A Review: On Human Short Stature
Rana Khalid Iqbal*, Nasrullah, Khizra Shoukat, Mehwish Sultana and Faiza Arif
Institute of molecular biology & biotechnology Bahauddin Zakariya University, Pakistan
Submission: May 21, 2019;Published: June 26, 2019
*Corresponding author: Rana Khalid Iqbal, Department of Institute of molecular biology & biotechnology Bahauddin Zakariya University, Pakistan
How to cite this article: Rana Khalid Iqbal, Nasrullah, Khizra Shoukat, Mehwish Sultana, Faiza Arif. A Review: On Human Short Stature. J of Pharmacol & Clin Res. 2019; 7(3): 555714. DOI: 10.19080/JPCR.2019.07.555714
Summary
Human height is a polygenic and complex character control by numerous genes, due to these many problems are related to it one of them is short stature, in which individuals do not achieve standard height compare to others. What are the genes and other factors involved in it, to know it we go thought resent and old published available data present and research articles, review articles, short and long communication and other data related to it. By going through research and literature we came to know that Short stature may be due genes or Short Stature homeobox (SHOX) region present on the pseudo autosomal region (PAR1) of both X and Y chromosome contains and major genes for human growth defect in it ultimately results in short stature or defect in protein which have effect on height and results of protein are on hormones which is also major contributor of Short stature and also that Growth hormones and insulin like growth factors are very important for achievement of normal height and defect in any one can leads to short stature. To get rid of Short stature researchers developed Recombinant human growth hormones therapy and recombinant insulin-like growth factor to treat short stature. we conclude that genes responsible for short stature are present in SHOX region, proteins are involved as well as hormones, we can treat the short stature with the help of Recombinant Human growth hormones and insulin like growth factors.
Keywords: Genes; IGF; rhGH; SHOX
Abbreviations: SS: Short Stature; GH: growth hormones; GHD: Growth hormone deficiency; ISS; idiopathic short stature; IGF: insulin-like growth factor; CNVs: Copy number variants; GWAS: Genome-wide association studies; SNPs: Single polymorphic nucleotides as predictors;
Introduction
Height is a heritable character which transfer from parents to offspring through genes. Due to genes involvement many problems are related to it, one of them is Short stature. Short Stature (SS) is disorder in which patients are not able to obtain his/her normal height (compare to his sex, age and people of community).
There are two types of SS, those which obtain their height in later condition known as familial SS in which growth hormones (GH) are produced in later stages and those which don’t reach to their normal height are called idiopathic short stature [1]. SS can be syndromic or dis-syndromic, proportionate or disproportionate and associated or not to bone malformation. Due to large problems associated with SS, researcher are being able to find all those factors due to which SS occur i.e. heredity, mutation in extracellular matrix protein, other types of mutation, certain types of infection, hormonal dis-balance in body, malnutrition and environmental conditions [2] which are things which have effects on height.
Here point to be noted that 75% growth is effect by inherence (DNA) and 180 loci are related to height and in human growth complex genes are involved, studies show that >600 variants are related with normal height and growth in human [3-5] As DNA is responsible for normal functioning of body and defects in DNA leads to abnormities same is the case with the SS in which Growth hormone deficiency (GHD), which is responsible for 55.56% of SS case worldwide and rest include idiopathic short stature (ISS) 30% and hypothyroidism 4.07% small gestational age (SGA) 2.59% and congenital ovarian hypoplasia 2.29% [5]. Growth hormones deficiency (GHD) is a common source in all SS cases (1 out of 4000) [6]. GH is not the only thing that play its role in SS but also insulin-like growth factor (IGF) as well play significant role in gaining normal height in a linear manner and both show combined effect in the development of body parts, defect in any one of them (GH, IGF ) may leads to SS [7-9].
SS as a genetically originated case was identified as short stature homeobox (SHOX) [10,11]. SHOX is pseudo autosomal region 1 (part1) with high reproducibility present on both X and Y chromosome containing genes pairs having fundamental role in linear growth of an individual and limb development [10] When SHOX is completely/partially absent or deleted or duplication upstream or downstream results are skeletal abnormalities like SS and abnormalities in other body parts like limbs remain short occurs [12-16]. Mutations of SHOX region occur which can either be Homozygous or heterozygous, X chromosome, hemizygous or its downstream accompaniment causing Langer mesomelic dysplasia in 75% of individuals which is the more unadorned in medical point of view, upstream/downstream Heterozygous mutations in SHOX region or its enhancer essentials are related to 2-22% of cases of ISS which is more damaging in females as compare to males [18] . Turner syndrome is also one cause of SS [20,19]. As damage of DNA is involved due to this protein and also hormones are damaged which are reveal in review and the effect of them on height and SS. In this review will look over the SS s important features and discuss them Figure 1.
Phenotype
Short stature is because of mutations which can either be homozygous, heterozygous, X chromosome, hemizygous or multipart disabling of SHOX region that results in either form of phenotype like Langer mesomelic dysplasia, milder skeletal dysplasia, Leri-Weill dyschondrosteosis which are caused by heterozygous mutations or deletions of SHOX region clinically known as ISS, Turner syndrome which is caused by heterozygous SHOX deletion also result in SS and growth failure, Mesomelia effecting the magnitudes of body [21-23] . Many other syndromes (known as rasopathies) includes Noonan, LEOPARD, Costello, cardio-facial-cutaneous, and neurofibromatosis-Noonan syndrome, all these results in SS and growth failure. Faciogenital dysplasia SS is caused by mutation in FGD1 and mutations in SOX 9 which give rise to skeletal dysplasia, campomelic dysplasia [24] and also skeletal abnormities and microcephaly [25-27] Russell-silver syndrome is also one of the phenotypes of SS and which individually don’t reach to optimize height [27,28,31] SS individuals suffer from asymmetry in body (limb), having triangular faces, short arms, well known forehead, in some cases feeding problems, changes are observed in skeletal like symbol of Vickers ligament which leads the lunate to the distal lot of the radius and patients with SHOX deficiency affect the level of estrogen hormones which cause abnormalities in skeletal formation [29-30] SHOX deficiency give rise to more acute skeletal damages in adult females as compare to kids and males Females with SHOX deficiency have normal ovarian shape and function [30].
Genes Responsible for Short Stature
The monogenic disorders normally related with short stature and bone dysmorphology and also more than 300 different monogenic disorders of bone as well. One study shows that deficiency of SHOX is monogenic disorders of bones but common one in many other shreds of evidence and studies is osteodysplasia [27]. The only treatment to overcome this disorder is that clinician and radiologists should recognize the phenotype of this disorder and then make the current diagnosis [28]. Two basic approaches are generally described to discover the genes involved in growth regulation. These two basic approaches include: one is GWAS (genome-wide association studies) [30] and the second is to perform genetic studies in a patient with enormously short or tall height and search for causative variations [31,32]. In the first approach, researchers found that more than 180 loci are related to adult height. Each loci contribute small amount of effect to height and contain various genes, about 10% of phenotypic variation is explained by cumulative loci [33] In an alternative of it if we use SNPs (single polymorphic nucleotides) as predictors in GWAS approach it can explain about up to 40% of the variance in height [32].
In the second approach, one can test the previously described gene defects, or genome-wide research for copy number variants (CNVs) is performed or for mutations wholeexome sequencing. A number of genes involved in monogenic defects related to short stature or tall height such as IGFI, STAT5B, IGFALS, and IGFIR are detected [33-35]. but the novel genes involved in or responsible for growth regulations are not detected or found out yet. Biochemical analysis of primary IGF- 1 deficiency showed that it is usually linked with mutations in growth hormone-IGF pathway, growth hormone receptor are also included in it as well as STAT5B, IGF1, and IGFALS and also seen in the RASopathies, also faults connected to nuclear factor-κ B pathway and several disorders (e.g., 3-M) [36,37]. For SS treatment has been developed (explain later in review) and detailed analysis has a great probability to help in making treatment decisions. i.e. defect in IGFALS individuals having reasonable growth deficiency, this conclusion leads to a decision that treatment did not cure all patients, because many patients have achieved naturally normal adult height [39-41]. When there is no treatment found several factors affect the poorer height for example short stature of parents and adult age that is based on stature or bone age prediction is lower.
The Molecular Mechanism Causing Short Stature
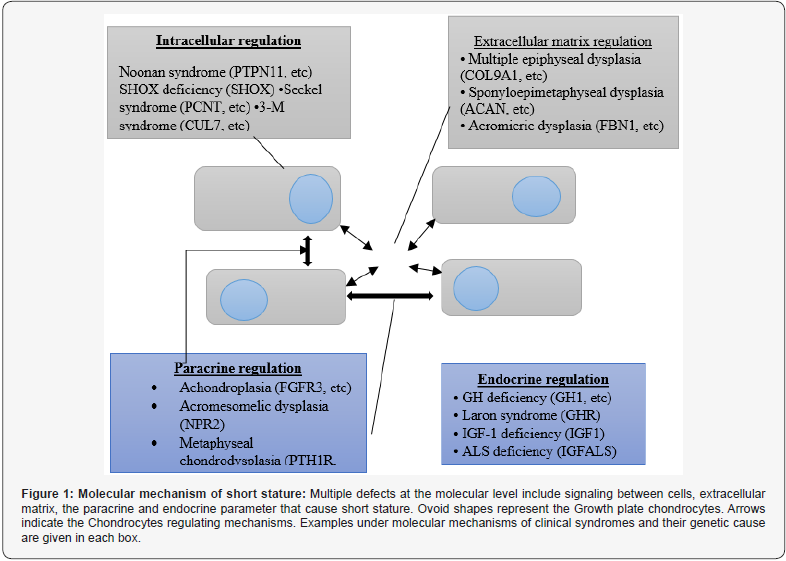
This following figure shows the molecular mechanism that causes short stature. This molecular mechanism includes different/ multiple molecular defects. These defects include signaling between cells, or intracellular signalling mechanism, extracellular matrix, paracrine and endocrine regulation. Different examples are shown by every molecular defect includes some clinical syndrome and theirsome genetic causes that comes underneath the molecular mechanisms. Growth plate chondrocytes are involved in this molecular mechanism which is represented by ovoid shapes. Every molecular defect and molecular mechanism directly or indirectly involved these growth plate chondrocytes through mechanisms regulating chondrocytes which are indicated by arrows [9,11] The intracellular signaling mechanism includes Noonan syndrome caused by gene PTPN11, etc, [ 12,42,43] SHOX deficiency caused by SHOX gene [27] Sickle syndrome caused by PCNT, etc [44,45] and 3-M syndrome by CUL7, etc [46]. Similarly, the extracellular matrix regulation includes multiple epiphyseal dysplasias (COL9A1, etc) [47,48], Sponyloepimetaphyseal dysplasia (ACAN, etc) [49,51] and Acromicric dysplasia [52]. The paracrine regulation includes Achondroplasia (FGFR3, etc Acromesomelic dysplasia (NPR2, etc) [53]. The endocrine regulation syndromes include GH deficiency (GH1, etc) [54,55] Laron syndrome (GHR) [56] IGF-1 deficiency by (IGF1) [54] ALS deficiency by (IGFALS) [55].
SHOX Genes
Short arms of chromosomes (X & Y) which are identical at the distal ends also known as pseudo autosomal regions contain genes which play their role in height. PHOG (pseudo autosomal homeobox- containing osteogenic gene) is a region which is found in this area exhibits an important function in bone development and growth, and also known as SHOX genes [57,58].Studies suggested that in embryo SHOX genes regulates the body structure development predominantly and also changes in development & limb maturation [59-61]. Regulatory regions (heterozygous mutations) of SHOX gene have been identified in 17 % of patients with ISS & 50-90% with LWS where a complex of ligation-dependent probe amplification analysis & sequencing were done in some cases [27]. Leri-Weill Dyschondrosteosis is because of SHOX gene Haploinssufiency is caused which is another type of mesomelic dysplasia which has SS and associated with Madelung deformity in the forearms & short forelegs [62].Variation in patients of Turner syndrome (which is due to X chromosomes aberrations) reveals that SHOX haploinsufficiency may be responsible for SS in the first part and see that syndrome and anomalies of the connected skeletal limb [57,63,64]Dyschondrosteosis is related to Turner syndrome, so patients of Turner syndrome may have dyschondrosteosis and SS. Molecular basis for the SHOX is involved in the whole or partial gene duplications and deletions of up & downstream enhancer sequence variations [12-14] [57,58] all these are involved in SS and skeletal abnormities.
In SHOX region genes encode for transcription factor for proteins which are related to cartilage development and [28,64] and any changes in them ultimately effect on height. Two alternative transcripts (isoforms) called SHOX(a) & SHOX (b) are encoded by seven exons and its length is about 40 kb, these transcripts are same at the last exon at 5end and different at 3end [63] they are translated into 2 proteins which are isoforms of 225 & 292 amino acids. Additionally, 2 alternative promoters in SHOX is transcribed were used which generate different mRNAs that code for proteins but with variant 5 sequences UTR55, both transcripts are translated efficiently, anticipated that the amount of SHOX protein can be changed and control translational and transcriptional with the help of control mechanisms. Patients with LWD have been identified to have 41 mutations out of [60] , in ISS it is 10, in Langer syndrome 5 whereas 3 are not interconnected with phenotype [63] Across full coding region mutations of SHOX were spread, mutations (21) were found in exons 44.7%, 9 in 2 exons, eight in 4, and 5 exons in 6, and 3 exons in 5, and 1 intron 2, there were fortyfour (44) substitutions in between them, 6 insertions and 9 deletions, 21 mutations which are linked with the homeodomain 1, 7 mutations that are in homeodomain two and 1 mutation in SHOX3 domain [17,27,65-69].
Protein formation
The SHOX protein contains a homeodomain, a structure commonly seen in transcription factors convoluted in body modeling [70] These proteins are rare and focus on SHOX comprises 292 amino acids [71,72]. SHOX alter the presence of extracellular matrix genes with Ctgf in the developing limbs which cause the abnormal limbs. In SHOX protein domain is appropriate for nuclear translocation and for dimerization of SHOX [72] which is also translates for the C-terminal-located OAR domain that are applicable for the transactivation activity of SHOX. The SHOX mRNA is encoded by 7 exons (1-5, 6a, and 6b) surrounding about 40 kb of genomic DNA. Alternative splicing results in 2 products: SHOX (a) and its shortened version SHOX (b) which give information for the OAR domain and is consequently not active as a transcriptional activator [56,57] Specific proteases like hormones/ protein proteinase pregnancyassociated plasma protein (PAPP)-A and PAPP-A2, selectively proteolyze IGFBP-4 or IGFBP-3 and IGFBP-5, respectively release free IGF-1 to activate receptor on target tissues (“endocrine, autocrine or paracrine actions of IGF-1) are products of SHOX protein and changes in anyone of them effect on height [7,72]. PAPP-A2 is stated excessively in human placenta development, the non-pregnant mammary gland and other tissues, including the kidney, fetal brain, and pancreas [33,73] PAPPA-2 (PAPPA2) is associated with mild short stature, apparently by inadequate disposal of free IGF-I [74] Other protein 65K is part of a molecular bridge that connects U11 and U12 snRNPs with di-snRNP which are connected with human height [74]. In previous studies have operated in vitro and in vivo analysis using chick micro mass culture to recognize putative target genes of SHOX [46,71,74]. These studies recommended that SHOX applies positive and negative effects on the expression of BNP and Fgfr3, respectively [17]. In addition, SHOX interrelate with SOX5, SOX6 and SOX9 they act as main chondrogenic factor [59].
Hormones
As Height is control by many factors in which few hormones and other factors are also involved, like GH, IGF and some other supporting hormones play a significant role in height gain. It is very vital for humans that their GH and IGF-1 and II work optimally to obtain maximum height, GH produce IGF which work on growth plats and result of which is in the form of height [75]In mammalian height hormones and IGF play central role, responsible for 70% of height do this reason rhGH and other factors are used for therapy to achieve maximum height [76]. Patients with ISS might have reduced sensation towards GH or not having enough concentration of GH or inheritance problems in GH or their receptor which in later stages results in the form of ISS (Hospital et al., 2005). Defects in JAK/STAB system which is one of downstream signaling pathway cause GH insensitivity and immunodeficiency and have lighter effect on growth which is exerted by heterozygous genetic variants at STAT5B, which support the hypothesis which says that rare pathogenic disparities expand to standard height heritability [77].GH deficiency is very important thought life that’s why it is very important to understand their release as well. GHs are release in an episodic manner thought day and night, pulses are more periodic and more secretion of hormones at night time as compare to day time, generally GH level is low during day time so liver starts releasing IGF-I and IGF-III which are more stable throughout the day and better checkpoints for hormones deficiency tests, but IGF-I and III are depended on age, physical development, prepubertal and pubertal stages of patients [1]. GH measurement can take place at many points with the help of GH secretagogue, like clonidine, arginine, L-Dopa, propranolol, glucagon, or insulin, a good method to measure GH in the bloodstream is radioimmunoassay and further clarification of GH signaling and mechanism of IGF and receptors of them [37].
With the help of the availability of rhGH, which clear the roles of growth hormone and IGF as main controller of skeletal growth, and also showed that both hormones releaser and acceptor exist along a continuous pathway and mutation in either one them (GHs) and their action (receptor) may cause ISS [20,21,78] GHs are not only responsible for SS but in cases such as Laron syndrome like appearance they are responsible for immunodeficiency as a defect in receptor occur in case of some SS cases [79] Some other hormones are also responsible for SS and gain in height for example Thyroid stimulating hormone (TSH), Prolactin, Luteinizing hormone(LH), Follicle stimulating hormone (FSH), Adrenocorticotropic hormone (ACTH) have their own effect on height [79] . Patients which are resistant towards TSH have normal stature but in many cases, SS is observed because of mutations caused in THRB [80]. Mutations in GH1, GHRHR, SOX3, and BTK leads to GH deficiency and mutations in GHR and STAT5B cause resistance to GH [3,81]. Homozygosity is also responsible for growth failure and also mutation in GH1 and GHRHR leads to development of anti GH antibodies due to jumping of EXON3, which results to form isomer of GH (17.5kDa) with a strong negative effects on skeletal development and mutation in BTK and SOX show same results on skeletal [4,11,82-84]. Defects in GHR or STAT5b, are clear right after birth or in childhood those defects are in IGF1or IGF1R, results in growth failure, as IGF-I is involved in the development of CNS utero, defects often described by microcephaly, delay developmental problems with hearing [23,24].
Treatment
GH therapy is resourceful and safe, suitable treatment for those who are suffering from short stature and it shows good effects in both short term and long term duration [49].In 1985 the US Food and drug administration (FDA) and EMEA allow the rhGH treatment for patients with GH deficiency and SS constructed on facts which were collected after randomized controlled experiments [110-120] and the mean aim and target of rhGH is to increase the height of those who have short height as compare to standard or other living community members . In countries which are approved by FDA, treatment against SS has been started and rhDH are being used to treat individuals with IGF,I,II and GH deficiency [50] Patients when treated with rhDH indicate decant results but still results are uneven and not clear, yet and shows defects when apply on others patients (patients to patients variations) [99] The treatment of GH depends upon the age of the patient, the dosage with which the patient is being treated who much the GH deficiency, IGFs deficiency and other parts like receptors working [100]Food and drug administration in 2003 permitted GH therapy dosage (0.3-0.37mg/kg/week) for individuals with SS for those countries in which treatment can be done in control conditions [1,121-130]. Mutation can block the receptor of GH and by finding mutation rhGH can be used to overcome that mutation in shape of blockage of receptors [144]. As in case of ACAN gene a trail treatment was used to see the effect of it [85] and results were pretty good as height was increased. In case of ISS the rhGH are used to treat up to 50% kids who have normal or even more GH secretion but having low IGF, in those cases higher rhGH are required for treatment or rhIGF-1 can also be used to treat the patients [86] With rhGH gestational age SS are treated and also silver Russell syndrome in children [120] [113] Treatment is more operative when started at earlier stages/young age and concluded that to have best results and increase in height the treatment of GH should be started in earlier stages with heavy doses [60] GH therapy is more effective, and treatment is more useful when there is deletion in enhancer (anything related to growth) as compare to those who have genetic abnormalities in them [131-140].
When patients were treated with rhGH antibodies are produced during treatment, [7] and also negative effects in the form of hyperglycemia, overgrowth of organs like kidney, spleen and anabolic effects by the high concentration of IGF [80,81] in these kind of cases rhIGF1 could be used, Defects in PAPP-A2 deficiency was treated with rhGH no negative effects were observed [82].Remember that rhGH can’t be apply all the time as in case of IGFALS defect the rhGH may not work and also in those in patients who obtain their height in later stages like (gestational age, blooms syndrome, Fanconi`s), and also remember that the use of rhGH treatment is conditional as risk of malignancy are involved [83]. Although rhGH show good results and increase in height was obtained but still there are many cases which show no results in height increase in GH therapy [34]. It is not easy to treat patients with GH, because GH treatment need a prepare monitoring of hormones, growth velocity and most important of all the dose of GH [141-151].
Conclusion
Height a heritable character that is transferred to offspring from parents is controlled by several genes. Any problems related to these genes leads to height problem i.e. tall height and short staure. One of the major genes that is cause for short stature is SHOX gene. It is concluded that it is very important that human’s hormone i.e. GH and IGF-1 and 2 work optimally to obtain maximum height. For short stature and height gain not only GHs are responsible but some other hormones are also responsible include thyroid stimulating hormone, prolactin, luteinizing hormone, follicle stimulating hormone, and adrenocorticotropic hormone.
Acknowledgments
No acknowledgments.
Conflict of Interest
The authors report no conflicts of interest.
References
- Feldman PM, Lee MM (2018) Normal Variant and Idiopathic Short Stature: pp. 61-79.
- Hawkes CP, Grasberg A (2015) Insulin-Like Growth Factor-I is a Marker for the Nutritional State. Pediatr Endocrinol Rev 13(2): 499-511.
- Silventoinen Karri, Sampo Sammalisto, Markus Perola, Dorret I, Boomsma, et all. (2003) Heritability of Adult Body Height: A Comparative Study of Twin Cohorts in Eight Countries 6(5): 399-408.
- Height H, Visscher PM, Yang J, Goddard ME (2010) A Commentary on Common SNPs Explain a Large Proportion of the Heritability for Twin Res Hum Genet 13(6): 517-524.
- Lango Allen H, Estrada K, Lettre G, Berndt SI, Weedon MN, et al. (2010) Hundreds of variants clustered in genomic loci and biological pathway 467(7317): 832-838.
- Hana Lango Allen, Karol, Estrada, Joel N, Hirschhorn (2010) Hundreds of variants clustered in genomic loci and biological pathways affect human height. Nature 467(7317): 832-838.
- Zhang Y, Zhang M, Chu Y, Shao Q, Ban B, et al. (2018) Association between Growth Hormone-Insulin-Like Growth Factor-1 Axis Gene Polymorphisms and Short Stature in Chinese Children. BioMed Research International 2018: 1-6.
- Rabbani Muhammad W, Khan Waqas I, Afzal Ahmad Bilal R (2013) Causes of short stature identified in children presenting at a tertiary care hospital in Multan Pakistan 29(1): 53-57.
- Manuel H, Aguiar Oliveira, Anita HO, Souza, Carla RP, et all. (2017) The multiple facets of GHRH/GH/IGF-I axis: Lessons from lifetime, untreated, isolated GH deficiency due to a GHRH receptor gene mutation 177(2): 1-42.
- Argente J (2017) One level up: abnormal proteolytic regulation of IGF activity plays a role in human pathophysiology (9): 1-8: 1338-1345.
- Argente J, Pérez jurado LA (2018) Genetic causes of proportionate short stature. Best Practice & Research Clinical Endocrinology & Metabolism 32(4): 499-522.
- Jee Y H, Andrade A C, Baron J, Kennedy E (2018) HHS Public Access 1-25.
- Ramachandrappa S, Kulkarni A, Gandhi H, Ellis C, Hutt R, et al. (2017) SHOX haploinsuf fi ciency presenting with isolated short long bones in the second and third trimester. European Journal of Human Genetics 26: 350-358.
- Jee Y H, Baron J (2016) The Biology of Stature. The Journal of Pediatrics 173: 32-38.
- Benito sanz S, Benito sanz S, Royo J L, Barroso E, Paumard Herna B, et al. (2015) Identification of the first recurrent PAR1 deletion in ri-Weill dyschondrosteosis and idiopathic short Le stature reveals the presence of a novel SHOX enhancer 49(7).
- Bunyan DJ, Baffico M, Capone L, Vannelli S, Iughetti L, et al. (2015) Duplications Upstream and Downstream of SHOX Identified as Novel Causes of Leri-Weill Dyschondrosteosis or Idiopathic Short Stature.
- Bunyan DJ, Baker KR, Harvey JF, Thomas NS (2013) Diagnostic Screening Identifies a Wide Range of Mutations Involving the SHOX Gene, Including a Common 47. 5 kb Deletion 160 kb Downstream ith a Variable Phenotypic Effect.
- Rica etxebarria I, Benito-sanz S, Aza carmona M, Rodrı A, et al. (2012) Identification of the first PAR1 deletion encompassing upstream SHOX enhancers in a family with idiopathic short stature 125-127.
- Zinn AR, Wei F, Zhang L, Elder FF, Scott CI, et al. (2002) Complete SHOX Deficiency Causes Langer Mesomelic Dysplasia. American journal of medical genetic banner 163 158-163.
- Seaver L H, Irons M, Committee G (2009) ACMG P RACTICE G UIDELINES ACMG practice guideline: Genetic evaluation of short stature 11(6): 465-470.
- With S, The E O F, Report C (1996) Retardation and postnatal growth failure associated with: pp. 1363-1367.
- Buckway C K, Tsubaki J, Pratt K L, Bezrodnik L, Jasper H, et al. (2003) Growth Hormone Insensitivity Associated with a STAT5b, 349(12): 1139-1147.
- Trial M, Blum WF, Crowe BJ, Quigley CA, Jung H, et al. (2015) Growth Hormone Is Effective in Treatment of Short Stature Associated with Short Stature Homeobox- Containing Gene Deficiency: Two-Year Results of a Randomized, Controlled, Multicenter Trial. The journal of clinical endocrinology and metabolism 92(1): 219-228.
- David A, Hwa V, Metherell L A, Camacho Hu C, Clark A JL, et al. (2011) Evidence for a Continuum of Genetic, Phenotypic, and Biochemical Abnormalities in Children with Growth Hormone Insensitivity 32(4): 472-497.
- Kim CA, Arnhold IJP, Mendonca BB (2007) SHOX mutations in idiopathic short stature and Leri-Weill dyschondrosteosis: frequency and phenotypic variability: pp. 130-135.
- Mendonca BB, Arnhold JP, Jorge AAL (2013) HOR MON E RE SE ARCH I N the Sitting Height / Height Ratio for Age in Healthy and Short Individuals and Its Potential Role in Selecting Short Children for SHOX Analysis. Horm Res Paediatr 80: 449-456.
- Rappold GA, Fukami M, Matsuo N, Hasegawa T, Sato S, et al. (2003) Longitudinal auxological study in a female with SHOX (short stature homeobox containing gene) haploinsufficiency and normal ovarian function. Eur J Endocrinol 149(4): 337-341.
- Niesler B, Schiller S, Zumkeller W, Bettendorf M, Genetics H (2015) Deletions of the Homeobox Gene SHOX (Short Stature Homeobox) Are an Important Cause of Growth Failure in Children with Short Stature 87: 1402-1406.
- Akiyama H (2011) Unraveling the transcriptional regulatory machinery in chondrogenesis Journal of Bone and Mineral Metabolism 29(4): 390-395.
- Pasteris NG, Cadle A, Logie LJ, Potteous MEM, Schwartz CE, et al. (1994) Isolation and Characterization of the Faciogenital Dysplasia (Aarskog-Scott Syndrome) Gene: A Putative Rho / Rat Guanine Nucleotide Exchange Factor 79: 669-678.
- Coman D, Irving M, Kannu P, Jaeken J, The SR (2008) The skeletal manifestations of the congenital disorders of glycosylation 73(6): 507-515.
- Morava E, Zeevaert R, Korsch E, Huijben K, Wopereis S, et al. (2007) ARTICLE A common mutation in the COG7 gene with a consistent phenotype including microcephaly, adducted thumbs, growth retardation, VSD and episodes of hyperthermia: pp. 638-645.
- Binder G, Eggermann TDP, Enders H, Ranke MB (2001) Tall stature, gonadal dysgenesis, and stigmata of Turners syndrome caused by a structurally altered X chromosome. The journal of pediatrics 138(2): 285-287.
- Binder G (2011) HORMONE RESEARCH IN Short Stature due to SHOX Deficiency: Genotype, Phenotype, and Therapy 75(2): 81-89.
- Kant S G, Kriek M, Walenkamp M JE, Hansson K BM (2007) Tall stature and duplication of the insulin-like growth factor I receptor gene 50(1): 1-10.
- Binder G, Renz A, Martinez A, Keselman ANA, Hesse V, et al. (2015). Dyschondrosteosis: Prevalence and Growth Failure in Relation to Mutation, Sex, and Degree of Wrist Deformity 89(9): 4403-4408.
- Allen HL, Estrada K, Lettre G, Berndt S I, Michael N, et al. (2011) NIH Public Access, 467(7317): 832-838.
- Fukami M, Matsuo N, Hasegawa T, Sato S, Ogata T (2003) Longitudinal auxological study in a female with SHOX (short stature homeobox containing gene) haploinsufficiency and normal ovarian function Eur J Endocrinol 149(4): 337-341.
- Cirulli E T, Goldstein D B (2010) Uncovering the roles of rare variants in common disease through whole-genome sequencing. Nature Publishing Group 11(6): 415-425.
- Yang J, Benyamin B, Mcevoy BP, Gordon S, Henders AK, et al. (2011) NIH Public Access 42(7): 565-569.
- Duyvenvoorde HA Van, Kempers M JE, Twickler TB, Doorn J Van, Gerver W J, et al. (2008) Homozygous and heterozygous expression of a novel mutation of the acid-labile subunit European Journal of Endocrinology 159(2): 113-120.
- Duyvenvoorde HA Van, Setten P A Van, Walenkamp M JE, Doorn J Van, Koenig J, et al. (2010) Short Stature Associated with a Novel Heterozygous Mutation in the Insulin-Like Growth Factor 1 Gene. The journal of clinical endocrinology and metabolism 95(11): 363-367.
- Woods K A, Health O, Savage M O, Mary Q (1996) Intrauterine Growth Retardation and Postnatal Growth Failure Associated with Deletion of the Insulin-Like Growth Factor I Gene 335: 1363-1367.
- Hanson D, Murray P G, Coulson T, Sud A, Omokanye A, et al. (2012). Mutations in CUL7, OBSL1 and CCDC8 in 3-M syndrome lead to disordered growth factor signaling. Journal of Molecular Endocrinology 49(3): 267-275.
- Hanson D, Murray P G, Sullivan J O, Urquhart J, Daly S, et al. (2011) Exome Sequencing Identifies CCDC8 Mutations in 3-M Syndrome, suggesting that CCDC8 Contributes in a Pathway with CUL7 and OBSL1 to Control Human Growth. The American Journal of Human Genetics 89(1): 148-153.
- Als A, Domené HM, Biology M (2011) Best Practice & Research Clinical Endocrinology & Metabolism 25(1): 101-113.
- Castro AS, Miachon AAS, Tartuci JS, Azrak R, Rey R A, et al (1969) No Title, 0-2.
- Domené HM, Argente J, Wit M (2009) Human Acid-Labile Subunit Deficiency: Clinica, Endocrine 72: 129-141.
- Grimberg A, Divall A, Allen DB (2016) Hormone Research In Guidelines for Growth Hormone and Insulin-Like Growth Factor-I Treatment in Children and Adolescents: Growth Hormone Deficiency, Idiopathic Short Stature, and Primary Insulin-Like Growth Factor-I Deficiency 86:361-397.
- Aoki Y, Niihori T, Banjo T, Okamoto N, Mizuno S, et al. (2013) Gain-of-Function Mutations in RIT1 Cause Noonan Syndrome, a RAS / MAPK Pathway Syndrome. The American Journal of Human Genetics 93(1): 173-180.
- Noonan JA, Child AJD, Genet A JM (2006) Noonan syndrome and related disorders: Alterations in growth and puberty Endocrine and Metabolic Disorders 7(4): 251-255.
- Borck G, Baumann C, Baujat G, Bieth E, Edery P, et al. (2010) Molecular analysis of pericentrin gene (PCNT) in a series of 24 Seckel / microcephalic osteodys plastic primordial dwarfism type II (MOPD II) families, (Mopd Ii) 47(12).
- Khetarpal P, Das S, Panigrahi I, Munshi A (2016) Primordial dwarfism: overview of clinical and genetic aspects. Molecular Genetics and Genomics 291(1): 1-15.
- Chernausek SD, Backeljauw PF, Frane J, Kuntze J, Underwood LE (2014) Long-Term Treatment with Recombinant Insulin-Like Growth Factor (IGF) -I in Children with Severe IGF-I Deficiency due to Growth Hormone Insensitivity Endocrinology & Metabolism 92(3): 902-910.
- Randomized A, Lucia ANA (2015) on Safety and Efficacy in Children.
- Argente Luis J ,Juradoef P (2018) Letter to the Editor: History and clinical implications of PAPP-A2 in human growth: When reflecting on idiopathic short stature leads to a specific and new diagnosis: Understanding the concept of “low IGF-I availability” Growth Hormone & IGF Research 40: 17-19.
- Jackson G C, Mittaz-crettol L, Taylor J A, Mortier G R, Spranger J, et al. (2011) Pseudo achondroplasia and Multiple Epiphyseal Dysplasia : A 7-Year Comprehensive Analysis of the Known Disease Genes Identify Novel and Recurrent Mutations and Provides an Accurate Assessment of Their Relative 33(1):p 144-157.
- Gleghorn L, Ramesar R, Beighton P, Wallis G (2005) A Mutation in the Variable Repeat Region of the Aggrecan Gene (AGC1) Causes a Form of Spondyloepiphyseal Dysplasia Associated with Severe, Premature Osteoarthritis, pp. 484-490.
- Nilsson O, Guo MH, Dunbar N, Popovic, J, Flynn D, et al. (2014) Short Stature, Accelerated Bone Maturation, and Early Growth Cessation Due to Heterozygous Aggrecan Mutations 99(8): 1510-1518.
- Wang L W, Allali S, Abhyankar A, Goff C Le, Jensen S, et al (2011). Mutations in the TGF b Binding-Protein-Like Domain 5 of FBN1 Are Responsible for Acromicric and Geleophysic Dysplasias 89(1): 7-14.
- Duyvenvoorde HA V, Kempers M J E, Twickler TB, Doorn J Van, Gerver W J, et al. (2008) Homozygous and heterozygous expression of a novel mutation of the acid-labile subunit 159(2): 113-120.
- Olney R C, Bartels CF, Prickett T CR, Espiner E A, Potter L R, et al. (2006) Heterozygous Mutations in Natriuretic Peptide Receptor-B (NPR2) Are Associated with Short Stature, Endocrinology & Metabolism 91(4): 1229-1232.
- Czarny-ratajczak M, Lohiniva J, Rogala P, Kozlowski K, Carter L, et al. (2001) A Mutation in COL9A1 Causes Multiple Epiphyseal Dysplasia: Further Evidence for Locus Heterogeneity, Am J Hum Genet 69(5): 969-980.
- Rao E, Blaschke R J, Marchini A, Niesler B, Burnett M, et al. (2001) The Leri- Weill and Turner syndrome homeobox gene SHOX encodes a cell-type specific transcriptional activator, Hum Mol Genet 10(26): 3083-3091.
- Ellison J W, Wardak Z, Young M F, Robey PG, Laig-webster M, et al. (1997) PHOG, a candidate gene for involvement in the short stature of Turner syndrome, Hum Mol Genet 6(8): 1341-1347.
- Huss-garcia Y, Duboule D, Cobb J (2006) A mouse model for human short-stature syndromes identifies Shox2 as an upstream regulator of Runx2 during long-bone development Proc Natl Acad Sci U S A 103(12): 4511-4515.
- Shears DJ, Guillen-navarro E, Sempere-miralles M, Domingo-jimenez, R, Scambler P J et al. (2002) Pseudodominant Inheritance of Langer Mesomelic Dysplasia Caused by a SHOX Homeobox Missense Mutation, Medical Genetics banner 110(2): 153-157.
- Kosho T, Muroya K, Nagai T, Fujimoto M, Ogata T (2015) Skeletal Features and Growth Patterns in 14 Patients with Haploinsufficiency of SHOX: Implications for the Development of Turner Syndrome 84(12): 4613-4621.
- Moises H W (1995) Nature Publishing Group.
- Grigorescu F, Lautier C, Keller E, Osgood D Chernausek S D (2014) IGF-I Receptor Mutations Resulting in Intrauterine and Postnatal Growth Retardation: 2211-2222.
- Eklöf O, Anders S, Otto I, Lo W (2000) Mutations in short stature homeobox containing gene (SHOX) in dyschondrosteosis but not in hypochondroplasia, pp. 145-149.
- Grigelioniene G, Schoumans J, Neumeyer L, Anders S, Ole I, et al (2001) Analysis of short stature homeobox-containing gene (SHOX) and auxological phenotype in dyschondrosteosis and isolated Madelung deformity Human Genetics 109(5): 551-558.
- Sanctis LDe, Pucarelli I, Radetti G, Wasniewska M, Weber G, et al. (2002) point mutations and deletions in Leri-Weill dyschondrosteosis, (table 1), pp. 1-5.
- Angeles L (1992) Homeobox Genes and Pattern Formation in the Vertebrate Limb (36): 26-36.
- Tiecke E, Bangs F, Blaschke R, Farrell E R, Rappold G, et al. (2006) Expression of the short stature homeobox gene Shox is restricted by proximal and distal signals in chick limb buds and affects the length of skeletal elements Developmental Biology 298(2): 585-596.
- Weiss B, Jantz K, Tiecke E, Sabherwal N, Bangs F, et al. (2007) Long-range conserved non-coding SHOX sequences regulate expression in developing chicken limb and are associated with short stature phenotypes in human patients 16(2): 210-222.
- Igfbp N (2001) JBC Papers in Press. Published on March 22, 2001 as Manuscript M102191200, 2.
- Jan M W, Wilma O, Monique L, Hermine A van D, Claudia A L, et al. (2016) Novel genetic causes of short stature. European Journal of Endocrinology 174(4): R145-R173.
- Liu J, Baker J, Perkins AS, Roberteon EJ (1993) Mice Carrying Null Mutations of the Genes Encoding Insulin-like Growth Factor I (Igf. 1) and Type 1 IGF Receptor (Igflr) 75(1): 59-72.
- Flores R, Guti A, Cusc I, Frilander M J, Luis, et al. (2014) Defective minor spliceosome mRNA processing results in isolated familial growth hormone deficiency 6(3): 299-306.
- Glaser A, Decker E, Durand C, Bender S, Ro C,et al. (2011) FGFR3 is a target of the homeobox transcription factor SHOX in limb development, Hum Mol Genet 20(8): 1524-1535.
- Burkitt EMM, Perveen R, Clayton P E, Hall C M, Costa T, et al. (2009) X-linked isolated growth hormone deficiency: expanding the phenotypic spectrum of SOX3 polyalanine tract expansions Clin Dysmorphol 18(4): 218-221.
- Fritez N, Sobrier M, Iraqi H, Netchine, I (2014) Molecular screening of a large cohort of Moroccan patients with congenital hypopituitarism Article Type: 2 Original Article - Europe, excluding UK 82(6): 876-884.
- Gkourogianni A, Andrew M, Tyzinski L, Crocker M, Dunbar N, et al. (2016) Clinical characterization of patients with autosomal dominant short stature due to aggrecan mutations, endocrinology. 102(2): 460-469.
- Andrew G, Clayton P, Bonnemaire M, Dutailly P, Maisonobe P,et (2013) Characterizing Short Stature by Insulin-like Growth Factor Axis Status and Genetic Associations: Results, endocrinology 98(6): 1122-1130.
- Suh JKBK, Lee CWKKH, Hwang CHSJS (2017) Recombinant growth hormone therapy for prepubertal children with idiopathic short stature in Korea: a phase III randomized trial, endocrinology. 41(4): 475-483.
- Donze SH, Meijer CR, Kant SG, Zandwijken GR, van der H, et al. (2015) The growth response to GH treatment is greater in patients with SHOX enhancer deletions compared to SHOX defects 173(5): 611-621.
- Steen M Van Der, Smeets CCJ, Kerkhof GF, Hokken-koelega ACS (2017) Metabolic health of young adults who were born small for gestational age and treated with growth hormone, after cessation of growth hormone treatment: a 5-year. THE LANCET Diabetes & Endocrinology 5(2): 106-116.
- Steen M Van Der, Hokken-koelega, ACS (2016) Growth and Metabolism in Children Born Small for Gestational Age Growth Small for gestational age Growth hormone treatment Metabolic health 45(2): 283-294.
- Scott K, Gadomski T, Kozicz T (2014) Congenital disorders of glycosylation: new defects and still counting.
- Sga S (n.d.) etiological aspects.
- Savage MO, Burren CP, Rosenfeld RG (2010) The continuum of growth hormone - IGF-I axis defects causing short stature: diagnostic and therapeutic challenges 72(6): 721-728.
- Ross JL, Scott C, Marttila PIA, Kowal K, Nass A, et al. (2015) Phenotypes Associated with SHOX Deficiency 86: 5674-5680.
- Rosilio M, Carel J, Blum WF (2012) Genotypes and Phenotypes of Children with SHOX Deficiency in France 97(2): 1257-1265.
- Roberts AE, Allanson JE, Tartaglia M, Gelb BD, Ematologia D (2013) Noonan syndrome. The Lancet 381(9863): 333-342.
- Renes JS, Steen M Van Der, Society E (2016) Metabolic Health and Long-term Safety of Growth Hormone Treatment in Silver-Russell Syndrome, 102(3): 983-991.
- Wit JM (2011) HOR MON E RE SE ARCH I N Mechanisms of Growth Plate Maturation and Epiphyseal Fusion 75: 383-391.
- Wit JM, Luca F De (2016) Growth Hormone & IGF Research Atypical defects resulting in growth hormone insensitivity. YGHIR 28: 57-61.
- Rappold GA, Batch JA, Hyland VJ, Glass I A (2002) Prevalence of mutations in the short stature homeobox containing gene.
- Oliveira CS, & Alves C (2011) The role of the SHOX gene in the pathophysiology of Turner. Medicina Intensiva (English Edition) 58(8): 433-442.
- Norppa AJ, Kauppala TM, Heikkinen HA, Verma B, Iwaï H, et al. (2018) Mutations in the U11 / U12 ‐ 65K protein associated with isolated growth hormone deficiency lead to structural destabilization and impaired binding of U12 snRNA 24(3): 396-409.
- Mutation GG, Secretion GA, Petkovic V, Godi M, Pandey AV, et al. (2015) Growth Hormone (GH) Deficiency Type II: A Novel and Action 95(2): 731-739.
- Högler W, Martin DD, Crabtree N, Nightingale P, Tomlinson J, et al. (2014) IGFALS Gene Dosage Effects on Serum IGF-I and Glucose Metabolism, Body Composition, Bone Growth 99(4): 703-712.
- Dysplasia E, Chapman KL, Mortier GR, Chapman K, Loughlin J, et al. (2001) Mutations in the Region Encoding the Von Willebrand Factor A Domain of Mutations in the region encoding the von Willebrand factor A domain of matrilin-3 are associated with multiple epiphyseal dysplasia (28): 393-396.
- Aza-Carmona M, Barca-Tierno V, Hisado-Oliva A, Belinchón, A, Gorbenko-del Blanco Det al (2014) NPPB and ACAN, two novel SHOX2 transcription targets implicated in skeletal development PLoS ONE, 9(1).
- Belin V, Cusin V, Viot G, Girlich D, Toutain A, et al. (1998) SHOX mutations in dyschondrosteosis (Leri-Weill syndrome). Nature genetics 19(1): 67-69.
- Belinl V, Cusin V, Viot G, Girlich D, Vekemans M et al. (1999) SHOX gene mutations and deletions in dyschondrosteosis or Leri-Weill syndrome 88 (433): 55-59.
- Bobick BE, Cobb J (2012) Shox2 regulates progression through chondrogenesis in the mouse proximal limb Accepted manuscript 125: 6071-6083.
- Kalf ME, Duyvenvoorde HA Van, Boot AM, Kant SG, White S J, et al. (2015) Successful Long-Term Growth Hormone Therapy in a Girl with Haploinsufficiency of the Insulin-Like Growth Factor-I Receptor due to a Terminal 93(6): 2421-2425.
- Lupu F, Terwilliger JD, Lee K, Segre G V, Efstratiadis A (2001) Roles of Growth Hormone and Insulin-like Growth Factor 1 in Mouse Postnatal Growth 229(1): 141-162.
- Scalco RC, Hwa V, Belgorosky A, Pereira AM, Tonelli, et al. (2015) Accepted Preprint first posted on 1 June 2015 as Manuscript EJE-15-0398, pp. 1-20.
- Chen J, Wildhardt G, Zhong Z, Ro R, Weiss B, et al. (2009) Enhancer deletions of the SHOX gene as a frequent cause of short stature : the essential role of a 250 kb downstream regulatory domain pp. 834-839.
- Cirulli ET, Goldstein DB (2010) Uncovering the roles of rare variants in common disease through whole-genome sequencing. Nature Publishing Group 11(6): 415-425.
- Conover CA, Boldt HB, Bale LK, Clifton KB, Grell JA, et al. (2015) Tissue Expression and Biological Consequences of Gene Knockout in Mice 152(7): 2837-2844.
- Cormier‐Daire V, Belin V, Cusin V, Viot G, Girlich D, et al. (1999) SHOX gene mutations and deletions in dyschondrosteosis or Leri‐Weill syndrome. Acta Paediatrica 88(433): 55-59.
- Dauber A, Rosenfeld R G, Hirschhorn JN (2014) Genetic Evaluation of Short Stature 99(9): 1-15.
- Durand C, Decker E, Roeth R, Schneider KU, Rappold G (2012) The Homeobox Transcription Factor HOXA9 Is a Regulator of SHOX in U2OS Cells and Chicken Micromass Cultures 7(9): 1-11.
- Fuqua JS, Rosenfeld RG (2012) Novel Insights from Clinical Practice HOR MON E RE SE ARCH I N Identification of a Novel Heterozygous IGF1 Splicing Mutation in a Large Kindred with Familial Short Stature 78: 59-66.
- HORMONE RESEARCH IN Heterozygous (2013) IGFALS Gene Variants in Idiopathic Short Stature and Normal Children: Impact on Height and the IGF System 80: 413-423.
- Hospital P, Fiorentino S, Cutler GB, Crowe B, Beck TJ, et al (2005) WITH IDIOPATHIC SHORT STATURE: EVIDENCE FOR A DOSE EFFECT 146 (1): 45-53.
- Huber C (2004) Génétique et gène SHOX SHOX gene and short - stature syndromes 11: 555-556.
- Dias-santagata D, Glaser A, Sullivan JO, Brauner R, Wu K, et al. (2005). Identification of mutations in CUL7 in 3-M syndrome 37(10): 1119-1124.
- Schiller S, Spranger S, Schechinger B, Fukami M, Knoblauch H (2000) Phenotypic variation and genetic heterogeneity in L ´ eri-Weill syndrome 8(1): 54-62.
- Phillips SA, Lazar J, Ando S, Hauser P, Skarulis MC (2015) Extreme Thyroid Hormone Resistance in a Patient with a Novel Truncated TR Mutant (86): 5142-5147.
- Laron Z (2015) Review Article 21(12): 1395-1402. Pozo, J, Chowen JA, Martos-moreno GÁ, Frystyk J (2016) Case Description 1-5. Treatment With Recombinant Human Insulin-Like Growth Factor-1 Improves Growth in Patients With PAPP-A2 Deficiency The Journal of Clinical Endocrinology & Metabolism 101(11): 3879-3883.
- Kamp GA, Waelkens JJ, de Muinck Keizer-Schrama SM, Delemarre-Van de Waal HA, Verhoeven-Wind L, et al (2002) High dose growth hormone treatment induces acceleration of skeletal maturation and an earlier onset of puberty in children with idiopathic short stature 87(3): 215-220.
- Alatzoglou KS, Turton JP, Kelberman D, Clayton PE, Mehta A, et al. (2015) Expanding the Spectrum of Mutations in GH1 and GHRHR: Genetic Screening in a Large Cohort of Deficiency 94(9): 3191-3199.
- Alatzoglou KS, Webb EA, Tissier PLe, Dattani MT (2014) Isolated Growth Hormone Deficiency (GHD) in Childhood and Adolescence: Recent Advances 35(3): 376-432.
- Woods KA, Camacho-Hübner C, Savage MO, Clark AJ (1996) RETARDATION AND POSTNATAL GROWTH FAILURE ASSOCIATED WITH 335(18): 1363-1367.
- Gardner M, Boshart ML, Yeguez CE, Desai KM, Sandberg DE (2016) Coming Up Short: Risks of Bias in Assessing Psychological Outcomes in Growth Hormone Therapy for Short Stature 101(1): 23-30.
- Ranke MB, Lindberg A (2009) Predicting growth in response to growth hormone treatment. Growth Hormone & IGF Research 19(1): 1-11.
- Yu L, Liu H, Yan M, Yang J, Long F (2007) Shox2 is required for chondrocyte proliferation and maturation in proximal limb skeleton Developmental Biology 306(2): 549-559.
- Wit JM, Reiter EO, Ross JL, Saenger PH, Savage MO, et al. (2014) Consensus Statement on the Diagnosis and Treatment of Children with Idiopathic Short Stature: A Summary of the Growth Hormone Research Society, the Lawson Wilkins Pediatric 93(1): 4210-4217.
- Wit JM, Reiter EO, Ross J L, Saenger PH, Savage MO, et al. (2008) Idiopathic short stature: Management and growth hormone treatment 18(2): 111-135.
- Walenkamp MJE, Kamp HJ Van Der, Pereira A M, Kant SG, Duyvenvoorde HA Van (2006) A Variable Degree of Intrauterine and Postnatal Growth Retardation in a Family with a Missense Mutation in the Insulin-Like Growth Factor I Receptor 91(8): 3062-3070.
- Walenkamp MJE, Karperien M, Pereira AM, Doorn J Van, Chen J W, et al. (2005) Homozygous and Heterozygous Expression of a Novel Insulin-Like Growth Factor-I Mutation 90(5): 2855-2864.
- Vidarsdottir S, Walenkamp MJE, Pereira AM, Karperien M, Doorn J Van, et al. (2006) Clinical and Biochemical Characteristics of a Male Patient with a Novel Homozygous STAT5b Mutation 91(9): 3482-3485.
- Varco-merth B, Feigerlová, E, Shinde U, Rosenfeld RG, Hwa V et al. (2013) Mutations that Disrupt Protein Folding and Activity, 27(1): 150-161.
- Trial P, Leschek EW, Rose SR, Yanovski JA, Troendle JF, et al. (2015) Effect of Growth Hormone Treatment on Adult Height in Peripubertal Children with Idiopathic Short Stature 89(7): 3140-3148.
- Andrew R, Wood Tonu E, Timothy M (2014) Defining the role of common variation in the genomic and biological architecture of adult human height. Nature Genetics volume 46: 1173-1186.
- Eirini M, Mariaelisa G, Guillaume L (2017) Rare and low-frequency coding variants alter human adult height. Nature volume 542: 186-190
- Huber C, Cusin V, Merrer M Le, Mathieu M, Sulmont V, et al (2001) Letters to the Editor SHOX point mutations in dyschondrosteosis Phenotypic heterogeneity of CYP1B1: mutations in a patient with Peters anomaly 323-351.
- Human growth hormone (somatropin) for the treatment of growth failure in children (2018).
- Hwa V, Nadeau K, Clinics A, Wit JM, Rosenfeld RG (2011) Best Practice & Research Clinical Endocrinology & Metabolism STAT5b de fi ciency: Lessons from STAT5b gene mutations. Best Practice & Research Clinical Endocrinology & Metabolism 25(1): 61-75.
- Kemp SF, Kuntze J, Attie KM, Maneatis T, Butler S, et al. (2005) Efficacy and Safety Results of Long-Term Growth Hormone Treatment of Idiopathic Short Stature, J Clin Endocrinol Metab 90(9): 5247-5253.
- Klammt J, Kiess W, Pfäf R (2011) Best Practice & Research Clinical Endocrinology & Metabolism IGF1R mutations as cause of SGA 25(1): 191-206.
- Lee BH, Kim J, Jin HY, Kim G, Choi J, et al. (2011) Spectrum of Mutations in Noonan Syndrome and Their Correlation with Phenotypes. The Journal of Pediatrics 159(6): 1029-1035.
- Li L, Gretz N, Beiser KU, Glaser A, Kleinschmidt K, et al. (2014) Identification of Novel SHOX Target Genes in the Developing Limb Using a Transgenic Mouse Model 9(6): 1-10.
- Marttila T, Hesse V, Emons J, Marchini A, Ha B, et al. (2007) BNP is a transcriptional target of the short stature homeobox gene SHOX, Endocrinology 16(24): 3081-3087.
- Murray PG, Clayton PE, Chernausek SD (2018) Review A genetic approach to evaluation of short stature of undetermined cause. THE LANCET Diabetes & Endocrinology 8587(18): 1-11.
- Kemp SF, Kuntze J, Attie KM, Maneatis T, Butler S, et al. (2015) Efficacy and Safety Results of Long-Term Growth Hormone Treatment of Idiopathic Short Stature, Endocrinology 90(9): 5247-5253.






























