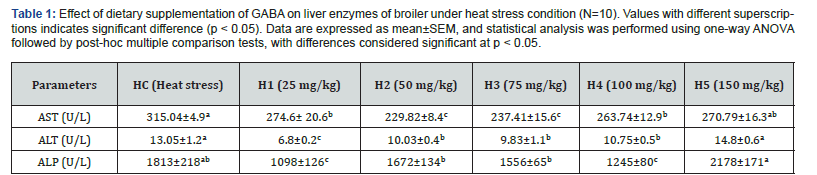
Effect of GABA on liver enzymes of chicken in heat stress condition-
The effects of dietary γ-aminobutyric acid (GABA) supplementation on liver function parameters-aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP) in broiler chickens subjected to heat stress are presented in Table-1.
Aspartate aminotransferase (AST)
Heat-stressed control birds (HC) exhibited significantly elevated serum AST activity (315.04±4.9U/L), indicating hepatocellular damage induced by thermal stress. Dietary GABA supplementation resulted in a significant reduction in AST activity (p<0.05) across most treatment groups. The greatest decrease was observed in H2 (229.82±8.4U/L), followed by H3 (237.41±15.6U/L), suggesting marked hepatoprotective effects at moderate GABA inclusion levels. Groups H1 (274.6±20.6U/L) and H4 (263.74±12.9U/L) also showed significantly lower AST values compared with HC, although the reduction was less pronounced. In contrast, H5 (270.79±16.3U/L) exhibited AST levels closer to the heat-stressed control, indicating diminished efficacy at higher GABA dosage.
Alanine aminotransferase (ALT)
Serum ALT activity was significantly influenced by heat stress and dietary GABA supplementation (p<0.05). The heat-stressed control group recorded elevated ALT levels (13.05±1.2U/L), reflecting hepatic cellular leakage under stress conditions. GABA supplementation significantly reduced ALT activity in H1 (6.8±0.2U/L), representing the most pronounced improvement among all groups. Moderate reductions were also observed in H2 (10.03±0.4U/L), H3 (9.83±1.1U/L), and H4 (10.75±0.5U/L) compared with HC. However, H5 (14.8±0.6U/L) exhibited significantly higher ALT levels than all other groups and exceeded the control value, suggesting possible hepatic overload or metabolic imbalance at excessive GABA supplementation.
Alkaline phosphatase (ALP)
ALP activity was markedly elevated in heat-stressed control birds (1813±218U/L), indicating hepatobiliary dysfunction associated with thermal stress. Dietary GABA supplementation significantly modulated ALP activity (p<0.05). The lowest ALP concentration was observed in H1 (1098±126U/L), followed by H4 (1245±80U/L), demonstrating substantial improvement in liver functional integrity. Groups H2 (1672± 134U/L) and H3 (1556±65U/L) showed moderate reductions relative to HC. In contrast, H5 (2178±171U/L) exhibited the highest ALP activity among all groups, exceedingly even the heat-stressed control, indicating a loss of hepatoprotective effect at the highest GABA dose.
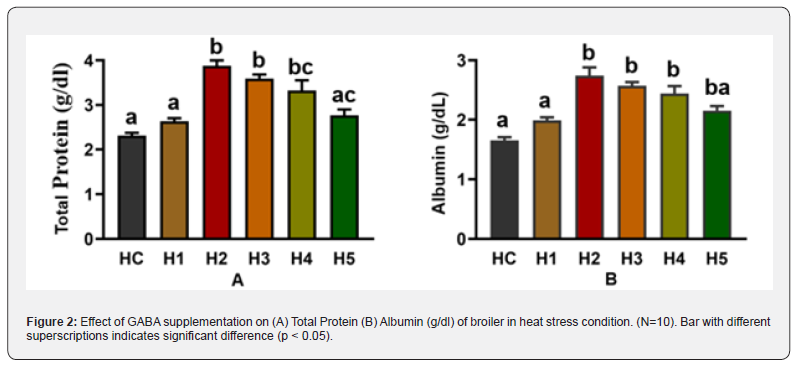
Effect of GABA on Total Protein (TP) and albumin of Chicken in heat stress condition-
We investigate the effect of GABA @ the dose of 0, 25, 50, 75, 100, and 150mg/kg of feed on broiler TP and albumin under heat stress condition. Heat stress markedly reduced serum protein indices in broiler chickens, as evidenced by significantly lower total protein and albumin concentrations in the heat stress control (HC) group. Dietary supplementation of γ-aminobutyric acid (GABA) significantly (p<0.05) improved both parameters in a dose-dependent manner up to an optimal level. Broilers in the HC group showed the lowest serum total protein concentration (2.31±0.07g/dL). GABA supplementation significantly increased total protein levels across all treated groups (p<0.05). The highest total protein concentration was observed in the H2 group (50mg/ kg GABA; 3.88±0.12g/dL), which was significantly higher than all other groups. The H3 (75mg/kg; 3.59±0.09g/dL) and H4 (100mg/ kg; 3.32±0.23g/dL) groups also showed significantly elevated values compared with HC and H1 but were lower than H2. At the highest dose (H5, 150mg/kg), total protein declined (2.77±0.13g/ dL) compared with H2–H4, though it remained significantly higher than HC (p<0.05). These results indicate that moderate GABA supplementation most effectively ameliorated heat stress– induced suppression of protein metabolism.
A similar trend was observed for serum albumin concentration. The HC group had the lowest albumin level (1.66±0.05g/dL). GABA supplementation significantly increased albumin in all treated groups (p<0.05), with the highest value recorded in the H2 group (2.74±0.14g/dL). Albumin concentrations in the H3 (2.57±0.06g/ dL) and H4 (2.44±0.12g/dL) groups were significantly higher than HC and H1 but lower than H2. The H5 group (2.15±0.08g/ dL) showed a reduction compared with H2–H4, suggesting a diminishing response at excessive GABA inclusion levels. Overall, one-way ANOVA followed by post hoc multiple comparison analysis demonstrated that 50mg/kg GABA supplementation (H2) was the most effective dose for improving serum total protein and albumin in broilers under heat stress conditions (p<0.05), highlighting its role in enhancing protein synthesis and maintaining hepatic functional integrity during thermal stress.
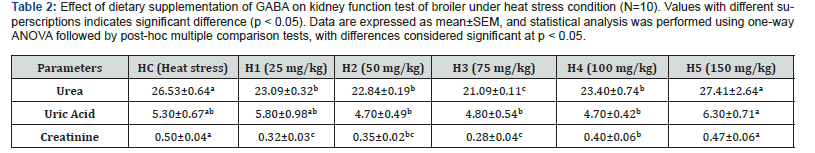
Effect of dietary GABA on kidney function test of chicken in heat stress condition-
Kidney function tests are crucial diagnostic tools used in both human and animal health (including poultry like broiler chickens) to assess how well the kidneys are performing their vital roles. The most common parameter used as diagnostic tools were urea, uric acid and creatinine. Under heat stress clearly disrupts kidney function and metabolic homeostasis, leading to increases in markers like creatinine and uric acid. The effects of dietary γ-aminobutyric acid (GABA) supplementation on kidney function parameters—serum urea, uric acid, and creatinine—in broiler chickens subjected to heat stress are presented in Table-2. Data are expressed as mean±SEM. Statistical analysis was performed using one-way ANOVA, followed by a post-hoc multiple comparison test, with significance considered at p<0.05.
Serum urea
Heat-stressed control birds (HC) exhibited a relatively elevated serum urea concentration (26.53±0.64), indicating impaired renal function under thermal stress. Dietary supplementation with GABA resulted in a significant reduction (p<0.05) in serum urea levels in most treatment groups. The lowest urea concentration was observed in group H3 (21.09±0.11), representing a marked improvement compared with the heat-stressed control. Groups H1 (23.09±0.32) and H2 (22.84±0.19) also showed significantly reduced urea levels relative to HC, suggesting a protective effect of moderate GABA supplementation on protein metabolism and renal clearance. In contrast, group H5 (27.41±2.64) exhibited urea levels comparable to or slightly higher than the control, indicating that excessive GABA supplementation did not confer additional renal benefits under heat stress.
Serum uric acid
Serum uric acid concentration, a primary indicator of nitrogen metabolism and kidney function in birds, was elevated in the heat-stressed control group (5.30±0.67). GABA supplementation significantly modulated uric acid levels (p<0.05). Groups H2 (4.70±0.49), H3 (4.80±0.54), and H4 (4.70±0.42) demonstrated lower uric acid concentrations compared with HC, reflecting improved renal excretory capacity and reduced protein catabolism. Conversely, the highest uric acid level was observed in H5 (6.30± 0.71), suggesting that higher GABA inclusion may adversely affect purine metabolism or renal handling under heat stress conditions.
Serum creatinine
Creatinine concentration, a sensitive marker of glomerular filtration rate, was significantly elevated in the heat-stressed control group (0.50±0.04). GABA-treated groups showed a significant decrease in serum creatinine levels (p<0.01) compared with HC. The most pronounced reduction was recorded in H3 (0.28± 0.04), followed by H1 (0.32±0.03) and H2 (0.35±0.02), indicating enhanced renal filtration efficiency and reduced muscle protein breakdown. Group H5 (0.47±0.06) exhibited creatinine values similar to the control, further supporting the observation that excessive GABA supplementation does not improve kidney function under heat stress.
Effect of dietary GABA on histopathological changes of liver and kidney in heat stress condition
Histopathological examination of kidney and liver tissues was performed using H&E staining (10× magnification) to assess the tissue-level protective effects of GABA supplementation under heat stress (Figure 3, 4).
Liver histopathology
The liver was severely affected by heat stress. Control birds exhibited extensive bleeding throughout the liver tissue, with deposits of breakdown products from red blood cells and multiple areas of dead hepatocytes. Adding 25mg/kgGABA reduced but did not eliminate cell death, while 50mg/kg still showed both necrosis and haemorrhage. At 75mg/kg and 100mg/kg, the liver showed immune cell infiltration, a sign of ongoing repair, but the 75mg/kg group had considerably less damage overall. The highest dose (150mg/kg) caused alarming hepatic deterioration: liver cells lost their normal organized arrangement, inflammatory cells clustered heavily, and large zones of dead tissue appeared around central veins throughout the liver.
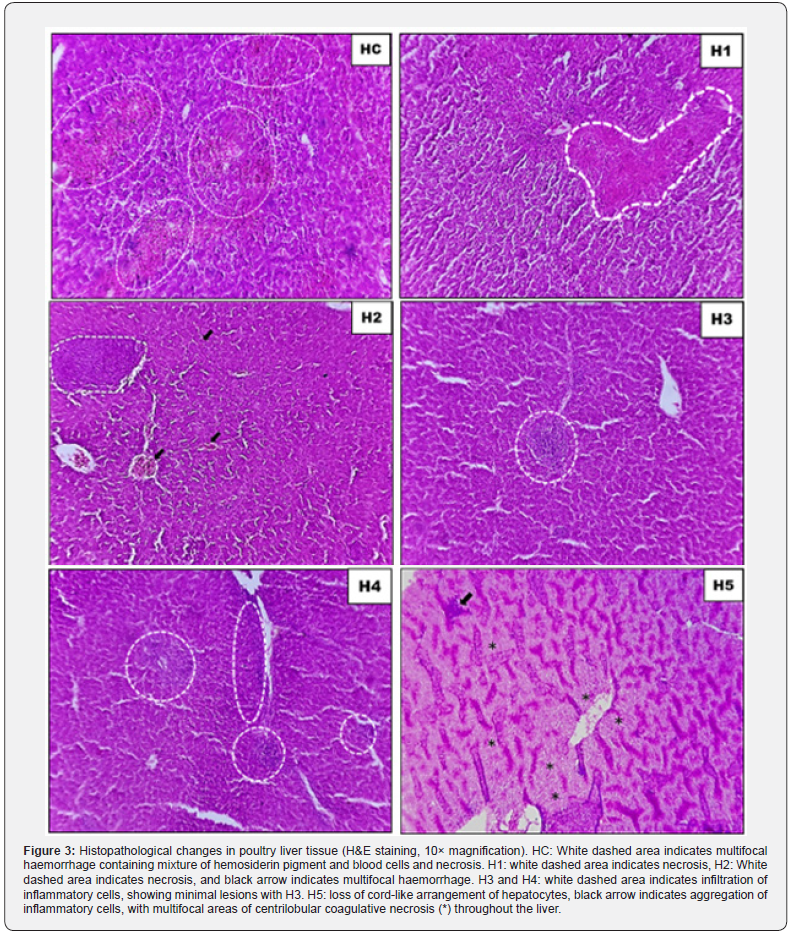
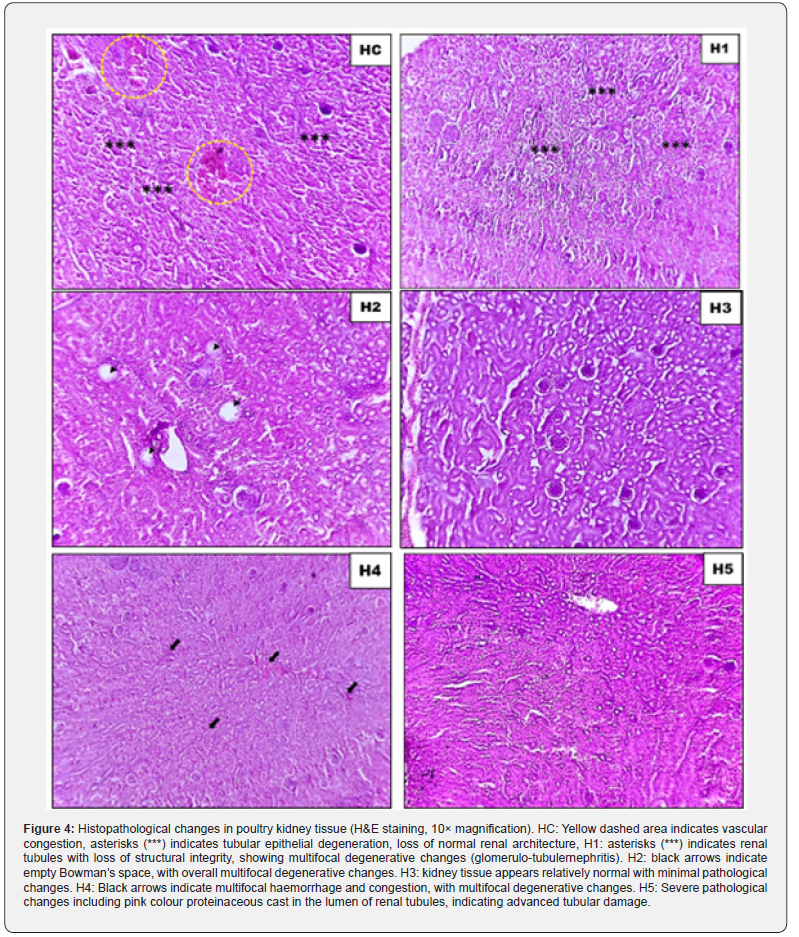
Kidney histopathology
Heat stress induced marked renal damage in the control group, characterized by severe blood vessel congestion and widespread breakdown of tubular epithelial cells. The normal kidney structure was largely disrupted in these birds. Low-dose GABA supplementation (25mg/kg) provided minimal protection, as kidney sections still showed extensive tubular damage and signs of inflammation affecting both glomeruli and tubules. At 50mg/ kg, kidney damage remained substantial—Bowman’s spaces were empty due to glomerular loss or atrophy, and scattered degenerative changes persisted throughout the tissue. The most striking improvement occurred at 75mg/kg, where kidney tissue appeared nearly normal with only minor abnormalities detected. Interestingly, this protection diminished at higher doses: 100mg/ kg supplementation showed a return of bleeding and congestion in multiple areas, while 150mg/kg produced severe kidney damage including protein accumulation inside the tubules, a sign of advanced renal failure.
Discussion
Heat stress is a major constraint to sustainable poultry production, particularly in tropical and subtropical regions, where prolonged exposure to elevated ambient temperatures disrupts physiological homeostasis and compromises productivity. Consistent with previous reports, broilers exposed to chronic heat stress in the present study exhibited reduced body weight gain (BWG), impaired feed efficiency, and marked alterations in biochemical indicators of liver and kidney function, confirming the multifactorial nature of heat stress–induced performance decline [18-20]. Dietary supplementation with γ-aminobutyric acid (GABA) significantly alleviated the adverse effects of heat stress on growth performance, with a clear dose-dependent response. Among the supplemented groups, 50mg/kgGABA produced the greatest improvement in final BWG, while 50– 75mg/kg yielded the most favourable feed conversion ratio (FCR). These findings indicate that moderate GABA inclusion enhances physiological resilience and nutrient utilization under thermal challenge, whereas higher doses confer no additional benefit and may disrupt metabolic balance. Similar improvements in BWG and FCR following GABA supplementation under heat stress have been reported previously, supporting the robustness of the present results [13,21].
The improvement in growth performance was most evident during the later stages of heat exposure, suggesting that GABA exerts its primary benefits under prolonged or cumulative stress conditions. Heat stress is known to alter hypothalamic–pituitary regulation, suppress appetite, and impair anabolic processes, partly through dysregulation of central neurotransmitters and endocrine hormones. Reduced endogenous GABA availability under thermal stress has been associated with impaired growth and feed efficiency. Dietary GABA supplementation may restore neuroendocrine balance, attenuate stress signaling, and indirectly support feed intake and muscle accretion during chronic heat exposure. Beyond its neuroregulatory role, GABA supplementation exerted pronounced hepatoprotective effects, which appear central to the observed improvements in productive performance. Heat-stressed control birds exhibited significantly elevated serum AST, ALT, and ALP activities, indicating hepatocellular and hepatobiliary damage. These alterations are consistent with oxidative stress–induced membrane instability and impaired hepatic metabolism under high ambient temperatures [8,22]. Moderate GABA supplementation (50–75mg/kg) significantly normalized these liver enzyme activities, suggesting preservation of hepatocyte integrity and improved hepatic metabolic function. In contrast, the highest inclusion level (150mg/kg) failed to maintain this protective effect, highlighting the importance of dose optimization.
Improved liver function was further reflected in the restoration of serum total protein and albumin concentrations in GABA-supplemented birds. Heat stress–induced reductions in these parameters are indicative of suppressed hepatic protein synthesis and increased protein catabolism [23,24]. Albumin, synthesized exclusively by the liver, is a sensitive marker of hepatic functional capacity; thus, its significant elevation following GABA supplementation—particularly at 50mg/kg—strongly supports a hepatocentric mechanism underlying improved growth performance. The decline in protein indices at the highest supplementation level again suggests that excessive GABA intake may disrupt metabolic homeostasis rather than enhance it.
Renal function was also adversely affected by heat stress, as evidenced by elevated serum urea, uric acid, and creatinine concentrations, reflecting increased protein catabolism, oxidative stress, and impaired renal filtration. Dietary GABA supplementation significantly ameliorated these alterations, with the most consistent improvements observed at 75mg/kg. The reduction in nitrogenous waste products suggests improved nitrogen utilization, reduced muscle proteolysis, and enhanced renal excretory efficiency. These effects may be mediated by GABA’s ability to modulate stress-related endocrine responses, reduce corticosterone secretion, and attenuate oxidative damage in renal tissues [25,26]. Histopathological examination confirmed that heat stress caused severe structural damage in both liver and kidney tissues, with tissue-level findings closely matching the biochemical results. The 75mg/kg group showed nearly normal kidney architecture and minimal hepatic lesions, while 150mg/kg resulted in catastrophic damage including proteinaceous casts in kidney tubules and widespread liver necrosis, demonstrating that optimal GABA dosing is critical for organ protection.
Collectively, the present findings demonstrate that dietary GABA supplementation mitigates heat stress–induced declines in broiler performance through integrated neuroendocrine, hepatic, and metabolic mechanisms. Liver health emerges as a central pathway linking GABA supplementation to improved growth performance, protein metabolism, and feed efficiency under thermal stress. Moderate inclusion levels (50–75mg/kg feed) consistently produced the most favourable outcomes across productive performance, liver enzyme activity, protein–albumin status, and renal function, whereas excessive supplementation diminished these benefits. These results support the application of GABA as a functional nutritional strategy for improving physiological resilience and sustaining productivity of broiler chickens under chronic heat stress conditions typical of tropical poultry production systems.
Summary
The present study demonstrates that dietary gammaaminobutyric acid (GABA) supplementation effectively mitigates the detrimental effects of heat stress on productive performance and physiological health of broiler chickens, primarily through improvements in liver function and metabolic homeostasis. Heat stress markedly impaired growth performance, feed efficiency, and organ function, as evidenced by reduced body weight gain (BWG), elevated feed conversion ratio (FCR), increased liver enzyme activities, suppressed serum protein indices, and compromised kidney function in heat-stressed control birds. Dietary inclusion of GABA significantly improved growth performance under heat stress conditions. Although no significant differences in BWG were observed during the early phase of heat exposure, GABA-supplemented broilers exhibited significantly higher final BWG compared with heat-stressed controls by the end of the experimental period. The most pronounced improvement was observed at 50mg/kg dietary GABA, indicating this level as optimal for maximizing growth performance. Correspondingly, feed efficiency was significantly enhanced in GABA-treated groups, with the lowest FCR values recorded at 50–75mg/kg inclusion levels, reflecting improved nutrient utilization during thermal challenge.
Heat stress induced pronounced hepatic dysfunction, as indicated by elevated serum AST, ALT, and ALP activities. GABA supplementation significantly attenuated these alterations in a dose-dependent manner, with moderate inclusion levels (50–75mg/kg) providing the greatest hepatoprotective effects. The normalization of liver enzyme activities suggests that GABA preserves hepatocellular integrity and hepatobiliary function, likely through its antioxidant, anti-stress, and metabolic regulatory properties. In contrast, excessive supplementation (150mg/ kg) failed to confer protection and, in some cases, exacerbated hepatic enzyme responses, highlighting the importance of dose optimization. Consistent with improved liver function, serum total protein and albumin concentrations—both markedly reduced by heat stress—were significantly restored by GABA supplementation. The highest levels of total protein and albumin were observed in broilers receiving 50mg/kg GABA, indicating enhanced hepatic protein synthesis and improved metabolic status under thermal stress. The decline in these parameters at higher supplementation levels further supports a non-linear, dosedependent response. Heat stress also disrupted renal function, as reflected by elevated serum urea, uric acid, and creatinine concentrations. Moderate GABA supplementation significantly ameliorated these alterations, with the most favourable kidney function profile observed at 75mg/kg inclusion.
Conclusion
Heat stress significantly compromises growth performance, feed efficiency, and hepato-renal function in broiler chickens. The present study demonstrates that dietary gamma-aminobutyric acid (GABA) supplementation is an effective nutritional strategy to mitigate these adverse effects. Moderate GABA inclusion levels (50–75mg/kg feed) significantly improved body weight gain and feed conversion ratio while restoring liver enzyme activities, serum total protein and albumin concentrations, and kidney function markers. Histopathological findings further confirmed that GABA at 75mg/kg preserved normal liver and kidney architecture under heat stress, whereas excessive supplementation (150mg/kg) failed to provide protection and induced tissue damage. Overall, GABA supplementation enhances physiological resilience to heat stress through integrated improvements in hepatic metabolism, protein synthesis, and renal homeostasis. These findings support the practical application of GABA at optimized inclusion levels as a functional feed additive to sustain broiler productivity in heatstressed tropical production systems.
References
- Lara LJ, Rostagno MH (2013) Impact of heat stress on poultry production. Animals 3(2): 356-369.
- Prates JAM (2025) Heat Stress Effects on Animal Health and Performance in Monogastric Livestock: Physiological Responses, Molecular Mechanisms, and Management Interventions. Vet Sci 12(5): 429.
- Slimen IB, Najar T, Ghram A, Abdrrabba M (2016) Heat stress effects on livestock: molecular, cellular and metabolic aspects. J Anim Physiol Anim Nutr (Berl) 100(3): 401-412.
- Habibian M, Ghazi S, Moeini MM, Abdolmohammadi A (2015) Effects of dietary selenium and vitamin E on growth performance and antioxidant status of broilers under heat stress. Biological Trace Element Research 165: 1-9.
- Nawab A, Ibtisham F, Li G, Kieser B, Wu J, et al. (2018) Heat stress in poultry production: mitigation strategies to overcome the future challenges facing the global poultry industry. J Therm Biol 78: 131-139.
- Cheng CY, Tu WL, Chen CJ, Chan HL, Chen CF, et al. (2018) Functional genomics study of acute heat stress response in broiler chickens. Poultry Science 97: 437-449.
- Khan RU, Naz S, Nikousefat Z, Selvaggi M, Laudadio V, et al. (2012) Effect of ascorbic acid supplementation in heat-stressed poultry. World’s Poultry Science Journal 68(3): 477-489.
- Lin H, Jiao HC, Buyse J, Decuypere E (2005) Strategies for preventing heat stress in poultry. World’s Poultry Science Journal 61(1): 71-86.
- Khan RU, Naz S, Dhama K, Swaminathan M, Tiwari R, et al. (2011) Modes of action and beneficial applications of chromium in poultry nutrition, production and health: A review. Int J Pharmacol 7(4): 357-368.
- Chand N, Naz S, Khan A, Khan S, Khan RU (2014) Performance traits and immune response of broiler chicks treated with zinc and ascorbic acid supplementation during cyclic heat stress. Int J Biometeorol 58(10): 2153-2157.
- Dhama K, Latheef SK, Mani S, Samad HA, Karthik K, et al. (2015) Multiple beneficial applications and modes of action of herbs in poultry health and production-A review. Int J Pharmacol 11(3): 152-176.
- Hu X, Guo Y, Huang B, Bun S, Zhang L, et al. (2015) Effect of chromium picolinate supplementation on growth performance, carcass characteristics, and immune responses of broilers under heat stress conditions. Poultry Science 94(5): 1143-1151.
- Park JH, Kim IH (2017) Effects of dietary γ-aminobutyric acid supplementation on growth performance, nutrient digestibility, blood profiles and meat quality in broiler chickens exposed to heat stress. Journal of Applied Poultry Research 26: 501-509.
- Al-Wakeel RA, Abdel-Daim MM, Alkahtani S (2017) Protective effects of gamma-aminobutyric acid against oxidative stress in animals. Environmental Science and Pollution Research 24: 12796-12803.
- Zhang MH, Li L, Wang XG, Gao F (2012) Effects of dietary GABA on growth performance and antioxidant capacity in broilers. Poultry Science 91: 2866-2872.
- Zhu YW, Li L, Zhang J, Wang ZX, Gao F (2015) Effect of dietary gamma-aminobutyric acid on intestinal morphology and digestive enzyme activities in broilers under heat stress. Poultry Science 94: 1-9.
- Jahan MS, Md Haque MI, Gautam M, Bhuiyan MER (2024) Comparative analysis of high-fat diets: Effects of mutton, beef, and vegetable fats on body weight, biochemical profiles, and liver histology in mice. Heliyon 10(20): e39349.
- Sahin K, Sahin N, Kucuk O (2003) Effects of chromium and ascorbic acid supplementation on growth, carcass traits, serum metabolites, and antioxidant status of broiler chickens reared at high ambient temperature. Nutrition Research 23(2): 225-238.
- Zaman QU, Mushtaq T, Nawaz H, Mirza MA, Mahmood S, et al. (2008) Effect of varying dietary energy and protein on broiler performance under high ambient temperature. Poultry Science 87: 1939-1944.
- Yan J, Qiu-Zhou X (2019) Heat stress affects poultry health and production. World’s Poultry Science Journal 75: 1-14.
- Xu Y, Lai X, Li Z, Zhang X, Luo Q (2018) Effect of dietary GABA supplementation on stress response and performance of broilers. Journal of Animal Physiology and Animal Nutrition 102: 145-153.
- Mujahid A, Akiba Y, Toyomizu M (2009) Oxidative stress and acute heat stress in broiler chickens. Journal of Poultry Science 46: 16-21.
- Habibian M, Ghazi S, Moeini MM, Abdolmohammadi A (2014) Effects of heat stress on growth performance and blood parameters of broilers. Animal Feed Science and Technology 193: 1-8.
- Akbarian A, Michiels J, Degroote J, Majdeddin, M, Golian A, et al. (2016) Association between heat stress and oxidative stress in poultry. J Anim Sci Biotechnol 28:7:37.
- Bhandage AK, Jin Z, Korpi ER (2018) GABAergic signaling and stress regulation. Neuropharmacology 136: 38-50.
- Wang J, Wang Z, Wang X, Gao F (2021) Dietary modulation of renal oxidative stress in heat-stressed broilers. Poultry Science 100: 101-110.






























