Anticonvulsant and Analgesic Assessment of Dichrostachys Cinerea Root Bark, an Ivorian Anti-Asthmatic Herbal, in Mice
Amenan Geneviève Irié-N’guessan1*, Sylvain Landry Kouakou1, Kadio Brou Donald Koua2, Kouakou Etienne Effo1, Ayoman Thierry Lenoir Djadji1, N’golo Diarrassouba1 and N’doua Gisèle Kouakou-Siransy1
1Laboratory of Pharmacology, Training and Research Unit of Pharmaceutical and Biological Sciences, Felix Houphouet-Boigny University, Africa
2Laboratory of Biochemical Pharmacodynamics, Training and Research Unit of Biosciences, Felix Houphouet-Boigny University, Africa
Submission: August 24, 2018;Published: September 19, 2018
*Corresponding author: Amenan Geneviève Irié-N’guessan, Laboratory of Pharmacology, Training and Research Unit of Pharmaceutical and Biological Sciences, Felix Houphouet-Boigny University, Africa
How to cite this article: A G Irié-N’guessan, S L Kouakou, KBD Koua, K E Effo, A T L Djadji, et al. Anticonvulsant and Analgesic Assessment of Dichrostachys Cinerea Root Bark, an Ivorian Anti-Asthmatic Herbal, in Mice. J of Pharmacol & Clin Res. 2018; 6(3): 555687. DOI: 10.19080/JPCR.2018.06.555687
Abstract
Root bark of Dichrostachys cinerea is widely used in Ivorian folk, especially for treatment of asthma. However, the experimental exploration of its potential pharmacological properties is not exhaustive.
Objective: This study aimed to evaluate anticonvulsant and analgesic activities of this plant.
Material and Methods: An aqueous-alcoholic extract was obtained from the root bark. Both activities were evaluated in mice by two tests i.e. pentylenetetrazol and isoniazid-induced convulsions for the anticonvulsant activity on one hand and acetic acid-induced abdominal contortions and tail-immersion test for the analgesic activity on the other hand.
Results: At 10, 100, and 1000 mg/kg b. wt. the extract inhibited pentylenetetrazol or isoniazid-induced convulsions at 6 to 34% and 15 to 50% respectively, delayed the onset of convulsions of 6 to 73 seconds and protected the animals of the death by convulsion from 50 to 100%. The same doses were not found to have inhibitory activity against the pain engendered by the warm water in the temperature from 50 to 60°C, in comparison with morphine, but they inhibited the abdominal contortions by 47% for at the dose of 10 mg/kg b. wt., 50% for 100 mg/kg b. wt., and 68% for 1000 mg/kg b. wt.
Conclusion: Dichrostachys cinerea root bark induced a low anticonvulsant effect that could be beneficial as anticonvulsant additive. The extract did not either induce morphine-type analgesic effect, however, this herbal developed non-morphine-type analgesic effect that could present an interest in the therapeutic coverage of asthma.
Keywords: Plant; Pentylenetetrazol-induced convulsions; Isoniazid-induced convulsions; Abdominal contortions; Tail immersion
Introduction
According to the World Health Organization, about 80% of African people resort to medicinal herbals for their needs for health [1], making of these plants the most importing therapeutic arsenal. In Côte d’Ivoire, populations traditionally use Dichrostachys cinerea (L.) Wight & Arn. (Fabaceae) root bark ground with water and administered in nasal instillations for asthma attacks management [2]. As smooth muscle relaxation is an anti-asthmatic beneficial mechanism, we previously investigated the ex vivo relaxant effect of a crude aqueous-alcoholic extract of the root bark of D. cinerea on isolated trachea of mice with complete inhibition of precontraction at 100 mg/mL concentration [3]. Furthermore, in addition to bronchospasm characterizing asthma attacks, asthma is a chronic inflammatory airways disease [4]. Then, the anti-inflammatory potential of the same plant extract was looked for in a previous study [5], showing a low effect by a non-specific test, but no effect by an anti-inflammatory specific test. In view of these data, we therefore wondered the potential effect of D. cinerea root bark aqueous-alcoholic extract on the other type of muscle, i.e. skeletal muscle on one hand, and the real activity it exhibited through the non-specific anti-inflammatory test on the other hand, justifying the current anticonvulsant and analgesic investigations.
Material and Methods
Plant Material
Dichrostachys cinerea was collected on April 15, 2017 from bushes near Grand-Bassam (south-eastern part of Côte d’Ivoire). The plant was authenticated by a taxonomist at the Centre National de Floristique d’Abidjan, i.e. National Floristic Center (Abidjan, Ivory Coast), in comparison with the voucher of the Centre’s herbaria (Dichrotachys cinerea (L.) Wight & Arn. Adjanohoun E. and Ake Assi L., 29, forest of Banco Ivory Coast on March 20, 1972). Bark was removed from the roots, washed with distilled water, air-dried at air-conditioning temperature for two weeks, and pulverized using an electric grinder (Retsch GM300®). The powder of root bark served as sample to be tested.
Extraction Procedure
The aqueous-alcoholic extract of Dichrostachys cinerea root bark was prepared with an ethanol-distilled water mixture (100 g dry powder material in 1 L; room temperature, under magnetic stirring, 24 h). After filtration twice on white cotton and once on what Mann filter paper No 3, the extract was dried under reduced pressure at 45°C temperature using a rotary evaporator (Heidoph RZ 2.5). Extraction yield was 9.14%. The extract powder was stored in a glass container covered with parafilm and kept in at 7-8 °C temperature.
Chemicals
a) Distilled water
b) Normal saline (NaCl 0.9%)
c) Pentylenetetrazol (SIGMA-ALDRICH, Germany)
d) Clonazepam 100 mg tablet (LUTIN Laboratory, India)
e) Isoniazid 100 mg tablet (ROCHE Laboratory SA, Switzerland)
f) Acetic acid 1% (ZEUS, Germany)
g) Doliprane (acetaminophen) 200 mg powder (AVENTIS PHARMA, France)
h) Aspegic (acetylsalicylic acid) 100 mg powder (SANOFIAVENTIS, France)
i) Acupan (nefopam) 10 mg/mL (BIOCODEX, France)
j) Morphine 10 mg/mL (RENAUDIN, France)
Experiment Animals
Mice belonging to Mus musculus species of either sex, weighing 28 ± 2 g were used. Animals were fasted for 4 hours prior to experimentations with free access to water. Animals were distributed in homogeneous groups in weight.
Pharmacological Tests
For all tests, the dry aqueous-alcoholic extract of Dichrostachys cinerea root bark was extemporaneously dissolved in normal saline at 100 mg/mL and subsequent dilutions were performed in distilled water to obtain a concentration range at 10 mg/mL and 1 mg/ml. The other products (pentylenetetrazol, isoniazid, clonazepam, acetic acid solution, acetaminophen, acetylsalicylic acid, nefopam, morphine) were also diluted in normal saline prior to their administration in mice.
Evaluation of Anticonvulsant Potential
Pentylenetetrazol-Induced Convulsions Test
The experience was led according to the method described by Bum et al. [6].
Animals were divided into five groups of six mice:
a) Group 1, as control, received normal saline (NaCl 0.9% at 10 ml/kg b.wt. per os);
b) Group 2 received standard drug (clonazepam 3 mg/kg b.wt. ip);
c) Groups 3, 4, and 5 received respectively graded doses of aqueous-alcoholic extract of D. cinerea (10, 100 and 1000 mg/ kg b.wt. per os).
Thirty min after clonazepam intraperitoneally administration and one hour after other substances orally administration, all animals were administered intraperitoneally with pentylenetetrazol (60 mg/kg b.wt.) and placed in isolated cages for 15 min observation. Animals that did not convulse within the complete period of observation were qualified as protected. In unprotected animals, time to onset of the first convulsion, as well as duration of convulsions and percentage of mortality were recorded.
Isoniazid-Induced Convulsions Test
The experience was led according to the method described by Raza et al. [7].
Animals were divided into five groups of six mice.
a) Group 1, as control, received normal saline (NaCl 0.9% at 10 ml/kg b. wt. per os);
b) Group 2 received standard drug (clonazepam 3 mg/kg b. wt. ip);
c) Groups 3, 4, and 5 received respectively graded doses of aqueous-alcoholic extract of D. cinerea (10, 100 and 1000 mg/ kg b. wt. per os).
Thirty min after clonazepam intraperitoneally administration, and one hour after other substances orally administration, all animals were administered orally with isoniazid (250 mg/kg b. wt.) and placed in isolated cages for 3 hours observation. Animals that did not convulse within the complete period of observation were qualified as protected. In unprotected animals, time to onset of the first convulsion, as well as duration of convulsions and percentage of mortality were recorded.
Evaluation of Analgesic Potential
Writhing Test or Acetic Acid-Induced Abdominal Contortions Test
The experience was led according to the method described by Koster et al. [8] and modified by Collier et al. [9].
The animals were divided according to the weight in seven groups of six mice with oral pretreatment in this way:
a) Group 1: mice treated with normal saline (NaCl 0.9% at 10 ml/kg b. wt. per os)
b) Group 2: mice treated with 10 mg/kg b. wt. of extract
c) Group 3: mice treated with 100 mg/kg b. wt. of extract
d) Group 4: mice treated with 1000 mg/kg b. wt. of extract
e) Group 5: mice treated with 100 mg/kg b. wt. of acetylsalicylic acid (standard drug)
f) Group 6: mice treated with 100 mg/kg b. wt. of acetaminophen (standard drug)
g) Group 7: mice treated with 10 mg/kg b. wt. of nefopam (standard drug).
Thirty min after the oral administration of drugs, the acetic acid 1% solution was intraperitoneally injected to mice. Writhes, as pain sign, consisted in contortions then digging of sides, outstretching of the hind limbs and twisting of the dorsalabdominal muscles. The number of writhes was recorded five min after acetic acid injection for a period of twenty minutes and the percentage of pain inhibition was calculated by the following equation:

Tail-Immersion Test
The experience was led according to the method described by Janssen et al. [10].
The animals were divided according to the weight in five groups of six mice with pretreatment in this way:
a) Group 1: mice treated with normal saline (NaCl 0.9% at 10 ml/kg b. wt. per os)
b) Group 2: mice orally treated with 10 mg/kg b. wt. of extract
c) Group 3: mice orally treated with 100 mg/kg b. wt. of extract
d) Group 4: mice orally treated with 1000 mg/kg b. wt. of extract
e) Group 5: mice peritoneally treated with 10 mg/kg b. wt. of morphine (standard drug).
Thirty min, then sixty min and ninety min after the oral or intraperitoneal administration of drugs, the lower extremity of mice tail is immersed in some warm water maintained in 55 ± 5°C susceptible to provoke a neurogenic pain. The tail withdrawal time, without exceeding fifteen seconds, was considered as deadline of pain perception. Animals that did not withdraw tail within 15 seconds were qualified as protected and the percentage of pain inhibition was calculated by the following equation:
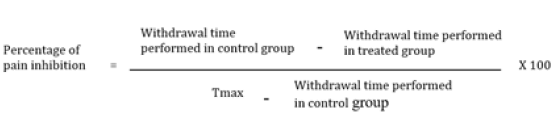
With T max = 15 seconds (time that could not be exceeded).
Statistical Method
Values were expressed as mean ± SD (standard deviation) with n = 6 rats per group. Data were analyzed with GraphPad Prism.7® software by Wilcoxon statistical test, with criterion set for statistical significance at p < 0.05 for risk of α = 0.05.
Ethical Approval
The experimental procedures were conducted after the approval of the Ethical Guidelines of the University (Ivory Coast) Committee on Animal Resources. All these procedures used, were in strict accordance with the guidelines for Care and Use of Laboratory Animals and the statements of the European Union regarding the handling of experimental animals (86/609/EEC) [11].
Results
Anticonvulsant Activity
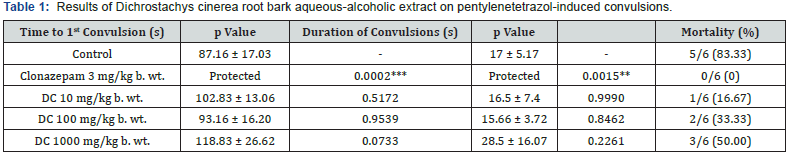
DC: Dichrostachys cinerea root bark aqueous-alcoholic extract.
**: 0.001≤p0.01 (significant difference between tested groups and control).
***: p0.001 (significant difference between tested groups and control).
Effect of D. cinerea Root Bark on Pentylenetetrazol- Induced Convulsions: Administration of the extract in the dose of 10 mg/kg b. wt. resulted in a deadline of appearance of the first convulsion postponed of 15.67 seconds, inhibition of convulsion onset at 16.87% and a low mortality at 16.67%, comparatively to the control group. In the dose of 100 mg/kg b. wt., the extract delayed the deadline of appearance of the first convulsion of only 6 seconds on average, inhibited appearance of convulsions at 6.82% and raised the mortality rate to border 33.33%. Concerning the dose of 1000 mg/kg b. wt., the deadline of appearance of the first convulsion was delayed of 31.67 seconds, onset of convulsions was inhibited at 34.11%, but the mortality rate was higher 50%. With clonazepam (standard drug) no death was observed and animals were completely protected against seizures induced by administration of pentylenetetrazol (Table 1).
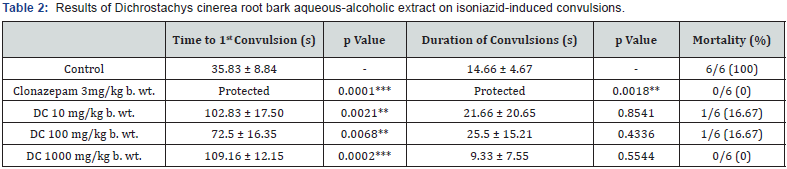
DC: Dichrostachys cinerea root bark aqueous-alcoholic extract.
**: 0.001≤p<0.01 (significant difference between tested groups and control).
***: p<0.001 (significant difference between tested groups and control).
Effect of D. cinerea Root Bark on Isoniazid-Induced Convulsions: Administration of the extract in the dose of 10 mg/kg b. wt. postponed time to onset of the first convulsion of 67 seconds, inhibited convulsions onset at 46.47% and induced a low mortality at 16.67%, comparatively to the control group. The dose of 100 mg/kg b. wt. also induced a low mortality at 16.67%, inhibited convulsions onset at 15.6% with a deadline of appearance of the first convulsion postponed of 36.67 seconds on average. In the dose of 1000 mg/kg b. wt., the deadline of appearance of the first convulsion was delayed of 73.33 seconds, convulsion onset was inhibited at 50.86% and no death was observed. Likewise, with clonazepam, no death nor convulsions were induced by the administration of isoniazid in animals (Table 2).
Anticonvulsant Activity
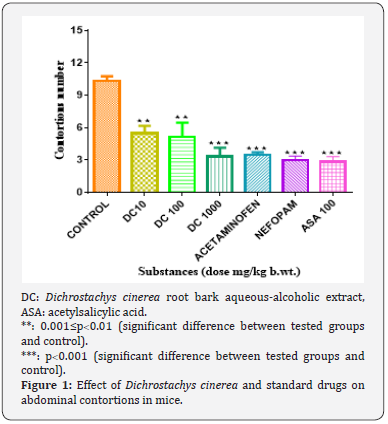
Effect of D. cinerea Root Bark on Acetic Acid-Induced Abdominal Contortions: In comparison with the control group that developed 10.33±1.03 writhes, all the tested doses of the extract reduced writhes (Figure 1). Percentages of writhes inhibition were 46.67% (p = 0.0036) in the dose of 10 mg/kg b. wt., 50% (p = 0.0048) in the dose of 100 mg/kg b. wt., and 67.74% (p = 0.0001) in the dose of 1000 mg/kg b. wt. Percentages of writhes inhibition in standard groups were 66.12% (p=0.0001) for acetaminophen, 70.96% (p = 0.0001) for nefopam, and 72.58% (p = 0.0001) for acetylsalicylic acid.
Effect of D. cinerea Root Bark on Warm Water-Induced Neurogenic Pain: Tail withdrawal times were not significantly different from those of control group (Figure 2). Percentages of neurogenic pain inhibition were 27.73% (p = 0.3725) in the dose of 10 mg/kg b. wt., 16.94% (p = 0.0554) in the dose of 100 mg/kg b. wt., and -0.46% seconds (p = 0.990) in the dose of 1000 mg/kg b. wt. in the first period observation (thirty min after administrations). Sixty min after extract administration, percentages of neurogenic pain inhibition were not augmented anyway: 2.22% (p = 0.9770) in the dose of 10 mg/kg b. wt., 7.19% (p = 0.6042) in the dose of 100 mg/kg b. wt., and -9.94% (p = 0.395) in the dose of 1000 mg/kg b. wt. Ninety min after extract administration, percentages of neurogenic pain inhibition stayed the same: 13.8% (p = 0.1776) in the dose of 10 mg/kg b. wt., 25.73% (p = 0.0852) in the dose of 100 mg/kg b. wt., and 1.79% (p = 0.9925) in the dose of 1000 mg/kg b. wt. For all periods (30, 60, and 90 min after administration). Percentages of neurogenic pain inhibition in standard group, using morphine as standard opioid analgesic drug, were very high, i.e. 100% (p = 0.0001), 71.55% (p = 0.000177), and 90.19% (p = 0.000127) respectively.

Discussion
Dichrostachys cinerea root bark is well known in Ivorian traditional medicine as anti-asthmatic medicine. However, the underlying pharmacological properties of this vegetable drug are little explored. Asthma is a chronic inflammatory airways disease associated with a widespread airflow obstruction [4]. Thus, we previously looked for and highlighted the antispasmodic properties of a polar aqueous-alcoholic extract of this part of plant on mice trachea [3], probably favorable in the management of airway obstruction that appears in asthma attacks. As antispasmodic properties concern smooth muscle, the current study questioned the probability of involvement of D. cinerea root bark in the relaxation of skeletal muscle, i.e. anticonvulsant activity. We noted that the extract did not significantly prevent appearance of convulsions in mice, neither delayed their onset, nor protected the animals of the death by convulsion induced by pentylenetetrazol. However, the extract significantly postponed first convulsion onset induced by isoniazid. But in looking at the gross data, the extract dose of 1000 mg/kg b. wt. protected 34% of the animals from pentylenetetrazol-induced convulsions, delayed the appearance of these convulsions of about 32 seconds, and in 10 mg/kg b. wt. death arisen was prevented at 83.33% in unprotected animals. It is well known that pentylenetetrazol exercises a convulsant effect by opposing the fixation of gamma aminobutyric acid (GABA) on its channel receptor GABA-A [12]. GABA is the major inhibitive neurotransmitter of the central nervous system, and the inhibition of its neurotransmission was considered as the underlying factor of epilepsy or convulsion [13]. Knowing that the anticonvulsant effect of benzodiazepines like clonazepam is mainly attributed to the allosteric potentializing of the receptor GABA-A [14], the low protection of mice from pentylenetetrazolinduced convulsions with Dichrostachys cinerea in the dose of 1000 mg/kg b. wt could have been bound to the presence of some components in the root bark aqueous-alcoholic extract that would have lowly acted on GABA neurotransmission. However, this interaction, either by direct activation of GABA-A or by strengthening the effect of GABA on GABA-A, seems to be insufficient to exert a real anticonvulsant effect. Concerning isoniazid-induced convulsions, the extract only in the dose of 1000 mg/kg b. wt. significantly protected the animals from convulsions onset, significantly delayed the appearance of these convulsions of about 73 seconds and prevented the death of all unprotected animals. The extract’s anticonvulsant effect was found to be superior against isoniazid-induced convulsions than against pentylenetetrazol-induced convulsions. Isoniazid induce convulsions by reducing the GABA content in the brain through inhibition of glutamic acid decarboxylase [14,15], enzyme that is involved in the neuronal synthesis of GABA. So, some components in the root bark aqueous-alcoholic of Dichrostachys cinerea could have promoted GABA synthesis. The findings of the current study, therefore, tend to suggest that Dichrostachys cinerea root bark aqueous-alcoholic extract has a little anticonvulsant activity through stimulating GABA synthesis interfering with GABAergic neurotransmission. However, further study is needed to find the accurate mechanism of interaction with GABA mediated pathway.
Besides, the inflammatory aspect of asthma was experimentally studied [5] and data brought out an antiinflammatory potential effect through formalin-induced rat paw irritation, a non-specific anti-inflammatory test that also measure analgesic activity. As the extract did not show any effect through carrageenan-induced edema, a specific antiinflammatory test, the current study also aimed to estimate the analgesic potential of D. cinerea root bark. The extract did not exert morphine-type analgesic effect in conformity with formalininduced rat paw irritation in which the first phase, related to pain mediated by substances that effects can be antagonized by opioids [16], was not inhibited. On the other hand, the extract developed an analgesic effect on acetic acid-induced writhes. Writhing test is not specific because it measures analgesic, antiinflammatory and skeletal muscle relaxant activities, with a greater sensibility to the evaluation of peripheral pain [8]. Not only D. cinerea previously did not show anti-inflammatory effect, and just exerted low relaxant effect on skeletal muscle (low anticonvulsant effect), but therefore acetic acid is involved in peripheral mechanisms of pain, including release of substances like histamine, prostaglandins, serotonin, bradykinin in high proportions in rodent’s peritoneal exudates [17]. Thus, it makes sense to evoke the strong probability of an analgesic effect with peripheral mediation such as that highlighted by the team of our laboratory [18].
Conclusion
Dichrostachys cinerea root bark, an Ivory Coast traditional medicine, induced a low anticonvulsant effect. In more, the level of protection from convulsion-induced death could be beneficial in epilepsy additive treatment as a new therapeutic indication of this plant outside the asthma. The extract did not either induce morphine-type analgesic effect. However, this herbal developed non-morphine-type analgesic effect that could present an interest in the therapeutic coverage of asthma in which contraction of smooth muscle is painful.
References
- WHO (2013) WHO Traditional Medicine Strategy: 2014-2023. World Health Organization, Switzerland p. 76.
- Adjanohoun EJ, Ake Assi L (1979) Contribution au Recensement des Plantes Médicinales en Côte d’Ivoire (Contribution to the Identification of Medicinal Plants in Cote d’Ivoire). Centre National de Floristique, Abidjan pp. 358.
- Irie N’guessan G, Champy P, Kouakou Siransy G, Koffi A, Kablan BJ, et al. (2011) Tracheal relaxation of five Ivorian anti asthmatic plants: Role of epithelium and K+ channels in the effect of the aqueous-alcoholic extract of Dichrostachys cinerea root bark. J Ethnopharmacol 138(2): 432-438.
- Bousquet J, Jeffery PK, Busse WW, Johnson M, Vignola AM (2000) Asthma from bronchoconstriction to airways inflammation and remodeling. Am J Respir Critic Care Med 161(5): 1720-1745.
- Irié-N’guessan AG, Kouakou SL, Effo KE, Adepo AA, Kouakou-Siransy NG, et al. (2017) Anti-inflammatory and antioxidant potential of Dichrostachys cinerea root bark, an Ivorian anti-asthmatic herbal. Int J Pharmacol Res 7(12): 248-254.
- Bum EN, Schmutz M, Meyer C, Rakotonirina A, Bopelet M, et al. (2001) Anticonvulsant properties of the methanolic extract of Cyperus articulatus (Cyperaceae). J Ethnopharmacol 76(2): 145-150.
- Raza M, Shaheen F, Choudhary MI, Suria A, Sombati S, et al. (2000) Anticonvulsant activities of the FS-1 subfraction isolated from roots of Delphinium denudatum. Phytother Res 15(5): 426-430.
- Koster R, Anderson M, de Beer EJ (1959) Acetic acid for analgesic screening. Fed Proc 18: 412-418.
- Collier HOJ, Dinneen LC, Jonhson CA, Schneider C (1968) The abdominal constriction response and its suppression by analgesic drugs in the mouse. Br J Pharmacol Chemother 32(2): 295-310.
- Janssen PA, Niemegeers CJ, Dony JG (1963) The inhibitory effect of fentanyl and other morphine-like analgesics on the warm water induced tail withdrawal reflex in rats. Arzneimittelforschung 1963(13): 502-507.
- Louhimies S (2002) Directive 86/609/EEC on the protection of animals used for experimental and other scientific purposes. Altern Laboratory Animals: Atla 30(2): 217-219.
- Vasconcelos SM, Lima NM, Sales GT, Cunha GM, Aguiar LM, et al. (2007) Anticonvulsant activity of hydroalcoholic extracts from Erythrina velutina and Erythrina mulungu. J Ethnopharmacol 110(2): 271-274.
- Silambujanaki P, Chitra V, Suman K, Sankari M, Raju D, et al. (2010) Anti-convulsant activity of methanolic extract of Butea monosperma leaves. Res J Pharm Biol Chem Sci 1(2): 431-435.
- Yemitan OK, Salahdeen HM (2005) Neurosedative and muscle relaxant activities of aqueous extract of Bryophyllum pinnatum. Fitoter 76(2): 187-193.
- Schousboe A, Waagepetersen HS (2007) GABA: homeostatic and pharmacological aspects. Progress in Brain Res 160: 9-19.
- Tjolsen A, Berge OG, Hunskaar S, Rosland JH, Hole K (1992) The formalin test: An evaluation of the method. Pain 4: 161-174.
- Negus SS, Vanderah TW, Brandt MR, Blisky EJ, Becerra L, et al. (2006) Preclinical assessment of candidate analgesic drugs: Recent advances and future challenges. J Pharmacol Exp Ther 319(2): 507-514.
- Kouakou-Siransy G, Effo KE, Irié-Nguessan G, Koua E (2017) Analgesic efficacy, quality and safety of “Sarenta”: An herbal preparation from Ivorian traditional medicine. Int J Pharmacol 13(3): 257-265.






























