A Systematic Review of the Beta-Blockers Carvedilol and Metoprolol for the Treatment of Chronic Heart Failure
Leo Buckley, Susie Choi, Kevin Padilla and Benjamin W. Van Tassell*
Department of Pharmacotherapy and Outcomes Science, Virginia Commonwealth University, USA
Submission: June 02, 2017; Published: June 27, 2017
*Corresponding author: Benjamin W. Van Tassell, Department of Pharmacotherapy and Outcomes Science Virginia Common wealth University, USA, Fax: (804] 28-0343; Email: bvantassell@vcu.edu
How to cite this article: Leo B, Susie C, Kevin P , Benjamin W. Van T. A Systematic Review of the Beta-Blockers Carvedilol and Metoprolol for the Treatment of Chronic Heart Failure. J of Pharmacol & Clin Res. 2017; 3(1): 555605. DOI: 10.19080/JPCR.2017.03.555605
Abstract
Due to various differences between the beta-blockers approved for the treatment of chronic heart failure with reduced ejection fraction (HFrEF), particularly carvedilol and metoprolol, the efficacy and safety of each agent relative to the other remains unresolved. In this review, we discuss the differences between carvedilol and metoprolol with respect to clinical endpoints and the differential effects of these agents in chronic HFrEF. We conducted a systematic review of phase 2, phase 3 and observational studies which compared metoprolol and carvedilol in patients with chronic HFrEF. Carvedilol exhibits more favorable effects on cardiac structure and function, hemodynamic, exercise capacity and metabolism compared to metoprolol. In the controversial COMET study, carvedilol reduced mortality to a greater extent than metoprolol. We suggest carvedilol as the preferred beta-blocker for the treatment of HFrEF in the majority of patients.
Introduction
Heart failure (HF) is one of the most deadly and costly diseases worldwide. In the United States alone, HF contributes to 1 in every 9 deaths and consumes over $24 billion in healthcare resources. Current estimates project nearly a doubling in the prevalence of HF and almost a tripling in costs between the years 2012 and 2030 [1,2]. While the development of novel therapeutics remains an important focus of research, the refinement of currently available therapies presents an immediate opportunity to achieve improvements in patient care.
After the discovery of adrenergic receptors by Raymond Ahlquist in 1948, the role of beta-adrenergic receptors in HF was defined over the following 40 years [3]. Seminal discoveries include the predominance of the beta-1 subtype of adrenergic receptors in the heart, cardio toxicity of circulating catecholamine and, most important, down regulation of beta receptors in the failing heart [4]. Landmark trials soon would confirm the clinical benefits of beta-receptor blockade in HF.
The beta-blockers carvediloland metoprolol have received Class IA recommendations for reducing morbidity and mortality in patients with chronic HF and a reduced ejection fraction(HFrEF) in both American and European guidelines [5,6]. While each of these agents has demonstrated reductions in all-cause mortality, whether one agent provides superior additional benefit remains debated. We sought to systematically review the comparative literature of metoprolol and carvedilol to determine the relative effects of each agent on cardiac structure and function, hemodynamic, exercise capacity and metabolism in HFrEF.
Methods
We conducted a literature search of the National Library of Medicine via PubMed with the following Medical Subject Headings (MeSH): ("adrenergic beta-antagonists"[MeSH] OR "carvedilol" OR "metoprolol") AND "heart failure"[MeSH]. We included English-language studies that randomized HFrEF patients to receive either metoprolol tartrate/succinate or carvedilol. In addition, two observational studies and two metaanalyses comparing metoprolol succinate and carvedilol were included due to their recent publication dates. Last, we reviewed the references of selected articles. The most recent search was conducted in March 2017. Data were extracted independently by the authors, with a focus on endpoints related to cardiac structure and function, hemodynamic, exercise capacity and metabolism.
Results
Beta-receptor Signaling in the Normal Heart
Increased cardiac adrenergic activity in response to decreased cardiac output leads to not only direct cardio toxicity but also beta-receptor down regulation [4,7,8]. Thus, beta-receptor antagonism re-sensitizes the myocardium to adrenergic signaling to improve myocardial reserve as well as protects against catecholamine overload.
Beta-1 receptors are the predominant adrenergic receptor in the heart, although beta-2 receptors represent 20-30% of the overall myocardial beta receptors [9]. Beta-2 receptors are also expressed in the lung, kidney and blood vessels. Beta-3 receptors, found primarily in the periphery where they mediate NO production, are also expressed in the heart [10]. The role of beta-3 receptors in HF and the effects of carvedilol and metoprololon these receptors remain under investigation [11].
Beta-adrenergic receptors are G-protein-coupled receptors which couple to and induce phosphorylation of an intracellular G-protein complex to initiate intracellular signaling (Figure 1). In the heart, norepinephrine and epinephrine are the primary beta-adrenergic receptor ligands, but intracellular signaling differs based upon the receptor (beta-1 vs. beta-2) and not the ligand.
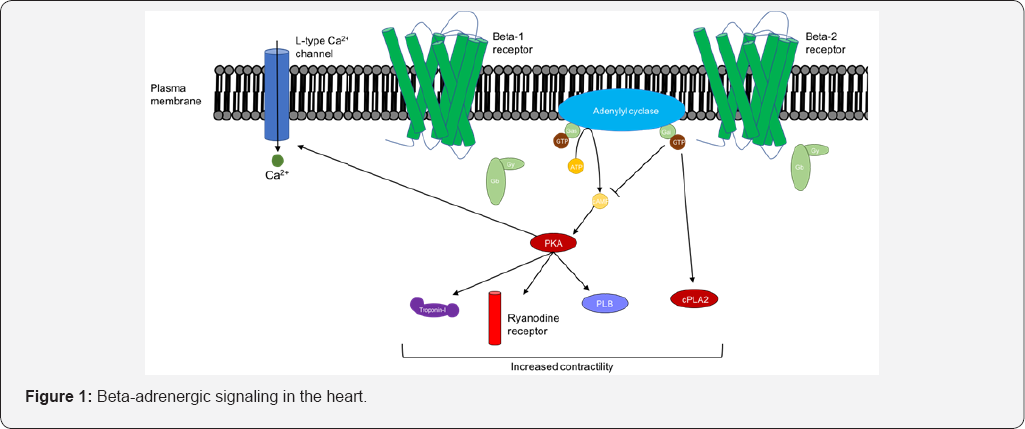
Ligand binding to the cardiac beta-1 adrenergic receptor leads to G-stimulatory (Gs) protein binding and phosphorylation of adenylyl cyclase, an enzyme which converts adenosine triphosphate to the second messenger cyclic adenosine monophosphate (cAMP). The extracellular message is then transferred from cAMP to protein kinase A (PKA), which phosphorylates troponin I, phospholamban (a regulator of the sarcoplasmic/endoplasmic reticulum Ca2+-ATPase [SERCA2a]) and the L-type calcium channel.Cardiac beta-adrenergic receptor Ligand binding also directly activates the L-type calcium channel to increase intracellular calcium levels. Phosphorylation of troponin I, phospholamban and the L-type calcium channel each increase cytoplasmic calcium concentrations and increase contractility.
Beta-2 receptor activation signals through both Gs protein and G-inhibitory (Gi) proteins. Gi protein activation blocks the activity of adenylyl cyclase but also activates phospholipase A2, which mediates calcium signaling and contractility independent of cAMP.The beta-2 receptor pathway serves as an alternative to a deficient beta-1 receptor pathway and its relative importance may be increased in chronic HF.
A negative feedback loop, characteristic of all G-protein- coupled receptors, is utilized to prevent excess beta-adrenergic activity. Desensitization is accomplished through inactivation of G-proteins or blocking G-protein coupling to the beta-adrenergic receptor, both of which interfere with intracellular signaling, and increased beta-receptor degradation, which decreases receptor density.
Beta-receptor Signaling in the Failing Heart
Abnormalities in beta-receptor signaling in the failing heart exist at several levels [4]. Increased beta-2/beta-1 receptor density, increased inhibitory G-protein activity and increased blockade of beta-adrenergic receptor-G-protein coupling each contribute to impaired myocardial function. In addition, circulating catecholamine levels are significantly increased andare capable of direct myocardial toxicity.
Pharmacology of Carvedilol and Metoprolol
Both carvedilol and metoprolol block the effects of catecholamines at the beta-adrenergic receptor in addition to exerting some activity as inverse agonists [12]. Metoprolol more selective lyinhibits the beta-1 receptor (particularly at lower doses), whereas carvedilol acts non-selectively on both the beta-1 and beta-2 receptors [13]. Notably, however, the beta-1 receptor selectivity of metoprolol is lost as doses approach 200 mg [14]. In addition to these beta-receptor effects, carvedilol also block the alpha-1 adrenergic receptor, providing vasodilation in addition to beta-blockade.
Contemporary descriptions of HF include a paradigm of beta- receptor desensitization—presumably due to norepinephrine overstimulation [7,8] and both metoprolol and carvedilol show evidence of restoring beta-receptor responsiveness over time [15-19]. Moreover, carvedilol may also improve responsiveness of peripheral alpha-1 receptors in HF patients [20]. Carvedilol's anti-adrenergic effects may be attributed in part to blockade of pre-junctional beta-2 receptors, which decreases norepinephrine levels [4].
Through more comprehensive beta-recepter blockade, carvedilol may produce greater reductions in sympathetic activity than metoprolol [9]. On the other hand, metoprolol may improve beta-1 receptor up regulation to a greater extent than carvedilol. Moreover, the effects of either beta-blocker on beta-3 receptors remain incompletely understood, although metoprolol appears to increase beta-3 receptor expression and carvedilol to decrease beta-3 receptor expression [10]. A recent clinical trial of a beta-3 receptor agonist, mirabegron, did not meet its primary endpoint [21].
By antagonizing the effects of catecholamines on myocardial adrenergic receptors, carvedilol and metoprolol reduce heart rate and inotropy. In addition, beta blockers may reduce kidney juxtaglomerular production of renin and therefore reduce systemic vasoconstriction [22].
Comparative Pharmacodynamics Effects of Carvedilol and Metoprolol
Cardiac function and structure
Carvedilol has shown more beneficial effects on resting cardiac function and structure than metoprolol in human clinical trials. Although left ventricular EF (LVEF) is improved by both agents, [23] multiple investigations have reported that carvedilol achieves a greater LVEF improvement than metoprolol tartrate [24-26]. Moreover, carvedilol appears to induce LV reverse remodeling to a greater extent than metoprolol tartrate as measured by LV end-diastolic and end-systolic diameters and volumes [24,27,28].
Hemodynamic Effects
Carvedilol and metoprolol tartrate appear to exhibit similar effects on heart rate (HR) reduction as both agents may be titrated to achieve a target HR [23,25]. Notably 24-hour HR monitoring did not find any differences in HR variability between metoprolol tartrate 50 mg twice daily and carvedilol 25 mg twice daily. In 30 patients with dilated cardiomyopathy, 24-hour Holter monitoring revealed a mean HR of 68 ± 3 beats per minute in carvedilol-treated patients and 69 ± 2 beats per minute in metoprolol tartrate-treated patients at a dose ratio of 1:2 [24]. In a similar manner, a study of 51 patients with HFrEF found no significant differences in mean 24-hour HR (70 ± 2 vs. 70 ± 2 beats per minute for carvedilol and metoprolol tartrate, respectively) [27]. While blood pressure reductions due to betablockade are usually mildin HF patients, carvedilol did produce a marginally greater blood pressure reduction than metoprolol in COMET (3.8 mmHg vs 2.0 mmHg, P<0.01), presumably due to alpha-1 blockade [25,27].
Metra et al. [25] conducted a randomized, double-blinded, placebo-controlled study of metoprolol tartrate versus carvedilol in 122 HFrEF patients to investigate the effects of each agent on invasively measured hemodynamic. Patients randomized to metoprolol tartrate achieved an average dose of 115 mg/ day and patients randomized to carvedilol achieved an average dose of 44 mg/day. Upon completion of 12 months follow-up, both metoprolol tartrate and carvedilol reduced left-sided filling pressures and increased stroke volume to similar extents. However, carvedilol produced more favorable changes from baseline in resting pulmonary capillary wedge pressure (27 → 19 mmHg [carvedilol] versus 25 → 20 mmHg [metoprolol], P=0.002) and resting mean pulmonary artery pressure (33 → 24 mmHg [carvedilol] versus 32 → 26 mmHg [metoprolol], P=0.049).
Metra et al. [29] also conducted provocative exercise testing and repeated these same invasive hemodynamic measurements in a separate study. Along with a lower peak heart rate, carvedilol-treated patients had a higher peak stroke volume index than metoprolol tartrate-treated patients (32 → 43 mL/ m2 verus35 → 42 mL/m2; p = 0.016).Pulmonary wedge pressure and mean pulmonary artery pressure were also lower in the carvedilol arm.
Maack et al. [28] conducted a randomized, open-label study in which HFrEF patients were initially assigned to carvedilol or metoprolol tartrate and titrated to maximum tolerable doses (carvedilol 47 mg/day; metoprolol 182 mg/day). After 12 months of treatment, patients were crossed over to the alternative treatment arm using an equivalent dose of metoprolol tartrate or carvedilol. Dobutamine (beta-1/beta-2/alpha-1 agonist) stress echocardiography was performed immediately prior to and 6 months after crossover. Investigators reported that patients experienced similar increases in cardiac output during treatment with either agent in response to dobutamine (+42% [carvedilol] versus +43% [metoprolol tartrate], P=NS), but the authors attributed this observation to greater responsiveness in HR for patients receiving metoprolol tartrate (+23% [carvedilol] versus +49% [metoprolol], P<0.05) and greater changes in stroke volume for patients receiving carvedilol (+16% [carvedilol] versus -3% [metoprolol], P=NS).
Exercise capacity
Metra et al evaluated exercise capacity at baseline and 12 months after randomization to either carvedilol or metoprolol tartrate [25]. Despite more favorable improvements in resting LV ejection fraction and LV systolic and diastolic volumes among patients randomized to carvedilol, metoprolol tartrate produced more favorable effects on exercise capacity, as measured by peak oxygen consumptionusing a stationary bicycle protocol (13.7 mL/kg/min → 15.0 mL/kg/min versus 14.2 mL/kg/min →14.0 mL/kg/min, P=0.035), but there were no differences in exercise capacity as measured by 6-minute walk test [25]. In this study, carvedilol produced greater reductions at peak exercise in HR (140 → 120 bpm [metoprolol] versus 143→ 116 bpm [carvedilol], P=0.006) coupled with greater improvements in SV index (35 → 42 mL/m2 [metoprolol] versus 32 → 43 mL/m2 [carvedilol], P=0.016) withoutany notable between group differences in cardiac index. These results suggest that carvedilol-induced improvements in contractility were counteracted by more pronounced reductions in peak HR [25]. Since the beneficial effects of beta-blockade in HF are independent of exercise capacity, the clinical relevance of the differences above is unknown.
Metabolism
Carvedilol has been associated with antioxidant effects. In study of 24 Chinese HF patients, erythrocyte superoxide dismutase activity and glutathione peroxidase activity, but not total antioxidant status, were reduced at week 12 compared to baseline in carvedilol-treated patients [30]. There was no difference compared to baseline in metoprolol tartrate-treated patients.
A study of Japanese patients showed that carvedilol treatment reduced plasma lipid peroxide levels [31]. Changes in thiobarbituric acid-reactive substances were reduced similarly by metoprolol tartrate and carvedilol in another study of HF patients [23].
Carvedilol may also switch myocardial energy substrate utilization from free fatty acids towards lactate, thus restoring homeostatic metabolism in myocytes [32]. Carvedilol also possesses anti-inflammatory properties, reducing levels of interleukin-1 beta [33] and C-reactive protein [31].
Additional support for the beneficial effects of carvedilol on metabolism can be found in the Glycemic Effects in Diabetes Mellitus: Carvedilol-Metoprolol Comparison in Hypertensives (GEMINI) trial [34]. Although conducted in patients with type 2 diabetes mellitus and hypertension—instead of patients with HF—metoprolol tartrate-treated patients had a significant worsening of hemoglobin A1c (+0.15%; 95% confidence interval, 0.08%-0.22%; P<0.001), whereas patients receiving carvedilol experienced no changes hemoglobin A1c (0.02%, 95% confidence interval -0.06%-0.10%, P=0.65). Carvedilol also showed beneficial effects on insulin resistance as measured by reduction in homeostatic model assessment-insulin resistance (-9.1% [carvedilol] versus -2.0% [metoprolol], P=0.004). This trial provides indirect evidence to support favorable metabolic effects of carvedilol (Table 1).

Landmark Clinical Trials of Carvedilol and Metoprolol in Heart Failure
a)Metoprolol vs. placebo
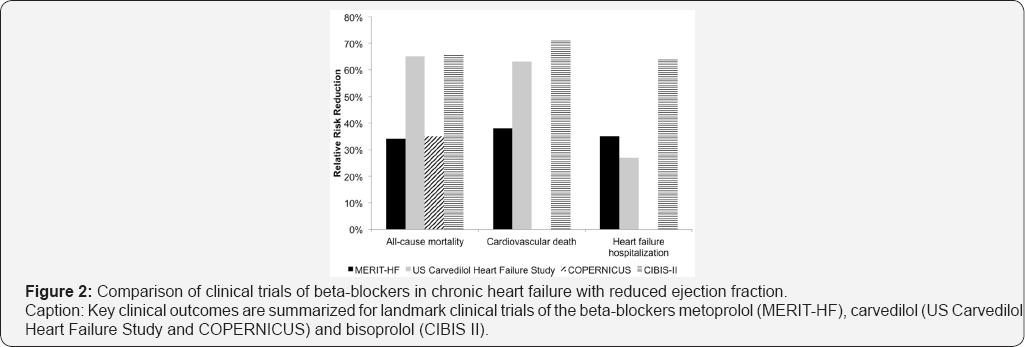
The efficacy of metoprolol succinate for the treatment of chronic HFrEF was demonstrated in the MERIT-HF study [35]. Investigators enrolled 3991 patients with NYHA Class II- IV symptoms and LVEF <40%. In addition to a background of diuretics, angiotensin-converting enzyme inhibitor (ACEI)/ angiotensin receptor blocker (ARB) and digitalis, patients were randomized to metoprolol succinate (mean daily dose 159 mg daily) or placebo. After a mean follow-up of 1 year, the study was terminated early. Metoprolol succinate significantly reduced the risk of all-cause mortality by 34% compared to placebo (7.2%/ patient-year vs. 11%/patient-year; 95% CI, 53-81%; p = 0.0062) (Figure 2).
Metoprolol tartrate was studied in the Metoprolol in Dilated Cardiomyopathy (MDC) trial [36]. In this study of 383 subjects with HFrEF and NYHA II-III symptoms, all-cause mortality or need for heart transplantation was numerically lower in metoprolol tartrate-treated patients compared with placebo (25% vs. 38%), although the p-value did not achieve statistical significance (p = 0.058).
b)Carvedilol vs. placebo
The US Carvedilol Heart Failure Study Group randomly assigned 1094 patients with chronic HF and an EF ≤35% to either carvedilol titrated to a target dose of 50 mg twice daily or placebo [37]. Patients received standard HF therapies, including an ACEI/ARB, a diuretic and digitalis, in addition to carvedilol (mean daily dose 45 mg) or placebo. The majority of patients had NYHA II-III symptoms. At the time of early termination of the study at the recommendation of the Data and Safety Monitoring Board, 3.2% of carvedilol patients as compared with 7.8% of placebo patients had died (relative risk, 65%; 95% CI, 39-80%; P<0.001).
In the COPERNICUS trial, 2289 patients with severe heart failure, defined as the presence of symptoms at rest (NYHA IV) and left ventricular ejection fraction less than 25%, were randomized to either carvedilol (mean daily dose 37 mg) or placebo [38]. The majority of patients were treated with diuretics and an ACEI or ARB. After treatment for a mean of 10 months, patients who received carvedilol had a 35% lower risk of all-cause mortality (95% CI 19-48%; P=0.0014) and a 24% lower risk of all-cause mortality or hospitalization (95% CI, 1333%; P<0.001).
c)Carvedilol vs. Metoprolol
To date, the Carvedilol or Metoprolol European Trial (COMET) remains the only randomized, head-to-head comparison of carvedilol and metoprolol outcomes in chronic HF [39]. Conducted exclusively in Europe, this trial enrolled patients with stable, NYHA Class II-IV HF, LVEF ≤35%, and a cardiovascular hospital admission within the previous 2 years. In addition, all patients had to be receiving stable doses of an ACEI for at least 4 weeks and a diuretic for at least 2 weeks. Patients were initiated either on carvedilol 3.125 mg twice daily or metoprolol tartrate 5 mg twice daily and titrated to target doses of 25 mg twice daily or 50 mg twice daily, respectively [40].
At the beginning of the maintenance phase, the mean daily doses for carvedilol and metoprolol tartrate were 41.8 mg and 95 mg, respectively, both similar to doses utilized in routine clinical practice [41]. The majority of carvedilol (75%) and metoprolol (78%) patients received the intended target dose. For approximately the first year, the mean HR was lower in the carvedilol group compared to the metoprolol tartrate group (between group difference = 1.6 beats per minute). However, there was no significant difference in HR between the two treatments for the remainder of the trial. At the 4-month mark, systolic blood pressure was 1.8 mm Hg lower in carvedilol patients than metoprolol patients (95% CI -3.2 to -0.4).
After mean study duration of 58 months, all-cause death was 35% in the carvedilol arm compared to 40% in the metoprolol tartrate arm. This 5% absolute reduction in all-cause mortality was statistically significant (HR 0.83; 95% CI 0.74-0.93; P=0.002). The majority of deaths in both carvedilol- and metoprolol tartrate-treated patients were due to a cardiovascular event and cardiovascular deaths occurred less frequently in the carvedilol group (29% vs. 35%; HR 0.80; 95% CI 0.70-0.90; P=0.0004). Carvedilol also reduced the risk of cardiovascular death, sudden death, and death due to stroke [42].
The percent of patients who experienced a serious adverse event was 55% in the carvedilol arm and 57% in the metoprolol tartrate arm. There were no differences in the rate of bradycardia. The authors concluded that the results of the COMET trial suggest that carvedilol extends survival compared to metoprolol. Interestingly, a post-hoc analysis of the COMET study suggested that transitioning from metoprolol to carvedilol was better tolerated than the reverse [43].
An additional post-hoc analysis examined the relationship between HR and outcomes in COMET [44]. In multivariate analysis adjusting for several confounders, including HR, carvedilol was associated with a significantly lower risk of death compared to metoprolol (relative risk, 0.767; 95% confidence interval, 0.663-0.887; p<0.0004), further supporting the overall study results. Carvedilol was associated with a significantly lower risk of death compared to metoprolol among patients with an achieved HR >68 beats per minute (hazard ratio, 0.77; 95% confidence interval, 0.62-0.95; p=0.0171) and those with an achieved HR≤68 beats per minute (hazard ratio, 0.78; 95% confidence interval, 0.64-0.96; p=0.019).
Similarly, mortality was significantly lower in carvedilol- treated patients as compared with metoprolol-treated patients whether the target dose was achieved or not (achieved target dose: hazard ratio, 0.77; 95% confidence interval, 0.64-0.91; p=0.0003; did not achieve target dose: hazard ratio, 0.70; 95% confidence interval, 0.54-0.92; p=0.010). The COMET investigators concluded that "The beneficial effects of carvedilol, when compared with metoprolol tartrate, at the pre-defined target doses of each compound, on mortality could not be explained by the differences in [blood pressure] and [heart rate] observed after 4 months of study treatment or by patients not achieving target study drug levels" [44].
Despite the apparent benefit of carvedilol, the COMET trial also raised many questions, most notably the use and dosing of metoprolol tartrate. While the MERIT-HF trial demonstrated that the succinate salt reduces mortality compared with placebo, the tartrate salt has not been studied in a randomized comparison with placebo. Moreover, the target dose of metoprolol used in COMET was 50 mg twice daily, less than half of the target dose used in MERIT-HF.
Comparative Effects of Metoprolol Tartrate and Metoprolol Succinate
Despite differences in pharmacokinetics, metoprolol tartrate and succinate exert similar Pharmacodynamics effects in HF patients. Kukin et al. [45]. Directly compared 3 months treatment with metoprolol tartrate and metoprolol succinate in 27 patients with HFrEF, concluding that these two agents "produce similar hemodynamic and clinical effects acutely and chronically". The target doses were 50 mg twice daily for metoprolol tartrate and 100 mg once daily for metoprolol succinate (or 100 mg twice daily and 200 mg once daily if >85 kg). All patients in the metoprolol succinate group achieved the target dose whereas 11/14 achieved the target metoprolol tartrate dose. Specifically, the following parameters were not statistically significantly different between each salt: LVEF, maximal oxygen consumption, plasma norepinephrine levels, resting HR (73 ± 13.7 vs. 73 ± 16.9 beats per minute for tartrate and succinate, respectively), pulmonary capillary wedge pressure, cardiac index, systemic vascular resistance, stroke volume index, and stroke work index.
Non-randomized Comparisons of Metoprolol and Carvedilol in Heart Failure
a) Observational Registries
Two recent large cohort analyses have provided additional insight into the clinical utility of metoprolol and carvedilol for the treatment of chronic HFrEF [46,47]. Pasternak et al. [46] included 11,664 patients from the Danish HF Registry who started carvedilol (n = 6026) or metoprolol succinate (n = 5638) within 60 days of an HF hospitalization and continued this therapy for a minimum of 120 days. To minimize residual bias, the authors matched patients with a propensity score calculated from 126 variables.
The majority of patients had NYHA II or III symptoms at inclusion. The proportion of patients with EF <25% was higher in the carvedilol group than the metoprolol succinate group (41% vs. 27%). Background therapy consisted of guideline- directed medical therapy such as ACEI or ARB, a diuretic and an aldosterone antagonist. In the propensity-matched analysis, there was no difference between carvedilol and metoprolol succinate with respect to all-cause mortality (18.3% vs. 18.8%; HR, 0.99; 95% CI, 0.88-1.11). These results did not differ when analysis was restricted to patients who reached the target dose.
A similar study was conducted by Frohlich et al. [47] using data combined from the Norwegian Heart Failure Registry and the outpatient HF clinic of the University of Heidelberg. Patients were eligible for inclusion in this study if they had systolic HF (EF <45%) and were receiving an ACEI/ARB with carvedilol or metoprolol succinate. Propensity scores were used to match patients who received carvedilol (n = 1118) or metoprolol succinate (n = 2898).The median dose of metoprolol succinate was 103 mg daily while the median dose for carvedilol was 38 mg daily. The univariate analysis of the general sample revealed significantly higher all-cause mortality among patients taking metoprolol (HR 1.49; 95% confidence interval 1.31-1.69; P<0.001). However, after adjustment for dose equivalency and propensity score matching, there was no significant difference between groups (HR, 1.00; 95% CI, 0.82-1.23; p = 0.99).
b)Meta-Analyses
Two meta-analyses have compared metoprolol and carvedilol. Chatterjee et al. [48] conducted a network metaanalysis of 21 trials (n = 23,122), establishing indirect comparisons of interventions from different studies.50In this analysis, carvedilol was the beta-blocker with the largest mortality benefit. Compared to metoprolol, carvedilol reduced the risk of mortality by 20% (odds ratio 0.83; 95% confidence interval 0.59-1.08), cardiovascular death by 41% (odds ratio 0.59; 95% confidence interval 0.20-1.39) and sudden death by 33% (odds ratio 0.67; 95% confidence interval 0.19-1.49).
Briasoulis et al. [49] utilized a different approach, conducting a traditional meta-analysis but including retrospective and post-hoc analyses. In the fixed effects model, carvedilol was associated with a significant mortality benefit (odds ratio 0.80; 95% confidence interval 0.70-0.91) that was not seen in the random effects model (odds ratio 1.02; 95% confidence interval 0.90-1.16). A similar pattern was seen for HF hospitalizations.
Discussion
The availability of large, randomized controlled trials has elevated the standard by which drugs and devices are evaluated in cardiovascular diseases. Unfortunately, useful data from well-designed clinical trials is often over-criticized and underappreciated [50]. Practicing clinicians are therefore left to sort through the 'imperfect' clinical data to make informed clinical decisions.
One common criticism of COMET has been that the short- acting metoprolol tartrate formulation does not provide the same benefits as the succinate formulation. Given that the metoprolol dose chosen (in COMET) was lower than the dose studied in the landmark MERIT-HF trial, the achievement of a highermetoprolol dose might be required for maximal benefit. Together, these criticisms imply that metoprolol-treated patients (in COMET) did not receive the same degree of beta-receptor antagonism as carvedilol-treated patients.
The evidence, however, suggests that the magnitude of differences in HR between metoprolol tartrate and carvedilol was minimal in COMET. For the first 16 months, the mean HR in the carvedilol arm was 1.6 beats per minute lower than in the metoprolol arm. For the study period from month 16 through month 58, there was no statistically significant difference in HR between the two groups. We believe that a transient HR reduction of 1-2 beats per minute (over the first 16 [28%] months of follow-up) does not explain the 20-30% reduction in outcomes observed in COMET over a 58-month follow-up. Furthermore, the Phase 2 clinical trials discussed above found that metoprolol tartrate and carvedilol demonstrated similar HR reductions when given in a 2:1 dose ratio as conducted in COMET.
Moreover, we argue that the pharmacokinetic differences between metoprolol tartrate and metoprolol succinate have no clinical implications beyond the convenience of once daily dosing with metoprolol succinate. The benefits of beta-blockade are largely mediated through long-term beta-receptor resensitization and there is no evidence that one salt formulation provides a superior impact on the negative feedback loop responsible for beta-receptor re-sensitization [51-53].
Within this context, we believe that the COMET results provide, at a minimum, evidence that carvedilol is likely superior to metoprolol tartrate or succinate, although the effect size may be overestimated.
Conclusion
Controversy regarding the preferred beta-blocker in patients with HFrEF remains ongoing. The available data therefore suggest that carvedilol is considered the first-line beta-blocker for the treatment of HF. While individual patient characteristics (e.g. severe obstructive pulmonary disease, low baseline BP) may favor the use of metoprolol in limited situations, the preponderance of evidence suggests that carvedilol should be the first-line beta-blocker in the general HFrEF population.
References
- Mozaffarian D, Benjamin EJ, Go AS, Moy Cs, Woo D, et al. (2016) Heart disease and stroke statistics - 2016 Update. Circulation 133(4): e38- 60.
- Heidenreich PA, Albert NM, Allen LA, Bluemke DA, Fonarow GC, et al. (2014) Forecasting the impact of heart failure in the United States: a policy statement from the American Heart Association. Circ Heart Fail 6(3): 606-619.
- Ahlquist RP (1948) A study of the adrenotropic receptors. Am J Physiol 153(3):586-600.
- Bristow MR (2011) Treatment of chronic heart failure with ß-adrenergic receptor antagonists: A convergence of receptor pharmacology and clinical cardiology. Circ. Res 109(10): 1176-11794.
- Yancy CW, Jessup M, Bozkurt B, A Geracie, W H Wilson Tang. et al. (2013) 2013 ACCF/AHA guideline for the management of heart failure. Circulation 128(16): 1810-1852.
- McMurray JJ V, Adamopoulos S, Anker SD, M Bohm, john J V M M, et al. (2013) ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012. Eur Heart J 33(14): 1787-1847.
- Vatner DE, Asai K, Iwase M, Homcy CJ, Vatner SF, et al. (1999) Beta- adrenergic receptor-G protein-adenylyl cyclase signal transduction in the failing heart. Am J Cardiol 83(12A): 80H-85H.
- Movsesian MA, Bristow MR (2005) Alterations in cAMP-mediated signaling and their role in the pathophysiology of dilated cardiomyopathy. Curr Top Dev Biol 68: 25-48.
- Bristow MR (1997) Mechanism of action of beta-blocking agents in heart failure. Am J Cardiol 80(11A): 26L-40L.
- Cannavo A, Koch WJ (2016) Targeting B-3 Adrenergic Receptors In The Heart-Selective Agonism And B-Blockade. J rdiovasc Pharmacol 69(2): 71-78.
- Flather MD, Shibata MC, Coats AJS, Toman J, Bohm M, et al. (2005) Randomized trial to determine the effect of nebivolol on mortality and cardiovascular hospital admission in elderly patients with heart failure (SENIORS). Eur Heart J 26(3): 215-225.
- Bauman JL, Talbert RL (2004) Pharmacodynamics of beta-blockers in heart failure: lessons from the carvedilol or metoprolol European trial. J Cardiovasc Pharmacol Ther 9(2): 117-128.
- Maack C, Cremers B, Flesch M, Höper A, Südkamp M, et al. (2000) Different intrinsic activities of bucindolol, carvedilol and metoprolol in human failing myocardium. Br J Pharmacol 130(5): 1131-1139.
- Zebrack JS, Munger M, Macgregor J, Lombardi WL, Stoddard GP, et al. (2009) Beta-receptor selectivity of carvedilol and metoprolol succinate in patients with heart failure (SELECT trial): a randomized dose-ranging trial. Pharmacother 29(8): 883-890.
- Sigmund M, Jakob H, Becker H, Hanrath P, Schumacher, et al. (1996) Effects of metoprolol on myocardial beta-adrenoceptors and Gi alphaproteins in patients with congestive heart failure. Eur J Clin Pharmacol 51(2): 127-132.
- Kubo H, Margulies KB, Piacentino V, Gaughan JP, Houser SR (2001) Patients with end-stage congestive heart failure treated with beta- adrenergic receptor antagonists have improved ventricular myocyte calcium regulatory protein abundance. Circulation 104(9): 1012-1018.
- Lowes BD, Gilbert EM, Abraham WT, Quaife RA, Bristow MR, et al.(2002) Myocardial gene expression in dilated cardiomyopathy treated with beta-blocking agents. N Engl J Med 346(18): 1357-1365.
- Heilbrunn SM, Shah P, Bristow MR, Valantine HA, Ginsburg R, et al. (1989) Increased beta-receptor density and improved hemodynamic response to catecholamine stimulation during long-term metoprolol therapy in heart failure from dilated cardiomyopathy. Circulation 79(3): 483-490.
- Gilbert EM, Olsen SL, Renlund DG, Bristow MR (1993) beta-adrenergic receptor regulation and left ventricular function in idiopathic dilated cardiomyopathy. Am J Cardiol 71(9): 23C-29C.
- Van Tassell BW, Rondina MT, Huggins F, Gilbert EM, Munger MA (2008) Carvedilol increases blood pressure response to phenylephrine infusion in heart failure subjects with systolic dysfunction: evidence of improved vascular alpha1-adrenoreceptor signal transduction. Am Heart J 156(2): 315-321.
- Bundgaard H, Axelsson A, Hartvig Thomsen J, Kaber L, Valeur N, et al. (2017) The-first-in-man randomized trial of a beta3 adrenoceptor agonist in chronic heart failure: the BEAT-HF trial. Eur J Heart Fail 19(4): 566-575.
- Man In't Veld A, Schalekamp M (1983) On the antihypertensive mechanisms of beta-adrenoreceptor antagonists in relation to intrinsic sympathomimetic activity cardioselectivity and hydrophilicity. J Pharmacol 14(11): 69-104.
- Kukin ML, Kalman J, Charney RH, et al. (1999) Prospective, randomized comparison of effect of long-term treatment with metoprolol or carvedilol on symptoms, exercise, ejection fraction, and oxidative stress in heart failure. Circulation 99(20): 2645-2651.
- Di Lenarda A, Sabbadini G, Salvatore L, et al. (1999) Long-term effects of carvedilol in idiopathic dilated cardiomyopathy with persistent left ventricular dysfunction despite chronic metoprolol. The Heart-Muscle Disease Study Group. J Am Coll Cardiol 33(7): 1926-1934.
- Metra M, Giubbini R, Nodari S, Boldi E, Modena MG, et al. (2000] Differential effects of beta-blockers in patients with heart failure: A prospective, randomized, double-blind comparison of the long-term effects of metoprolol versus carvedilol. Circulation 102(5): 546-551.
- Packer M, Antonopoulos G V, Berlin JA, Chittams J, Konstam MA, et al. (2001) Comparative effects of carvedilol and metoprolol on left ventricular ejection fraction in heart failure: results of a meta-analysis. Am Hear J 141(6): 899-907.
- Sanderson JE, Chan SK, Yip G, Yeung LY, Woo KS, et al. (1999) Betablockade in heart failure: a comparison of carvedilol with metoprolol. J Am Coll Cardiol 34(5): 1522-1528.
- Maack C, Elter T, Nickenig G, La Roseeet k, Wuttke, et al. (2001] Prospective crossover comparison of carvedilol and metoprolol in patients with chronic heart failure. J Am Coll Cardiol 38(4): 939-946.
- Metra M, Nodari S, D'Aloia A, Muneretto C, Dei cas L, et al. (2002) Betablocker therapy influences the hemodynamic response to inotropic agents in patients with heart failure: a randomized comparison of dobutamine and enoximone before and after chronic treatment with metoprolol or carvedilol. J Am Coll Cardiol 40(7): 1248-1258.
- Arumanayagam M, Chan S, Tong S, Sanderson JE (2001) Antioxidant properties of carvedilol and metoprolol in heart failure: a double-blind randomized controlled trial. J Cardiovasc Pharmacol 37(1): 48-54.
- Nagatomo Y, Yoshikawa T, Kohno T, Yoshizawa, Mequro T, et al. (2007] Effects of beta-blocker therapy on high sensitivity c-reactive protein, oxidative stress, and cardiac function in patients with congestive heart failure. J Card Fail 13(5): 365-371.
- Al-Hesayen A, Azevedo ER, Floras JS, Hollingshead S, Lopaschuk GD, et al. (2005) Selective versus nonselective beta-adrenergic receptor blockade in chronic heart failure: differential effects on myocardial energy substrate utilization. Eur J Heart Fail 7(4): 618-623.
- Cinquegrana G, D'Aniello L, Landi M, Grande G, D Prisco,et al. (2016] Effects of different degrees of sympathetic antagonism on cytokine network in patients with ischemic dilated cardiomyopathy. J Card Fail 11(3): 213-219.
- Bakris GL, Fonseca V, Katholi RE, Oakers R, Lukas MA,et al. (2004] Metabolic effects of carvedilol vs metoprolol in patients with type 2 diabetes mellitus and hypertension: a randomized controlled trial. JAMA 292(18): 2227-2236.
- (1999) MERIT-HF Study Group. Effect of metoprolol CR/XL in chronic heart failure. Lancet 353: 2001-2007.
- Waagstein F, Bristow MR, Swedberg K, Camerini F, Silver MA,et al. (1993) Beneficial effects of metoprolol in idiopathic dilated cardiomyopathy. Metoprolol in Dilated Cardiomyopathy (MDC) Trial Study Group. Lancet 342(8885): 1441-1446.
- Packer M, Bristow MR, Cohn JN, Edward M. Gilbert, Wilson S. Colucci, et al. (1996) The effect of carvedilol on morbidity and mortality in patients with chronic heart failure. N Engl J Med 334: 1349-1355.
- Packer M, Coats AJ, Fowler MB, Hugo A. Katus, Jean L Rouleau, et al.(2001) Effect of carvedilol on survival in severe congestive heart failure. N Engl J Med 344: 1651-1658.
- Poole-Wilson PA, Swedberg K, Cleland JGF, Hanrath P, Lutiquer, et al.(2003) Comparison of carvedilol and metoprolol on clinical outcomes in patients with chronic heart failure in the Carvedilol Or Metoprolol European Trial (COMET): randomised controlled trial. Lancet 362(9377): 7-13.
- Poole-Wilson PA, Cleland JGF, Di Lenarda A, Hanrath P, Metra M, et al.(2002) Rationale and design of the carvedilol or metoprolol European trial in patients with chronic heart failure: COMET Eur J Heart Fail 4(3):321-329.
- Butler J, Khadim G, Belue R, Chomsky D, Dittus RS, et al. (2003] Tolerability to beta-blocker therapy among heart failure patients in clinical practice. J Card Fail 9(3):203-209.
- Remme WJ, Cleland JG, Erhardt L, Metra M, Moullet C, et al. (2007] Effect of carvedilol and metoprolol on the mode of death in patients with heart failure. Eur J Heart Fail 9(11): 1128-1135.
- Di Lenarda A, Remme WJ, Charlesworth A, Cleland JG, Metra M, et al. (2005) Exchange of ß-blockers in heart failure patients. Experiences from the poststudy phase of COMET (the Carvedilol or Metoprolol European Trial). Eur J Heart Fail 7(4): 640-649.
- Metra M, Torp-Pedersen C, Swedberg K, Cleland JG, Komajda M, et al. (2005) Influence of heart rate, blood pressure, and beta-blocker dose on outcome and the differences in outcome between carvedilol and metoprolol tartrate in patients with chronic heart failure: results from the COMET trial. Eur Heart J [Internet] 26(21): 2259-2268.
- Kukin ML, Mannino MM, Freudenberger RS, Kalman J, Buchholz-Varley C, et al. (2016) Hemodynamic comparison of twice daily metoprolol tartrate with once daily metoprolol succinate in congestive heart failure. Jam Coll Cardiol 35(1): 45-50.
- Pasternak B, Svanström H, Melbye M, Hviid A. (2014) Association of treatment with carvedilol vs metoprolol succinate and mortality in patients with heart failure. JAMA Intern Med [Internet] [cited 2016 Mar 11];174(10): 1597-604.
- Fröhlich H, Zhao J, Täger T, ebola R, katus, et al. (2015) Carvedilol Compared With Metoprolol Succinate in the Treatment and Prognosis of Patients With Stable Chronic Heart Failure: Carvedilol or Metoprolol Evaluation Study. Circ Heart Fail [Internet] 2015 [cited 2016 Mar 11];8(5):887-896.
- Chatterjee S, Biondi-Zoccai G, Abbate A, Fobrizo, B V Tassell et al. (2013) Benefits of ß blockers in patients with heart failure and reduced ejection fraction: network meta-analysis. BMJ 346: f55.
- Briasoulis A, Palla M, Afonso L (2015) Meta-analysis of the effects of carvedilol versus metoprolol on all-cause mortality and hospitalizations in patients with heart failure. Am J Cardiol [Internet] 2015 115(8): 1111-1115.
- Packer M (2016) Unbelievable Folly of Clinical Trials in Heart Failure: The Inconvenient Truth About How Investigators and Guidelines Weigh Evidence. Circ Heart Fail 9(4): e002837.
- Flannery G, Gehrig-Mills R, Billah B, Krum H. (2008) Analysis of randomized controlled trials on the effect of magnitude of heart rate reduction on clinical. Am J Cardio 101(6): 865-869.
- Falskov B, Hermann TS, Rauns� J, Buris C, Lars K, et al. (2011) Endothelial function is unaffected by changing between carvedilol and metoprolol in patients
- Hryniewicz K, Androne AS, Hudaihed A, Katz SD (2003) Comparative effects of carvedilol and metoprolol on regional vascular responses to adrenergic stimuli in normal subjects






























