Current scenario and distribution of NABL Accreditated laboratories in India
Maurya Mitesh R*1 and Maurya Bindulata2
1Department of Clinical Pharmacology, Seth GS Medical College & Hospital, India
2Department of material medical, G.D. Pol foundation, Yerala medical college & PG institute, India
Submission: September 23, 2017; Published: October 05, 2017
*Corresponding author: Dr. Maurya Mitesh R, DM resident from department of Clinical Pharmacology, Seth GS Medical College & Hospital, parel, Mumbai-12, India, Tel: 9271790018; Email: mitesh.maurya4@gmail.com
How to cite this article: Maurya Mitesh R , Maurya Bindulata. Current scenario and distribution of NABL Accreditated laboratories in India. J of Pharmacol 007 & Clin Res. 2017; 3(5): 555625. DOI: 10.19080/JPCR.2017.03.555625.
Abstract
Medical laboratories can seek recognition as being compliant with particular standards through a number of different systems. Recognition of medical laboratories handling clinical specimens as distinct from those dealing with the results of pre-clinical studies is in their infancy in most countries. A laboratory accreditation system contains three elements, the organisation or authority which conducts the inspections or assessments and grants accreditation (and may also set the standards), the inspection or assessment which seeks to establish compliance with the standards, and crucial to the whole process the standards themselves. In most countries the emphasis has been on development of standards and quality manuals and practical experience in all three elements of accreditation is, with a few exceptions, is very limited. In other parts of the world, such as USA, Canada and Australia there is depth of experience from which other countries can learn. In this we have conducted audit study on current scenario and distribution of National Accreditation Board for Testing and Calibration Laboratories (NABL) accreditation in India up to as on 30 August 2017. The data is very helpful in analysing the trend of NABL accreditation in India and strengthening the new accreditation practices and also renewal of expired accreditation [5-7].
Keywords: Accreditation; ISO; NABL; Suspended; Expired; Voluntary; Calibration; Laboratories
Introduction
Accreditation is the third party attestation related to a conformity assessment body conveying the formal demonstration of its competence to carry out specific conformity assessment task. Conformity Assessment Body (CAB) is a body which includes Testing including medical Laboratory, Calibration Laboratory, Proficiency Testing Provider, and Certified Reference Material (CRM) Producer. The liberalization of trade and industry policies of the Government of India has created quality consciousness in domestic trade and provided greater thrust for export. As a consequence, testing centres and laboratories have to demonstrably operate at an internationally acceptable level of competence. Laboratory accreditation is a procedure by which an authoritative body gives formal recognition of technical competence for specific tests/ measurements, based on third party assessment and following international standards. Similarly, Proficiency testing Provider accreditation gives formal recognition of competence for organizations that provide proficiency testing. Reference Material Producers Accreditation gives formal recognition of competence to carry out the production of reference materials based on third party assessment and following international standards [1-3].
NABL is a Constituent Board of Quality Council of India. National Accreditation Board for Testing and Calibration Laboratories (NABL), New Delhi is a Society registered under Societies Registration Act, 1860 and is an autonomous body under the aegis of Department of Science & Technology (DST). NABL has been established with the objective to provide Government, Industry Associations and Industry in general with a scheme for third-party assessment of the quality and technical competence of Conformity Assessment Bodies (Testing including Medical, Calibration Laboratories, Proficiency Testing Providers and Reference Material Producers).The laboratory accreditation services to testing and calibration laboratories are provided in accordance with ISO/ IEC 17025 'General Requirements for the Competence of Testing and Calibration Laboratories' and ISO 15189 'Medical laboratories - Requirements for Quality and Competence' The accreditation to Proficiency testing providers are based on ISO/IEC 17043 “Conformity Assessment - General Requirements for Proficiency Testing” and to Reference Material Producers based on ISO Guide 34 “General Requirements for the Competence of Reference Material Producers.”These services Medical Laboratories are offered in a non-discriminatory manner and are accessible to all Conformity Assessment Bodies (CABs) in India and abroad, regardless of their ownership, legal status, size and degree of independence. NABL has established its Accreditation System in accordance with ISO/IEC 17011 “Conformity Assessment - General requirement for accreditation bodies accrediting conformity assessment bodies”, which is followed internationally [1-3].
NABL maintains linkages with the international bodies like International Laboratory Accreditation Co-operation (ILAC) and Asia Pacific Laboratory Accreditation Co-operation (APLAC). NABL is signatory to ILAC as well as APLAC Mutual Recognition Arrangements (MRA) for accreditation of Testing including Medical and Calibration laboratories, which is based on mutual evaluation and acceptance of other MRA Partner accreditation systems. Such international arrangements facilitate acceptance of test/ calibration results between countries which MRA partners represent [1-3].
Scope and Areas of Accreditation
NABL Accreditation is currently given in the following fields and disciplines. The multi-disciplinary CABs shall have to apply in relevant discipline separately depending upon to which discipline the scope belongs. For more details on scope of accreditation please refer the relevant specific criteria [1-3].
Testing Laboratories
Testing Laboratories
a) Biological
b) Chemical
c) Electrical
d) Electronics
e) Fluid-Flow
f) Mechanical
g) Non-Destructive Testing
h) Radiological
i) Forensic
j) Thermal
Calibration Laboratories
a) Electro-Technical
b) Mechanical
c) Fluid Flow
d) Thermal & Optical
e) Radiological
Medical Laboratories
a) Clinical Biochemistry
b) Clinical Pathology
c) Hematology & Immunohaematology
d) Microbiology & Serology
e) Histopathology
f) Cytopathology
g) Genetice
h) Nuclear Medicine (in-vitro tests only)
Proficiency Testing Providers
a) Testing
b) Calibration
c) Medical
d) Inspection
Reference Material Producers
a) Chemical Composition
b) Biological & Clinical Properties
c) Physical Properties
d) Engineering Properties
e) Miscellaneous Properties
Methodology
Since the data lies in public domain, the study was conducted under ethical exemption. The website http://www.nabl-india.org/ was accessed and following url link http://www.nabl-india.org/nabl/index.php?c=search&m=index&Itemid=177was explored on 30/08/2017 and laboratory searches was performed in order to get information regarding total number of accreditated laboratories (region wise, zonal distribution, various fields and discipline wise), suspended accreditation, forceful withdrawal accreditation, voluntary withdrawal accreditation and expired accreditation (fields and year wise distribution).
Results
Some laboratories were NABL accredited, some had suspended accreditation, and some had forced withdrawal accreditation while some had voluntary withdrawal accreditation. Few accreditations were expired and require renewal. On going through the data available, following were the figures of distribution of these accreditation obtained.
NABL Accreditated laboratories
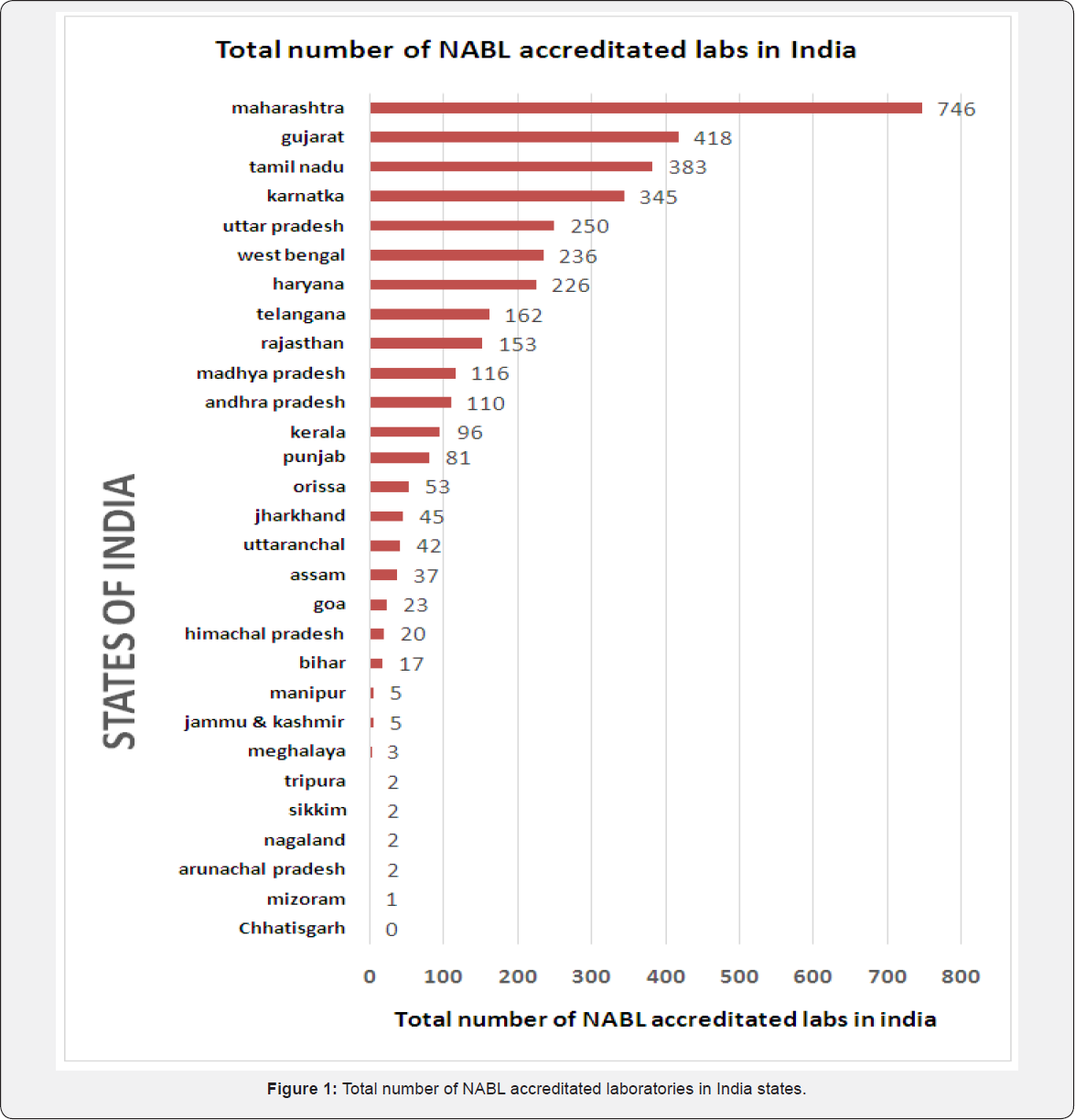
There are total 3873 NABL accreditated laboratories in India. Out of these 3581 laboratories were accreditated in states, 264 NABL accreditated laboratories belonged to union territory under purview of central government and remaining 28 accreditated laboratories that was classified to unknown category comprised of laboratories from different countries. From 3873 NABL accreditated laboratories, maximum laboratories were accreditated from states of Maharashtra (746), Gujarat (418), Tamil Nadu (383) and Karnataka (345) while there were no labs accreditated in state of Chhattisgarh and very few from states of north-eastern region like Mizoram (1), Arunachal Pradesh (1), Nagaland (2), Sikkim (2), Tripura (2) [1] (Figure 1).
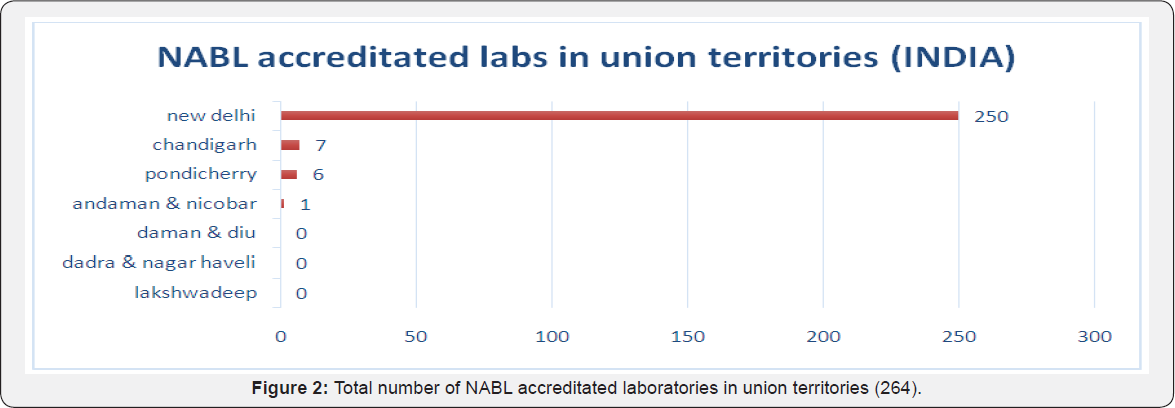
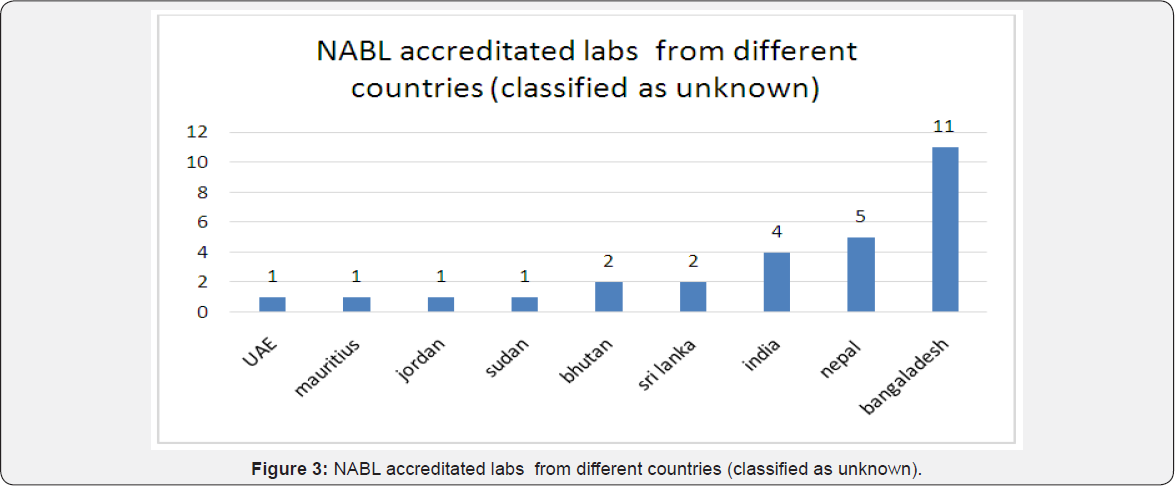




Among Union territories, maximum laboratories received accreditation from capital New delhi (250) followed by Chandigarh (7), Pondicherry (7), Andaman and nicobar (1) while others had none (Dadra and Nagar haveli, Lakshadweep and Daman & Diu) [1] (Figure 2). Remaining 28 accreditated laboratories that was classified to unknown category comprised of laboratories from different countries like India(4) that included two from Chandigarh, one from New Delhi and one from Uttarakhand, Nepal(5), Bhutan(2), Sudan(1), Jordan(1), Sri Lanka(2), UAE(1), Mauritius(1) and Bangladesh(11) [1] (Figure 3). When studied according to 5 zones, the distribution of laboratories recognized with NABL accreditation were maximum from west zone (1278 labs) to be followed by south zone (1113), north zone (967 labs), east zone (421 labs) and central zone with 155 laboratories only [1] (Figures 4-11).
Among NABL accreditated testing laboratories (3551) as shown above, following were the distribution of various disciplines. Maximum testing laboratories that received accreditation belongs to disciplines of chemical (1717), electrical (928) and non-destructive laboratories (332) while very few labs from disciplines such as electronics (101), photometry (21), forensic (8), radiology (6), fluid flow (4), thermal (2) contributed to NABL accreditation [1] of the total number of accreditated laboratories in India, 3679 laboratories provide permanent facility while 38 labs has on site facility while only 1 laboratory has mobile facility.
Suspended accreditation
In all, 17 laboratories from following disciplines were suspended of NABL accreditation w.e.f year 2016-17 of which maximum belonged to chemical labs (53%) followed by mechanical labs (17%). Only one medical lab was suspended of NABL accreditation w.e.f 30/6/17 (Xcelris Labs Limited) [1].
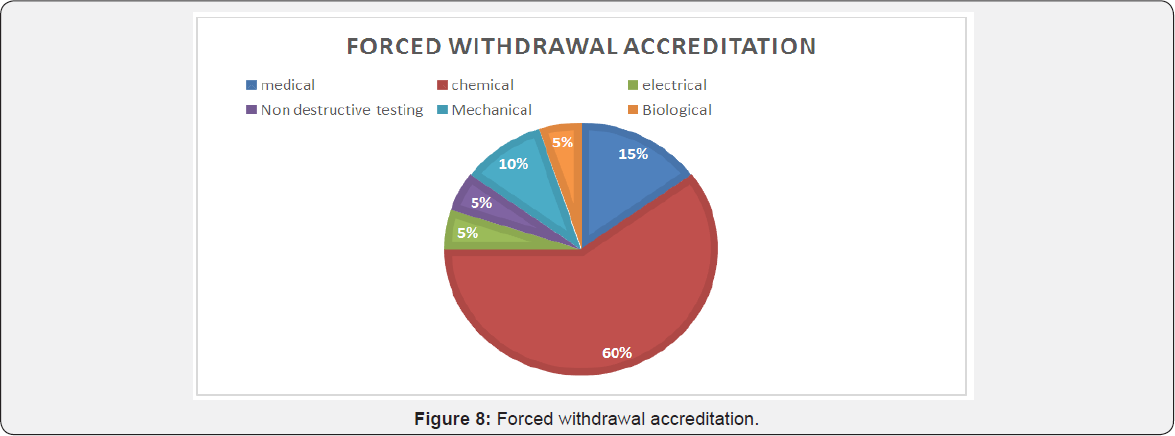
Forced withdrawal accreditation
Following were the distribution of various disciplines that saw forced withdrawal of NABL accreditation of which maximum were the chemical laboratories and medical laboratories while less labs lost accreditation from electrical, biological and nondestructive testing labs [1].
Voluntary withdrawal accreditation
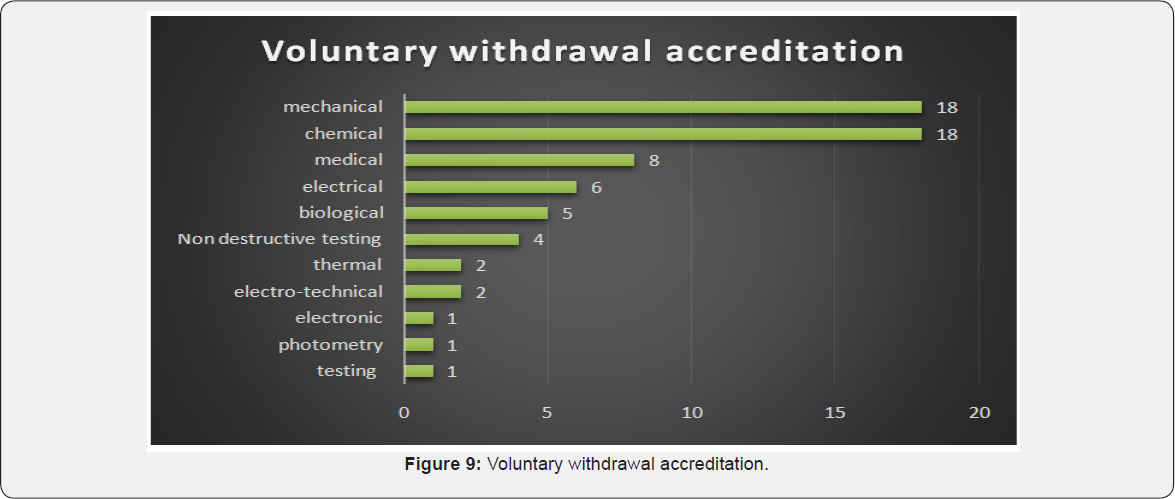
Many labs were withdrawn NABL accreditation voluntarily from mechanical and chemical labs followed by medical labs while certain disciplines like electro-technical, electronic, photometry and testing labs shown least voluntary withdrawal of accreditation [1].
Expired accreditation
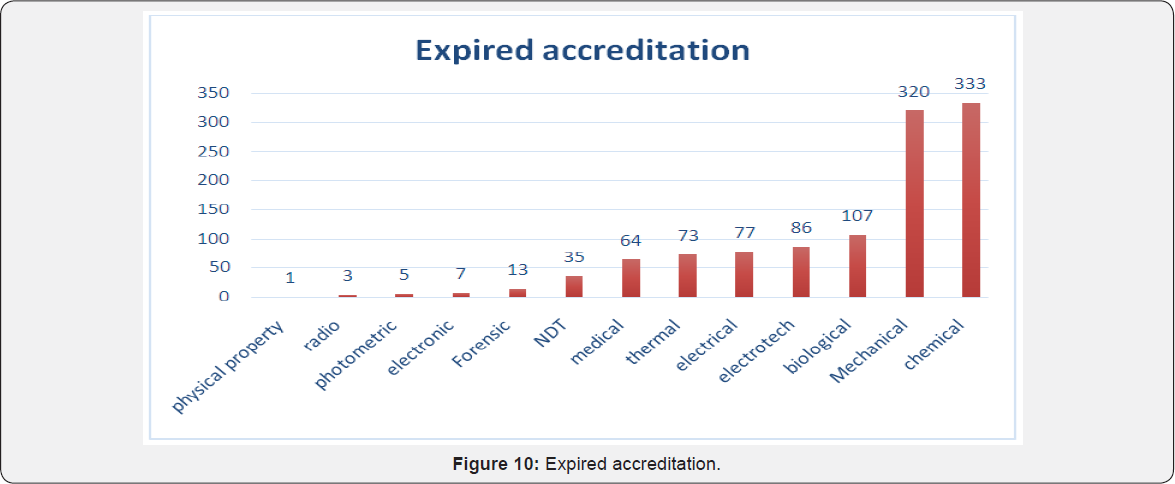
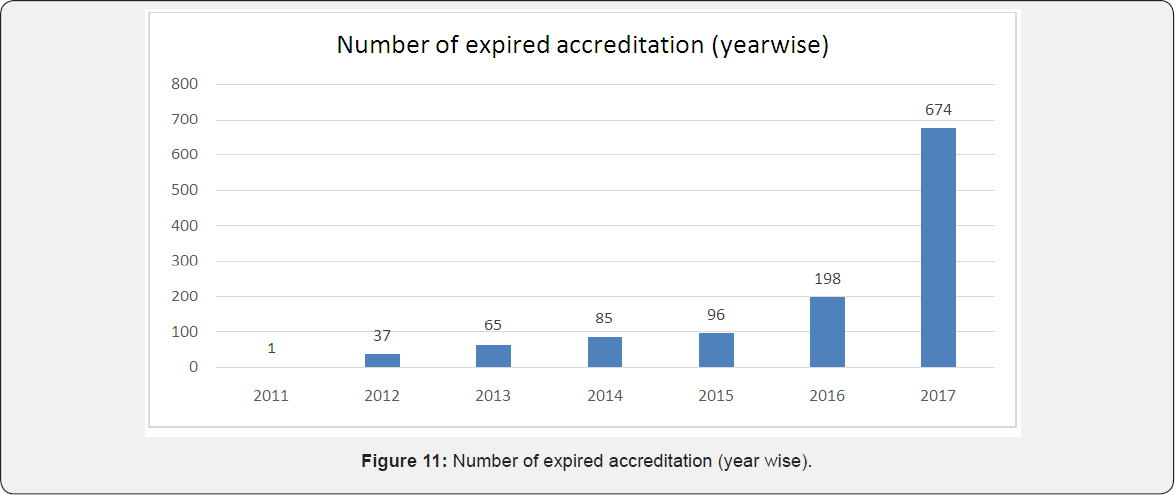
Again as per analysis of data up to 30 august 2017, maximum laboratories accreditation were expired from chemical, mechanical, biological and electro technical disciplines. Very few accreditation were expired from fields of physical property, radiology, photometric and electronics. Almost 64 medical labs shown their accreditation to be expired [1] of the expired accreditation, year wise analysis was done and following data was obtained. As per data maximum labs were expired in year 2017 (674 labs). Also there seems to be increasing trend in more laboratories getting expired of its NABL accreditation in succeeding years and needs to apply for renewal for NABL accreditation [1].
Conclusion
The process of NABL accreditation is gaining momentum in recent years. More and more NABL certified labs are upcoming meeting required standards and criteria. The NABL website of India is actively updating the registration of NABL accreditated labs and suspending those substandard ones. Expired accreditation has to get renewed its NABL accreditation. Now the process of NABL accreditation will encourage and facilitate in raising standards in various disciplines promoting consumer safety and satisfaction by meeting their expectations and also competing with international standards.
Acknowledgement
This work was supported in part by beloved wife Dr. Bindulata Maurya who has contributed in collection and data analysis with valuable comment being the very part of this study and making it worth complete in short span of time.
References
- Nabl India org (2017).
- National Accreditation Board for Testing and Calibration Laboratories (NABL).
- National Accreditation Board for Testing & Calibration Laboratories New Delhi (2017)
- List of NABL Accredited Laboratories in India (2017)
- Burnett D (1996) Understanding Accreditation in Laboratory Medicine. ACB Venture Publications, London, UK
- Burnett D (1998) Healthcare accreditation in Hospital Healthcare Europe. London.
- Jansen RTP, Blaton V, Burnett D (1997) European Communities Confederation of Clinical Chemistry: Essential Criteria for Quality Systems of Medical Laboratories. Eur J Clin Chem Clin Bio chem 35(2): 123-132.






























