Therapeutic Alternatives for The Treatment and Prevention of Dilated Cardiomyopathy (DCM): The Era of Biological Agents
Alexandros Sarantopoulos11* and Ioannis Efthimiadis2
12nd Department of Internal medicine, Aristotle University of Thessaloniki, Greece
2 Department of Cardiology, Bioclinic Thessaloniki, Greece
Submission: July 2, 2019;Published: July 23, 2019
*Corresponding author: Alexandros Sarantopoulos, 2nd Department of Internal medicine, Aristotle University of Thessaloniki, Greece
How to cite this article: Alexandros Sarantopoulos, Ioannis Efthimiadis. Therapeutic Alternatives for The Treatment and Prevention of Dilated Cardiomyopathy (DCM): The Era of Biological Agents. J of Pharmacol & Clin Res. 2019; 7(4): 555720. DOI: 10.19080/JPCR.2019.07.555720
Abstract
Dilated Cardiomyopathy is a rare condition that can be caused by a variety of common morbidities. It is an immune mediated disease, characterized by chronic stimulation of inflammatory responses. The so far available treatment consists of agents that ameliorate only the functional burden off the circulatory system. Biological therapies have been successfully applied in several diseases derived from hyper activation of the immune system. In this mini review we investigate whether abatacept, a biological agent targeting co-stimulation, could be beneficial in inhibiting pathophysiological progression of DCM.
Dilated Cardiomyopathy–Introduction
Dilated cardiomyopathy is the most common cardiomyopathy. It reflects a functional state where the anatomic structures of the heart cannot support a proper blood circulation. Dilated cardiomyopathy is not a common disease. Nevertheless, its progression is crucial for the survival of the patient, since it can lead even rapidly to the need of transplantation. The a etiopathogenesis of the disease includes some very common morbidities of the general population, such as hypertension, coronary heart disease and heart attack. Metabolic diseases such as diabetes mellitus, and thyroid disease have been associated with DCM. Also, infections predominately viral can lead to DCM. All these conditions can affect the heart muscle, which in genetically predisposed patients can favor the development of DCM [1].
Pathophysiology of DCM: The Role of The Immune System
The role of the immune system has been long ago documented in the aetiopathogenesis and evolution of the disease. A proinflammatory microenvironment favors the development of Th1 and Th17 responses of the acquired immunity, This polarization of the adaptive immunity leads, thru a type IV hypersensitivity reaction (delayed, cell mediated type of tissue damage), to chronic inflammation, resulting in functional deregulation of the heart muscle. On this regard, insufficient mobilization of the homeostatic anti-inflammatory Treg response confers to further propagation of tissue damage and disease progression [2].
Therapy: Current Available Agents
Therapeutic interventions consist of cardiovascular agents that regulate the equilibrium of systemic circulation, thus compensating the functional impairment of the heart. The so far therapeutic approach only retards the progression to a non-compensative heart failure and does not intervene in the pathophysiological evolution of the disease. It has been already pointed out that DCM is an immune mediated condition. As a result, several immunosuppressive drugs have been appointed as potential therapeutic regimes for achieving a certain degree of disease repression. Nevertheless, the subclinical progression of the disease is a crucial factor for which the onset of clinical symptoms is often associated with substantial damage, annulling practically any benefits of a classical immunosuppressive intervention, which either way, per se, needs a certain degree of time in order to produce clinical amelioration.
Biological Therapy
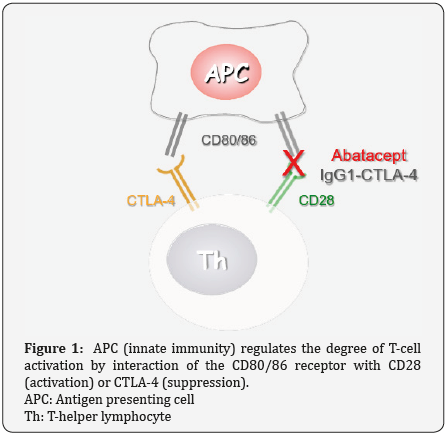
Beyond classical immunosuppression, the era of biological agents permitted pharmacological manipulation of several aspects of the immune response, in order to favor the rearrangement of an immune effective environment that will retard disease progression in several diseases. Central role in the establishment of a chronic inflammatory microenvironment holds the co- stimulatory pathway. This pathway consists of receptors and ligands that regulate the level of activation of the adaptive immune response from cells of the innate immune compartment. The main receptor of this pathway on antigen presenting cells is CD80/86 that can bind to either CTLA-4 (suppressive) or CD28 (activating) receptors on the surface of T-cells. The fact that the same receptor on APCs can either activate of suppress T-cell function, is a sophisticated homeostatic evolution of the immune system acting as a dimmer that can properly regulate the level of activation of T-cells (Figure 1).
Abatacept for DCM
In autoimmune diseases, suppression of T-cell activation, by inhibiting CD80/86-CD28 interaction has been proven clinically beneficial in conditions such as Rheumatoid Arthritis. Could the application of such a therapeutic strategy have beneficial effects of DCM? First of all, it has been documented that DCM is associated with an enhanced overexpression of constitutive molecules (CD25) on behalf of T-cells presupposing CD28 overexpression, both propagating continuous activation [1]. Furthermore, other reports discuss that the alternative CD80/86-CTLA-pathway is associated with down regulation of the inflammatory Th17 response [2], a helper T-cell response that contributes to the development of DCM. Therefore, the authors suggest that potential use of the monoclonal antibody abatacept in DCM, could indeed down regulate T-cell activation in patients manifesting premature clinical symptoms of DCM (Figure 1). Blockade of the CD80/86-CD28 pathway with abatacept will also leave unaffected the suppressive part of the dimmer complex (CD80/86-CTLA4) further favoring suppression of inflammatory Th responses (Th17).
Conclusion
Manipulation of the immune system has been proven so far effective for the treatment of autoimmune diseases and certain types of malignancies. Sophisticated application of current biological therapies may still provide therapeutic solutions in other immune mediated diseases such as DCM.






























