Cpap Treatment Promotes the Closure of a Patent Foramen Ovale in Subjects with Obstructive Sleep Apnea Syndrome -Results from a Pilot study
Manolo Beelke*
Worldwide Clinical Trials, Medical & Scientific Affairs, Germany
Submission: May 6, 2017; Published: June 27, 2017
*Corresponding author: Manolo E. Beelke, Senior Medical Director, Worldwide Clinical Trials, Germany Email: manolo.beelke@worldwide.com
How to cite this article: Manolo B. Cpap Treatment Promotes the Closure of a Patent Foramen Ovale in Subjects with Obstructive Sleep Apnea Syndrome Results from a Pilot study. J of Pharmacol & Clin Res. 2017; 3(1): 555604. DOI: 10.19080/JPCR.2017.02.555604.
Abstract
Study Objectives: Patent foramen ovale (PFO) is an independent risk factor for ischemic stroke by means of paradoxical embolization, due to right-to-left shunt (RLSh). The higher prevalence of PFO found in OSAS respect to the general population could be due to the chronic effect of the intrathoracic pressure changes during periods of obstructive sleep apnea. This study aimed to re-evaluate the magnitude of RLSh in subjects with obstructive sleep apnea syndrome (OSAS) and diagnosed PFO after a long period of CPAP treatment.
Design and Setting: Assessment of PFO and concomitant OSAS. Application of CPAP treatment with control of compliance. Re-evaluation of RLSh through the assessed PFO at follow-up (after 41 ± 6 months) by means of Transcranial Doppler with contrast medium injected in antecubital vein.
Participants: Eighteen OSAS subjects affected by PFO (mean age 56 ± 11 years).
Interventions: They underwent Transcranial Doppler, with injection of agitated saline solution mixed with air during normal breathing and Valsalva maneuver.
Measurements and Results: CPAP treatment has chronically applied by 15 of 18 subjects (83%). RLSh magnitude did not change in the 3 subjects, who had not applied the CPAP treatment. In 4 of the 15 subjects, who used the CPAP treatment, no RLSh could be recorded (PFO closure). The difference between CPAP-user and CPAP-non-users was significant (p>0.01). In the other 11 of this subgroup RLSh magnitude was reduced respect to baseline recording. Multiple regression analysis modelling, magnitude of RLSh correlated mainly to the weekly CPAP use (days/week) negatively and the condition of atrial fibrillation positively.
Conclusion: In the nocturnal sleep period RLSh can occur during single obstructive apneas in subjects with OSAS and concomitant presence of PFO. This can be a risk factor for cerebrovascular diseases. This risk could probably increase proportionally to the respiratory disturbance index of these subjects. Compliance to CPAP treatment is able to reduce the magnitude of RLSh.
Keywords: Intra-cardiac Shunting; Right-to-left Shunt; Obstructive Sleep Apnea; OSAS; Patent Foramen Ovale; PFO; Transcranial Doppler Sonography; Stroke; Continuous Positive Airway Pressure; CPAP
Introduction
Although the prevalence of PFO is about 25 percent in the general population, this increases up to a third in subjects who have stroke of unknown cause, referred to as cryptogenic stroke, and in young subjects, actually half of strokes are considered as cryptogenic[1]. For quite some time, obstructive sleep apnea syndrome (OSAS) has been recognized as a condition which increase the risk for stroke in these subjects respect to the general population, especially in young to mid-aged subjects [2,3]. Indeed, the relative risk for developing new vascular problems in untreated OSAS has been estimated in an odds ratio of 2.3 [4]. How frequent the occurrence of stroke in the natural story of OSAS is can furthermore emphasized by data which report that 70-90% of subjects with acute stroke present also OSAS [4-7] The high prevalence of stroke in OSAS has been correlated in the past principally cardiovascular and hematological factors [8-15] The most important aspect of all these hematological alterations is characterized by reactive polycythemia in these subjects with an increased likelihood to form microemboli [9,16-17].
This condition of high risk for the occurrence of stroke could be particularly adverse for those subjects with OSAS, who present simultaneously a patent foramen ovale (PFO). Patent foramen ovale (PFO) has been found to be the most common intertribal communication [18-23] and it has been shown in previous studies that the prevalence of PFO is significantly higher in subjects with stroke [24-31] Furthermore, also in subjects with OSAS the prevalence of PFO is significantly higher than in the normal population [32,33].
The impact of a PFO as risk factor for stroke has been recently additionally stressed, when the diameter of PFO individualized as an independent risk factor for ischemic events [34], especially for recurrent strokes. Because of its valve-like nature, PFO may allow right to left shunting (RLSh) between the right and the left atria during the cardiac cycle as a result of a transient, instantaneous pressure gradient [35,36] (at rest shunting PFO), and micro emboli can, as venous clots, easily reach in this way the arterial circulation, thus favoring cerebral embolism [37].
More frequently RLSh through a PFO can be observed only in the presence of provocative Valsalva-like maneuvers, which increase the right cardiac chamber pressure (provocative shunting PFO). Such a condition of higher right compared to left atrial pressure can also occur during every day events such as coughing, sport efforts, heavy weight lifting, and breath instrument playing, which are all similar to the provocative Valsalva maneuver. However a recent study showed that RLSh also occurs in subjects with OSAS during periods of nocturnal apnea, if the apnea length is longer than 17 s [38]. The frequency of these respiratory events in subjects with OSAS is high. In fact, clinical practice shows that among these subjects respiratory events can range from ten to more than 100 apneas per hour.
Based on the hypothesis that the effort of the intrathoracic pressure changes on the right atrium wall due to sleep apneas could contribute in increasing the size of a revealed PFO in subjects with OSAS, a controlled ventilatory treatment with continuous positive airway pressure (CPAP), consequently, could help to reduce the magnitude of the RLSh through this PFO, with effects persisting also outside of the periods of nocturnal apnea. This study investigates, therefore, if a controlled CPAP treatment with high compliance could reduce the magnitude of PFO during wakefulness, reducing by this means the general risk for stroke in these subjects.
Subjects and Methods
The subjects of this study have been enrolled from a previous study in which 78 consecutive subjects with documented medium-severe OSAS have been evaluated for the concomitant presence of a PFO [33] for all subjects of this sample CPAP treatment was the best eligible therapy. In 21 of 78 screened subjects a PFO has been diagnosed (Figure 1). For the follow- up recording 18 of 21 subjects (4 F and 14 M, mean age: 56 ± 11 years), in who the presence of an RLSh through a PFO has been shown, was asked to underwent a follow-up session for the evaluation of the magnitude of RLSh after a sufficient long period of CPAP treatment. Three of 18 subjects never applied the CPAP treatment, and 15 subjects (83 %) used the CPAP treatment for the whole time from diagnosis to follow-up (mean period 41 ± 6 months, range [30-46 months], mean treatment pressure: 11 ± 2 mbar). Sample features are listed in table 1. Compliance to CPAP treatment has been controlled every 3 months, controlling thedata about daily CPAP use memorized in the microchip of the CPAP. Furthermore, as ulterior control of treatment efficacy, Epworth sleepiness scale has been administrated at the moment of diagnosis for OSAS (mean ESS score 13 ± 4) and at the moment of Follow-up assessment for RLSh magnitude.
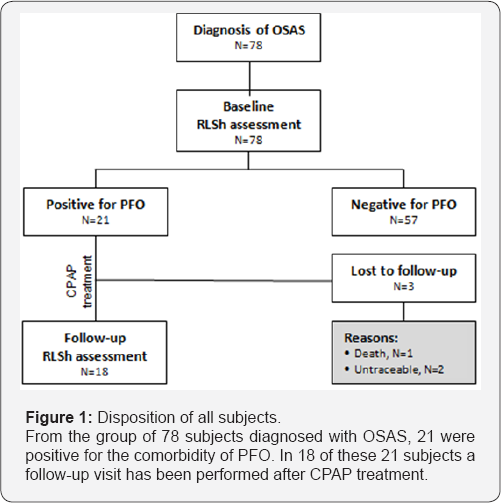
During the observational period, no subject had had neither myocardial infarction, unstable angina, decompensated congestive heart failure, nor TIA or stroke, prior pulmonary embolism nor pulmonary infarction.
At the Follow-up visit the same procedure as during the previous assessment of RLSh magnitude through PFO at baseline [33] has been applied.
In all subjects the presence of RLSh was evaluated by means of TcD (Multidop DWL, Sipplingen, Germany) with injection of microcavitation saline contrast medium. The technique for the PFO assessment has been described more detailed in previously [33,39,40]. Briefly, with the subject in supine position and the blood flow on middle artery recorded bilaterally through the temporal bone window by two transcranial pulsed-wave 2 MHz ultrasound probes, the contrast medium (a mixture of 9 ml physiologic solution and 1 ml air) will rapidly injected into the cubital vein. The examination will done in two sessions: the first one with physiological respiration, followed by the second one with Valsalva maneuver. The recording of so-called high intensity transient signals (HITS) during the TcD examination allows highlighting an RLSh due to the presence of a PFO. This diagnosis is based on the principle that a gaseous contrast medium injected into a peripheral vein is expired normally at pulmonary level, but will pass from the right to the left circulation if a PFO is present.
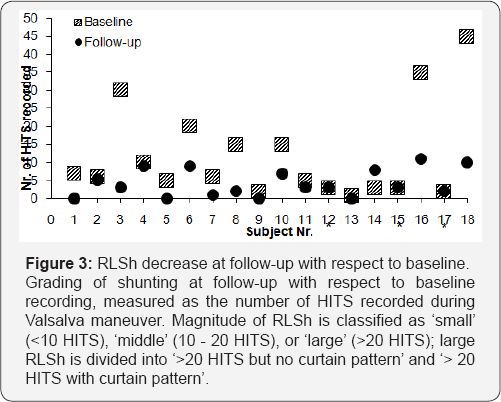
The occurrence of RLSh has been distinguished in "at rest shunt" and "provocative shunt". Magnitude of RLSh was classified as 'small' (>10 HITS), 'middle' (10 - 20 HITS), or 'large' (>20 HITS); large RLSh was divided into ‘>20 HITS but no curtain pattern’ and '> 20 HITS with curtain pattern’ [33,39,40].
Statistical analysis
Differences in the rating of the ESS, the BMI, number of microembolic signals during normal respiration and during Valsalva maneuver has been evaluated applying paired t-test procedure. This test has been applied for the whole sample and for the two sub-groups: subjects with already at rest shunting (group A) and subjects with shunting during Valsalva maneuver, provocative shunting, (group B).
Then backward multiple regression analysis has been applied to evaluate the relationship between the magnitude of RLSh reduction on the follow-up respect to the baseline recording (dependent variable) and the following independent variables: CPAP use as months of treatment, daily use (hours) and weekly use (days/week), the CPAP treatment pressure (mbar), age, the condition of atrial fibrillation, the number of HITS at baseline recording.
All computations were performed using the SAS package software version 6, SAS Inc. [41].
Results
Though for years, many experts have postulated a cause-and- effect relationship between migraine headaches and PFO [42,43] only one subject referred episodes of migraine without aura. As far as increased risk for strokes regards due to comorbidity of atrial fibrillation, only one subject of the study sample suffered this condition.
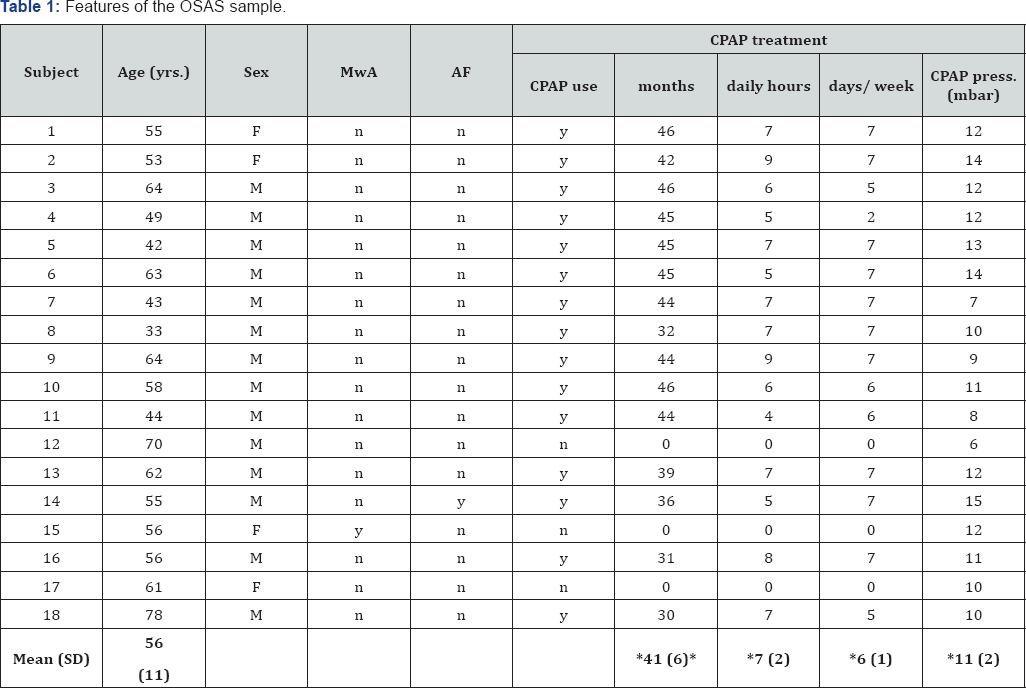
Note: Highlighted in grey are those subjects, who have not used CPAP during the follow-up period.
*Mean value and Standard deviation calculated only for subjects, who used the CPAP
MwA, migraine without aura; AF, atrial fibrillation, F, female; M, male; y, yes; n, no; SD, Standard Deviation
Among the 15 subjects who used CPAP treatment, compliance was good with a mean daily use of 6.6 ± 1.5 hours and a meanly weekly use of 6 ± 1 day a week (Table 1).
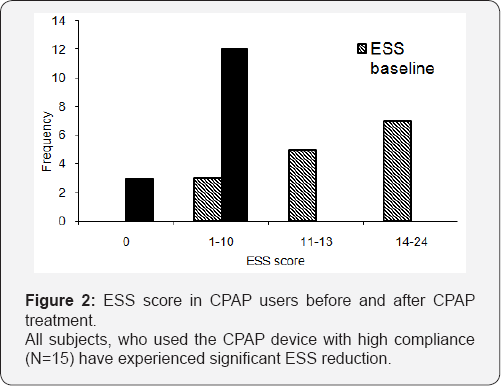
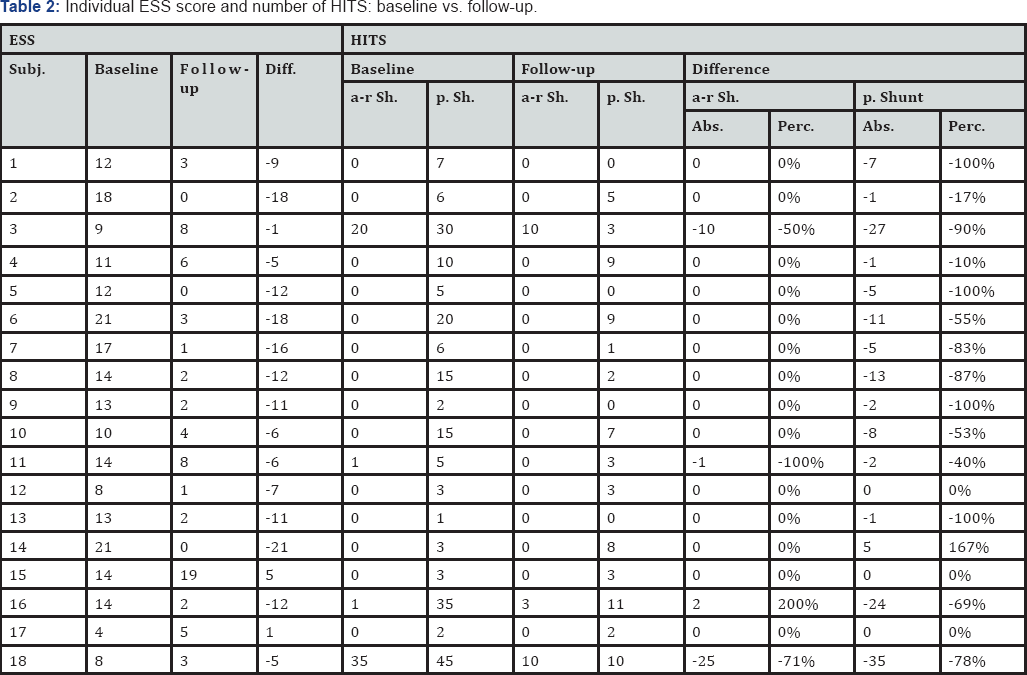
Despite the small sample size, analysis by paired t-test showed at follow-up respect to baseline significant ESS score decrease for the CPAP users (p>0.0001); (Figure 2). In the group of the 3 subjects, who have not used the CPAP device, 2 of 3 subjects showed increased ESS scores at follow-up (Table 2). In addition all CPAP users showed an ESS score lower than 10 (mean ESS score 3 ± 3) vs. non-users (mean ESS score 8 ± 10).
Furthermore at follow-up in the CPAP user group the number of HITS was significantly reduced during Valsalva maneuver, whereas it was not in the non-user group (p>0.01), (Table 2& Figure 3). No significant differences could be shown for the number of HITS recorded during physiologic respiration (Table 2). In particular, during Valsalva maneuver in 4 of 15 subjects (27%) no HITS could be recorded at follow-up (100% reduction). A reduction in HITS during Valsalva maneuver at follow-up with respect to the baseline recording of at least 50% has been seen in 11 of 15 subjects (73%). An increase of HITS during Valsalva maneuver at follow-up has been observed only in 1 subjects, although the ESS score decreased from 21 at baseline to 0 at follow-up (Table 2).
No statistically significant differences were present for the BMI neither between groups and nor between time (follow-up respect to baseline).
No significant differences have been found for the latency to the microembolic signal and for the BMI.
When multiple regression analysis was applied, the weekly CPAP use (days/week) and the condition of atrial fibrillation were recognized as these factors, which mainly influence the amount of HITS at follow-up. In this model the number of days of weekly CPAP use was negatively and the condition of atrial fibrillation was positively correlated to the number of HITS recorded at follow-up.
Discussion
In this study it has been shown for the first time that the daily application of CPAP treatment reduce the magnitude of RLSh in those OSAS subjects, who present concomitantly a PFO, where the non-use do not change the PFO size. In the study sample, the treatment effect of the CPAP device use appears to be related more to the weekly CPAP use (days/week) instead of the daily hours of use. The lack of an impact of the daily use of the CPAP device might however not see in this sample due to the high daily use (7 ± 2 hours daily).
a-r Sh, at-rest Shunt; p. Sh., provocative Shunt; Abs., absolute frequency; Perc., percentage
Note: Highlighted in grey are those subjects, who have not used the CPAP device during the follow-up period
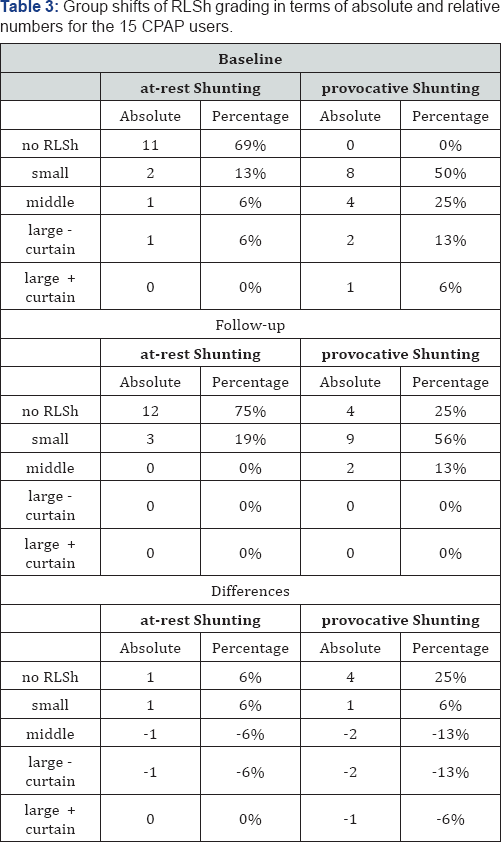
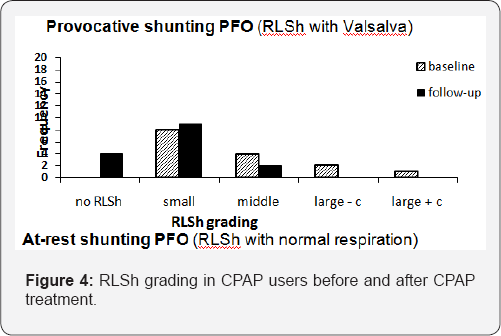
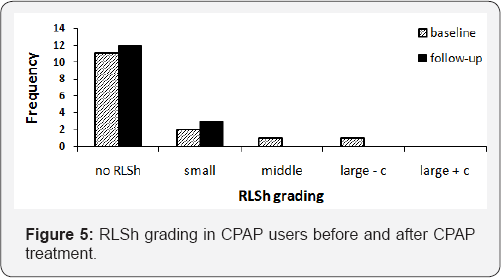
OSAS subjects with PFO of little size can reveal at follow- up an absolute complete closure of the PFO (Table 2). PFO's of middle and large size, however show only a partial closure, shifting generally in the next lower class (Table 3) & (Figure 4). In subjects with PFO of large size and shunting already at rest, the decrease of the magnitude of RLSh at follow-up is not characterized by a shift in the next lower class (Figures 4-6).
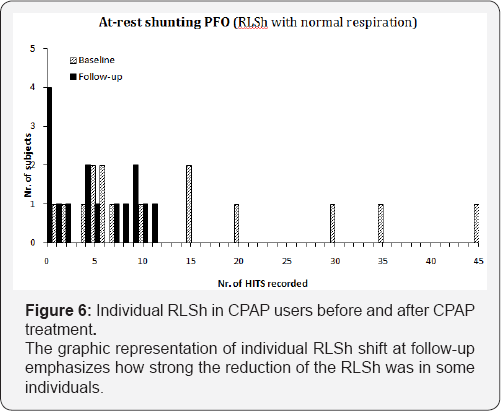
In previous studies, the diameter of a PFO has been shown to be an independent risk factor for ischemic events, especially recurrent strokes [31,44]. The occurrence of PFO has been associated to genetic factors, especially to mutations of the homeobox gene NKX2-5, but the importance of these factors in sporadic PFO is still open to debate [45,46].
In a previous study [38], it has been shown that nocturnal sleep apnea could act like the Valsalva maneuver in opening of a provocative shunting PFO. This study suggests therefore that the chronic effect of apnea-dependent mechanical factors may influence the size of a PFO. In fact, these mechanical factors start to act already at the beginning of an obstructive apnea, where an inspiratory effort against a closed upper airway is present, inducing a decrease in end-inspiratory pleural pressure. This event is followed by swings of increasing end decreasing effects on pleural pressure, influencing directly the interatrial pressure balance and the increase of inspiratory venous return. The consequent right-sided pressure increase will then be at the origin of the RLSh through a PFO. The chronic effect of such a mechanism could also explain the higher prevalence of PFO in subjects with OSAS respect to the control group [32,33].
The well reported high morbidity and mortality in OSAS [2,3,47,48], related particularly to stroke, can be explained by the sequential path physiological events, which, occurring during periods of nocturnal obstructive apneas, can compromise the brain circulation [8-14]. Chronic CPAP treatment has commonly seen to reduce the risk for stroke [49-51]. Data from this study suggest, indirectly, that this stroke-protective effect may not only be linked to hematologic and arteriosus pressure features, but also to the ability to reduce the size of a PFO, or absolutely close it. However, to obtain this effect, a high compliance to the CPAP treatment seems to be a prerequisite.
Conclusion
This study shows furthermore, that in subjects with a PFO of big size before treatment and in those subjects with an RLSh already at rest with normal respiration, the decrease of the RLSh at follow-up will be only partially. Considering, that at the moment of diagnosis of PFO in a subject with OSAS it is impossible to preview the magnitude of RLSh decrease due to the CPAP treatment, additional therapeutic approaches as surgical closure of PFO [52] or life-long treatment with anticoagulants or antiplatelet agents should be considered at least, till a follow-up TcD examination do not show a spontaneous PFO closure or a PFO of little size.
References
- Amarenco P (2005) Cryptogenic stroke, aortic arch atheroma, patent foramen ovale, and the risk of stroke. Cerebrovasc Dis 20(suppl2): 6874.
- Bassetti C, Aldrich MS, Quint D (1997) Sleep-disordered breathing in subjects with acute supra- and infratentorial strokes:A prospective study of 39 subjects. Stroke 28: 1765-1772.
- Pressman MR, Schetman WR, Figueroa WG, VAN Uitert B, Caplan HJ, et al. (1995) Transient ischemic attacks and minor stroke during sleep. Relationship to obstructive sleep apnea syndrome. Stroke 26(12): 2361-2365.
- Partinen M, Guilleminault C (1990) Daytime sleepiness and vascular morbidity at seven-year follow-up in obstructive sleep apnea subjects. Chest 97(1): 27-32.
- Parra O, Arboix A, Bechich S, Garcia-Eroles L, Montserrat JM, et al. (2000) Time course of sleep-related breathing disorders in first-ever stroke or transient ischemic attack. Am J Respir Crit Care Med 161(2 Pt 1):375-380.
- Clarenbach P and Wessendorf T (2001) Sleep and stroke. Rev Neurol 157: S46-S52
- Neau JP, Paquereau J, Meurice JC, Chavagnat JJ, Gil R (2002) Stroke and sleep apnoea: cause or consequence? Sleep Med Rev 6(6): 457-469.
- Szucs A, Vitrai J, Janszky J, Migleczi G, Bodizs R, et al. (2002) Pathological sleep apnoea frequency remains permanent in ischaemic stroke and it is transient in haemorrhagic stroke. Eur Neurol 47(1):15-19.
- Nobili L, Schiavi G, Bozano M, De Carli F, Ferrillo F, et al. (2000) Morning increase of whole blood viscosity in obstructive sleep apnea syndrome. Clin Hemorheol Microcirc 22: 21-27.
- Kiely JL, McNicholas WT (2000) Cardiovascular risk factors in subjects with obstructive sleep apnoea syndrome. Eur Respir J 16(1): 128-133.
- Geiser T, Buck F, Meyer BJ, Bassetti C, Haeberli A, et al. (2002) In vivo platelet activation is increased during sleep in subjects with obstructive sleep apnea syndrome. Respiration 69(3): 229-234.
- Placidi F, Diomedi M, Cupini LM, Bernardi G, Silvestrini M (1998) Impairment of daytime cerebrovascular reactivity in subjects with obstructive sleep apnoea syndrome. J Sleep Res 7(4): 288-292.
- Beelke M, Angeli S, Del Sette M, De Carli F, Canovaro P, et al. (2002) Obstructive sleep apnea can be provocative for right-to-left shunting through a patent foramen ovale. Sleep 25(8): 856-862.
- Trzebski A, Smietanowski M (2001) Non-linear dynamics of cardiovascular system in humans exposed to repetitive apneas modeling obstructive sleep apnea: aggregated time series data analysis. Auton Neurosci 90(1-2): 106-115.
- Sanner BM, Konermann M, Tepel M, Groetz J, Mummenhoff C, et al. (2000) Platelet function in subjects with obstructive sleep apnoea syndrome. Eur Respir J 16(4): 648-652.
- Eisensehr I, Noachtar S (2001) Haematological aspects of obstructive sleep apnoea. Sleep Med Rev 5(3): 207-221.
- Biswas M, Prakash PK, Cossburn M, Myers K, Hanna F (2003) Life- threatening thrombotic complications of relative polycythaemia. J Intern Med 253(4): 481-483.
- Hagen PT, Scholz DG, Edwards WD (1998) Incidence and size of paten foramen ovale during the first 10 decades of life: an autopsy study of 965 normal hearts. Mayo Clin Proc 59: 17-20.
- Schneider B, Zienkiewicz T, Jansen V, Hofmann, Noltenius H, et al. (1996) Diagnosis of patent foramen ovale by transesophageal echocardiography and correlation with autopsy findings. Am J Cardiol 77: 1202-1229.
- Lynch JJ, Schuchard GH, Gross CM, Wann LS (1984) Prevalence of right to left atrial shunting in a healthy population: detection by Valsalva maneuver contrast echocardiography. Am J Cardiol 53:1478-1480.
- Movsowitz C, Podolsky LA, Meyerowitz CB, Jacobs LE, Kotter MN (1992) Patent foramen ovale: a functional embryological remnant or a potential cause of significant pathology? J Am Soc Echocadiogr 5: 259270.
- Siostrzonek P, Lang W, Zangeneh M, Georg R, Andreas S, et al. (1992) Significance of left-sided heart disease for the detection of patent foramen ovale by transesophageal contrast echocardiography. J Am Coll Cardiol 19: 1192.
- Fisher DC, Fisher EA, Budd JH, Rosen SE, Goldman ME (1995) The incidence of patent foramen ovale in 1,000 consecutive subjects. A contrast transesophageal echocardiography study. Chest 107: 15041509.
- Lechat P, Mas JL, Lascault G, Theard M, Drobinsk G, et al. (1988) Prevalence of patent foramen ovale in subjects with stroke. N Engl J Med 318(18): 1148-1152.
- Itoh T, Matsumoto M, Handa N, Tsukamoto Y, Kamada T, et al. (1994) Paradoxical embolism as a cause of ischemic stroke of uncertain etiology. A transcranial Doppler sonographic study. Stroke 25(4): 771775.
- Yeung M, Khan KA, Shuaib A (1996) Transcranial Doppler ultrasonography in the detection of venous to arterial shunting in acute stroke and transient ischaemic attacks. J Neurol Neurosurg Psychiatry 61(5): 445-449.
- Webster MW, Chancellor AM, Smith HJ, Smith HJ, Sharpe DN, et al. (1988) Patent foramen ovale in young stroke subjects. Lancet 2(8601): 11-12.
- Di Tullio M, Sacco RL, Gopal A, Mohr JP, Homma S (1992) Patent foramen ovale as a risk factor for cryptogenic stroke. Ann Intern Med 117(6): 461-465.
- Ranoux D, Cohen A, Cabanes L, Amarenco P, Mas JL et al. (1993) Patent foramen ovale: is stroke due to paradoxical embolism? Stroke 24: 31
- Klötzsch C, Janssen G, Berlit P (1994) Transesophageal echocardiography and contrast-TCD in the detection of a patent foramen ovale: experiences with 111 subjects. Neurology 44: 16031606.
- Cujec B, Mainra R, Johnson DH (1999) Prevention of recurrent cerebral ischemic events in subjects with patent foramen ovale and cryptogenic strokes or transient ischemic attacks. Can J Cardiol 15: 57-64.
- Shanoudy H, Soliman A, Raggi P, Liu JW, Russell DC, et al. (1998) Prevalence of patent foramen ovale and its contribution to hypoxemia in subjects with obstructive sleep apnea. Chest 113: 91-96.
- Beelke M, Angeli S, Del Sette M, Gandolfo C, Cabano ME, et al. (2003) Prevalence of patent foramen ovale in subjects with obstructive sleep apnea: a transcranial Doppler ultrasound study. Sleep Medicine 4(3): 219-223.
- Schuchlenz HW, Weihs W, Horner S, Quehenberger F (2000) The association between the diameter of a patent foramen ovale and the risk of embolic cerebrovascular events. Am J Med 109: 456-462.
- Strunk BL, Cheitlin MD, Stulbarg MS, Schiller NB (1987) Right-to-left interatrial shunting through a patent foramen ovale despite normal intracardiac pressures. Am J Cardiol 60: 413-415.
- Maraj R, Ahmed O, Fraifeld M, L E Jacobs, M N Kotler, et al.(1999) Hypoxia due to patent foramen ovale in the absence of pulmonary hypertension. Tex Heart Inst J 26(4): 306-308.
- Chant H, McCollum C (2001) Stroke in young adults: the role of paradoxical embolism. Thromb Haemost 85(1): 22-29.
- Beelke M, Angeli S, Del Sette M, De Carli F, Canovaro P, et al. (2002) Obstructive sleep apnea can be provocative for right-to-left shunting through a patent foramen ovale. Sleep 25(8): 856-862.
- Jauss M, Zanette E (2000) Detection of rightto-left shunt with ultrasound contrast agent and transcranial Doppler sonography. Cerebrovasc Dis 10: 490-496.
- Angeli S, Del Sette M, Beelke M, Anzola GP, Zanette E (2001) Transcranial Doppler in the diagnosis of cardiac patent foramen ovale. Neurol Sci 22(5): 353-356.
- SAS Institute Inc. User's guide: statistics. Version 6. Cary, NC: SAS Institute Inc; 1990.
- Schwedt TJ, Demaerschalk BM, Dodick DW (2008) Patent foramen ovale and migraine: a quantitative systematic review. Cephalalgia 28: 531-540.
- Kurth T, Tzourio C, Bousser (2008) MG Migraine: a matter of the heart? Circulation 118: 1405.
- Serena J, Segura T, Perez-Ayuso MJ, Bassaganyas J, et al. (1998) The need to quantify rightto-left shunt in acute ischemic stroke. A case- control study. Stroke 29: 1322-1328.
- Elliott DA, Kirk EP, Yeoh T, Chandar S, McKenzie F, et al. Cardiac homeobox gene NKX2-5 mutations and congenital heart disease:associations with atrial septal defect and hypoplastic left heart syndrome. J Am Coll Cardiol. 2003 Jun 4;41(11): 2072-2076.
- Schott JJ, Benson DW, Basson CT, Pease W, Silberbach GM, Moak, et al. (1998) Congenital heart disease caused by mutations in the transcription factor NKX2-5. Science 281(5373): 108-111.
- Dyken ME, Somers VK, Yamada T, Ren ZY, Zimmerman B (1996) Investigating the realtionship between stroke and obstructive sleep apnoea. Stroke 27(3): 401-407.
- Wessendorf TE, Teschler H, Wang YM, Konietzko N, THilmann AF (2000) Sleep-disordered breathing among subjects with first-ever stroke. J Neurol 247: 41-47.
- Diomedi M, Placidi F, Cupini LM, Bernardi G, Silvestrini M (1998) Cerebral hemodynamic changes in sleep apnea syndrome and effect of continuous positive airway pressure treatment. Neurology 51(4): 1051-1056.
- Voogel AJ, van Steenwijk RP, Karemaker JM, van Montfrans GA (1999) Effects of treatment of obstructive sleep apnea on circadian hemodynamics. J Auton Nerv Syst 77(2-3): 177-183.
- Akashiba T, Minemura H, Yamamoto H, Kosaka N, Saito O, et al.(1999) Nasal continuous positive airway pressure changes blood pressure "non-dippers” to "dippers” in subjects with obstructive sleep apnea. Sleep 22(7): 849-853.
- Thaler D, Carroll J, Saver J PFO closure reduces long term recurrence of ischemic stroke: Final primary and secondary population results from the RESPECT multicenter trial. ISC Abstract 71.






























