Incentives to Increase Pharmacovigilance Practices from an Educational and Ethical Point of View
Müberra Devrim Güner*1 and Perihan Elif Ekmekci2
1Department of Medical Pharmacology, TOBB Economy and Technology University, Turkey
2Department of History of Medicine and Ethics, TOBB Economy and Technology University, Turkey
Submission: September 20, 2017; Published: September 22, 2017
*Corresponding author: Müberra Devrim Güner, TOBB Economy and Technology University, Medical Faculty Department of Medical Pharmacology. Söğützöü Cad. No: 43 06560, Ankara, Turkey, E-mail: devrimguner@etu.edu.tr
How to cite this article: Guner M D. Ekmekci P E. Incentives to Increase Pharmacovigilance Practices from an Educational and Ethical Point of View. J of Pharmacol & Clin Res. 2017; 3(5): 555624. DOI: 10.19080/JPCR.2017.03.555624
Abstract
An adverse drug reaction (ADR) is a burden both to healthcare system and has economic, medical and ethical dimensions. ADR reporting is a responsibility of all healthcare professionals and is essential for the effectiveness of Pharmacovigilance system. ADR reporting in Turkey is still below the average of similar income countries. The main reason for low reporting rates can be lack/insufficiency of reporting culture and limited awareness about the importance of reporting. This can be improved by incorporating pharmacovigilance practices and ADR reporting procedures education and training to both undergraduate and continuous education of healthcare professionals.
Keywords: Pharmacovigilance; Medical education; Adverse drug reaction reporting
Abbreviations: ADR: Adverse Drug Reaction; WHO: World Health Organization; TÜFAM: Turkish Pharmacovigilance Center; TADMER: Turkish Drug Adverse Event Monitoring and Evaluation Center
Introduction
The World Health Organization (WHO) defined adverse drug reaction (ADR) as "a response to a drug which is noxious and unintended, and which occurs at doses normally used in man for the prophylaxis, diagnosis, or therapy of disease, or for the modification of physiological function" in 1972. Pharmacovigilance was defined as "the science and activities relating to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problem" in 2002 by WHO. WHO initiated a program for international drug monitoring which is based in Uppsala since 1978 Turkey became the 27th member of the program in 1987.
In Turkey, pharmacovigilance activities were institutionalized by the establishment of "Turkish Drug Adverse Event Monitoring and Evaluation Center" (TADMER) in 1985. However, a regulation on the Monitoring and Assessment of the Safety of Medicinal Products for Human Use was published 20 years later in 2005 by Turkish Republic Ministry of Health, and was updated as "Regulation on safety of drugs" in 2014 [1]. Pharmaco vigilance practices were reevaluated by this regulation and activities carried by "Turkish Pharmacovigilance Center" (TÜFAM) such as data collection, risk evaluation, trainings, and inspections were increased. Since 2005, hospitals have been required to assign a healthcare professional to function as a "contact point" for pharmacovigilance activities within the hospital. "Good Pharmacovigilance Practices" guidelines were also published in 2014 [2].
These efforts are all in line with the current activities in European Union (EU) and the United States of America (USA), but as proven by scarce literature on this issue, ADR reporting rates are still below the expected numbers Turkey[3-6]. ADR reporting is a responsibility of all healthcare professionals and is essential for the effectiveness of pharmacovigilance system [1]. According to a current study reporting rates gradually increased since 2005 [3], however still below the average of similar income countries: the ADR reporting rate of Turkey is 2 reports/million inhabitants/year [7]. Most of the ADR reports from Turkey were reported by physicians (59.8%), followed by pharmacists (9.1%) and other healthcare professionals (28.7%) [3].
ADR is an important public health problem. According to a meta-analysis of 39 USA studies, the overall incidence of serious ADRs is 6.7% and fatal ADRs is 0.32% making ADRs 4th -6th leading cause of death [8]. Hospital admission rate is 5.1% and increases with age up to 9.8% (>75 years) and 40% of these admissions are considered to be avoidable [9]. According to a current meta-analysis, the median prevalence of ADR is 6.3% and median proportion of preventable ADRs accounts to 71.7% [10]. ADRs also bring an important economic burden to the health system, causing an average cost of 260$ per patient and total estimated cost per year 588 million $ for Germany [11]. Hospitalizations due to ADR cost about 1840-3000€/hospital admission for France [12]. Drug-related morbidities cost about 30 - 130 billion $/year in USA [13].
ADR reporting also carries a very important ethical dimension besides the health and economic burden. Considering the main ethical principles of medicine, underreporting of ADR is problematic in terms of two principles: beneficence and respect for autonomy. The concept of benefit includes avoiding any potential risk of harm as well [14]. Reluctance in reporting ADR inarguably augments health risks for other patients who are subject to the same treatment. It also avoids the accumulation of scientific knowledge regarding effects the drug, which would lead to the dominance of false data in literature and misuse of resources. This would breach the prima facie duty of the physicians' to respect the autonomy of their patients as well since the information they provide during informed consent would lack some information essential to their patients' health. Therefore, it is plausible to say that the principle of benefit and respect to autonomy require the physicians to take active action to report ADR.
Physicians’ responsibility in reporting ADR is justifiably broadened by utilitarian ethical theory, which suggests that an action that produces maximum utility for a maximum number of people is ethically right and therefore should be done [15]. This approach enhances the responsibility of the physician to provide benefit not only for her particular patient but for all patients suffering from the same health problem and have the potential to use the same drug, as well as contributing to the accumulation of scientific data. Moreover underreporting ADRs may lead significant negative economic and public health consequences by diverting health expenditures to futile or even harmful treatments.
The scarce reporting rates can be attributed to the lack/ insufficiency of reporting culture and limited awareness about the importance of reporting. According to several surveys although healthcare professionals have knowledge of pharmacovigilance and ADRs, the reporting rates are around 10-15% [16]. The awareness and knowledge, although not sufficient, is highest among medical doctors, and decreases significantly for other healthcare professionals [4,6,17,18]. Moreover, the healthcare professionals other than medical doctors think that suspecting an ADR, detecting and reporting it is only a responsibility for medical doctors [6,17,18]. Competent and confident health professionals can be accomplished by increasing the knowledge of pharmacovigilance and prioritizing medical safety and ethics at each step of daily medical practice. The necessary incentives to obtain the goals are to develop a more comprehensive pharmacovigilance syllabus underlining the ethical aspects of individual patients' and public’s health and wellbeing, medical professionals 'competency, pharmaceutical companies and regulatory authorities' roles and responsibilities. Moreover, this education must also not be limited to graduate schools but must be expanded to continuous professional education.
The numbers of Turkish medical schools are 81 as of 2017. In order to harmonize and increase the quality, standardization of the medical education is required. Council of Higher Education published "National Core Education Program" in 2002 and updated in 2014 [19]. Medical schools are required to set their curricula in coherence with the core program. According to the updated program of 2014, pharmacovigilance related competencies of a medical student graduate can be listed as:
a) To plan, apply and monitor a rational medical treatment
b) To comply with principles of treatment of special populations (pediatric, geriatric, pregnant, lactating and patients with hepatic and renal diseases)
To calculate drug doses accurately
Moreover "adverse effects of drugs and drug interactions" is listed in "symptoms and clinical conditions". The subject of "drug side effects" is classified as a multisystem problem and the skill objectives defined as follows in National Core Education Program [19]:
i. To diagnose and treat
ii. To define and treat the emergency condition
iii. To refer to a specialist in case of need
iv. To monitor and control for long term under primary health care conditions
v. To apply the prevention methods
The activity of ADR and other related pharmacovigilance practices are not mentioned in the "record keeping, reporting and communication" part of the basic medical practices defined by National Core Education Program [19]. We suggest that next update must include a comprehensive handling of ADR and pharmacovigilance practices, as "reporting an ADR" is defined as "responsibility of healthcare professional" by above mentioned national pharmacovigilance regulations.
Beckmann et al. defined elements of pharmacovigilance training and stated that comprehensive pharmacovigilance syllabus can be achieved only by multidisciplinary collaboration and involvement of all stakeholders in order to achieve a custom- made curriculum covering the needs of each party. With this background, these experts created a comprehensive, detailed and balanced curriculum of pharmacovigilance [20]. Moreover, the guideline on good pharmacovigilance practices, which is a set of measures drawn up to facilitate the performance of pharmacovigilance in the EU can also be consulted while preparing a comprehensive and continuing education for pharmacovigilance [21] (Table 1).
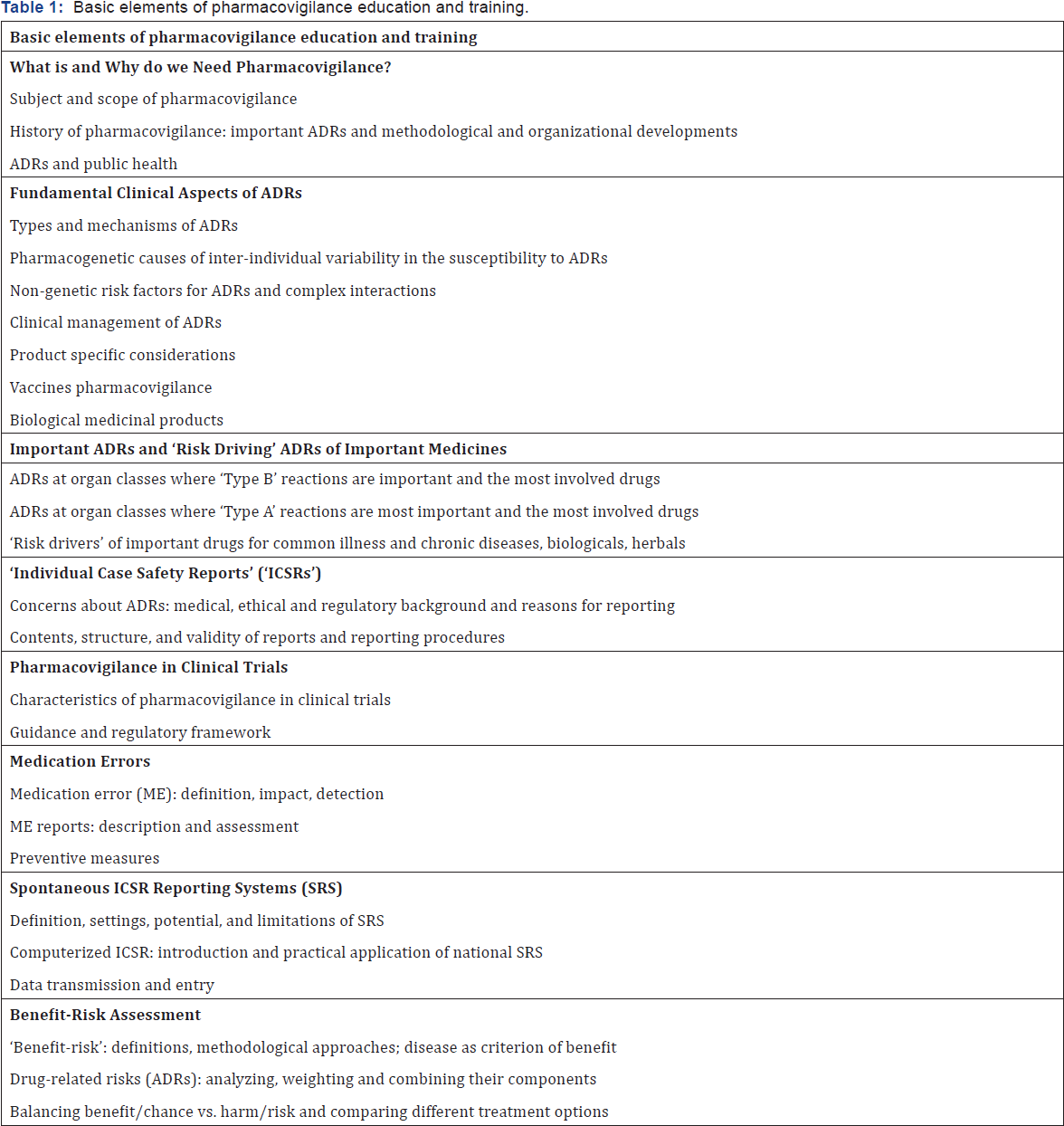
We believe that elements of this curriculum can be incorporated into the education of healthcare professionals both in undergraduate and continuing professional training, with a tailor-made approach. Profound knowledge of subjects such as rational drug use, prescription skills, drug use in special populations such as geriatric, pediatric and pregnant and lactating women are also essential to minimize the risk of ADRs and other prescription and medication errors. We also believe that both pharmacovigilance and these subjects should be covered in theoretical and practical courses and should be integrated to medical education program spirally, and the learning objectives and the outcomes should be defined adequately. This will not only increase the quality of diagnostic, prescription and treatment skills of future healthcare professionals but also will increase the knowledge, ability, and skills sufficient to perform pharmacovigilance activities properly.
Conclusion
In conclusion, realization of the centrality of pharmacovigilance to public health and its ethical importance by the regulatory authority, educators and pharmaceutical industry is the first step to improve the quality and quantity of pharmacovigilance activities and prevention of ADRs. Adding a comprehensive pharmacovigilance curriculum both to graduate and professional continuing education of all healthcare professionals, measuring of pharmacovigilance performance by governments and policymakers consistently and comprehensively on individual and institutional basis will also contribute to the idealization of pharmacovigilance activities. Raising not only professional but public awareness of the need and importance of pharmacovigilance through campaigns, seminars, workshops and, the creation of online and/or other means of user-friendly reporting mechanisms are also important elements of improvement.
References
- Turkish Drug and Medical Device Agency (2014) Regulation on safety of drugs.
- Turkish Drug and Medical Device Agency (2014) Good Pharmacovigilance Practices.
- Ozcan G, Aykac E, Kasap Y, Nemutlu NT, Sen E, et al. (2016) Adverse Drug Reaction Reporting Pattern in Turkey: Analysis of the National Database in the Context of the First Pharmacovigilance Legislation. Drugs Real World Outcomes 3(1): 33-43.
- Vural F, Ciftci S, Vural B (2015) The knowledge, attitude and behaviours of nurses about pharmacovigilance, adverse drug reaction and adverse event reporting in a state hospital. North ClinIstanb 1(3): 147-152.
- Usta C, Cilli A, Celenk B, Uzun R (2012) A pharmacovigilance study in chest diseases outpatient clinic. Tuberk Toraks 60(2): 123-128.
- Toklu HZ, Uysal MK (2008) The knowledge and attitude of the Turkish community pharmacists toward pharmacovigilance in the Kadikoy district of Istanbul. Pharm World Sci 30(5): 556-562.
- Aagaard L, Strandell J, Melskens L, Petersen PS, Holme Hansen E (2012) Global patterns of adverse drug reactions over a decade: analyses of spontaneous reports to VigiBase™. Drug Saf 35(12): 1171-1182.
- Lazarou J, Pomeranz BH, Corey PN (1998) Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA 279(15): 1200-1205.
- Van der Hooft CS, Dieleman JP, Siemes C, Aarnoudse A-J LHJ, Verhamme KMC, et al. (2008) Adverse drug reaction-related hospitalisations: a population-based cohort study. Pharmacoepidem Drug Safe 17: 365371.
- Angamo MT, Chalmers L, Curtain CM, Bereznicki LR (2016) Adverse-Drug-Reaction-Related Hospitalisations in Developed and Developing Countries: A Review of Prevalence and Contributing Factors. Drug Saf 39(9): 847-57.
- Goettler M, Schneeweiss S, Hasford J (1997) Adverse drug reaction monitoring-cost and benefit considerations. Part II: cost and prevent- ability of adverse drug reactions leading to hospital admission. Phar- macoepidemiol Drug Saf 6(Suppl 3): S79-90.
- Lagnaoui R, Moore N, Fach J, Longy-Boursier M, Bégaud B (2000) Adverse drug reactions in a department of systemic diseases-oriented internal medicine: prevalence, incidence, direct costs and avoidability. Eur J ClinPharmacol 56(2): 181-186.
- White TJ, Arakelian A, Rho JP (1999) Counting the costs of drug-related adverse events. Pharmacoeconomics 15(5): 445-458.
- Beauchamp TL, Childress JF (2001) Principles of biomedical ethics. Oxford University Press, USA, 454.
- Munson R (2013) Intervention and reflection: basic issues in bioethics. Cengage Learning. Boston, Massachusetts, USA, 863-875.
- Desai CK, Iyer G, Panchal J, Shah S, Dikshit RK (2011) An evaluation of knowledge, attitude, and practice of adverse drug reaction reporting among prescribers at a tertiary care hospital. Perspect Clin Res. 2(4): 129-1236.
- Sencen N, Altinkaynak M, Ferah I, Clark PM (2010) The Knowledge and Attitudes of Physicians and Nurses Towards Adverse Event Reporting and the Effect of Pharmacovigilance Training: A Hospital Experience. Journal of the Faculty of Pharmacy 30(1): 25-40.
- Alan S, Ozturk M, Gokyildiz S, Avcibay B, Karataç Y (2013) An evaluation of knowledge of pharmacovigilance among nurses and midwives in Turkey. Indian JPharmacol 45(6): 616-618.
- Turkish Council for High Education (2014) Undergraduate Medical Education National Core Competencies program.
- Beckmann J, Hagemann U, Bahri P, Bate A, Boyd IW, et al. (2014) Teaching pharmacovigilance: the WHO-ISoP core elements of a comprehensive modular curriculum. Drug Saf 37(10): 743-759.
- Guideline on good pharmacovigilance practices (2017).






























