Study on the Hepatoprotective Activity of Embelia Ribes on D- Galactosamine Induced Hepatotoxic Rats
Parimala P1*, Dayanand Reddy G2, Sudha N3 and Tripurasundari B4
1Assistant Professor, Gojan School of Business and Technology, Chennai, India
2Department of Pharmacology, Siddha Central Research Institute, Anna Hospital Campus, Arumbakkam, Chennai 600106.Tamilnadu, India
3Assistant Professor, Ethiraj College for Women, Chennai, India
4Assistant Professor, Valliammal College for Women, Chennai, India
Submission: July 27, 2023; Published: August 14, 2023
*Corresponding author: Parimala P, Assistant Professor, Gojan School of Business and Technology, Chennai, India. Email id: drpparimala@gmail.com
How to cite this article: Parimala P*, Dayanand Reddy G, Sudha N and Tripurasundari B. Study on the Hepatoprotective Activity of Embelia Ribes on D- Galactosamine Induced Hepatotoxic Rats. J of Pharmacol & Clin Res. 2023; 9(2): 555765. DOI: 10.19080/JPCR.2023.09.555765
Abstract
Hepatic disease is considered as one of the foremost health troubles worldwide. It is the foremost leading cause of death in western and developing countries. Treatment with synthetic drugs during hepatic injury may also induce adverse liver damage. Therefore, the need of effective therapeutic agents with low incidence of side effect is required. Herbal extracts have been proved to have effective hepatoprotective activity with minimal side effects. E.ribes, commonly known as Vedanga is one such traditional medicine of plant origin, reported to be useful in the jaundice treatment. However studies have not so far explored the E.ribes efficacy on hepatic damages. Hence, present study ascertained the medicinal value of E.ribes in D- galactosamine (D-GalN) induced hepatotoxic rats by analyzing biochemical parameters such as sugar, cholesterol, total bilirubin, total protein, albumin, globulin, SGPT, SGOT, ALP along with assessment of hematological parameters such as WBC, RBC, HGB and platelet. Silymarin a potent hepatoprotective drug used as reference standard. D-galactosamine (D-GalN), a well-known hepatotoxicant used as toxic drug. Results of the study showed that E.ribes extract exhibit hepatoprotective activity against the D-galactosamine (D-GalN) induced hepatotoxicity. It also necessitates the demand for executing further characterization of E.ribes to explore the component imparting hepatoprotection.
Keywords: E ribes (Embelia ribes); Silymarin; D- Galactosamine; Liver; Hepatoprotective
Abbreviations: D-GalN: D-Galactosamine; RNA: Ribonucleic Acid; ALP: Alkaline Phosphatase; TP: Total Protein; TC: Total Cholesterol; TB: Total Bilirubin; CCRS: Central Council for Research in Siddha
Introduction
Liver has a pivotal role in regulation of physiological processes of all living organisms. It is involved in several vital functions such as metabolism, secretion and storage. Furthermore, detoxification of a variety of drugs and xenobiotics occurs in liver. Liver diseases are among the most serious ailment. They may be classified as acute or chronic hepatitis (inflammatory liver diseases), hepatitis (non-inflammatory diseases) and cirrhosis (degenerative disorder resulting in fibrosis of the liver). Liver diseases are mainly caused by toxic chemicals (certain antibiotics, chemotherapeutics, peroxidized oil, aflatoxin, carbon-tetrachloride, chlorinated hydrocarbons, etc.), excess consumption of alcohol, infections and autoimmune/disorder [1]. In the present work D-GalN is used as a hepatotoxic drug as it is a well-known hepatotoxicant. It induces liver injury closely resembling human viral-hepatitis with necrosis, inflammation and regeneration. The toxicity of D-GalN is associated with the depletion of uridine pools, limited ribonucleic acid (RNA) and protein synthesis thus overall affecting hepatocellular function [2]. Most of the hepatotoxic chemicals damage liver cells mainly by inducing lipid peroxidation and other oxidative damages in liver. Enhanced lipid peroxidation may result in hepatitis and cirrhosis.
Silymarin has been used as a standard for comparison in our study as it is a standardized hepatoprotective extract of Silybum marianum (Compositae). It reverses hepatotoxin induced alterations of biochemical parameters. It has been for long the most thoroughly investigated of all hepatoprotective medicinal plants in preventing liver damage induced by carbon tetrachloride, D-GalN and paracetamol in rat models [3]. Beside expensive and ineffectual modern therapeutic agents like steroids and chemotherapy, south east Asian countries like India and China have an edge in treating hepatic disorders by means of invention of new herbal products with minimal side effect and better than the existing botanicals. Of them E.ribes is known for its efficiency against jaundice, commonly known as vidanga belongs to the family Myrisinacea. It is a well-known herbal for its antioxidant, antidiabetic [4], neuroprotective [5,6], cardioprotective, antihypertensive activity [7] and wound healing property [8]. The dried fruit was considered anthelmintic, astringent, carminative, alternative and stimulant. Hence in the present work, we have demonstrated the hepatoprotective effect of the E.ribes in the D-GalN induced hepatotoxic rats where silymarin used as the standard for substantiating the hepatoprotective role of vidanga.
Materials and Methods
Animals
Healthy male and female wistar rats (150-200gm) were used for the study. The rats were housed in polypropylene cages and maintained under standard conditions (temperature range: 65-75%F and humidity range: 40-70%). The animals had free access to a standard pellet diet (Amrut laboratory animals feed, Maharashtra house, Pune) and water utilizing aqua guard facility. This study was conducted after obtaining Institutional Animal Ethical Committee clearance (76/Pharma/SCRI/2010).
Preparation of plant extract
E.ribes (vidanga) dried fruits bought at siddha medicine shop at Redhill’s and got the authentication from siddha research center at Arumbakkam. The ethanolic extract of E.ribes was obtained by using 80% ethanol and by making it to syrupy nature after 72 hours incubation at room temperature, the last traces of solvent present was removed by vacuum.
Study design
All the animals 30 were weighed and randomly divided into five groups comprising of 6 animals in each group. The animals were classified into 5 groups for the present study.
a) Group 1: Representing normal control received only water and feed for 10days.
b) Group 2: Representing hepatotoxic control group, received D-GalN on 9th day (IM) at a dose of 400mg /kg and remaining days received Tween 80.
c) Group 3: Given silymarin at a dose of 100mg/kg once daily for 10 days orally and D-GalN at a dose of 400mg /kg injected (IM) on the ninth day.
d) Group 4: Given E.ribes in Tween 80 at a dose of 100mg/ kg orally for 10 days and D-GalN at a dose of 400mg/kg injected (IM) on the ninth day.
e) Group 5: Given E.ribes in Tween 80 at a dose of 200mg/ kg orally for 10 days and D-GalN at a dose of 400mg/kg injected (IM) on the ninth day.
Chemicals
The solvents used for extraction of the plant material were of analytical grade. D-Galactosamine (D-GalN) was purchased from Merck India Ltd., Mumbai, India. Autopax Siemens assay kits for serum aspartate aminotransferase (SGOT), alanine aminotransaminase (SGPT), alkaline phosphatase (ALP), total protein (TP), Albumin, Globulin, total cholesterol (TC), total bilirubin (TB), and Blood sugar were obtained from Healthcare Diagnostics Ltd. Gujarat, India and Silymarin from sigma, USA. All the other chemicals used were of analytical grade.
Biochemical and hematological parameters assay
On the eleventh day, blood samples were collected through retro- orbital sinus of all thirty animals. The blood was allowed to clot and centrifuged at 2500 rpm for 10 minutes. The serum was separated and used for the analysis. Biochemical parameters were estimated by semi-automated analyzer for analyzing glucose, cholesterol, bilirubin, total protein, albumin, globulin, SGOT, SGPT and ALP. Hematological analysis of WBC, RBC, HGB and platelets is done by hematological analyzer.
Histopathology
The animals were sacrificed by cervical dislocation & liver was excised, washed with saline and dried with tissue paper. Liver kept aside for histopathological studies were transferred to Forma saline solution. The liver tissues were processed for paraffin embedding and sections 5μm thick were taken in a microtome. After staining with hematoxylin and eosin, slides were examined under the microscope for histo-pathological changes [9].
Statistical Analysis
Data were analyzed using statistical software package version 7.0. Student’s t-test was used to ascertain the significance of variations between different groups. All data were presented as mean ± S.E.M. Differences were considered significant at p<0.05 and p<0.01.
Results
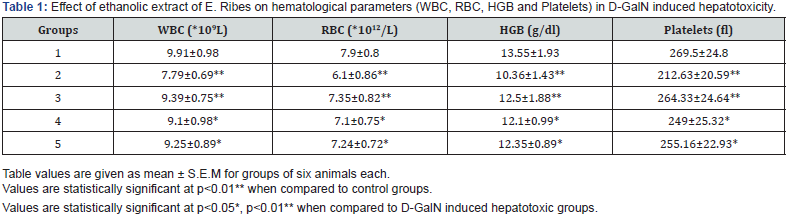
(Figure 1-5) & (Table 1)
Discussion
Since ancient times, herbal products been utilized for the treatment of various health problems. Plants and plant products are the most important herbal sources of drug discovery and development. E.ribes is one of the plant source, used in this study which has been excessively utilized in traditional medicine to cure a variety of hepatic diseases. In the present study effect of E.ribes is determined in the D-GalN induced hepatotoxic rats using silymarin as the standard drug. Glucose, cholesterol, Total protein, albumin, globulin, Total bilirubin were analyzed in the serum of all the specified groups. Glucose level was found to be decreased in the hepatotoxic group (group 2) when compared to control group (group 1), whereas pretreatment with alcoholic extract of E.ribes reduce the glucose level than the hepatotoxic group (group 2). Similar result was observed in the standard drug which received the silymarin. Significant depletion of liver glycogen and hypoglycemia is a striking feature of d-GalN/LPS-induced hepatitis. Since liver contain a full complement of the necessary enzymes involved in glucose homeostasis, toxic drugs may alter the glucose level. Arai [10], has suggested that, in D-GalN induced liver failure model, hepatic glucose production is completely arrested and the liver switches to a glycolytic mode even in a fasted animal [10]. D-GalN causes the activation of pyruvate kinase and the glycolysis pathway. The switch from gluconeogenesis to glycolysis may be due to the depletion of ATP and probable accumulation of AMP in liver tissue as a result of fulminant hepatic failure. Hypoglycemia during liver injury is a consequence of impaired gluconeogenesis and inability to mobilize glycogen stores. In the present study the depleted glucose level in group 2 rats was appreciably improved in group 4 and 5 rats pretreated with E.ribes suggesting that it may improve gluconeogenesis.
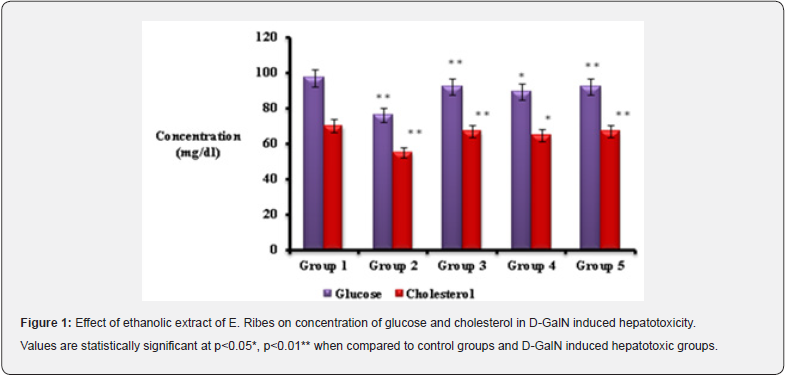

Cholesterol level was found to be increased in hyperlipoproteinemia, hypothyroidism, nephrosis, diabetes mellitus and in various liver diseases. In the present study decreased level of cholesterol was observed in hepatotoxic rats (group 2). Any liver disease will show an elevated blood cholesterol level [11]. The significant increase of cholesterol noted in the present study might be due to the inability of the diseased liver to remove cholesterol from circulation. This finding could be correlated with the results of the other studies [12]. There was observed bringing up of normal level of cholesterol using the treatment with E.ribes extract when compared to the D-GalN induced hepatoxic groups. Similarly, in silymarin pretreated groups was exhibited the significant cholesterol regulation. The liver is the major metabolic organ that synthesizes not only the protein it needs, but also produces numerous export proteins. Among the latter, plasma albumin is the most important. Export proteins are synthesized on polyribosomes bound to the rough endoplasmic reticulum of the hepatocytes. In contrast, protein destined for intracellular use is synthesized on free polyribosomes rather than bound polyribosomes [13]. Any alteration in the level of total protein, albumin and globulin leads to hepatocytes impairment. Total protein, albumin and globulin level were analyzed in the present study, and they were found to be decreased in D-GalN induced hepatotoxic rats (group 2). During hepatocellular damage protein level was found be decreased in the D-GalN induced hepatotoxic rats [14]. A finding similar to the present study was reported by Mangeney [15] in isolated rat hepatocytes indicating D-GalN induces a decrease of TG secretion and inhibits protein synthesis and secretion [15]. However pretreatment with E.ribes brought the level of protein, albumin and globulin nearly normal when compared to hepatotoxic groups, which is concomitant to the level observed in control and silymarin pretreated groups.
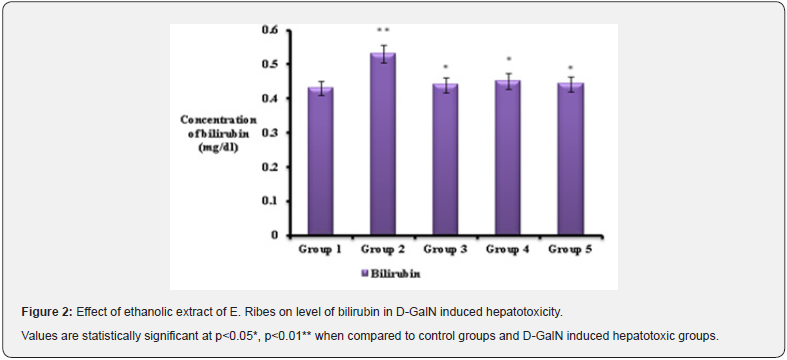
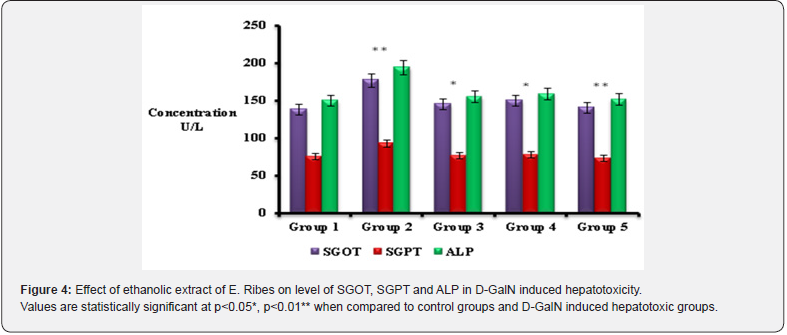
Total bilirubin levels were also measured in all the 5 groups where increased bilirubin level was noted in hepatotoxic control (group 2). Determination of serum bilirubin serves as an index for the assessment of hepatic function and any abnormal increase in the levels of bilirubin in the serum indicate hepatobiliary disease and severe disturbance of hepatocellular function. Hyperbilirubinemia is accompanied by increase in ALP [16]. Similar result was achieved in the study suggesting that increase in bilirubin levels may be due to induction of necrosis and apoptosis in liver parenchymal cells or by reduction of liver specific function in hepatotoxic control group [17]. The increased levels of bilirubin could result from an impairment of uptake or conjugation coupled with decreased excretion of the pigment. Increased levels of bilirubin in this study are in agreement with the reports of the other studies that D-GalN induced hepatitis is characterized by increased levels of bilirubin in serum [18]. In E.ribes pretreated groups (group 4 & 5) total bilirubin levels was significantly (p<0.05) decreased compared to the D-galactosamine induced hepatotoxic groups (group 2). Researchers also noted the increased bilirubin level during hepatotoxic condition which is suggesting D-GalN serves as the hepatotoxic agent [19] and E.ribes acts as the better hepatoprotective drug in rats [20]. This might be due to antioxidant activity of E.ribes extract (embelin) [21]. In hepatotoxic rats (group 2) liver marker enzymes (SGOT, SGPT and ALP) were increased compared to the normal rats. Liver marker enzymes were found to be increased in hepatotoxic condition due to xenobiotics, suggesting hepatocytes impairment and it causes leakage of serum enzymes (SGOT, SGPT and ALP) into blood stream. D-GalN intoxication is known to cause marked elevation in liver enzyme levels [22]. Similarly in our present study also increased liver marker enzymes were noted, proposing the hepatotoxic role of D-GalN in causing hepatocytes damage. The way by which D-GalN produces necrosis in hepatic parenchymal tissues is by its metabolization into UDP- hexosamine and UDP-N acetyl hexosamines in the hepatocytes and then it causes hepatic injury.
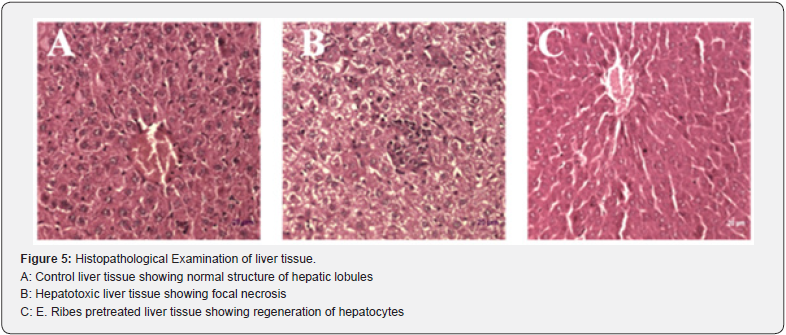
However, E.ribes pre-treatment at the dose of 100mg/kg and 200mg/kg had significantly (p<0.05 & p<0.01) prevented the D-GalN induced alteration in serum liver marker enzymes such as SGOT, SGPT and ALP in group 4 & 5 compared to the D-GalN alone induced hepatotoxic rats (group 2). The protective effect might be due to the flavanols present in the E.ribes. A study suggested that Embelin from E.ribes scavenges free radicals and results in hepatic glutathione antioxidant defense which decreases lipid peroxidation and minimizes the histological alteration in the liver [21]. Hence it might be the reason for the prevention of the cellular leakage of enzymes (SGOT, SGPT & ALP), and imparting the enzymes level nearly normal which is similar to the levels observed in control (group 1) and silymarin induced rats (group 3). Hematological parameters such as WBC, RBC, HGB and platelets were also analyzed in the present study. In hepatotoxic rats there was found to be slight increase in the level of WBC, RBC, HGB and platelets. Treatment with the ethanolic extract of E.ribes brought their levels normal similar to the silymarin induced rats. Both E.ribes and silymarin treated hepatotoxic rats showed the regulation of hematological parameters when compared to that of hepatotoxic groups. Hepatoprotective activity was studied with the alcoholic extract of E.ribes. There was a significant hepatoprotective effect using E.ribes extract with the dose of 200mg/kg than the dose of 100mg/kg. It was proved by comparing it with the D-GalN induced liver damaged rats. The hepatoprotective activity of E.ribes was also supported by histopathological studies of liver tissue which shows the necrosis in the hepatotoxic rats. However the necrosis was minimum in the E.ribes pretreated groups which is similar to the control groups.
Conclusion
Current results showed a significant increase in the levels of serum enzyme activities in D-GalN treated rats, reflecting the liver injury caused by D-GalN. The animals pretreated with the ethanol extracts of E.ribes elucidate its hepatoprotective activity against D-GalN induced. It also predicts the extracts of the E.ribes could afford significant dose-dependent protection against D-GalN induced hepatocellular injury.
Acknowledgment
The authors thank the Director General, Central Council for Research in Siddha (CCRS) for permitting us to do our research work.
- Research Article
- Abstract
- Introduction
- Materials and Methods
- Statistical Analysis
- Results
- Discussion
- Conclusion
- Acknowledgment
- References
References
- Pradeep KS (2013) Hepatoprotective activity of Ardisia solanacea in CCl4 induced Hepatoxic albino rats. Asian J Res Pharm Sci 3(2): 79-82.
- Padmanabhan P, Jangle SN (2014) Hepatoprotective Activity of Herbal Preparation (HP-4) Against D-Galactosamine Induced Hepatotoxicity in Mice International. J Pharm Sci Drug Res 6(1): 31.
- Ahmad Zubaidi Bin AL, Mainul Haque CS, Mahadeva R (2012) Clinical Study of Preventive Potentials of Consumption of Buah naga [Cactaceae] Against Paracetamol–Induced Hepatotoxicity as well as the Other Associated Biological Effects. Asian J Res Pharm Sci 2(1): 6-23.
- Bhandari U, Ansari MN (2008) Antihyperglycemic activity of aqueous extract of Embelia ribes Burm in streptozotocin-induced diabetic rats. Ind J of Exp Biol 46(8): 607-613.
- Ansari MN, Bhandari U (2008) Protective effect of Embelia ribes Burm on methionine-induced hyperhomocysteinemia and oxidative stress in rat brain. Ind J of Exp Biol 46(7): 521-527.
- Ansari MN, Bhandari U, Islam F, Tripathi CD (2008) Evaluation of antioxidant and neuroprotective effect of ethanolic extract of Embelia ribes Burm in focal cerebral ischemia/reperfusion-induced oxidative stress in rats. Fundam Clin Pharmacol 22(3): 305-314.
- Bhandari U, Ansari MN, Islam F (2008) Cardioprotective effect of aqueous extract of Embelia ribes Burm fruits against isoproterenol-induced myocardial infarction in albino rats. Ind J of Exp Biol 46(1): 35-40.
- Kumara Swamy HM, Krishna V, Shankarmurthy K, Abdul Rahiman B, Mankani KL, et al. (2007) Wound healing activity of embelin isolated from the ethanol extract of leaves of Embelia ribes Burm. J Ethnopharmacol 109(3): 529-534.
- Bancroft JD, Cook HC (1984) Chapter: Routine morphological staining. Book: Manual of Histopathological Techniques. 1st Edition Publisher: Churchill Livingstone Singapore p: 18-25.
- Arai K, Lee K, Berthiaume F, Tompkins RG, Yarmush ML (2001) Intrahepatic amino acid and glucose metabolism in a D-galactosamine-induced rat liver failure model. Hepatol 34(2): 360-371.
- McIntyre N, Rosalki S (1992) Biochemical Investigations in the Management of Liver Disease. Springer-Verlag p: 39-71.
- Dhanabal SP, Syamala G, Satish Kumar MN, Suresh B (2006) Hepatoprotective activity of the Indian medicinal plant Polygala arvensis on D-galactosamine-induced hepatic injury in rats. Fitoterapia 77(6): 472-474.
- Lin WC, Lin WL (2006) Ameliorative effect of Ganoderma lucidum on carbon tetrachloride-induced liver fibrosis in rats. World J Gastroenterol 12(2): 265-270.
- Ferencokov R, Cervinkov Z, Drahota Z (2003) Hepatotoxic Effect of D-Galactosamine and Protective Role of Lipid Emulsion. Physiol Res 52(1): 73-78.
- Mangeney M, Sire O, Montagne J, Nordmann J (1985) Effect of D-galactosamine in vitro on [U-14 C] palmitate oxidation, triacylglycerol synthesis and secretion in isolated hepatocytes. Biochem Biophys Acta 833: 119.
- Velayudham LS, Farrell GC (2003) Drug-induced cholestasis. Expert Opin Drug Saf 2(3): 287-304.
- Gujral HS, Guardiola I, Carbonell JV, Rosell CM (2003) Effect of cyclodextrin glycoxyl transferase on dough rheology and bread quality from rice flour. J of Agri and Food Chem 51(13): 3814-3818.
- Monika S, Meenakshi B, Razdan BK (2013) Hepatoprotective activity of a polyherbal preparation against D-galactosamine induced hepatoxicity in rats. Research J Pharm Tech 6(9): 1079-1082.
- Pushpendra KP, Narendra KP, Dubey BK (2012) Hepatotoxicity: Causes, Symptoms and Herbal Remedies. Research J Pharmacognosy and Phytochemistry 4(2): 104-111.
- Vaidya AB, Antarkar DS, Doshi JC, Bhatt AD, Ramesh VV, et al. (1996) Picrorhiza kurroa (Kutaki) Royle ex Benth as a hepatoprotective agent-experimental & clinical studies. J postgrad Med 42(4): 105-108.
- Sree Priya M, Bali G (2006) Effects of administration of Embelin and curcumin on lipid peroxidation ,hepatic glutathione antioxidant defense and hematopoietic system during Nitrosodiethylamine/phenobarbital –induced hepatocarcinogenesis in wistar rats. Mol Cell Biochem 284(1-2): 49-55.
- Sheik Abdulazeez Sheriff, Thiruvengadam Devaki (2012) Effect of Lycopeneon General Clinical Parameters During D-galactosamine/Lipopolysaccharide (D-GalN/LPS) Induced Hepatitis in Rats. Research J Pharm Tech 5(3): 398-403.






























