Abstract
Background: Hemorrhoid is a common anorectal disease that seriously affects people’s physical and mental state. However, the precise etiopathology and immunopathogenic mechanism of hemorrhoids are still not clear. Methods: In this study, 15 pairs of hemorrhoidal and
adjacent normal tissues were analysed for bacterial composition and immune cells infiltration investigation, the gene expression profile of
hemorrhoidal tissue was also analysed to explore the molecular mechanism of hemorrhoid pathogenesis. Finally, a peptide inhibiting matrix
metalloproteinase-9 (MMP-9) was designed and tested on macrophages.
Results: The results showed that Gram-negative bacteria were more abundant inside hemorrhoidal tissues than in adjacent normal tissues.
M1 macrophages infiltration was significantly higher in hemorrhoids than in adjacent normal tissues. The main component of Gram-negative
bacteria cell wall, lipopolysaccharide (LPS), polarized M0 macrophages into M1. RNA sequencing results indicated a strong correlation of VCAM-
1 and MMP-9 with M1 macrophages infiltration in hemorrhoid sites. After VCAM-1 interference, macrophage M1 polarization was inhibited, and
MMP-9 expression was downregulated. Importantly, an MMP-9 inhibitory peptide P2 inhibited LPS-induced M1 polarization and ROS generation.
Conclusion: This study elucidated the microbial distribution and immunological pathogenesis in hemorrhoidal tissues, offering a possible
strategy for the treatment of hemorrhoids.
Keywords: Hemorrhoids; Bacterial Composition; M1 Macrophage; Polarization; MMP-9; Peptide Inhibitor.
Introduction
Hemorrhoids are a common anorectal condition characterized by enlargement and displacement of anal cushions accompanied by a swelling of the veins or blood vessels in and around the anus and lower rectum [1]. The exact function of hemorrhoids is not completely understood, but hypotheses suggest that they may perceive sensations like fullness, pressure, and the presence of solid content in the anus. The incidence of hemorrhoids is challenging to be accurately determined, Johanson et al.[2] estimated the incidence of hemorrhoids among Americans to be approximately 4.4%, while supplementary data from the National Health Interview Survey in the United States indicated that the incidence of hemorrhoids is 13% [3], and there is an old saying in China that “nine out of ten people have hemorrhoids”. In the United States, the annual cost associated with the treatment of hemorrhoids is about 800 million USD [4], imposing a significant burden on the healthcare system and society. There are many risk factors for the development of hemorrhoids, including increased intra-abdominal pressure, prolonged sitting, constipation, diarrhoea, and high perfusion of the hemorrhoidal arteries. However, the process of hemorrhoid development typically involves four core physiological and pathological events: sliding of the anal cushions, relaxation of the connective tissue of the anal cushions, reduced venous return, and the formation of thrombosed hemorrhoidal veins [5].
Currently, the widely recognized mechanism for the development of hemorrhoids is the displacement of the anal cushions, although the specific mechanism for this movement is not yet clear. After the occurrence of hemorrhoids, various pathological changes such as bacterial infection, inflammation, vascular alterations, and connective tissue changes can be observed. The location of hemorrhoids is adjacent to the intestine, and previous works have extensively focused on the study of intestinal microbiota in various health issues such as tumours [6], atherosclerosis [7], and aging [8]. Gram-negative bacterial walls are primarily composed of peptidoglycan, lipopolysaccharide (LPS), and lipoproteins. Compared to Gram-positive bacteria, the components of Gram-negative bacterial cell walls are more likely to induce inflammatory reactions, with LPS playing a significant role in these effects. It has been reported that, in individuals with type 2 diabetes, intestinal dysbiosis revealed an increase in Gram-negative bacteria, and elevated levels of LPS can lead to the generation and release of pro-inflammatory factors, accelerating the progression of the disease [9]. Study by Cani et al. [10] reported that prebiotics can promote gut microbiota balance in obese mice, reduce LPS production, and inhibit liver inflammation. However, there hasn’t been a systematic study on the microbial content and types in hemorrhoids. This study conducted sequencing on human hemorrhoid tissues and revealed new research findings in this regard.
We observed a close association between inflammation and the occurrence and development of hemorrhoids, with macrophage polarization playing a central role in hemorrhoid development. Macrophages are crucial members of the innate immune system that can respond to different microenvironment signals and polarize into different phenotypes, such as M1 (classically activated macrophages) and M2 (alternatively activated macrophages) [11, 12]. The LPS present in Gramnegative bacteria can induce macrophages to polarize toward the M1 phenotype [13], leading to pro-inflammatory reactions and the production of inflammatory factors such as IL-6 and TNF-α [14]. Previous reports indicated that M1 macrophages promote inflammatory responses in periodontitis, resulting in tissue damage. However, research on immune cells in the occurrence and development of hemorrhoids is extremely scarce. Tawe visit et al. found an increased density of mast cells in hemorrhoidal veins, which may contribute to the development of hemorrhoids [15]. The relationship between macrophages and other immune cells and hemorrhoids has not been thoroughly investigated. This study analysed the composition of immune cells in the hemorrhoidal tissues and found that polarization of M0 macrophages into M1 macrophages leads to changes in certain proteins or factors, with a significant correlation observed between the levels of vascular cell adhesion molecule-1 (VCAM-1), MMP-9, and macrophages’ infiltration.
Typically, inflammatory reactions are initiated by TNF-α released by immune cells, including macrophages. TNF-α stimulates adhesion molecules like selectins, ICAM-1, and VCAM- 1 to recruit more immune cells to the site of inflammation [16]. Studies have shown that in a mouse model of dextran sulfate sodium-induced colitis, antibodies against VCAM-1 prevented white blood cells from adhering to the surface of vascular endothelial cells, significantly alleviating inflammation symptoms [17]. This suggests the crucial role of VCAM-1 in inflammation. However, there is no research specifically exploring the correlation between VCAM-1 and the occurrence and development of hemorrhoids, and its contribution need to be investigated. In addition, Serra et al. [18] detected an increase in MMP-9 activity in patients with hemorrhoids. Whether this activity can degrade elastic fibers, promote tissue remodeling, and contribute to the occurrence and development of hemorrhoids has not been totally investigated.
This study found that macrophages play a crucial role in regulating the inflammatory process during the onset of hemorrhoids. Specifically, a designed metalloproteinase inhibitor peptide P2 was shown to inhibit the polarization of macrophages to M1. This research provides new insights into the causes and mechanisms of hemorrhoid development, with the hope of offering new perspectives for the treatment of hemorrhoids.
Material And Method
RNA sequencing
Fifteen pairs of hemorrhoid tissue and corresponding adjacent tissue were collected for analysis. These samples were obtained from patients with Grade III or Grade IV hemorrhoids who received treatment at Jiangsu Cancer Hospital. This research was approved by Ethics Committee of Jiangsu Cancer Hospital with Ethical Review No. KY-2024-144. After haemorrhoidectomy, the collected specimens were preserved in liquid nitrogen. All specimens were obtained with the informed consent of the patients or their family members. RNA was then extracted by Trizol/chloroform method and an OD260/OD280 ratio was measured to determine the purity of the RNA (typically maintained between 1.8-2.0). Patient’s hemorrhoid tissues were used as the sample group, and the corresponding adjacent normal tissues were used as the control group. After total RNA extraction, ribosomal RNA (rRNA) was removed to enrich for mRNA. The isolated RNA was fragmented into smaller pieces and then used to create a sequencing library by attaching sequencing adapters to the RNA fragments. This library preparation step was followed by PCR amplification and subjected to high-throughput sequencing using the Illumina Novaseq™ 6000 sequencing platform.
For details of RNA extraction and reverse transcription, approximately 20mg of tissue was taken from liquid nitrogen and placed in a mortar. After adding liquid nitrogen, it was crushed, followed by the addition of 1 ml of Trizol for rapid grinding. The mixture was allowed to stand for 5 minutes, then 200μL of chloroform was added and left to stand for another 3 minutes. Centrifugation was performed at 12,000 rpm for 15 minutes at 4°C. The supernatant was collected, and an equal volume of isopropanol was added. The mixture was gently mixed and allowed to stand for 10 minutes. Centrifugation was then carried out at 12,000 rpm for 10 minutes at 4°C. The supernatant was discarded, and the precipitate was resuspended in 1mL of 75% ethanol. Centrifugation was repeated at 7,500 rpm for 8 minutes at 4°C. The supernatant was again discarded, and 20μL of DEPC water was added to dissolve the precipitate at 55°C for 13 minutes. To determine the purity of the RNA, 1μL of the RNA solution was taken to measure the OD260/OD280 ratio, which is typically controlled between 1.8 and 2.0.
Detecting the microbial composition of hemorrhoidal tissue through 16S rRNA gene sequencing
Microbial total DNA was extracted, and the quality of the extracted DNA was assessed through agarose gel electrophoresis. Simultaneously, the DNA was quantified using a UV-Vis spectrophotometer. Different primers were selected for PCR amplification based on specific project requirements. The PCR products were confirmed by electrophoresis on a 2% agarose gel. The PCR products were purified using AMPure XT beads (Beckman Coulter Genomics, Danvers, MA, USA) and quantified with a Qubit (Invitrogen, USA). The purified PCR products were then analysed using an Agilent 2100 Bioanalyzer (Agilent, USA) and a library quantification kit from Illumina (Kapa Biosciences, Woburn, MA, USA). Acceptable library concentrations should be above 2nM. Qualified sequencing libraries (with non-repeating Index sequences) were diluted to gradients and mixed in proportions according to the required sequencing volume. They were then denatured into single strands with NaOH for sequencing. Sequencing was performed using a NovaSeq 6000 sequencer with 2×250 bp paired end reads and corresponding NovaSeq 6000 SP reagents. For the paired-end sequencing data obtained, the samples were first demultiplexed based on barcode information, and adapter and barcode sequences were removed. Length filtering and denoising were conducted using DADA2, invoked through qiime dada2 denoise-paired. ASV (feature) sequences and ASV (feature) abundance tables were obtained, and singleton ASVs (i.e., ASVs (features) with a total sequence count of only 1 across all samples) were removed as a default step.
Diversity analysis for assessment of the differences in microbial communities between hemorrhoid and adjacent normal tissues
Diversity analysis was performed to evaluate and compare the diversity of microbial communities within and between hemorrhoid and adjacent normal tissues. This analysis was conducted based on the obtained ASV (Amplicon Sequence Variant) feature sequences and ASV abundance tables. Alpha and beta diversity analyses were conducted based on the obtained ASV feature sequences and the ASV feature abundance table. Alpha diversity analysis primarily employed seven key indices, including observed species, Shannon, Simpson, Chao1, goods coverage, and Pielou’s evenness, to assess the diversity within the habitat. Beta diversity analysis, on the other hand, primarily involved the calculation of four dissimilarity metrics (weighted_unifrac, unweighted_unifrac, Jaccard, and Bray-Curtis) and employed six major analytical approaches to assess diversity between different habitats or sample groups.
Analysis of 16S rRNA Gene Sequence Results
Species annotation was performed based on ASV (feature) sequence files using the SILVA (Release 138, https://www.arbsilva. de/documentation/release138/) database with the NT-16S database. The abundance of each species in each sample was then tallied according to the ASV (feature) abundance table. The confidence threshold for annotation was set at 0.7.
Cell culture and treatment
RAW264.7 cells were obtained from Cell Research (Shanghai, China) and were cultured in Dulbecco’s Modified Eagle Medium (DMEM) containing 10% fetal bovine serum (FBS), under standard conditions of 37°C and 5% CO2.
When the cell confluence reached 80%, the cells were collected, counted, and then seeded in a 6-well plate at a concentration of 2×105 cells/ml. When the cell confluence reached 60%, the cells were stimulated with LPS and different doses of P2 for 12 hours. Normal culture medium containing an equivalent volume of PBS was used as a negative control. Peptide P2 (more than 96% purity) were chemically synthesized by GL Biochem Ltd (Shanghai). The sequence of peptide P2 is Pro-(D-Pyr)-(D-Cys)-Bip-Arg- Gly-Glu-Gly-Gly-Gly-Gly-Ile-Val-Arg-Arg-Ala-Asp-Arg-Ala-Ala- Val-Pro, of which D-Pyr is 3-(3-pyridyl)-D-alanine, Bip is L-4,4’- biphenylalanine. siRNAs targeting VCAM-1 mRNA were used to silence VCAM-1 in mouse RAW264.7 cells. The sequences of the siRNAs were as follows: Forward, 5′-ggagcagacagcuaaauaatt-3′, Reverse, 5′-uuauuuagcugucugcucctt-3′. Following the manufacturer’s instructions, RAW264.7 cells were transfected with 20 nmol/L VCAM-1 siRNA using Lipofectamine 8000 reagent (Beyotime, Shanghai, China).
Victoria Blue Staining
Sections were routinely dewaxed and hydrated. A hydrophobic circle was drawn, and the sections were cleaned with distilled water. The sections were oxidized with potassium permanganate solution for 5 minutes and rinsed thoroughly with running water. They were then bleached with oxalic acid solution for 2-3 minutes until the specimens were colourless, followed by thorough rinsing with running water. The sections were briefly washed with 95% alcohol and stained with Elastin stain by adding a few drops of the stain, covering the sections, and incubating them in a 37°C incubator for 1 hour. Differentiation was performed with 95% alcohol by quickly dipping the sections until no excess stain was removed. The sections were rinsed with fine running water for 3-5 minutes and stained with Van Gieson’s stain for 1 minute. The stain was poured off without rinsing with water. The sections were differentiated with 95% alcohol for a few seconds, dehydrated with absolute alcohol, and cleared with xylene. They were mounted with neutral balsam and observed under a microscope.
Real-time Fluorescent Quantitative PCR (qRT-PCR)
RNA was extracted from hemorrhoid tissue using Trizol and its concentration was determined using a UV spectrophotometer. cDNA was synthesized according to the instructions of the 5X All- In-One Master MIX kit, with the reaction conditions set at 25℃ for 10 minutes, 42℃ for 15 minutes, 85℃ for 5 minutes, followed by storage at -80℃. The qRT-PCR system consisted of 20μL of cDNA, including 0.8μL of cDNA product, 0.6μL of each upstream and downstream primer (as shown in Table 1), and water added to make up to 20μL. The reaction conditions were as follows: initial denaturation at 95℃ for 10 minutes, followed by 40 cycles of denaturation at 95℃ for 15 seconds and annealing/extension at 60℃ for 60 seconds. The experiment included two parallel replicate wells, and all samples were tested in triplicate. Relative quantification was employed, with the relative expression levels of the target genes represented by -△Ct and 2^-△Ct, respectively. All samples were handled on ice, and all items used were treated with RNase-free reagents. The housekeeping gene GAPDH was used for normalization.

Western Blot
After protein extraction and denaturation with loading buffer at 100°C, 20μg of protein samples were separated on a 10% SDSPAGE gel and then transferred to a PVDF membrane (Millipore, Billerica, MA, USA). The membrane was blocked in 5% milk for 3 hours and then incubated overnight at 4°C with primary antibodies. It was then incubated with anti-mouse or anti-rabbit secondary antibodies (1:5000, Protein tech, Chicago, USA) on a shaker at room temperature for 1 hour. Protein bands were visualized using ECL luminescent liquid (Beyotime, Shanghai, China). ImageJ 1.44 was used to analyse protein expression. The primary antibodies used were as follows: CD86 (1:1000, Protein tech, Chicago, USA), iNOS (1:1000, WanleiBio, Shenyang, China), VCAM1 (1:1000, WanleiBio, Shenyang, China), MMP9 (1:1000, Protein tech, Chicago, USA), and GAPDH (1:1000; MULTISCIENCES, Hangzhou, China).
Immunohistochemistry
Briefly, all fresh tissues were immersed in 4% paraformaldehyde (Sigma-Aldrich, St. Louis, USA) for 30 minutes at room temperature. The tissues were dehydrated through an ascending ethanol gradient, embedded in paraffin, sectioned (6μm thickness), and immersed in xylene. The tissue sections were blocked with immunohistochemical blocking solution (Beyotime, Shanghai, China) at 37°C for 30 minutes. The blocking solution was discarded, and the slides were washed three times with immunohistochemical washing solution (Beyotime, Shanghai, China) for 5 minutes each at room temperature. Next, the primary antibody (Lipopolysaccharide Core, clone WN1 222- 5, HycultBiotech (#HM6011-20UG)) was added and incubated at 37°C for 45 minutes. The antibody was discarded, and the slides were washed three times with immunohistochemical washing solution (Beyotime, Shanghai, China) for 5 minutes each at room temperature. Then, the secondary antibody (anti-rabbit IgG (H+L), F(ab’)2 fragment (Alexa Fluor 488 conjugate) antibody (#4412) or anti-rabbit IgG (H+L), F(ab’)2 fragment (Alexa Fluor 555 conjugate) antibody (#4413)) was added and incubated at 37°C for 45 minutes. The antibody was discarded, and the slides were washed three times with immunohistochemical washing solution (Beyotime, Shanghai, China) for 5 minutes each. Finally, the slides were mounted using immunofluorescence mounting solution (Sigma-Aldrich, St. Louis, USA).
ROS detection
DCFH-DA (Beyotime, Shanghai, China) was diluted in serumfree culture medium at a ratio of 1:1000 to achieve a final concentration of 10μM. The cell culture medium was removed, and an appropriate volume of the diluted DCFH-DA was added. The volume added should be sufficient to completely cover the cells. After incubation 37°C in a cell culture incubator for 20 minutes, cells were washed three times with serum-free cell culture medium to thoroughly remove DCFH-DA that has not entered the cells and observe under a fluorescence microscope (OLYMPUS, IX53P1F).
Statistical analysis
All results are presented as mean ± SE (standard error). Statistical analyses were conducted using GraphPad Prism 8.0.2 software (GraphPad Software, San Diego, CA). To perform statistical comparisons, we initially assessed whether the data followed a normal distribution. Subsequently, the Brown- Forsythe test was employed to evaluate similarity of variances among normally distributed data. If variances were similar, Student’s t-test was utilized for comparisons between two groups, or ANOVA was employed for comparisons involving more than two groups. In all cases, a two-tailed probability less than 0.05 was considered indicative of statistically significant differences. Details of the statistical analyses applied to each experiment are provided in the corresponding figure legends.
Results
Gram-negative bacteria are significantly more abundant in hemorrhoid tissues

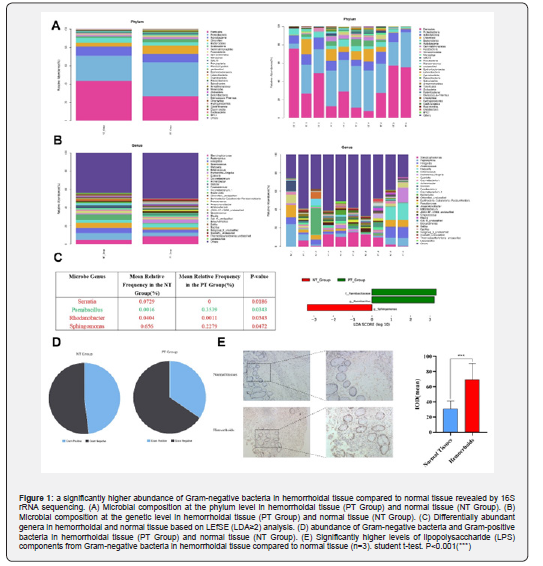
In this study, 16S rRNA sequencing was conducted on 5 pairs of hemorrhoidal tissues and their adjacent normal tissues. As shown in the rarefaction curve, the 16S rRNA sequencing in this study reached a sufficient sequencing depth, detecting a high abundance of 30 microbial genes, accounting for 49.81% to 55.93% of the total gene content. The most detected bacteria are shown in Fig.1A and B. At the genetic level, differences in microbial content between the PT (hemorrhoid tissue) group and NT (normal tissue) group were compared, the frequencies of Serratia, Rhodanobacter, and Sphingomonas in the PT group were lower than in the NT group (P < 0.05), while Paenibacillus was more abundant in the PT group. Based on the results of LEfSE (LDA≥2), Sphingomonas and Paenibacillus exhibited significant differences, with Sphingomonas population being significantly decreased and Paenibacillus bacteria being significantly increased (Fig. 1C).
Proteobacteria is the largest and most diverse phylum of bacteria, and are all Gram-negative bacteria, characterized by an outer membrane mainly composed of LPS. By calculating the abundance of Gram-positive and Gram-negative bacteria in hemorrhoid tissues and normal tissues, it was found that in comparison to normal tissues, the abundance of Gram-negative bacteria was higher in hemorrhoidal tissues than Gram-positive bacteria (Fig.1D). This view is evidenced by the observation of the presence of LPS, a component of the cell wall of gram-negative bacteria, within hemorrhoidal tissues by immunohistochemistry (Figure 1E). Research has shown a close association between the Proteobacteria and inflammation. An increase in Proteobacteria is indicative of ecological imbalance and an unstable structure of the intestinal microbiota. This study found that in hemorrhoid tissues, Proteobacteria is the dominant phylum, suggesting that the increase in Proteobacteria may be involved in the development of hemorrhoids.
Significant inflammatory environment within hemorrhoid tissues
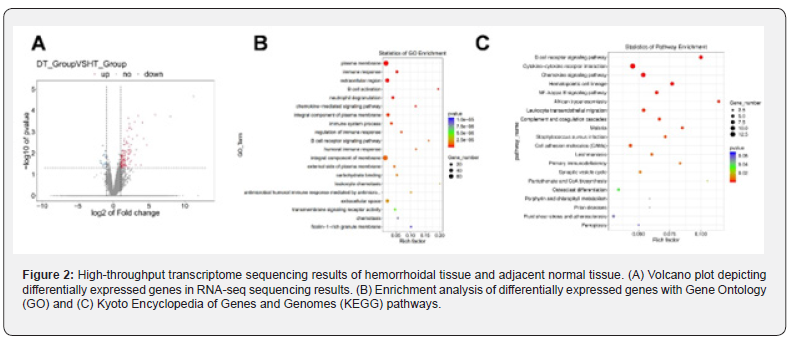
To explore the specific regulatory mechanisms by which Gramnegative bacteria are involved in the development of hemorrhoids, this study conducted high-throughput transcriptomic sequencing on 10 pairs of human hemorrhoid tissue and adjacent normal tissue. A total of 151 differentially expressed mRNAs were detected (Fig. 2A). Among them, 135 mRNAs were significantly upregulated in hemorrhoid tissues compared to normal tissues (FC > 2, P value < 0.05). Functional enrichment analysis of the differentially expressed mRNAs was conducted. GO analysis showed that the mRNAs that are mainly enriched are related to immune response, B cell activation, neutrophil degranulation, chemokine-mediated signaling pathway, and immune system processes (Figure 2B). KEGG pathway analysis revealed the involvement of these mRNAs in inflammatory-related pathways such as B cell receptor signaling pathway, cytokine-cytokine receptor interaction, chemokine signaling pathway, and NF-kappa B signaling pathway (Fig.2C). All these results suggest the presence of a significant inflammatory response in hemorrhoid tissues.
Presence of M1 macrophage polarization and an inflammatory response in hemorrhoidal tissues
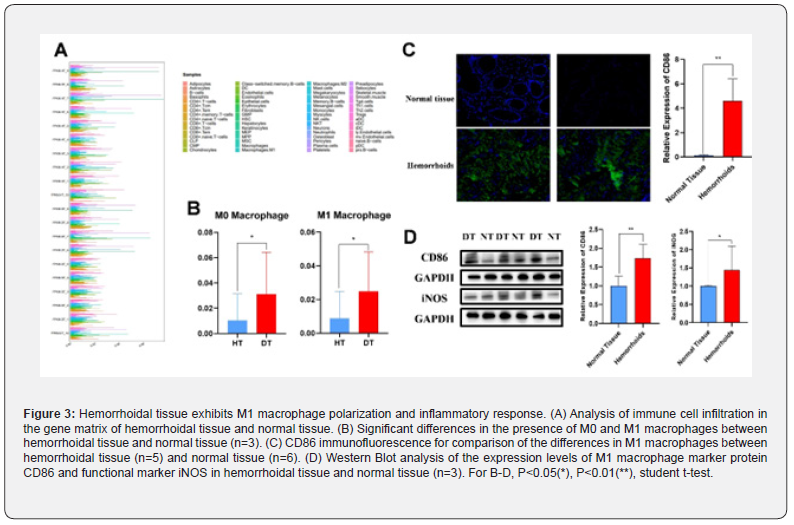
CIBERSORTx algorithm was used to analyse the differential gene expression matrix from the sequencing data. Results revealed that, compared to normal tissues, the infiltration of M0 and M1 macrophages in hemorrhoid tissues was significantly increased (Fig. 3A, B). This result was confirmed by CD86 immunofluorescence analysis of hemorrhoidal tissue sections in comparison with normal tissue (Fig.3C). In addition, Western Blot analysis of CD86 and the functional macrophages marker iNOS in hemorrhoid tissue showed that the expression levels of the M1 macrophage markers CD86 and iNOS were significantly higher in the hemorrhoidal tissues than in normal tissues (Fig.3D). It is evidenced that Gram-negative bacteria cell wall component LPS can induce macrophages polarization toward the M1 phenotype, leading to the production of pro-inflammatory responses and pro-inflammatory factors such as IL-6 and TNF-α. Thus, the enrichment of Gram-negative bacteria and the occurrence of inflammation in hemorrhoids may be closely linked through the polarization of M1 macrophages.
High expression of VCAM-1 promotes inflammation and M1 macrophage polarization
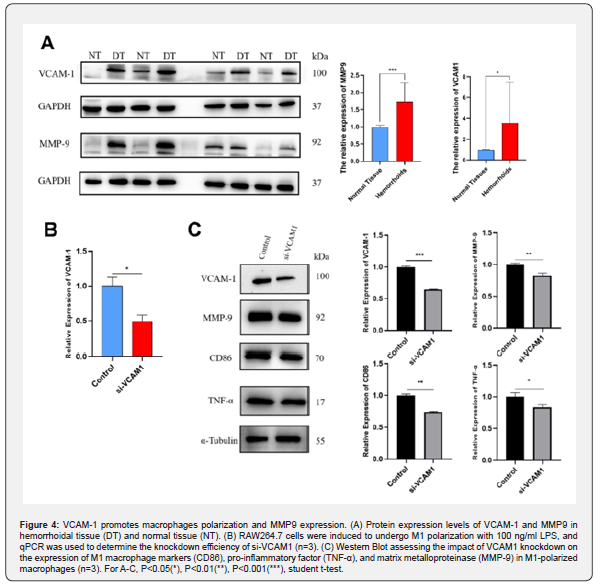
Through PPI (Protein-Protein Interaction) analysis, the top 10 differentially expressed genes from the transcriptome sequencing data were selected for correlation analysis with M0 and M1 macrophages. The results indicated that VCAM-1 and MMP-9 have the highest correlation with the infiltration of M0 and M1 macrophages (Table.2) highlighting their important role in the polarization of M1 macrophages and the pathogenesis of hemorrhoids. Immunoblotting results confirmed that the expression levels of VCAM-1 and MMP-9 in hemorrhoid tissues were significantly higher than in normal tissues (Fig.4A). RAW264.7 cells were polarized to M1 with LPS, interference with the expression of VCAM-1 using RNA interference (si-VCAM1) resulted in downregulation of M1 macrophage markers, including CD86 and the pro-inflammatory cytokine TNF-α in the si-VCAM1 group (Fig.4B and C). Moreover, the expression level of MMP-9, which was predicted to be significantly correlated with VCAM-1, also showed a decrease (Fig.4C). This demonstrates that the high expression of VCAM-1 in the hemorrhoidal tissues may promote the polarization of macrophages.
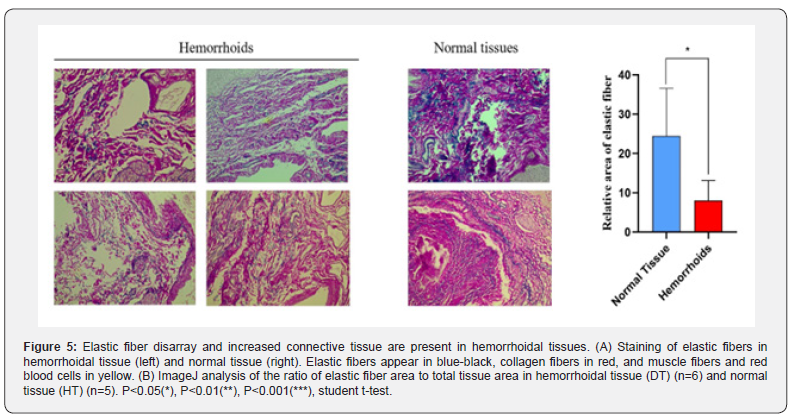
Disarray in elastic fibers and high expression of MMP9 in hemorrhoidal tissues
Collagen and elastic fibers are major components of connective tissue and play a crucial role in maintaining normal tissue function. As previously mentioned, high levels of activated MMPs are rapidly produced and released in inflammatory sites. MMP-9 degrades type IV collagen and gelatine and may play an important role in the degradation of elastic fibers and tissue remodeling and disarray [18]. A significant correlation between MMP-9 and the infiltration of polarized macrophages in hemorrhoidal tissues was observed in this study (Fig.3D). Victoria blue staining revealed that, compared to normal tissues, hemorrhoid tissues showed a decrease in collagen volume and a significant reduction in the relative content of elastic fibers (Fig. 5). This suggests that the deterioration of connective tissue in hemorrhoids is associated with marked fragmentation of collagen and elastic fibers, which is related to the inflammation triggered by M1 polarization, leading to the promotion of MMP-9 expression.
MMP-9 inhibitor peptide P2 alleviates the development of hemorrhoids by suppressing M1 polarization
RAW264.7 cells was stimulated with 100 ng LPS to induce M1 polarization, the induction was verified by qPCR analysis of M1 macrophage markers CD86 and iNOS, as well as functional markers MCP-1, IL-1β, IL-6, and MMP-9 (Fig.6A). Peptide P2, an MMP-9 inhibitor designed and synthetized by our team [19], was then added to the induced M1 macrophages at different doses (0, 2, 4, 8, 16, and 32μM), then, cells phenotype and functional status were analysed by Western Blot. The results showed that P2 significantly reversed LPS-induced CD86 expression and inhibited the expression of the pro-inflammatory factor TNF-α (Fig.6B). This indicates that peptide P2 can inhibit M1 polarization in LPS-induced RAW264.7 cells. The impact of peptide P2 on the expression levels of CD86 was further confirmed (Fig.6C). ROS levels were also measured using the ROS Assay Kit, observations under a fluorescence microscope showed a significant reduction in the green fluorescence area after administration (Fig.6D). These results suggest that peptide P2 can exert its effects by reducing ROS levels and oxidation in cells.
In conclusion, the MMP-9 inhibitor peptide P2 mitigates the development of hemorrhoids by inhibiting M1 macrophage polarization and the inflammatory response in hemorrhoid tissues, providing a potential new strategy for exploring novel treatment approaches.
The expression levels of CD86 in response to peptide P2 were examined. (D) ROS levels were measured using the DCFH-DA method, and fluorescence was observed under a microscope. The results indicate that peptide P2 inhibits the generation of ROS in a dose-dependent manner.
Discussion
Bacterial infection was previously considered as one hypothesis for the development of hemorrhoids. However, a high bacterial load within the anal canal did not impair wound healing [20], and the local application of antibiotics did not improve wound healing or reduce infections in hemorrhoids [21]. A recent meta-analysis showed that the post-haemorrhoidectomy wound infection rate was only 1.7% in 1326 patients [22]. It is generally believed that good blood supply leads to proper wound healing [23], which led scientists to believe that microorganisms play a very limited role in the occurrence and development of hemorrhoids. However, these data only assessed postoperative recovery and did not delve into the process of hemorrhoid development. Moreover, due to traditional methods, some low-abundance microorganisms were challenging to be investigated.

This study, for the first time, conducted a comprehensive analysis of the microbial composition inside hemorrhoidal tissue using 16S rRNA gene sequencing. The study found that there was no significant difference in microbial diversity between the hemorrhoidal tissue and normal adjacent tissue. Interestingly, the study noted a high abundance of Gram-negative bacteria inside hemorrhoids and confirmed the widespread presence of Gram-negative bacteria in clinical samples through immunohistochemistry (Figure 1D and E). The major component of the Gram-negative bacterial cell wall, LPS, is known to widely participate in the human immune response [24]. This investigation has, for the first time, discovered and confirmed the high abundance of Gram-negative bacteria inside hemorrhoids (Figure 1D). When bacteria invade anal tractus, human macrophages play a role in phagocytosis. Dead Gram-negative bacteria release a significant amount of LPS, which promotes the polarization of macrophages into the M1 state. This study found that, compared to normal tissue, there was severe disruption of elastic fibers within hemorrhoid tissues (Figure 5). Subsequent transcriptome data also indicated the presence of a widespread inflammatory environment inside hemorrhoids (Figure 2 and 3). Through immune infiltration analysis, it was observed that there was a high infiltration of M0 and M1 macrophages within hemorrhoids, further supporting the significant role of Gram-negative bacteria (Figure 3).
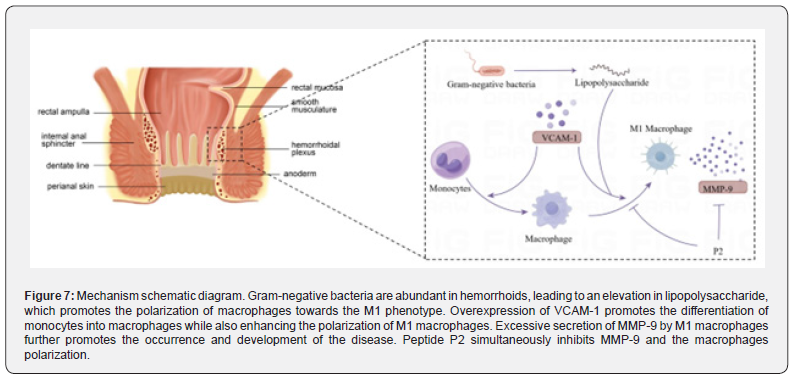
The degradation of connective tissue within the anal cushion is considered a key factor in the development of hemorrhoids. S. Willis and colleagues found that collagen metabolism is deteriorated in hemorrhoid patients [25]. Serra R. and collaborates found an increased activity of MMP-2 and MMP-9 within hemorrhoid tissues, which led to the degradation of collagen and elastic fibers, resulting in connective tissue deterioration [18]. Fox A and colleagues discovered a familial inheritance in hemorrhoid patients, leading to the breakdown of the Treitz muscle and supporting connective tissues degradation [26], a viewpoint further confirmed by a recent study by Zheng T et al.[27], who found a genetic susceptibility in hemorrhoids [27]. They identified gene mutations in ELN, SRPX, and COL5A2, which code for elastic proteins, and overexpression of the MYH11 gene, which codes for smooth muscle myosin, resulting in functional impairments of smooth muscle, epithelial, and connective tissues. However, the molecular pathology of hemorrhoids still requires further research.
This study further reveals that the polarization of M1 macrophages leads to aggravated inflammation, changes in connective tissue, and the subsequent deterioration of hemorrhoids. In addition, a significant positive correlation between the high expression of genes VCAM-1 and MMP-9 and the infiltration of M1 macrophages was revealed by gene analysis (Figure 7). Subsequent interference with VCAM-1 expression revealed a significant correlation with M1 polarization of macrophages and a reduction in MMP-9 expression (Figure 4). This research also, for the first time, demonstrated the therapeutic potential of inhibiting MMP-9 through the use of a related inhibitory peptide, Peptide P2, an MMP-9 inhibitor designed independently in our laboratory. P2 can inhibit the expression of CD86 and TNF-α, confirming suppression of macrophage polarization into the M1 state. Additionally, it can significantly inhibit the formation of reactive oxygen species (ROS) (Figure 6). Therefore, peptide P2 has the potential to alleviate symptoms in various ways and could be applied in the treatment of hemorrhoids.
Conclusion
This study, for the first time, employed 16SrRNA gene sequencing combined with transcriptome sequencing to classify the microbial community in hemorrhoid tissues. It revealed a high abundance of Gram-negative bacteria in hemorrhoids, leading to an increase in LPS content, which promotes the polarization of macrophages into the M1 state. This, in turn, elevated the expression of VCAM-1 and MMP9, thereby exacerbating the deterioration of hemorrhoidal tissue. On one hand, this increased the degree of inflammation, and on the other hand, it intensified the deterioration of connective tissue. The research team designed the MMPs inhibitory peptide P2, which can inhibit M1 macrophage polarization and reduce ROS production, thus mitigating the development of hemorrhoids. In summary, this study has provided an insight into the mechanisms underlying the development of hemorrhoids, offering new strategies for their treatment.
References
- Duthie HL, Gairns FW (1960) Sensory nerve-endings and sensation in the anal region of man. Br J Surg 47: 585-595.
- Johanson JF, Sonnenberg A (1990) The prevalence of hemorrhoids and chronic constipation. An epidemiologic study. Gastroenterology 98(2): 380-386.
- LeClere FB, Moss AJ, Everhart JE, Roth HP (1992) Prevalence of major digestive disorders and bowel symptoms, 1989. Adv Data 24(212): 1-15.
- Yang JY, Peery AF, Lund JL, Virginia P, Robert SS (2019)Burden and Cost of Outpatient Hemorrhoids in the United States Employer-Insured Population, 2014. Am J Gastroenterol 114(5): 798-803.
- Margetis N (2019) Pathophysiology of internal hemorrhoids. Ann Gastroenterol 32(3): 264-272.
- Wong SH, Yu J (2019) Gut microbiota in colorectal cancer: mechanisms of action and clinical applications. Nat Rev Gastroenterol Hepatol 16(11): 690-704.
- Björkegren JLM, Lusis AJ (2022) Atherosclerosis: Recent developments. Cell 185(10): 1630-1645.
- O'Toole PW, Jeffery IB (2015)Gut microbiota and aging. Science 350(6265): 1214-1215.
- Chen K, Gao Z, Ding Q, Cheng T, Haiyu Z, et al. (2022) Effect of natural polyphenols in Chinese herbal medicine on obesity and diabetes: Interactions among gut microbiota, metabolism, and immunity. Front Nutr 28:9:962720.
- Cani PD, Amar J, Iglesias MA, Claude K, Delphine B, et al. (2007) Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 56(7): 1761-1772.
- Locati M, Curtale G, Mantovani A (2020) Diversity, Mechanisms, and Significance of Macrophage Plasticity. Annu Rev Pathol 15: 123-147.
- Li M, Wang M, Wen Y, Hongfei Z, Guang NZ, et al. (2023) Signaling pathways in macrophages: molecular mechanisms and therapeutic targets. MedComm 4(5): e349.
- Liu YC, Zou XB, Chai YF, Yong MY (2014) Macrophage polarization in inflammatory diseases. Int J Biol Sci10(5): 520-529.
- Murray PJ, Allen JE, Biswas SK, Edward AF, Derek WG, et al. (2014) Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41(1): 14-20.
- Taweevisit M, Wisadeopas N, Phumsuk U, Thorner PS (2008) Increased mast cell density in haemorrhoid venous blood vessels suggests a role in pathogenesis. Singapore Med J 49(12): 977-979.
- Pober JS (2002) Endothelial activation: intracellular signaling pathways. Arthritis Res Suppl 3: S109-116.
- Soriano A, Salas A, Salas A, Sans M, Gironella M, et al. (2000) VCAM-1, but not ICAM-1 or MAdCAM-1, immunoblockade ameliorates DSS-induced colitis in mice. Lab Invest 80(10): 1541-1551.
- Serra R, Gallelli L, Grande R, Bruno A, Giovanni DC, et al. (2016) Hemorrhoids and matrix metalloproteinases: A multicenter study on the predictive role of biomarkers. Surgery 159(2): 487-494.
- Qiu Z, Hu J, Xu H, Wenjing W, Caihui N, et al. (2013) Generation of antitumor peptides by connection of matrix metalloproteinase-9 peptide inhibitor to an endostatin fragment. Anticancer Drugs 24(7): 677-689.
- Barrett TJ (2020) Macrophages in Atherosclerosis Regression. Arterioscler Thromb Vasc Biol 40(1): 20-33.
- de Paula PR, Speranzini MB, Hamzagic HC, Bassi DG, Chacon SMA, et al. (1991)Bacteriology of the anal wound after open hemorrhoidectomy. Qualitative and quantitative analysis. Dis Colon Rectum 34(8): 664-669.
- Ala S, Saeedi M, Eshghi F, Parastou M (2008) Topical metronidazole can reduce pain after surgery and pain on defecation in postoperative hemorrhoidectomy. Dis Colon Rectum 51(2): 235-238.
- Bhatti MI, Sajid MS, Baig MK (2016) Milligan-Morgan (Open) Versus Ferguson Haemorrhoidectomy (Closed): A Systematic Review and Meta-Analysis of Published Randomized, Controlled Trials. World J Surg 40(6): 1509-1519.
- Gosselink MP, Harman AN, Ctercteko G (2019)Why is a haemorrhoid never infected? Colorectal Dis 21(4): 385-386.
- Park BS, Lee JO (2013) Recognition of lipopolysaccharide pattern by TLR4 complexes. Exp Mol Med 45(12):e66.
- Willis S, Junge K, Ebrahimi R, Prescher A, Schumpelick V (2010) Haemorrhoids - a collagen disease? Colorectal Dis 12(12): 1249-1253.
- Zheng T, Ellinghaus D, Juzenas S, François C, Greta B, et al. (2021) Genome-wide analysis of 944 133 individuals provides insights into the etiology of haemorrhoidal disease. Gut 70(8): 1538-1549.






























