A Brief Overview of Nerve Gas
David M. Benjamin*
Clinical Pharmacologist & Toxicologist, Affiliate Associate Professor, Department of Pharmaceutical Sciences, School of Pharmacy, Northeastern UniversityBoston, MA, USA
Submission: August 24, 2018;Published: September 10, 2018
*Corresponding author: David M Benjamin, Clinical Pharmacologist & Toxicologist, Affiliate Associate Professor, Department of Pharmaceutical Sciences, School of Pharmacy, Northeastern University, Boston, MA, USA
How to cite this article: David M Benjamin. A Brief Overview of Nerve Gas. J of Pharmacol & Clin Res. 2018; 6(2): 555684. DOI: 10.19080/JPCR.2018.06.555684
History
In March 1995, a terrorist cult group led by an individual named Aum Shinrikyo (“Supreme Truth”) terrorized the people of Tokyo by releasing an organophosphate nerve gas called Sarin into the Tokyo subway system.This terrorist attack killed 12 people and injured more than 5500 people.The gas had been concealed in lunchboxes and soft drink containers and was released at several points along the Tokyo subway system.Because the gas was poorly volatilized, many victims first experienced ophthalmic and respiratory signs and symptoms due to direct contact.Had the Sarin been more completely volatilized, many more people would have been injured or killed. If the attack had been carried out during the summer when people wore sleeveless shirts and shorts, the toxic agent could have been absorbed through the skin and, once again, many more people would have been injured or killed. This was the same toxic nerve gas allegedly used by the Syrian government a few weeks ago against its citizens who were protesting against the government.
Organophosphate nerve gases Like Sarin, Tabun, Soman and diisopropyl fluorophosphate (DFP) were developed in Germany during the 1930s and used by the Nazis against American and allied troops.Because these chemicals are heavier than air, they tend to stay close to the ground and can easily incapacitate people at ground level or in trenches.
Chemistry
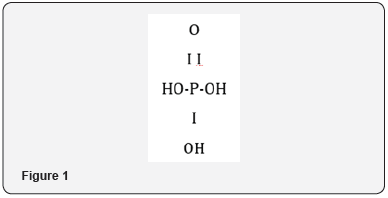
Organophosphate nerve gases are structurally related to a similar class of pesticides which include Malathion and Parathion.The organophosphates derive their name because their structures represent modifications of phosphoric acid.A schematic representation of the simple phosphoric acid molecule, H3PO4, is shown belowFigure 1.
In order to convert the simple phosphoric acid molecule to a deadly nerve gas, the hydroxyl groups (OH) are substituted or replaced by different organic substituents such as a methyl, ethyl or isopropyl side chains or a sulfur (S) or fluorine (F) molecule.
Mechanism of Action (Toxicity)
Organophosphate compounds produce their toxic effects by interfering with normal nerve conduction. When nerves send messages to one another, they communicate by releasing a neurotransmitter from the end of the first (pre-synaptic) neuron which diffuses across a space between the neurons called the synapse or synaptic cleft. When the neurotransmitter reaches the postsynaptic neuron, it stimulates or depolarizes the postsynaptic neuron causing it to generate an “action potential” which travels down the neuron and then synapses either with another neuron or with its end-organ, which can be a muscle or gland.Although there are many different neurotransmitters in human nervous system, the organophosphates specifically interfere with those neurons using acetylcholine (Ach) as a neurotransmitter. These neurons are called cholinergic neurons. In order to inactivate the acetylcholine to avoid continued stimulation and allow the neuron to return to its resting state, postsynaptic cholinergic neurons possess an enzyme called cholinesterase (AchE) which hydrolyzes (cleaves with water) the acetylcholine and permits the neuron to repolarize and return to its resting state.It is the cholinesterase enzyme that is the target of organophosphate chemical weapons.When AchE is inactivated, the cholinergic neurons continue to fire producing a constellation of cholinergic signs and symptoms throughout the body, the most common of which are excessive salivation and increased secretions in the lungs as well as constriction of the pupils of the eye accompanied by an urge to urinate and defecate.The most common cause of death is paralysis of the diaphragm and the intercostal muscles involved with breathing which leads to suffocation.However, depending on the physicalchemical properties of the particular nerve gas, distribution to the brain can also occur which may result in seizures.
Antidotes
The immediate antidotes for organophosphate nerve gas toxicity consist of the administration of atropine or scopolamine which block the effect of acetylcholine at the junction where the nerves synapse with the muscles involved with respiration. In order to regenerate the AchE, another type of agent called an oxime must be administered. The most common of these is pralidoxime, aka 2-PAM. If only atropine is administered, victims can go on to develop both intermediary and long-term neurologic toxicity of both the peripheral nervous system in the body and the central nervous system in the brain. Damage to the brain can be devastating and cognitive function declines significantly, and severely exposed victims can develop degenerative changes in the brain which mimic severe Alzheimer’s disease.
Closing Statement
Organophosphate nerve gases are horrible weapons.They create panic, horror, and pandemonium among exposed victims. They are not easily focused, like shooting a gun, and depending upon the vehicle used to deploy the weapon and its degree of volatilization, the liquid form or gas may be carried into far ranging areas by the wind or prevailing weather conditions. It is no wonder that the civilized world has enacted strong measures to deter their use under any conditionsand requires offending nations to submit to inspections which will ensure that these highly toxic agents have been destroyed.






























