Hepatobiliary and Cardiovascular Effects Limit the Utility of Systemic TGR5 Agonists in Diabetes
B Ganesh Bhat*1, LeeAnne McLean2, Dean P Phillips1, Judit Markovits3, Shufang Zhao3, Haisong Ju3, Ricardo E Chatelain3, Heather Sullivan1, Gus Welzel1, Carol Trotter1, Yali Chen1, Yang Yang1, Sudeep Chandra3, Shifeng Pan1, Peter McNamara1, H Martin Seidel1,2 and Deborah G Nguyen1
1Genomics Institute of the Novartis Research Foundation, USA
2Novartis Pharmaceuticals Corporation, USA
3Novartis Institutes for Bio Medical Research, USA
Submission: September 11, 2017; Published: September 20, 2017
*Corresponding author: B. Ganesh Bhat, PhD, RAC, Genomics Institute of the Novartis Research Foundation, 10675 John J Hopkins Dr, San Diego, CA, 92121, USA, Tel: 858-775-2824; Email: bgbhatl@gmai1.com
How to cite this article: Bhat B G, LeeAnne M, Dean P P, Judit M, Shufang Z, et al. Hepatobiliary and Cardiovascular Effects Limit the Utility of Systemic TGR5 Agonists 0012 in Diabetes. J of Pharmacol & Clin Res. 2017; 3(5): 555623. DOI: 10.19080/JPCR.2017.03.555623.
Abstract
There is considerable interest in the bile acid receptor TGR5 as a drug target for type 2 diabetes. Activation of TGR5 in intestinal enteroendocrine cells leads to increased cAMP and subsequent enhanced GLP-1 secretion, which is a clinically validated approach to control blood glucose levels. In addition, its expression and anti-inflammatory activity in monocytes/macrophages and dendritic cells suggested that it could also address the chronic inflammation characteristic of obesity and diabetes, and thus provide a better clinical benefit versus glucose lowering agents alone. We have identified a TGR5 agonist, FC-92-EC85, with suitable in vitro and pharmacokinetic properties. Acute non-clinical efficacy was demonstrated in both GLP-1 secretion and LPS-induced cytokine release models. However, treatment with FC-92-EC85 and other TGR5 agonists in rodents also led to increased bile volume, gall bladder wall thickening and hyperplasia. To determine whether these effects would be seen in higher species, we tested FC-92-EC85 in dogs. Unlike what was seen in mice, oral administration of FC-92-EC85 led to diminished GLP-1 response due to delayed gastric emptying. In addition, FC-92-EC85 delayed gallbladder emptying and induced acute wall thickening, as well as dose dependent increases in heart rate. These deleterious effects may limit the clinical application of TGR5 agonists in diabetes.
Keywords: TGR5; Gall bladder; Diabetes
Abbreviations: TGR5: Takeda G-protein-coupled Receptor 5; GLP-1: Glucagon-Like Peptide 1; cAMP: 3'-5'-cyclic Adenosine Mono Phosphate; GPBAR1: G-protein-Coupled Bile Acid Receptor 1; CFTR: Cystic Fibrosis Transmembrane Conductance Regulator; LPS: lipoPolySaccharide; TNF: Tumor Necrosis Factor; FXR: Farnesoid X Receptor; PBMC: Peripheral Blood Mononuclear Cells
Introduction
TGR5 is a Gs-coupled GPCR activated by both primary and secondary bile acids (i.e. taurolithocholic acid) encoded by the G protein-coupled bile acid receptor 1 (GPBAR1) gene. The biological role of TGR5 is a function of both its pattern of expression and its ability to increase cellular cAMP [1-3]. Activation of TGR5 in intestinal enteroendocrine cells leads to secretion of GLP-1 and other related incretins, through a mechanism similar to GPR119 [4]. In monocytes/macrophages and dendritic cells (Mo/Ma)/DC], TGR5 activation leads to decreases in LPS-induced Th1 cytokines like TNF-a and IL- 12 [5]. Together, these functions make TGR5 an attractive target in the treatment of type 2 diabetes, where strategies to enhance GLP-1 [6] and mitigate chronic inflammation [7-9] are already demonstrating benefit in the clinic. While several pharmaceutical organizations have pursued TGR5 agonists, no TGR5 drug has been approved for diabetes or metabolic diseases indication.
TGR5 expression is not limited to the gastrointestinal and immune systems. In the mouse, the highest expression of TGR5 is in the gallbladder [10]. Stimulation of TGR5 in gall bladder epithelium leads to increased membrane permeability to chloride, bicarbonate and water, mediated in part by CFTR [11]. In this way, TGR5 serves as a sensor of bile acid concentration in the bile, enabling dilution if the concentration is too high. Considering the toxicity of high concentrations of bile acids on the liver, TGR5 sensor activity in the gall bladder is a key function. Evidence also suggests that TGR5 is localized to gall bladder smooth muscle, where its activation leads to inhibition of contraction and subsequent gall bladder filling [12]. These functions may make development of a systemic TGR5 agonist challenging, especially in type 2 diabetics who are already prone to gall bladder dysfunction [13]. This may explain efforts by several companies to develop non-systemic TGR5 agonists, with the goal of activating TGR5 in the GI tract and avoiding gall bladder exposure [14-17]. However, this presents a challenging path for drug development, and it is still not clear if the distribution and function of TGR5 are the same in the gall bladder of human and the various pre-clinical species where studies have been conducted to date.
Considering the potential to treat type 2 diabetes, we identified systemic low molecular weight agonists of TGR5. Despite significant challenges in optimization of clinical candidates, potent compounds with suitable pharmacokinetic profiles were achieved [18]. The TGR5 agonist FC-92-EC85 strongly increased glucose mediated GLP-1 secretion and decreased LPS-induced TNF-a both in vitro and in vivo, as expected. However, acute treatment also led to increases in bile volume. Two week treatment with TGR5 agonists representing several chemo types induced changes in the gall bladder of mice, including increased wall thickness mostly due to mucosal hyperplasia. Subsequent acute testing of FC-92-EC85 in dogs showed a diminished GLP-1 response and delayed gastric emptying. In addition, acute changes in gall bladder morphology and function were seen by ultrasound imaging. Taken together, these data suggest that the clinical utility of systemic TGR5 agonists to treat type 2diabetes may be limited.
Materials and Methods
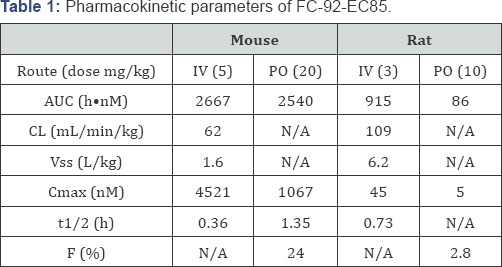
TGR5 cellular cAMP assay. Jurkat cells overexpressing human or mouse TGR5 were seeded at 50,000 cells/25|il/well in 384-well plates (Greiner) in AIM V media (Life Technologies) supplemented with 1mM IBMX (Sigma) and 10mM HEPES. Immediately after plating, 200nL of compounds in DMSO were transferred using the Pintool (GNF systems). Following a 1hr at 37 °C, cryptate and D2 conjugates from the cAMP HiRange HTRF kit (Cisbio) were added (12.5|il each). Plates were then incubated 1hr at room temperature and emission at 620nM and 665nM were read on the Envision (Perkin Elmer) as suggested by the manufacturer. Data was expressed as a ratio of emission at 665/620, and then normalized to a DMSO alone control. Dose- response data points were curve fitted using the standard logistic regression model, implemented with an in-house developed Matlab (mathworks.com) program. The resultant EC50 values correspond to the compound concentration that displaces 50% of the binding of the d2-labeled cAMP.
FXR reporter gene assay. 293T cells stably expressing GAL4-luciferase-FXR receptor were maintained in Dulbecco's Modified Eagles Medium (Hyclone) containing 10% fetal bovine serum (Sigma), 1% penicillin/streptomycin (Hyclone), and 200 μg/ml hygromycin (Hyclone). Cells were detached and seeded into 384-well white plates (Greiner Bio-one) at 4,000 cells/50 Hl/well in Dulbecco's Modified Eagles Medium (Hyclone) containing 3% charcoal/dextran treated fetal bovine serum (Sigma) and 1% penicillin/streptomycin. Following 24 hour incubation at 37°C, 50 μl of 1000X compound diluted in DMSO was transferred by Pintool (GNF Systems) into the assay plate and incubated for 24 hours at 37°C. 25 |il Bright Glo (Promega) was added per well and flash luminescence was detected on the Viewlux (PerkinElmer Life Sciences). Dose-response data points were curve fitted using the standard logistic regression model, implemented with an in-house developed Matlab (mathworks. com) program. The resultant IC50 values correspond to the compound concentration that inhibited activity by 50%.
GLUTag GLP-1 secretion assay. Murine GLUTag cells were maintained in Dulbecco's Modified Eagles Medium (Hyclone) containing 10% fetal bovine serum and 1% penicillin/ streptomycin. On day one of the assay, cells from a confluent flask were detached using 0.25% trypsin (Life Technologies) and seeded in 96 well poly-D lysine plates (BD Biosciences) at 42,000 cells /150 μl/well in maintenance medium. After incubation at 37°C overnight, medium was changed to phenol red free DMEM with low glucose (Sigma) and then incubated for another 24hrs at 37 °C. GLUTag cells were then washed and incubated in glucose free EBSS buffer with 0.1% BSA (Sigma) at 37 °C to starve, followed by two additional EBSS buffer washes. Following the last wash, EBSS buffer containing 15 mM glucose, DPP4 inhibitor PKF 275 (600 nM final concentration) and agonist compounds was added to the cells and incubated for another 2hr at 37 °C to allow for GLP-1 secretion. 6 μl of supernatant was then transferred to HEK293 cells stably expressing a GLP-1 luciferase reporter construct, seeded the morning of GLUTag cell treatment at 12,000 cells/40 μl/well in a 384-well plate (Greiner). After overnight incubation, 15 μl of 25% Bright Glo (Promega) was added to the reporter cells, incubated at room temperature for 2 min and luminescence was then read on Envision per the manufacturer's instructions (Perkin Elmer). Data were normalized to a DMSO alone control. Dose-response data points were curve fitted using the standard logistic regression model, implemented with an in-house developed Matlab (mathworks. com) program. The resultant EC50 values correspond to the compound concentration that induced half-maximal activity.
PBMC LPS assay. Peripheral blood mononuclear cells (PBMC) were isolated from healthy donors using Ficoll Hypaque per the manufacturer's recommendation (GE Healthcare). Cells were seeded at 30,000 cells/10 μL/well in a 384-well proxiplate (Perkin Elmer) in AIM V media (Life Technologies) supplemented with 3% FBS (Hyclone) and 1X Pen/Strep/ Glutamine. Following an overnight incubation at 37 °C, 50 nL of compound in DMSO was added per well by Pintool (GNF systems). Plates were incubated for 30 min at 37 °C, followed by the addition of 2μL/well of 6X LPS (EC80 final, determined during LPS dose titration for each donor) and incubation overnight at 37 °C. To detect TNFα secretion by HTRF, 2.5μL/weli anti-TNFα cryptate, then 2.5μL/well of anti-TNFα XL665 were added (Cisbio) and subsequently incubated for 3 hrs at room temperature. The emission at 620 nM and 665 nM were read on the Envision (Perkin Elmer) as suggested by the manufacturer. Data was expressed as a ratio of emission at 665/620, and then normalized to a DMSO alone control. Dose-response data points were curve fitted using the standard logistic regression model, implemented with an in-house developed Matlab (mathworks. com) program. The resultant IC50 values correspond to the compound concentration that inhibited activity by 50%.
Animal studies. All aspects of animal care, use, and welfare for all animals used for in vivo studies were performed in compliance with U.S. Department of Agriculture regulations and the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Furthermore, all animals were handled in accordance with Novartis Animal Care and Use Committee protocols and regulations. For mouse studies, animals were obtained from the Jackson laboratories (C57BL/6) or Taconic (Diet-Induced Obese C57BL/6 mice). For dog studies, male beagles were obtained from Marshall Farms, NY.
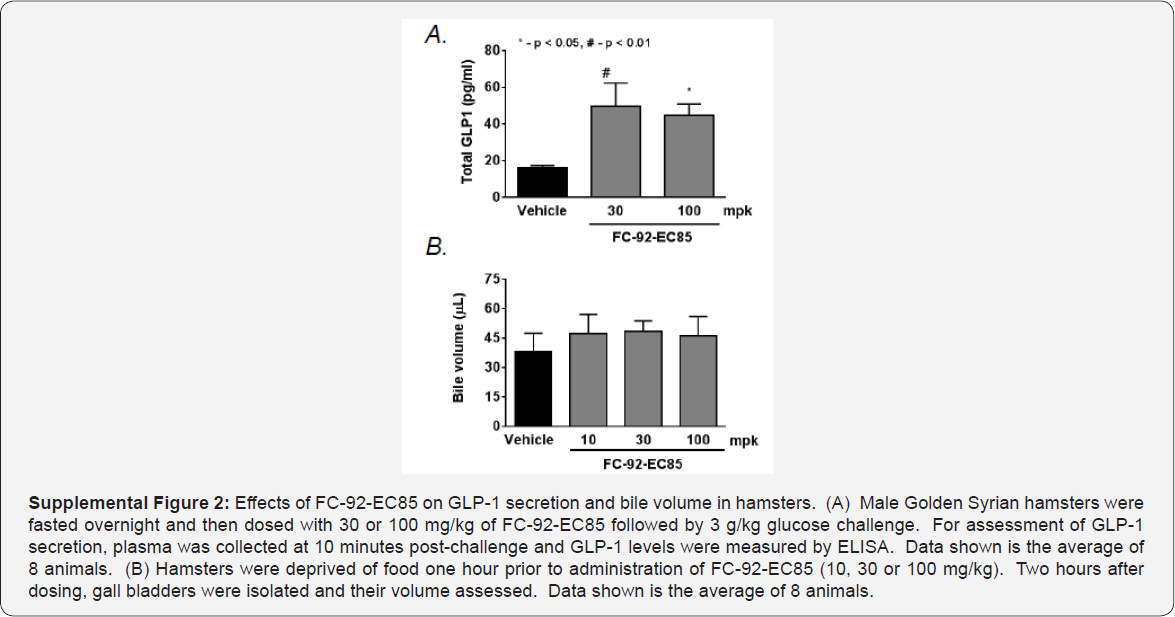
Mouse in vivo GLP-1 secretion assay. Male C57BL/6 mice were fasted overnight and then dosed orally with 10, 30, or 100 mg/ kg of FC-92-EC85. After 30 minutes, animals were challenged with by 3 g/kg oral dextrose (Sigma). For assessment of GLP- 1 secretion, plasma was collected at 2 min. post-challenge into EDTA coated tubes containing 8μL of a DPP4 inhibitor solution (EMD Millipore) and GLP-1 levels were measured by ELISA according to the manufacturer's instructions (Meso Scale Discovery). Data was analyzed using One-way ANOVA followed by Dunnett's post-test in Graphpad Prism. Values of p < 0.05 were considered statistically significant. Data shown is the average +/- SEM of 8 animals.
Mouse in vivo LPS challenge assay. Male C57BL/6 mice were fasted overnight and then dosed with 10, 30, or 100 mg/ kg of FC-92-EC85 one hour prior E. Coli LPS challenge (40 ng/ kg; Sigma). Serum was collected at 90 minutes post-challenge and TNFα or IL-10 levels were measured by ELISA according to the manufacturer's instructions (Meso Scale Discovery). Dexamethasone (Dex) dosed 2 hours prior to LPS at 5 mg/kg was also tested as a control (Sigma). Data was analyzed using Oneway ANOVA followed by Dunnett's post-test in Graphpad Prism. Values of p < 0.05 were considered statistically significant. Data shown is the average +/- SEM of 8 animals.
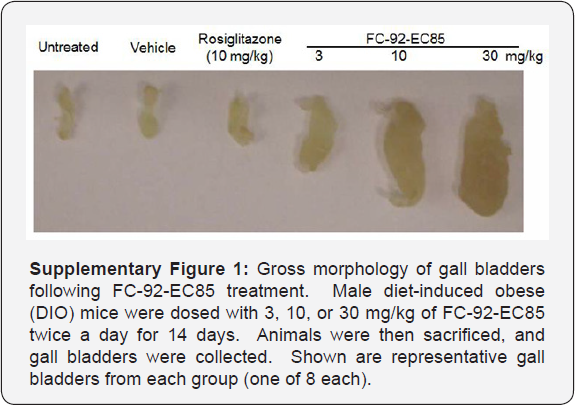
Mouse gall bladder morphology studies. Approximately 16 weeks old male DIO C57BL/6 mice were maintained on high fat diet and received BID oral doses of the TGR5 agonist FC-92- EC85 for 14 days. At the termination of the study gall bladders were saved frozen at -80°C after being separated from the liver. In order to assess the morphology of selected gall bladders, the frozen tissues were transferred into 10% neutral buffered (NB) formalin for 48h and then processed routinely. The gallbladders of two animals per group were serial sectioned and stained with hematoxylin and eosin (HE). Representative sections were evaluated microscopically. In order to further characterize the changes observed on HE, selected sections (representing upper, mid and lower portions of each gall bladder) were stained with Puchtler's picrosirius red, smooth muscle-specific actin (mouse monoclonal, Clone 1A4, Cat.# AM128-5M, Biogenex, Fremont, CA) and Ki67 (rabbit monoclonal, clone SP #6, catalog number VP- RM04, Vector, Burlingame, CA). For the second study comparing diverse chemotypes in lean vs DIO mice, approximately 14 weeks old male DIO or lean C57BL/6 mice (Taconic) were maintained on high fat diet or regular chow (lean mice) and received BID oral doses of TGR5 agonists for 16 days. At the termination of the study, liver and gall bladder were saved in 10% NB formalin for 48h and then processed routinely and stained with HE. In order to further characterize the changes observed on HE, selected gall bladders were stained with Puchtler's picrosirius red, smooth muscle-specific actin, and Ki67.
TGR5 mRNA expression analysis. Tissues samples of gall bladder, spleen and brain for mouse were collected from wild type C57BL/6 mice and immediately snap frozen in liquid nitrogen followed by storage at -80 °C until processing. mRNA was then isolated by Trizol extraction (Life Technologies) followed by purification on an affinity resin (RNeasy, Qiagen) according to the manufacturer's instructions. RNA samples for human and cynomolgus monkey were obtained commercially (Biochain). For human, mouse, and cynomolgus TGR5 analysis, quantitative RT-PCR was performed using the RNA to Ct kit (Life Technologies), run on the ABI Prism 7700 (Applied Biosystems) per the manufacturer’s instructions. Validated primer/probe master mixes were obtained from Life Technologies: human and cynomolgus Hs00544894_m1, mouse Mm00558112_s1. Ct values were normalized to an internal standard (36B4) to obtain a delta Ct, and relative quantity was calculated according to the formula: RQ = 2^- deltaCt. Data was then expressed as the average RQ +/- standard deviation.
Samples of dog gall bladder, spleen and brain (portion of frontal cortex) were obtained from untreated or vehicle-control animals used for internal preclinical toxicity studies at Novartis Pharma AG (Basel, Switzerland). Tissues were immediately snap-frozen in liquid nitrogen and stored at approximately -80°C until processing. Total RNA was obtained by acid guanidinium thiocyanate-phenol-dichloromethane extraction from each frozen tissue section and the total RNA was then purified on an affinity resin (RNeasy, Qiagen) according to the manufacturer's instruction. Total RNA was quantified by the absorbance at 260 nm, and the quality was determined using an Agilent 2100 Bioanalyzer (Agilent Technologies). The GeneChip® microarray experiments were conducted at CiToxLabs (Evreux, France) using Canine Genome 2.0 Arrays (Affymetrix, Inc., Santa Clara, CA, USA). A starting amount of approximately 100 ng of total RNA was used to obtain mRNA using the 3' IVT express kit according to manufacturer's instruction (Affymetrix). The Affymetrix MAS 5.0 software was used to scale the average intensity of each chip to a target intensity of 150. The probe set CfaAffx.22533.1.S1_ at was used to interrogate the expression of TGR5. The target sequence for this probe set maps to the coding region of the GPBAR1 (TGR5) gene at nucleotides 322-900 of reference sequence XM_545641.2.
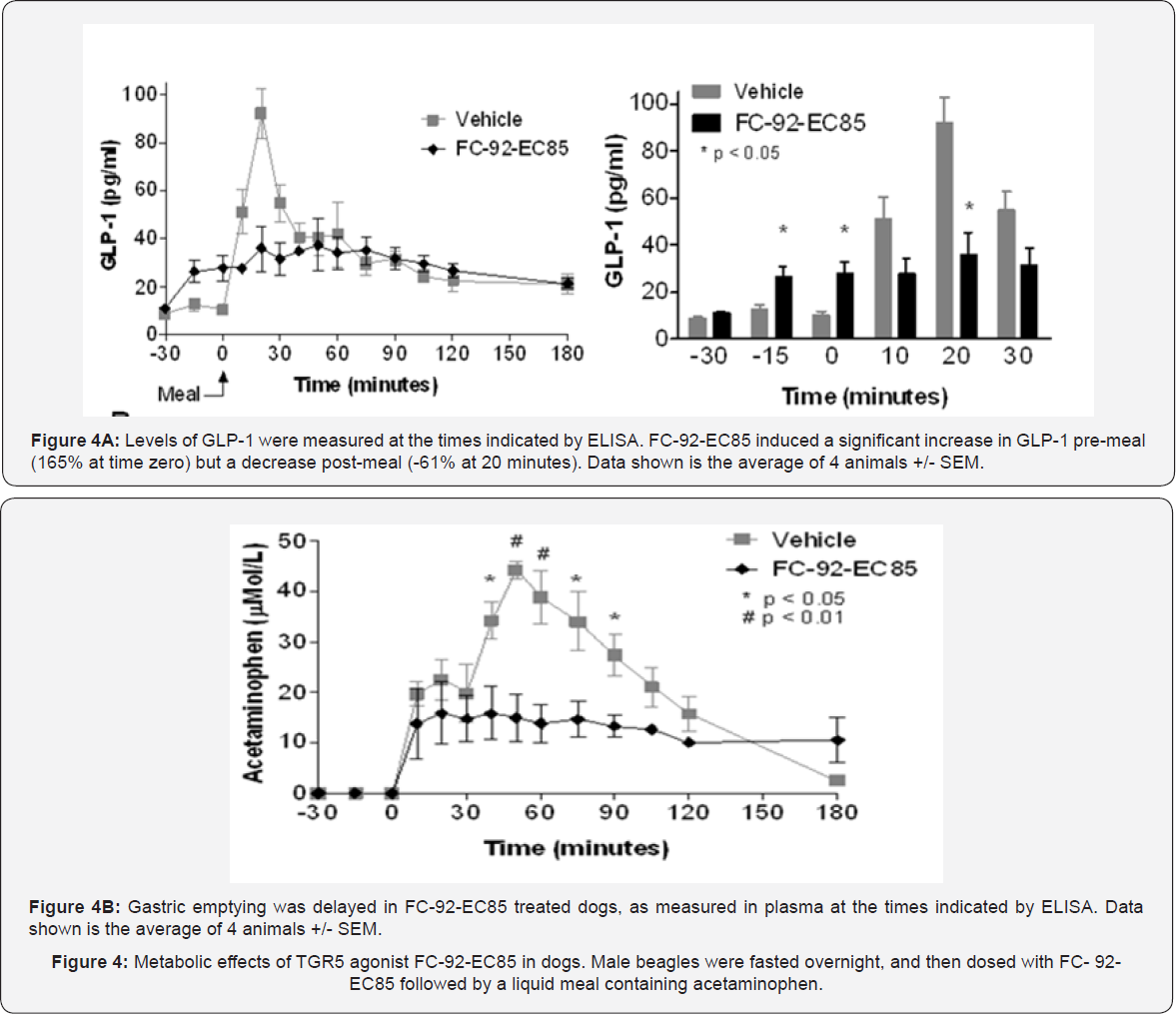
Effect of FC-92-EC85 on metabolic endpoints in dogs. Male Beagles with reproducible postprandial triglyceride and glucose excursions were selected for the study. A cream-glucose-water- saline mixture (= liquid meal) was prepared with heavy cream (2.7 ml/kg, fat 0.9 g/kg), glucose (1 g/kg) and water (5.3 ml/kg). A solution of acetaminophen (20 mg/kg) in saline (2 ml/kg) was added to the mixture. The dogs were fasted (16h) and an initial baseline blood sample was obtained at 30 min before the meal (-30 min). Immediately thereafter the vehicle was administered via gavage and after 15 min a second baseline blood sample was drawn (-15 min). A third baseline blood sample was obtained before the meal (0 min) and the meal (10 mL/kg) was given via gavage. Subsequent blood samples were obtained after 10, 20, 30, 40, 50, 60, 75, 90, 105, 120, 180 and 240 min from the ingestion of the meal. After a washout period, FC-92-EC85 was tested using the same dogs and the same sampling regimen. The compound was administered orally at 50 mg/kg in PEG300 and dH2O in a proportion of 3:1. Blood samples (3 mL) were drawn from the cephalic vein via syringe and transferred into tubes with EDTA, aprotinin (Trasylol, 500 kallikrein inhibitor units/ mL blood) and diprotin A (110 μmol/mL blood). Blood samples were placed on ice until centrifugation at 3,000 rpm for 20 min at 4°C. Plasma samples were divided into aliquots and stored at -70°C. Plasma acetaminophen was analyzed using an enzyme immunoassay procedure (Olympus Emit Tox Acetaminophen Assay, Olympus America Inc., Center Valley, PA) and absorbance was measured in an Olympus au400e spectrophotometer. Plasma GLP-1 was measured by ELISA per the manufacturer's instructions (Meso Scale Discovery). The results were analyzed using one-way repeated measures ANOVA followed by Dunnett’s post-tests. The entire acetaminophen curves (0 to 240 minutes) of the vehicle and the FC-92-EC85 phases were compared to each other using paired T tests. Paired T tests were also used to compare isolated time points. Values of P< 0.05 were considered statistically significant.
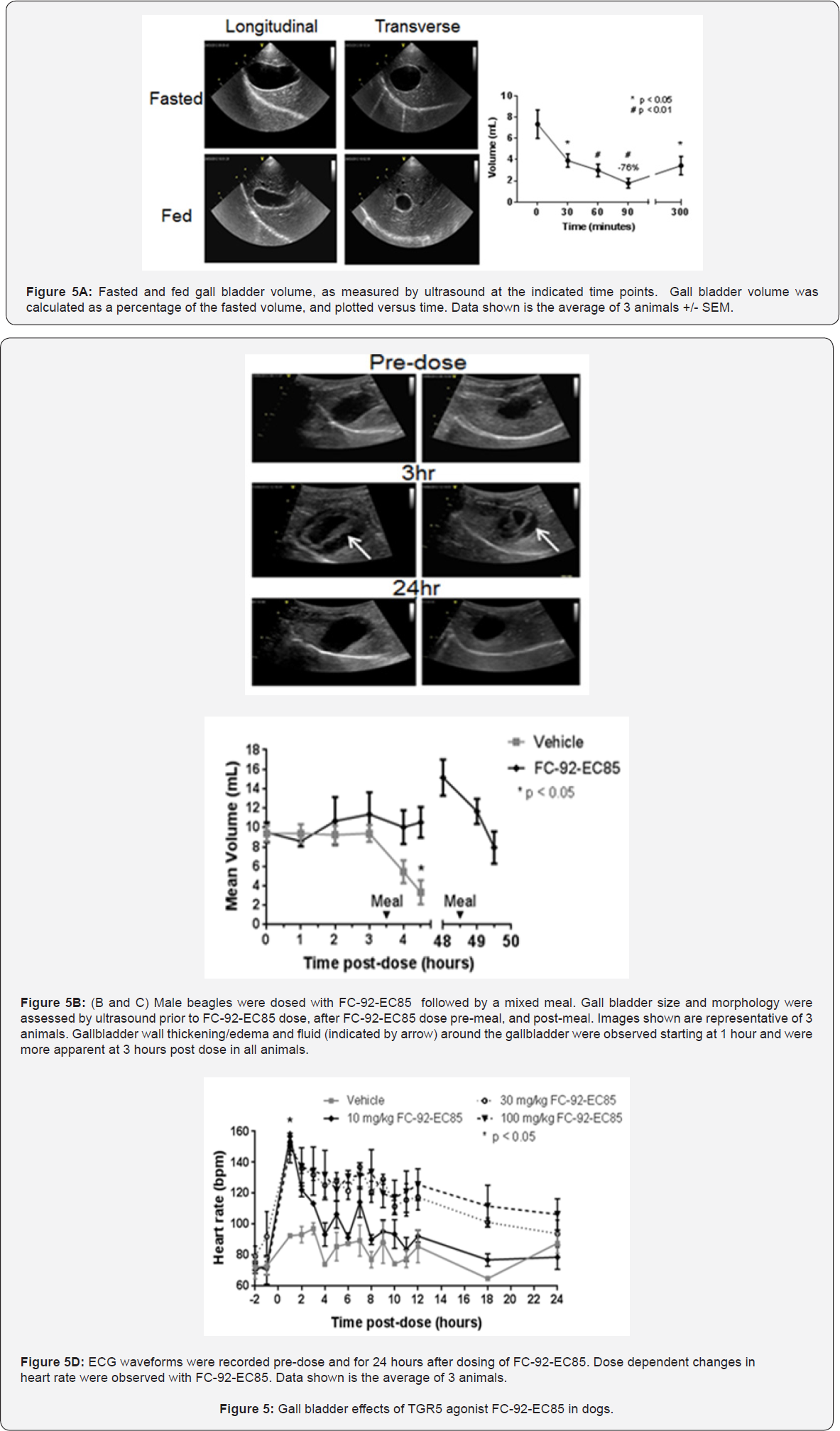
Effect of FC-92-EC85 on gall bladder morphology and function in dogs. Three male beagle dogs (body weight: 8-12 kg) were fasted for approximately 20h prior to the study. Both longitudinal and transverse section gall bladder images were then collected at fasting (baseline), and at 30, 60, 90 min and 5h after meal challenge (11 oz. of Hill's prescription diet - a/d Canine/Feline). For imaging, dogs were shaved locally and placed supine on a table specifically designated for ultrasound examination with manual restraint. Images were acquired and stored using the GE Vivid E9 ultrasound system (GE Medical Systems, Milwaukee, WI) using a 12S-DLP phased array sector transducer. For evaluation of FC-92-EC85, compound was administered to three beagle dogs (9-10 kg) at 0 and 30 mg/kg on study days 1 and 6, respectively, after approximately 19h of fasting. Ultrasound examinations were conducted predose and approximately 1, 2 and 3h after dosing. A meal challenge (i.e., approximately 11 oz. of Hill’s prescription diet - a/d Canine/Feline) was then administered and additional ultrasound examinations were conducted approximately 30 and 60 min after challenge. To evaluate recovery, an additional ultrasound examination was conducted on study day 7 approximately 24h post-dose. Animals were then fasted again for approximately 21.5h beginning on study day 7 followed by another meal challenge on study day 8. Ultrasound examinations were conducted prior to and approximately 30 and 60 min after administration of the meal challenge on study day 8 (approximately 48h after administration of FC-92-EC85). All images were analyzed offline using the GE Echo-Pac workstation (GE Medical Systems, Milwaukee, WI). The maximum length (L) and depth (DL) were measured from longitudinal section. The maximum width (W) and depth (DT) were measured from transverse section. Gallbladder volume was calculated based on Hackenberg's formula [19].
Effect of FC-92-EC85 on heart rate and QT interval in dogs. Three male beagle dogs (body weight 9.5 to 14.7 kilograms) were dosed with vehicle on study day 1. A single dose of 10, 30, 100 or 300 mg/kg FC-92-EC85 in rising dose fashion was given on separate days with at least 5 days between doses. ECG waveforms were recorded using non-invasive jacket telemetry for 2h prior to dose and 24h post dose at pretest and each dose. Animals were acclimated to telemetry jacket procedures at least three times on separate days. ECG waveforms were recorded using the emka ECG system when animals were in their home cage. For heart rate and ECG intervals, the third 15-min period of every hour were reported for after each treatment to minimize the influence of TK blood sampling procedures on animals and for pretest. The heart rate and ECG intervals (RR, PR, QRS and QT intervals) were analyzed by emka ECG-Auto computer software. QT interval was corrected for heart rate using Van de Water QT correction formula QTc or QTcVW=QT-0.087 (RR-1000).
Results
FC-92-EC85 is a TGR5 agonist suitable for proof-of-concept studies. We began a campaign to develop oral low molecular weight agonists of TGR5. While a diverse array of chemotypes was identified from high throughput screening, several key challenges became evident during initial medicinal chemistry. Human and mouse TGR5 are highly homologous at the protein level (83%), yet the majority of compounds showed preferential activation at the human receptor. This type of profile makes pre- clinical efficacy and toxicity studies challenging, so the effort focused on chemotypes with some degree of cross-reactivity. Lead optimization led to the identification of FC-92-EC85, a potent and selective TGR5 agonist with no activity on the nuclear bile acid receptor FXR [18] (Figure 1A). FC-92-EC85 increased glucose dependent GLP-1 secretion both in vitro, as measured in GLUTag cells, and in vivo following glucose bolus (Figures 1B &1C). In addition, FC-92-EC85 was able to decrease LPS-induced TNF-alpha in human PBMC and in vivo in mice (Figures 1D &1E). In pharmacokinetic studies in mice, oral dosing resulted in moderate exposure and short half-life due to rapid clearance (Table 1). Because of its acute efficacy and adequate mouse exposure, FC-92-EC85 was deemed a suitable tool compound for pre-clinical efficacy studies.
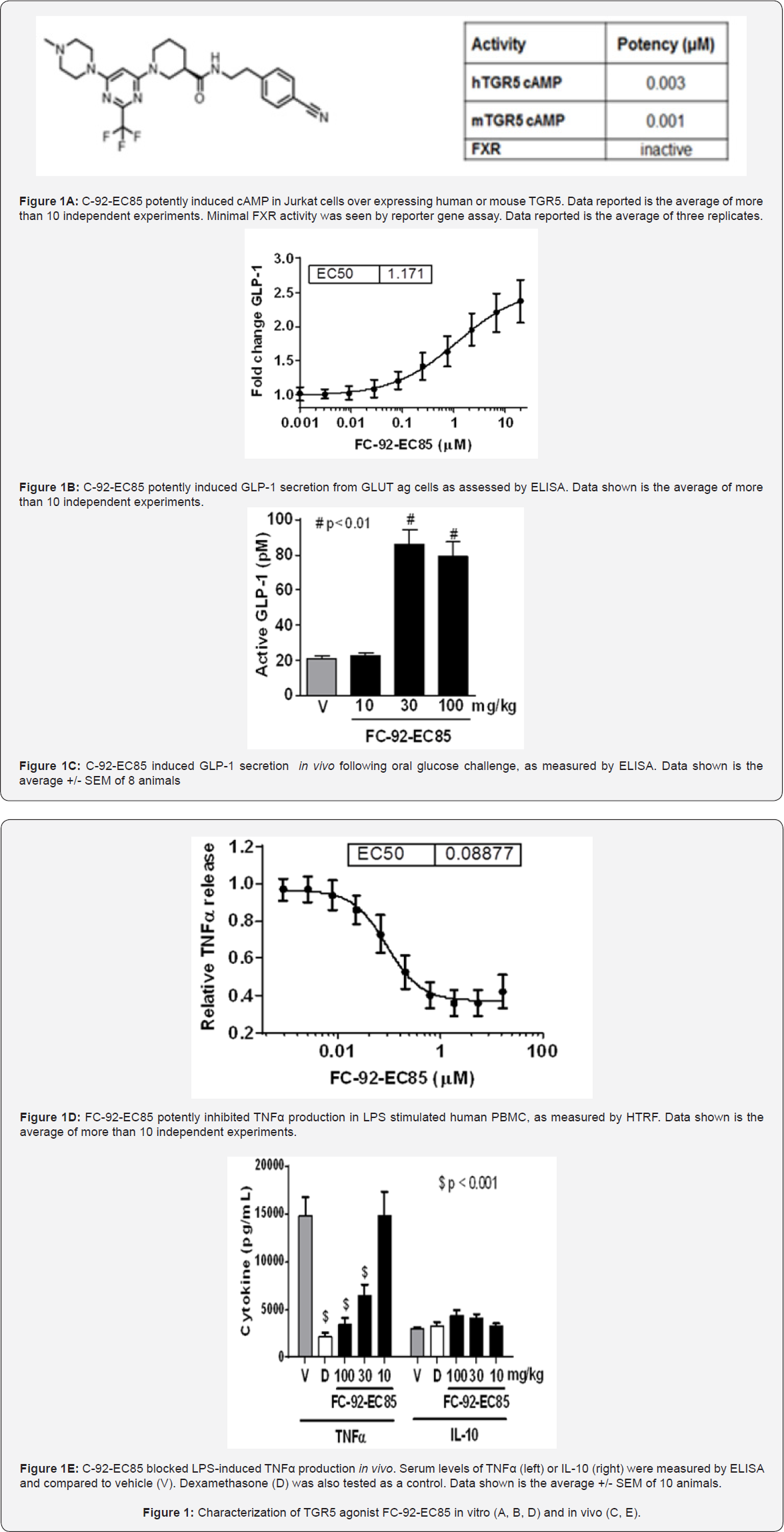
TGR5 agonists induce changes in gall bladders of obese mice. As shown for other TGR5 agonists in the literature [20], oral dosing of FC-92-EC85 led to a 2-3 fold increase in bile volume (Figure 2). This finding was less of a concern since bile volume changes 3-4 fold during regular periods of fasting and refeeding in humans [21] and such a change can be easily tracked by ultrasound. However, following 14 days of dosing in diet- induced obese (DIO) mice, animals dosed with FC-92-EC85 had distended, yellow, thickened gallbladders (Supplemental Figure 1). In control animals, gall bladders were small, their walls were relatively simple with occasional folds and their epithelial lining was uniformly low columnar, and there was no evidence of a cellular infiltrate in the lamina propria.
By contrast, gall bladders from animals in the high dose group had a thicker wall than controls, were more complex, and contained multiple in-folding/villus-like structures and occasional inflammatory cells in the lamina propria. Epithelial cells appeared to be tall columnar and nuclei were somewhat crowded; generally the elongated oval nuclei were oriented along their long axis. The morphology of gall bladders from the low dose animals was intermediate between controls and highdose (Figure 2B). Analysis of actin levels via immunostaining and fiber type using Sirius red revealed an increased fibro- muscular supportive tissue in animals that were dosed with FC- 92-EC85. Additionally, an increased immune-reactivity to Ki67 was recognized at the base of the mucosal folds even without the benefit of image analysis (Figure 2C) [22]. Other TGR5 agonists representing different chemotypes had similar, albeit less severe changes (Figure 2C) .
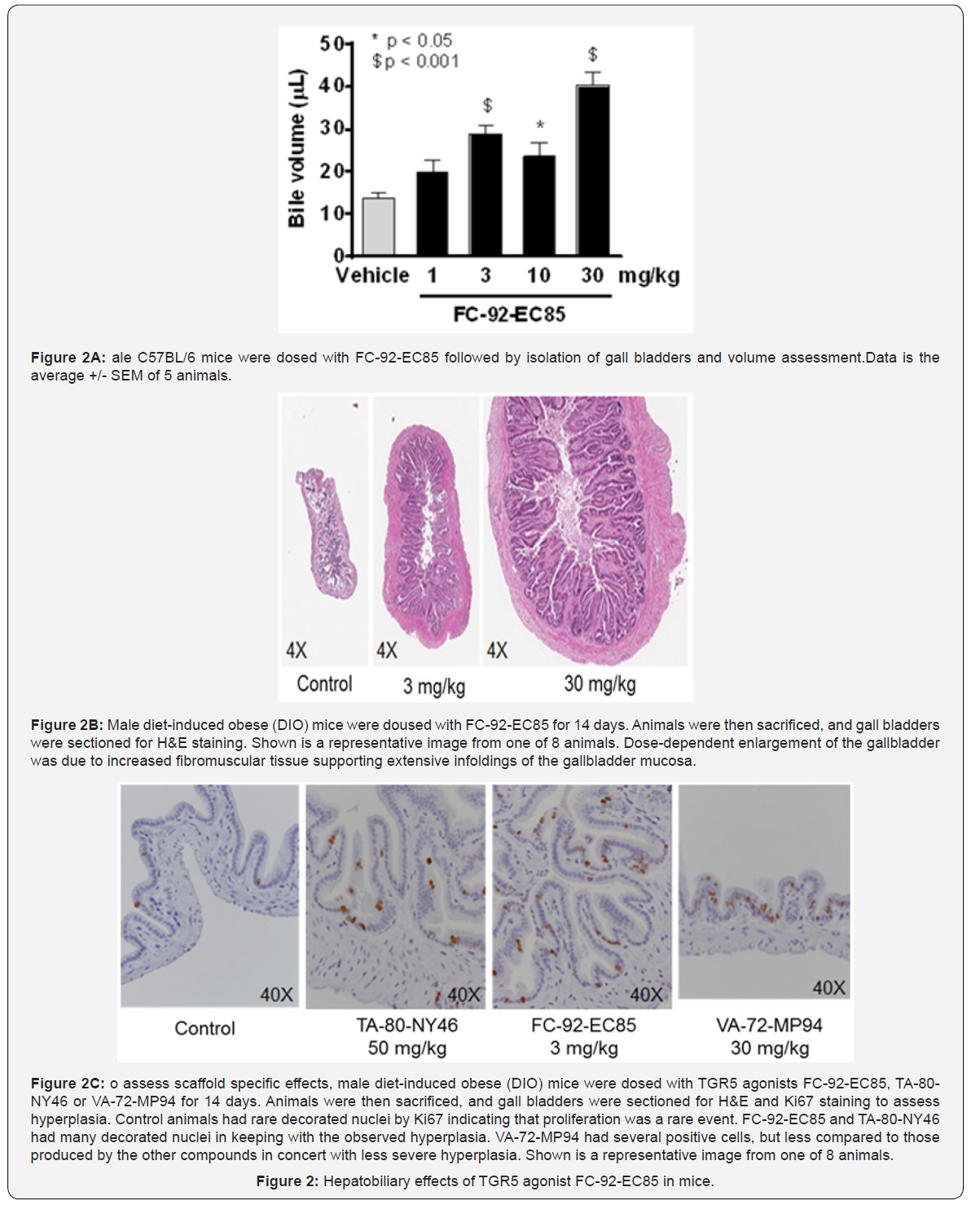
Expression of TGR5 in the mouse gall bladder dwarfs that of in gall bladder morphology with FC-92-EC85 were observed. other tissues [10], which may explain why such marked changes Interestingly, the mRNA levels in gall bladder of other non-rodent species are more comparable to the spleen, suggesting there may be species differences in response (Figure 3). Hamsters dosed with FC-92-EC85 showed expected increases in GLP-1 but without significant bile volume increases, further supporting the idea of species differences (Supplemental Figure 2). However, TGR5 has been detected in human gall bladder epithelium [11]. Because it was unclear whether the mouse findings would translate to higher species, we decided to test FC-92-EC85 in dogs, looking for acute effects both on metabolic endpoints and on gall bladder morphology and contractility.
TGR5 agonist FC-92-EC85 decreases gastric motility in dogs. Pharmacokinetic profiling of FC-92-EC85 in dogs showed adequate oral exposure for acute studies (data not shown). Oral administration of FC-92-EC85 led to an increase in prechallenge basal GLP-1 levels (165% prior to meal challenge), but greatly diminished GLP-1 levels in response to mixed meal challenge (61% decrease at 20 minutes post-meal; (Figure 4A).Acetaminophen was added to the meal challenge and tracked over time to determine if changes in gastric motility were responsible for this altered profile. Analysis of plasma acetaminophen levels showed significant delays in absorption following FC-92-EC85 administration (Figure 4B). These findings are consistent with recent data showing decrease in gastric smooth muscle contractility with TGR5 agonists [23] (Figure 4).
TGR5 agonist FC-92-EC85 impedes gall bladder contractility in dogs. In order to evaluate hepatobiliary effects in dogs, a method using ultrasound to measure gall bladder volume and morphology was developed. During fasting, the gall bladder stores bile in preparation for the next meal. When a meal is ingested, the gall bladder contracts to release the stored bile into the duodenum, thus solubilizing nutrients for absorption. To validate the canine ultrasound methodology, gall bladder volume was measured following overnight fasting, where the volume should be largest, and then again following a liquid meal challenge, where gall bladder contraction should decrease overall volume. Normal changes in gall bladder size were demonstrated by ultrasound during fasting and re-feeding, with decreasing volume in response to food challenge over a one hour time period (Figure 5a). Having validated the methodology, we next determined the effects of FC-92-EC85 on this process. Single administration of FC-92-EC85 at 30 mg/kg produced gall bladder wall thickening/edema and fluid around the gallbladder starting at 1h post-dose and were more apparent at 3h post dose in all animals (Figure 5B). The wall edema and surrounding fluid disappeared in all animals by 24h after administration of FC-92- EC85, suggesting a quick recovery. FC-92-EC85 also produced a marked decrease in meal induced gallbladder emptying suggesting impaired contractile function of the gall bladder (Figure 5C).
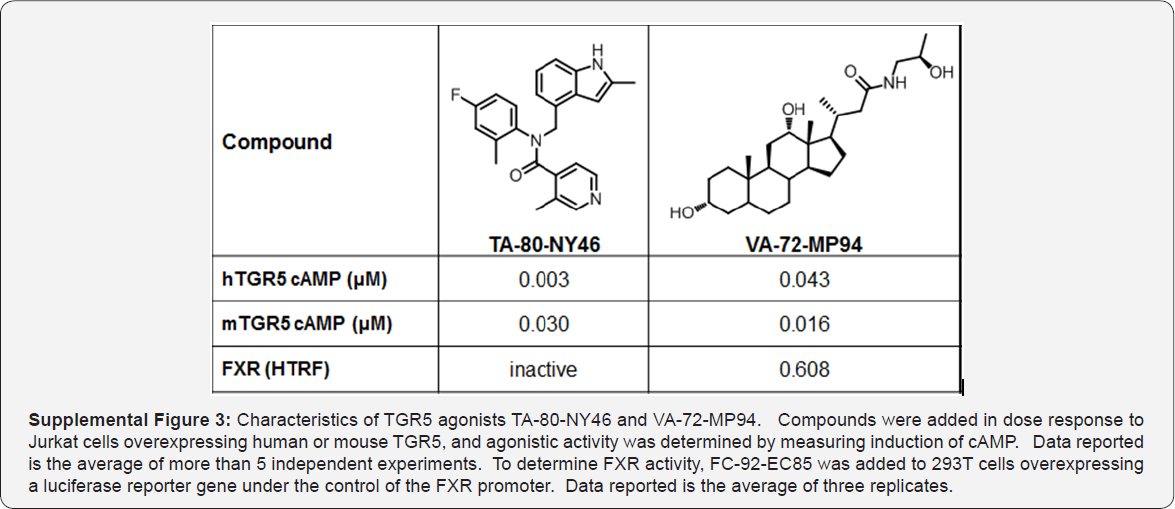
In addition, FC-92-EC85 was associated with apparent enlargement of the gallbladder at approximately 48h post dose, compatible with an increase in overall bile volume. Meal induced gall bladder emptying was partially recovered at 48 hours post dose. Perhaps more troubling, dose dependent increases in heart rate were also noted with FC-92-EC85, even at the lowest dose tested (Figure 5D). Together, these effects may limit the utility of systemic TGR5 agonists for metabolic disease (Supplemental Figure 3).
Discussion
There has been considerable interest in the potential of TGR5 as a drug target for type 2 diabetes. Activation of TGR5 in gastrointestinal enteroendocrine cells leads to increased cAMP and subsequent enhanced GLP-1 secretion, which is a clinically validated approach to control glucose levels. These effects can be accomplished without the inconvenience of protein injections and induces levels higher than those seen with DPP4 inhibitors. However, because of a wealth of treatments already available to decrease glucose levels, agents entering the clinic must demonstrate either superior chronic efficacy or bring additional benefits beyond glucose control. In the case of TGR5, its expression and anti-inflammatory activity in myeloid cells suggests it could also address the chronic inflammation characteristic of obesity and diabetes, and thus provide a benefit versus glucose lowering agents alone. This promising profile has led to the pursuit of systemic TGR5 agonists by several pharmaceutical companies.
Shortly after the role of TGR5 in GLP-1 secretion was published, there were reports that suggested that systemic TGR5 activation could have negative consequences. Tissue expression analysis shows widespread expression of the receptor in tissues beyond the GI tract and myeloid compartments, most notably in gall bladder, the heart, and the brain [10]. It has also been demonstrated that administration of bile acids leads to increases in bile volume that is not solely mediated by FXR [24], suggesting that the gall bladder expression may serve a physiological function at steady state. More recently, several authors have described that TGR5 agonists can mediate smooth muscle relaxation, both in the gall bladder and the GI tract [12,23]. Since TGR5 is a Gs-coupled receptor that increases cellular cAMP, this fits with studies showing that general elevations in cAMP lead to smooth muscle relaxation [25,26]. Dilation of gall bladder has been shown to cause mucosal hyperplasia [27]. Consistent with these findings, we found profound effects of chronic TGR5 activation on the mouse gall bladder, including hyperplasia and increased wall thickness. These effects were seen with multiple scaffolds, suggesting this may be a general consequence of TGR5 activation. However, they were more pronounced in mice on high fat diet compared to wild type mice.
Together with the lack of bile volume effect in hamsters, we questioned whether the gall bladder effects were species specific. Despite differences in absolute quantity of TGR5 mRNA in the gall bladder, these deleterious effects could also be witnessed acutely in dogs by ultrasound and were accompanied by defects in contractility. Decreased contractility was also noted in the gastrointestinal tract, where delayed gastric emptying was seen following FC-92-EC85 administration.
The gall bladder findings shed considerable doubt on the utility of systemic TGR5 agonists in type 2 diabetes, a population that already exhibits an increase in gall bladder dysfunction. Moreover, dogs treated with FC-92-EC85 showed elevated heart rate and QT prolongation. While a compound specific effect is conceivable due to the in vitro hERG inhibition, Piotrowski et al. has recently reported similar effects on heart rate and blood pressure with two TGR5 agonists from different compound classes and concluded that the observations were an on-target effect [28]. More recently, Fryer et al. reported that TGR5 activation shows a strong and pronounced effect on arterial vasodilation in a Kca1.1 dependent manner [29]. The cardiovascular effects observed with FC-92-EC85 in our studies are consistent with these reports and demonstrate that potent, bioavailable agonists will likely have low therapeutic indices.
To address the issues with systemic agonists, several small companies are pursuing GI-restricted agonists of TGR5] [14-17]. These would hopefully activate GLP-1 secretion in the intestine without significant systemic exposure. This concept faces several challenges. First, TGR5 is enriched in specific segments of the intestine, including the jejunum, ileum, and colon. Ensuring that enough compounds is delivered selectively and retained long enough in this location represents a significant challenge. Second, we have witnessed effects on bile volume at compound exposures below what is required for GLP-1 secretion. This suggests that a gastrointestinal-restricted compound may need to avoid all absorption. Since it is unclear whether TGR5 resides most in the luminal or basal membranes of the enteroendocrine cells, it remains to be seen whether strict luminal exposure will be efficacious. Third, regardless of where it is delivered, chronic delivery of TGR5 agonists could lead to loss of receptor expression from the cell surface and subsequent loss of activity, also known as tachyphylaxis. Indeed, studies with our more optimized candidates have demonstrated a loss of activity during chronic treatment [30,31]. Thus, even a gastrointestinal- restricted agonist will need to be carefully selected to avoid desensitization. Finally, focusing only on the GLP-1 secretion aspect of TGR5 agonism reduces it to being just another glucose lowering agent, a profile that does not meet the current patient needs. Coupled with the severity of the on-target side effects seen with systemic exposure, these challenges suggest that systemic TGR5 agonists may never realize their clinical potential.
References
- Krebs MG, Hou JM, Ward TH, Blackhall FH, Dive C (2010) Circulating tumour cells: their utility in cancer management and predicting outcomes. Therapeutic advances in medical oncology 2(6): 351-365.
- Senovilla L, Aranda F, Galluzzi L, Kroemer G (2014) Impact of myeloid cells on the efficacy of anticancer chemotherapy. Current opinion in immunology 30: 24-31.
- Keizman D, Gottfried M, Ish-Shalom M, Maimon N, Peer A, et al. (2012) Pretreatment neutrophil-to-lymphocyte ratio in metastatic castration- resistant prostate cancer patients treated with ketoconazole: association with outcome and predictive nomogram. The oncologist 17(12): 1508-1514.
- Templeton AJ, Pezaro C, Omlin A, McNamara MG, Leibowitz-Amit R, et al. (2014) Simple prognostic score for metastatic castration-resistant prostate cancer with incorporation of neutrophil-to-lymphocyte ratio. Cancer 120(21): 3346-3352.
- Cao J, Zhu X, Zhao X, Li XF, Xu R (2016) Neutrophil-to-Lymphocyte Ratio Predicts PSA Response and Prognosis in Prostate Cancer: A Systematic Review and Meta-Analysis. PloS one 11(7):e0158770.
- (2017) American Cancer Society. Hormone Therapy for Prostate Cancer.
- Parsons JK, Carter HB, Platz EA, Wright EJ, Landis P, et al. (2005) Serum testosterone and the risk of prostate cancer: potential implications for testosterone therapy. Cancer epidemiology, biomarkers & prevention 14(9): 2257-2260.
- Morgentaler A (2006) Testosterone and prostate cancer: an historical perspective on a modern myth. European urology 50(5): 935-939.?
- Stattin P, Lumme S, Tenkanen L, Alfthan H, Jellum E, et al. (2004) High levels of circulating testosterone are not associated with increased prostate cancer risk: a pooled prospective study. International journal of cancer 108(3): 418-424.
- Schatzl G, Madersbacher S, Thurridl T, Waldmuller J, Kramer G, et al. (2001) High-grade prostate cancer is associated with low serum testosterone levels. The Prostate 47(1): 52-58.
- Antonarakis ES, Lu C, Wang H, Luber B, Nakazawa M, et al. (2014) AR- V7 and resistance to enzalutamide and abiraterone in prostate cancer. The New England journal of medicine 371(11): 1028-1038.
- Antonarakis ES, Lu C, Luber B, Wang H, Chen Y, et al. (2015) Androgen Receptor Splice Variant 7 and Efficacy of Taxane Chemotherapy in Patients With Metastatic Castration-Resistant Prostate Cancer. JAMA oncology 1(5): 582-591.
- Bubendorf L, Schopfer A, Wagner U, Sauter G, Moch H, et al. (2000) Metastatic patterns of prostate cancer: an autopsy study of 1,589 patients. Human pathology 31(5): 578-583.
- Fohr B, Dunstan CR, Seibel MJ (2003) Clinical review 165: Markers of bone remodeling in metastatic bone disease. The Journal of clinical endocrinology and metabolism 88(11): 5059-5075.
- Coleman RE (2001) Metastatic bone disease: clinical features, pathophysiology and treatment strategies. Cancer treatment reviews 27(3): 165-176.
- Saad F, Lipton A, Cook R, Chen YM, Smith M, et al. Pathologic fractures correlate with reduced survival in patients with malignant bonedisease. Cancer 110(8): 1860-1867.
- Brown JE, Sim S (2010) Evolving role of bone biomarkers in castration- resistant prostate cancer. Neoplasia 12(9): 685-696.
- Lipton A, Fizazi K, Stopeck AT, Henry DH, Brown JE, et al. (2012) Superiority of denosumab to zoledronic acid for prevention of skeletal- related events: a combined analysis of 3 pivotal, randomised, phase 3 trials. European journal of cancer 48(16): 3082-3092.
- Nilsson S, Franzen L, Parker C, Tyrrell C, Blom R, et al. (2007) Bone- targeted radium-223 in symptomatic, hormone-refractory prostate cancer: a randomised, multicentre, placebo-controlled phase II study. The Lancet Oncology 8(7): 587-594.
- Rao SR, Snaith AE, Marino D, Cheng X, Lwin ST, et al. (2017) Tumour- derived alkaline phosphatase regulates tumour growth, epithelial plasticity and disease-free survival in metastatic prostate cancer. British journal of cancer 116(2): 227-236.
- Hammerich KH, Donahue TF, Rosner IL, Cullen J, Kuo HC, et al. (2017) Alkaline phosphatase velocity predicts overall survival and bone metastasis in patients with castration-resistant prostate cancer. Urologic oncology 35(7):460e21- 460e28.
- Ness N, Andersen S, Khanehkenari MR, Nordbakken CV, Valkov A, et al. (2017) The prognostic role of immune checkpoint markers programmed cell death protein 1 (PD-1) and programmed death ligand 1(PD-L1) in a large, multicenter prostate cancer cohort. Oncotarget 8(16): 26789-26801.






























