Effect of a Novel Curcumin Formulation on Adaptogenic and Endogenous Anti-oxidant /Oxidative Stress in Chronic Mild Unpredictable Stress Model in Rats
*Shankaranarayanan Jeyakodi and Jayant Deshpande
Department of Pharmacy, Omni Active Health Technologies Ltd, Indian
Submission: July 13, 2016; Published: September 01, 2016
*Corresponding author: Shankaranarayanan Jeyakodi, Omni Active Health Technologies Ltd, Wagle Industrial Estate, Thane West, Maharashtra, India.
How to cite this article: Shankaranarayanan J, Jayant D. Effect of a Novel Curcumin Formulation on Adaptogenic and Endogenous Anti-oxidant /Oxidative Stress in Chronic Mild Unpredictable Stress Model in Rats. J of Pharmacol & Clin Res. 2016; 1(4): 555567. DOI: 10.19080/JPCR.2016.01.555567
Abstract
This study investigates the anti-oxidant effect of Curcumin formulation (UltraSol CurcuWin) on stress induced rats. Fisher 344 N male rats that were 6-8 weeks old were chosen for the study. 30 animals were divided into 5 groups: I, II, III, IV and V. Group I was taken as the control group and was administered only with vehicle. All other groups were administered with various stressors every 24 hours over a period of 3 weeks. Group II which is administered with stressors and vehicle is used as a positive control. Curcumin powder 95% was administered to Group III with a dose of 100 mg/kg/day. UltraSol CurcuWin is administered per orally at 100 mg/kg/day and 200 mg/kg/day to groups IV & V respectively
Anti-oxidant effect was evaluated based on behavioral studies and tissue necropsy studies. Data revealed that UltraSol CurcuWin provided significant protection against loss of body weight (p<0.01), ameliorated anxiety (p<0.01) and depression behavior (p<0.01). Tissue necropsy studies on UltraSol CurcuWin20 % administered animals indicated a significant lower levels of Glutathione (GSH) in brain (p<0.01), heart (p<0.01) and liver (p<0.05). Lipid Peroxide (LPO) was significantly higher in brain (p<0.05), heart (p<0.01), liver (p<0.05) and kidney (p<0.01). Activity of Catalase decreased in the brain (p<0.05), heart (p<0.01) and kidney (p<0.01). The levels of super oxide dismutase (SOD) were significantly lower (p<0.01) in brain, heart and kidney. There is no significant difference between the plasma cortisol levels of unstressed rats and those that were administered with high dose UltraSol CurcuWin 20 % (Group V). There is no effect on protein content between the control and treatment groups. Histology studies on the hypothalamus indicated no stress related or treatment related changes.
In conclusion UltraSol CurcuWin showed a potential role on endocrine function and also demonstrated the ability in alleviating the stress induced changes. UltraSol CurcuWin can act as a potent adaptogen.
Keywords: Curcumin longa, Antioxidant activity, glutathione, cortisol, lipid peroxide, super oxide dismutase, catalase. Gum Acacia; Erythrocyte in the blood; Inflammations.
Introduction
Curcumin is a versatile compound having a plethora of pharmacological activities. Curcumin has been shown to exhibit antioxidant, anti-inflammatory, [1-4] anti microbial, and anticarcinogenic [5-9] activities. Additionally, the hepato- and nephro-protective, [10-12] thrombosis supressing, [13] myocardial infarction protective, [14-16]. hypoglycemic, [17-20] and anti rheumatic [21] effects of curcumin are also well established. Various animal models [22,23] or human studies [24-27] proved that curcumin is extremely safe even at very high doses. For example, three different phase I clinical trials indicated that curcumin, when taken as high as 12 g per day, is well tolerated [25-27]. Similarly, the efficacy of curcumin in various diseases including cancer has been well established [28]. Several clinical studies dealing with the efficacy of curcumin in humans can also be cited [29,30]. The pharmacological safety and efficacy of curcumin makes it a potential compound for treatment and prevention of a wide variety of human diseases.
Curcumin has been proved to be less bio available [28-33]. The absorption, bio distribution, metabolism, and elimination studies of curcumin have, unfortunately, shown only poor absorption, rapid metabolism, and elimination of curcumin as major reasons for poor bioavailability of this interesting poly phenolic compound [33-54].
Different strategies have been pursued to improve the absorption of curcumin including nanocrystals, emulsions, liposomes, self-assemblies and nanogels [55]. In animals, coadministration of curcumin with an extract obtained from the black pepper has been shown to increase the absorption (AUC) of curcumin by 1.5-fold. Whereas, a complex of curcumin with phospholipids increased absorption by 3.4-fold [56] and a formulation of curcumin with a micellar surfactant (polysorbate) has been shown to increase the absorption of curcumin in mice 9.0-fold [57]. A micro emulsion system of curcumin, which consists of Capryol 90 (oil), Cremophor RH40 (surfactant), and Transcutol P aqueous solution (co-surfactant) has been shown to increase the relative absorption in rats by 22.6-fold [58]. Polylactic-co-glycolic acid (PLGA) and PLGA-polyethylene glycol (PEG) (PLGA-PEG) blend nanoparticles increased curcumin absorption by 15.6- and 55.4-fold, respectively, compared to an aqueous suspension of curcumin in rats [59].
Food-grade formulations to enhance the absorption of curcumin have been studied in human clinical trials [60-61]. A proprietary formulation of curcumin has been developed retaining and utilizing more components of the raw turmeric root which are usually eliminated during extraction. The combination of curcuminoids and volatile oils of turmeric rhizome (CTR) resulted in a 6.9-fold increase in human absorption of curcumin [60]. The inclusion of curcumin in a lipophilic matrix (Phytosomes, Curcumin: Soy Lecithin: Microcrystalline Cellulose 1:2:2, CP) has been shown to increase the relative human absorption of curcumin by 19.2-fold [61].
A formulation made by mixing curcumin with glycerin, gum ghatti, and water, followed by wet milling and dispersion by highpressure homogenization has been shown the increase curcumin appearance in the blood by 27.6-fold [62]. A novel curcumin formulation which was made water soluble by dispersing curcumin and antioxidants (tocopherol and ascorbyl palmitate) on a water-soluble carrier such as polyvinyl pyrrolidone has been shown to have greater antidepressant action compared to conventional curcumin [63]. One study conducted at our research center demonstrated that a combination of hydrophilic carrier, cellulosic derivatives and natural antioxidants significantly increases curcuminoid appearance in the blood in comparison to unformulated standard curcumin [64].
Although molecular mechanisms of action of curcumin are not fully understood, curcumin has proven to be a safe agent for the treatment of various ailments. Our formulation has demonstrated significantly improved solubility in vitro and therefore is expected to have more bioavailability. Under routine conditions curcumin proved to be an ailment for various disorders that arise due to oxidative stress. Obviously a formulation with improved bioavailability must eliminate or decrease stress induced changes [1-30]. In this study we investigated the effectiveness of our Curcumin formulation (UltraSol CurcuWin) on adaptogenic and endogenous antioxidant /oxidative stress in chronic mild unpredictable stress model in rats and compared with that of the curcumin powder.
Materials and Methods
Chemicals & Reagents
Sodium pyrophosphate, NBT, PMS, NADH, glacial acetic acid, n-butanol, Griess reagent(sulphanilamide, orthophosphoric acid and naphthyl ethyl diamine), potassium chloride, Bradford reagent, trichloro acetic acid, 2-nitrobenzoic acid, sodium chloride , Sodium phosphate monobasic, Sodium phosphate dibasic, TBA, H2O2, Potassium dichromate, HCL, Sodium hydroxide all the chemicals purchased from SRL, Mumbai, India. All other reagents used were of analytical grade unless specified. Carboxy methyl cellulose (at 0.5% w/v is used as a vehicle, Ultrasol CurcuWin (20% with & purity 22.23 % is prepared in our facility. Curcumin Powder with purity 95.36 % is prepared in our facility.
Instrumentation
Spectrophotometer (25, Thermo Fisher Scientific, USA) is used for all the estimation. Microscopical examination is made using a Motic DMB1-2MP, China. Data analysis is performed using Graph Pad Prism Version 5.0 software for deriving statistical parameters.
The elevated plus maze was made of wood painted in black, and consisted of two opposite open arms (50 × 10 × 0.5 cm), an open platform (10 ×10 cm) in the center and two opposite closed arms (50 ×10 ×40 cm). Open field maze was made of black wood and consisted of a floor (96 × 96 cm) with 50 cm walls The box floor was painted with white lines (6 mm) to form 16 equal squares.
Management of animals
The institutional animal ethics committee of Sri Ramachandra University, Chennai approved the study protocol. The study was performed at CEFT, which is an approved laboratory vide registration number 189/PO/bc/1999/CPCSEA. All ethical practices as laid down in the guidelines for animal care were followed during the study. Fisher 344 N male rats aged between 6-8 weeks were used in this study, with the experiment lasting for a period of 3 weeks. The animals were procured from the breeding stock from National Institute of Nutrition, Hyderabad, India. Upon procurement, the rats were kept at the Center for toxicology and developmental research, Sri Ramachandra, University, Chennai. Animals are randomized based on stratified body weight method and divided into 5 groups of 6 animals each (I, II, III, IV and V) and were acclimatized for 5 days. The animals were marked with black permanent market at the base of the tail. Standard rodent feed purchased from M/s Provimi Animal Nutrition India was provided ad libitum. Mili Q RiOs filtered water was provided ad libitum.
The animal room was well ventilated with 12-15 air exchanges/hour and maintained with a temperature range of 19-23 ºc and humidity ranging from 30-70% throughout the study. Animal room was built with automatic 12hr light and dark cycles. Animals were housed in polypropylene cages with de-dusted and autoclaved husk as bedding material. Caging and bedding materials were changed on alternate days.
Administration of compounds and Induction of stress
Group I was taken as the control group and was administered with vehicle. All other groups were administered with various stressors every 24 hours over a period of 3 weeks. Group II which is administered with stressors and vehicle is used as a positive control. Curcumin powder 95% was administered with 100 mg/kg/day per orally to Group III. UltraSol CurcuWin20 % is administered per orally at 100 mg/kg/day and 200 mg/ kg/day to groups IV & V respectively [65-66]. Test /Reference compounds were formulated freshly prior to administration every day using 0.05% w/v CMC as vehicle. Test / reference items were administered 30-45 minutes before the induction of stress to the animals by oral gavage using ball tipped 18G needle and polypropylene syringe. The dose volume is fixed at 10 ml/ kg body weight.
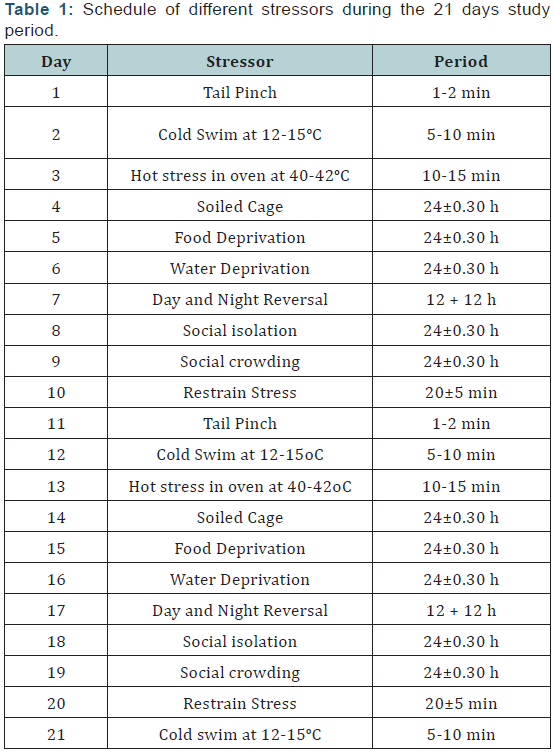
Stressors were administered using the procedure described by Wu et al. [67]. Accordingly, except for the 24 hour stressors, all stressors were administered once daily in the morning between 9.30 and 12.30. A complete schedule of different stressors administered during the 21 day study was given in (Table 1). The different stressors were administered at an interval of at least 7 days. With the exception of cold swim stress that was administered 3 times during the study, all other stressors were administered at least twice during the study period.
Behavioral studies
Measurement of anxiety in elevated plus maze
Anxiety behavior was assessed using elevated plus maze test following Lister method [68]. The elevated plus maze was made of wood painted in black, and consisted of two opposite open arms (50 × 10 × 0.5 cm), an open platform (10 ×10 cm) in the center and two opposite closed arms (50 ×10 ×40 cm). The entire maze was elevated 50 cm from the floor in a dimly lit room (20 Lux). Rat was placed on the central platform facing one of the open arms. The time spent and the numbers of entries in the open and closed arms were recorded for a period of three minutes. An entry was considered when all the four paws placed in one arm. The maze was cleaned following each trial to remove any residue or odors.
Measurement of depression in open field exploratory test
Depression was assessed using open field test following Lister [69]. The open field maze was made of black wood and consisted of a floor (96 × 96 cm) with 50 cm walls. The apparatus was illuminated with a low intensity diffuse light (45 W) situated 45 cm above the floor level. The box floor was painted with white lines (6 mm) to form 16 equal squares. During a three min observation period, the rat was placed at one corner of the apparatus facing the wall. The number of squares crossed (ambulation), rearing, grooming, immobility period, urination and fecal pellets were recorded. The maze was cleaned following each trial to remove any residue or odors.
Histology studies
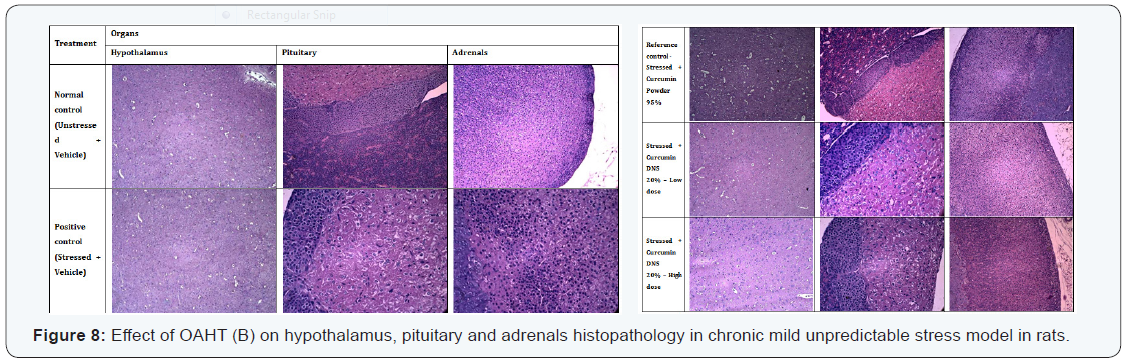
All animals were euthanized at the end of the experimental period using anesthetic ether in closed chamber. Adrenal glands, Pituitary gland and brain was collected and fixed in 10% neutral buffered formalin for 48 hours. The tissues were processed for paraffin embedment, sectioned and then stained with Hematoxylin and Eosin [70] for microscopic evaluation.
Biochemical Studies
After completion of the study, the enzymatic antioxidants levels in the brain, heart, liver and kidney were estimated using standard protocols. The antioxidant markers such as Superoxide dismutases Kakkar et al. [71], Catalase Beers & Sizer [72], lipid peroxidation Ohkawa et al. [73] and reduced Glutathione Jollow et al. [74] were estimated along with Total protein Lowry et al. [75] were available in literature. All methods were standardized in the laboratory before testing the study samples. The stress hormone cortisol is estimated by ELISA method [76]. Values were expressed in mean ± SEM; n= 6 animals/group; Mean difference between the groups were analyzed using one way ANOVA followed by tukey’s multiple comparison test in graph pad prism 5.0.
Results and Discussion
Behavioral studies
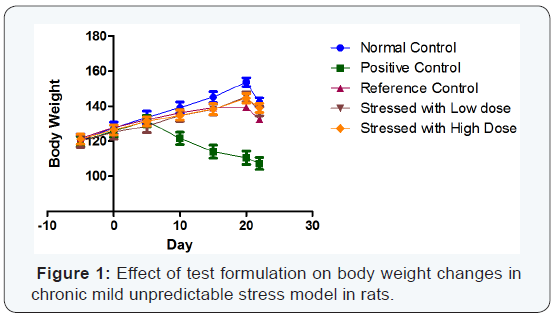
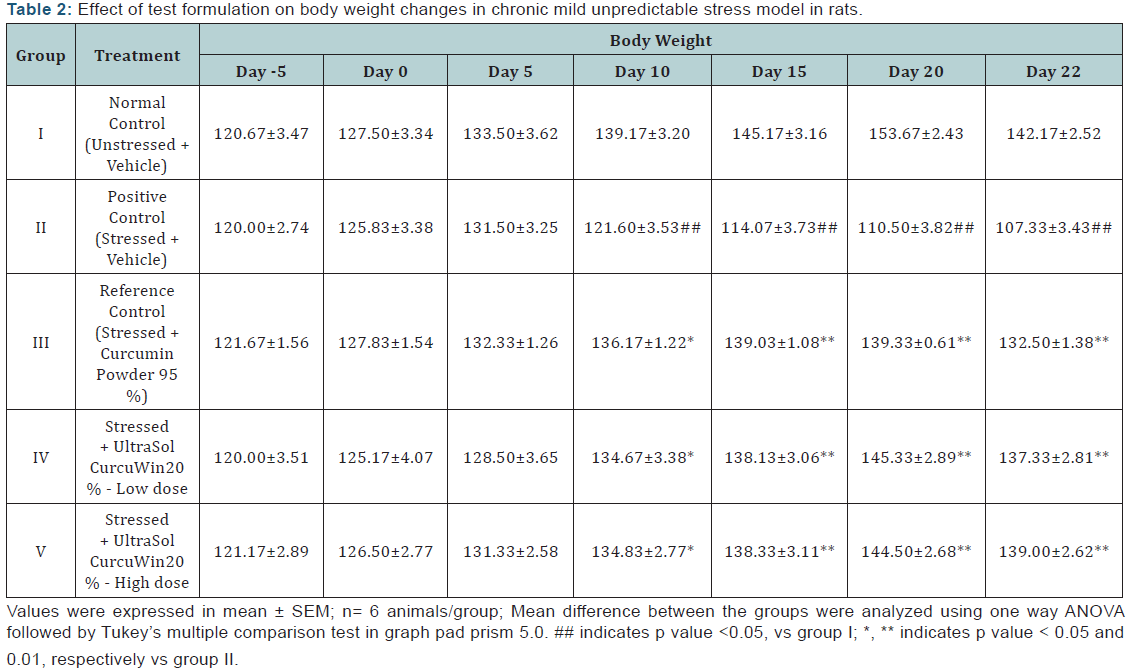
Body weight: A significant decrease (p< 0.01) in body weight was observed in stress induced rats when compared to unstressed rats from day 10 till the completion of the study. UltraSol CurcuWin treatment significantly ameliorated these changes in comparison to vehicle treated stressed rats [Day 10 (p < 0.05), 15 (p<0.01), 20 (p<0.01) and 22 (p<0.01)]. The values were found to be comparable with Curcumin Powder 95%. The drop in body weight on day 22 in all the groups is due to fasting. The results are given in (Table 2) & (Figure 1).
Open field exploratory behavior: A significant increase (p<0.01) in immobility period and decrease in ambulation (p<0.01), rearing (p<0.01) and grooming (p<0.05) were observed in vehicle treated stressed rats when compared to unstressed rats. UltraSol CurcuWin– low and high dose treatment significantly and dose dependently increased (p<0.05 and 0.01, respectively) ambulatory behavior in comparison to vehicle treated stressed rats. However, treatment with Curcumin Powder 95% and UltraSol CurcuWin– low and high dose produced a non-significant decreased in immobility period and increase in rearing and grooming behavior when compared to the vehicle treated stressed rats. The results are given in (Table 3) & (Figure 2)


Elevated plus maze: A significant increase (p<0.01) in number of entries and time spent in closed arms and significant decrease number of entries (p<0.05) and time spent (p<0.01) in open arms were observed in stressed rats when compared to unstressed rats. UltraSol CurcuWin treatment significantly decreased the number of entries (p<0.01) and time spent
(p<0.01 in high dose) in closed arms in comparison to stressed rats. In comparison to the vehicle treated stressed rats, rats treated with both the doses of UltraSol CurcuWin produced a non-significant increase in number of entries and time spent in open arms. The results are given in (Table 4).

Biochemical studies
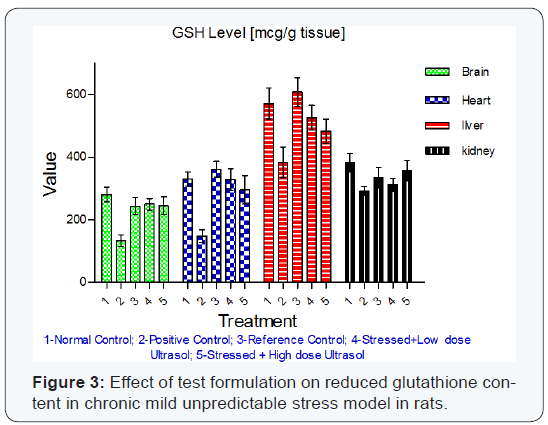
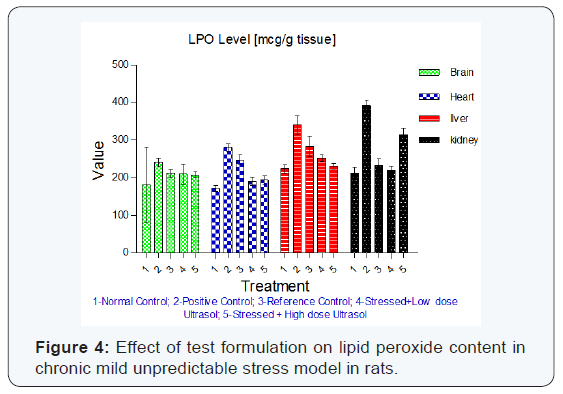
Reduced glutathione: Reduced glutathione content was found to be significantly decreased in brain (p<0.01), heart (p<0.01) and liver (p<0.05) tissues of stressed rats when compared to unstressed rats. Treatment with UltraSol CurcuWin significantly increased reduced glutathione content in brain [p<0.05 in UltraSol CurcuWin– low and high dose] and heart [p<0.01 and 0.05 in UltraSol CurcuWin– low and high dose, respectively] tissues when compared to vehicle treated stressed rats. However, a non-significant increase in liver glutathione content was observed in UltraSol CurcuWin treated rats in comparison to vehicle treated stressed rats. The values were found to be comparable with Curcumin Powder 95%. The results are given in (Table-5) & (Figure 3).
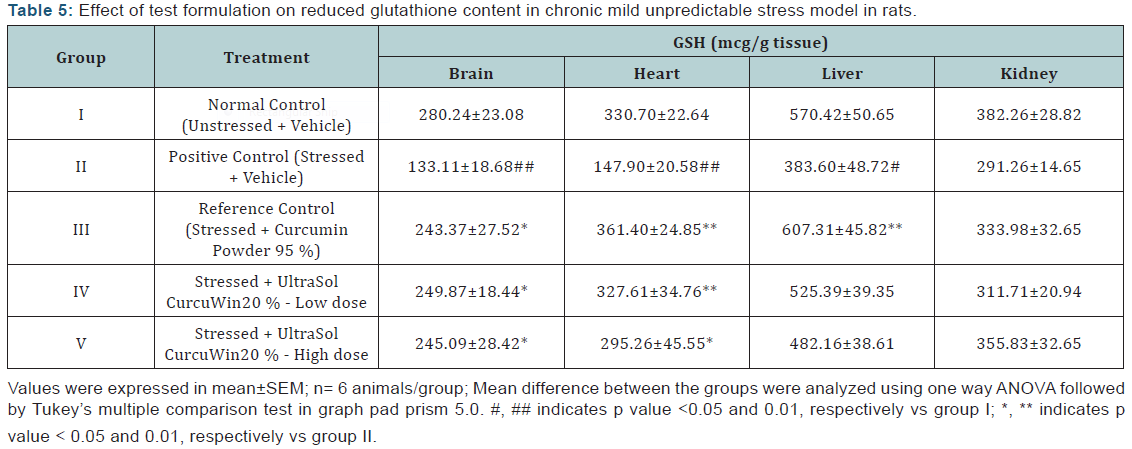
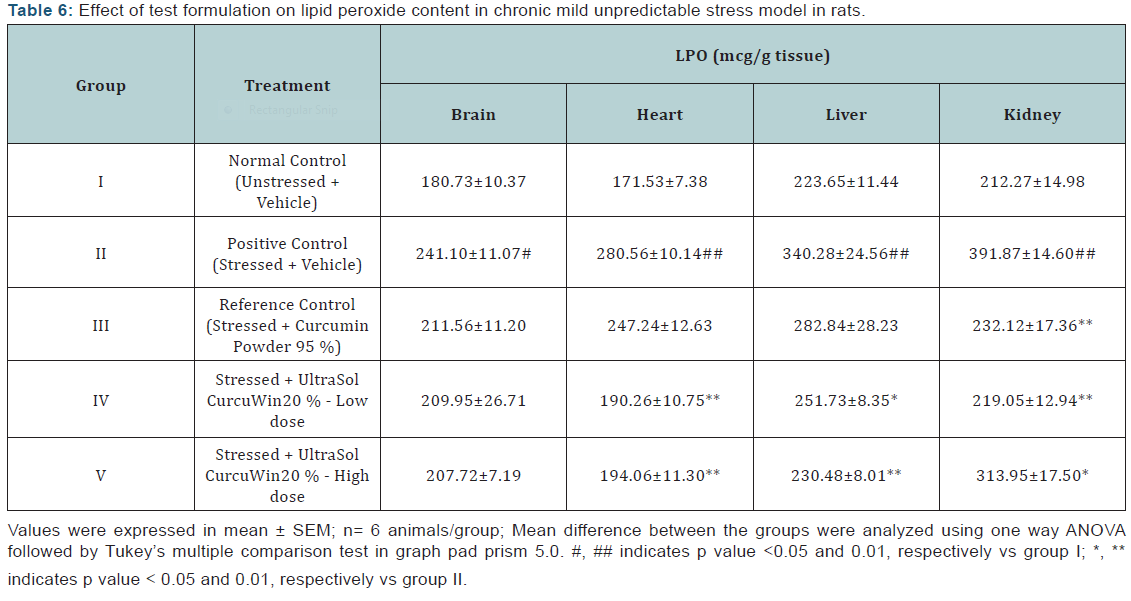
Lipid peroxide (LPO): A significant increase in LPO content was observed in brain, heart, liver and kidney (p<0.05, 0.01, 0.01 and 0.01, respectively) tissues of stressed rats when compared to unstressed rats. In comparison to vehicle treated stressed rats, UltraSol CurcuWin treatment significantly decreased these alterations in heart [p<0.01 in UltraSol CurcuWin– low and high dose], liver [p<0.05 and 0.01 in UltraSol CurcuWin– low and high dose, respectively] and kidney [p<0.05 and 0.01 in UltraSol CurcuWin– high and low dose, respectively] tissues. The protective effect of UltraSol CurcuWin was found to be better than Curcumin Powder 95% in heart, liver and kidney tissues. The results are given in (Table 6) & (Figure 4).
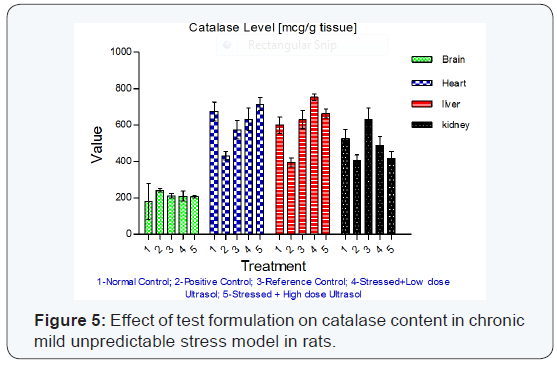
Catalase activity: A significant decrease in catalase activity was observed in brain, heart and kidney (p<0.05, 0.01 and 0.01, respectively) tissues of vehicle treated stressed rats when compared to unstressed rats. UltraSol CurcuWin treatment significantly increased the catalase activity in brain [p<0.01 in UltraSol CurcuWin– high dose], heart [p<0.01 in UltraSol CurcuWin– low and high dose] and kidney [p<0.01 in UltraSol CurcuWin– low and high dose] tissues when compared to vehicle treated stressed rats. The values were found to comparable with that of reference control, Curcumin Powder 95%. UltraSol CurcuWin increased catalase activity better than Curcumin Powder 95% in brain, heart and kidney tissues. The results are given in (Table 7) & (Figure 5).

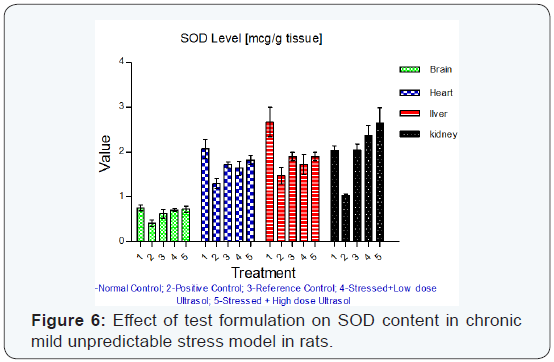
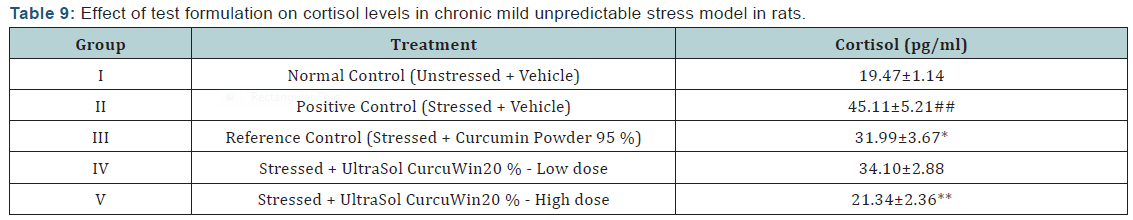
Superoxide dismutase (SOD) activity: Superoxide dismutase activity was found to be significantly decreased (p<0.01) in brain, heart, liver and kidney tissues of stressed rats when compared to unstressed rats. In comparison to vehicle treated stressed rats, UltraSol CurcuWin treatment significantly increased SOD activity in brain [p<0.05 in UltraSol CurcuWin– low and high dose], heart [p<0.05 in UltraSol CurcuWin– high dose] and kidney [p<0.01 in UltraSol CurcuWin– low and high dose] tissues. However, a non-significant increase in SOD activity was observed in liver tissues of UltraSol CurcuWin treatment when compared to vehicle treated stressed rats. Further, UltraSol CurcuWin increased SOD activity better than Curcumin Powder 95% in brain and kidney tissues. The results are given in (Table 8) & (Figure 6).
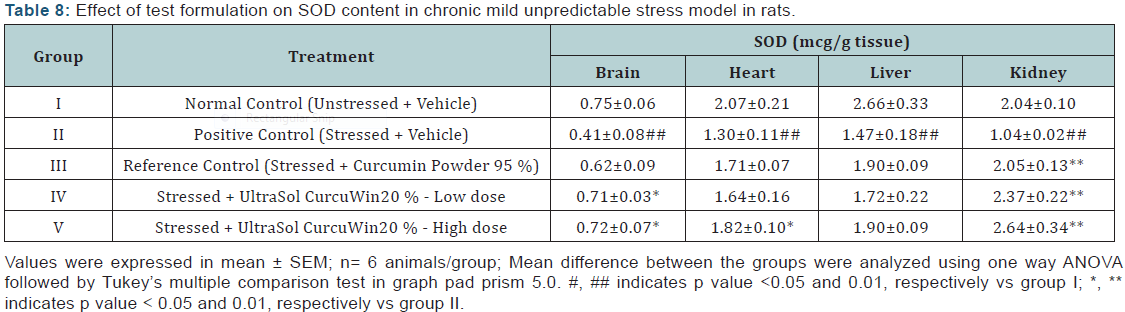
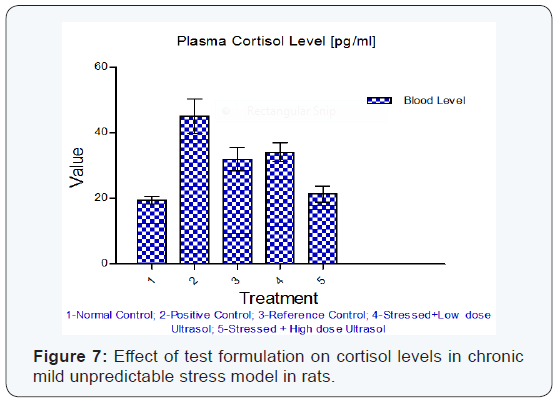
Cortisol: A significant increase (p<0.01) in plasma cortisol level was observed in stressed rats when compared to unstressed rats. High dose of OAHT (B) treatment significantly decreased (p<0.01) the cortisol level in comparison to vehicle treated stressed rats. The values were found to be comparable with that of reference drug, OAHT (A). OAHT (B) decreased plasma cortisol levels better than OAHT (A) stressed animals. The results are given in (Table 9) & (Figure 7).
Total Protein: The levels of total protein quantified using the Lowry method [75] indicated no significant differences in the organs of treated and untreated groups. This indicates that turmeric safe for administration and that damage to the DNA or protein synthesis is not impaired by the administration of curcumin and/or due to the presence of stress.
Histopathology Studies
Histopathological examination (Figure 8) of the three organs (hypothalamus, pituitary and adrenal glands) from the stressed rats and Curcumin Powder 95%, UltraSol CurcuWin – low and high dose treated rats revealed no stress related or treatment related changes and remained apparently normal in comparison to the unstressed rats. Reason for the no alterations in the pathology may be due to less intensity and duration of the stress administered to the animals.
Summary and Conclusion
Each cell in the human body maintains a condition of homeostasis between the oxidant and antioxidant species [76]. Up to 1–3% of the pulmonary intake of oxygen by humans is converted into ROS [77]. Under conditions of normal metabolism, the continuous formation of ROS and other free radicals is important for normal physiological functions like generation of ATP, various catabolic and anabolic processes and the accompanying cellular redox cycles. However, stress condition is known to cause the excessive release of free radicals either due to endogenous biological or exogenous environmental factors, such as chemical exposure, pollution, or radiation. The overall physiological impact of these factors and the adaptation ability of the body determine the variations in growth, development, productivity, and health status of the animals [78-80]. Strong and sustained exposure to stress [79,81,82] may result in higher energy negative balance and may ultimately result in reduction in adaptation mechanisms, increase in the susceptibility to infection by pathogens, decline in productivity, and finally a huge economical loss [79,83,84]. ROS can attack the lipids of cell membranes and DNA and protein content of the cells with lipid peroxidation of cellular membranes, calcium influx, and mitochondrial swelling and lysis [85-86].
In several studies, Curcumin showed significantly poor bioavailability. While pharmacological actions are usually dose dependent, the efficacy can also impaired by poor absorption and distribution characteristics. Our test formulation of Curcumin is aimed to increase the bioavailability and thus the overall pharmacological effect. In several animal models it has been demonstrated to exert potent anti-inflammatory and anti-tumor and hypolipidemic properties. In view of these properties, we have investigated the effectiveness of Curcumin as an antioxidant under stress conditions [87-88].
Stress can be acute, episodic or chronic depending on its occurance, duration and treatment approaches. Symptoms such as emotional distress, anger or irritability and depression are most commonly attributed to acute stress. Episodic stress is mostly due to disorderliness and lack of proper planning leading to chaos and misery. Chronic stress arise from traumatic, prolonged experiences and generally end up with suicide, violence, heart attack, stroke and, perhaps, even cancer. Keeping in view of the various types of stress and their impact on the physiological functions, we had designed the protocol to monitor the behavioral, biochemical and histological effects. From (Table 2), it is clearly evident that animals prone to stress have eventually demonstrated a loss of body weight. In comparison to the unstressed group, the group treated with Curcumin powder showed relatively less body weight growth. Animals treated with our Curcumin test formulation displayed significant and comparable levels of recovery from stress. From the behavioral studies (Table 3 & 4) it can be inferred that the curcumin test formulation ameliorated anxiety and depressive behavior and thus Curcumin can act as a potent adaptogen.
Curcumin has strong free radical scavenging activity and it is evident from this study that it can protect the biological systems against oxidative stress. Comparitive analysis of the curcumin formulations with that of the Curcumin powder revealed that the content of reduced glutathione did not vary significantly in the brain. In the heart and kidney, the amount of reduced glutathione is relatively low in the animals treated with Curcumin test formulation. In comparison with the animals treated with Curcumin powder, there is a drastically lower level of reduced glutathione in the liver of animals treated with test formulation. This can attributed to the hepatic metabolism of Curcumin.
Comparison of the LPO content indicated that Curcumin test formulation displayed high effectiveness in lowering the LPO in the heart & liver. The LPO content in the brain for rats treated with either the test formulation or the powder did not show significant difference. Except in the liver, the activity of the catalase is significantly higher in the animals treated with test formulation as compared with the animals treated with Curcumin powder. The animals treated with the test formulation of Curcumin showed increased SOD activity than those treated with Curcumin powder.
In response to stress, cortisol is released into the body as a homeostatic mechanism. A comparison of the cortisol levels in the positive control and treated groups indicated that Curcumin is effective in reducing the levels of cortisol. There is insignificant difference between the levels of cortisol in animals treated with high dose of test formulation and the animals of the normal control group. Therefore Curcumin has a potential role on endocrine function and also the ability to alleviate the stress induced changes. Histological studies indicated no stress related or therapy related changes and remained apparently normal in comparison with the unstressed rats.
As expected the levels of biochemical and histological markers due to stress induced changes are significantly less. Also, our formulation proved to be effective even in chronic conditions of stress. Therefore Curcumin can act as a potent antioxidant and as an adaptogen.
References
- Sharma OP (1976) Antioxidant activity of curcumin and related compounds. Biochem Pharmacol 25(15): 1811-1812.
- Ruby AJ, Kuttan G, Babu KD, Rajasekharan KN, Kuttan R (1995) Antitumour and antioxidant activity of natural curcuminoids. Cancer Lett 94(1): 79-83.
- Sugiyama Y, Kawakishi, S, Osawa T (1996) Involvement of the β-diketone moiety in the antioxidative mechanism of tetrahydrocurcumin. Biochem Pharmacol 52(4): 519-525.
- Srimal RC, Dhawan BN (1973) Pharmacology of diferuloyl methane (curcumin), a non-steroidal anti-inflammatory agent. J Pharm Pharmacol 25(6): 447-452.
- Jordan WC, Drew CR (1996) Curcumin–a natural herb with anti-HIV activity. J Natl Med Assoc 88(6): 333.
- Mahady GB, Pendland SL, Yun G, Lu ZZ (2002) Turmeric (Curcuma longa) and curcumin inhibit the growth of Helicobacter pylori a group 1 carcinogen. Anticancer Res 22(6C): 4179-4181.
- Kim MK, Choi GJ, Lee HS (2003) Fungicidal property of Curcuma longa L. rhizome-derived curcumin against phytopathogenic fungi in a greenhouse. J Agric Food Chem 51(6): 1578-81.
- Reddy RC, Vatsala PG, Keshamouni VG, Padmanaban G, Rangarajan PN (2005) Curcumin for malaria therapy. Biochem Biophys Res Commun 326(2): 472-474.
- Kuttan R, Bhanumathy P, Nirmala K, George MC (1985) Potential anticancer activity of turmeric (Curcuma longa). Cancer Lett 29(2): 197-202.
- Kiso Y, Suzuki Y, Watanabe N, Oshima Y, Hikino H (1983) Antihepatotoxic principles of Curcuma longa rhizomes. Planta Med 49(3): 185-187.
- Venkatesan N (1998) Curcumin attenuation of acute adriamycin myocardial toxicity in rats. Br J Pharmacol 124(3): 425-427.
- Venkatesan N, Punithavathi D, Arumugam V (2000) Curcumin prevents adriamycin nephrotoxicity in rats. Br J Pharmacol 129(2): 231-234.
- Srivastava R, Dikshit M, Srimal RC, Dhawan BN (1985) Antithrombotic effect of curcumin. Thromb Res 40(3): 413-417.
- Dikshit M, Rastogi L, Shukla R, Srimal RC (1995) Prevention of ischaemia-induced biochemical changes by curcumin & quinidine in the cat heart. Indian J Med Res 101: 31-35.
- Nirmala C, Puvanakrishnan R (1996) Protective role of curcumin against isoproterenol induced myocardial infarction in rats. Mol Cell Biochem 159(2): 85-93.
- Nirmala C, Puvanakrishnan R (1996) Effect of curcumin on certain lysosomal hydrolases in isoproterenol-induced myocardial infarction in rats. Biochem Pharmacol 51(1): 47-51.
- Srinivasan M (1972) Effect of curcumin on blood sugar as seen in a diabetic subject. Indian J Med Sci 26(4): 269-270.
- Babu PS, Srinivasan K (1995) Influence of dietary curcumin and cholesterol on the progression of experimentally induced diabetes in albino rat. Mol Cell Biochem 152(1): 13-21.
- Babu PS, Srinivasan K (1997) Hypolipidemic action of curcumin the active principle of turmeric (Curcuma longa) in streptozotocin induced diabetic rats. Mol Cell Biochem 166(1-2): 169-175.
- Arun N, Nalini N (2002) Efficacy of turmeric on blood sugar and polyol pathway in diabetic albino rats. Plant Foods Hum Nutr 57(1): 41-52.
- Deodhar SD, Sethi R, Srimal RC (1980) Preliminary study on antirheumatic activity of curcumin (diferuloyl methane). Indian J Med Res 71: 632-634.
- Shankar TN, Shantha NV, Ramesh HP, Murthy IA, Murthy VS (1980) Toxicity studies on turmeric (Curcuma longa): acute toxicity studies in rats, guineapigs & monkeys. Indian J Exp Biol 18(1): 73-75.
- Qureshi S, Shah, AH, Ageel AM (1992) Toxicity studies on Alpinia galanga and Curcuma longa. Planta Med 58(2): 124-127.
- Lao CD, Demierre MF, Sondak VK (2006) Targeting events in melanoma carcinogenesis for the prevention of melanoma. Expert ReV Anticancer Ther 6(11): 1559-1568.
- Lao CD, Ruffin MT, Normolle D, Heath DD, Murray SI, et al. (2006) Dose escalation of a curcuminoid formulation. BMC Complement Altern Med 6 10.
- Cheng AL, Hsu CH, Lin JK, Hsu MM, Ho YF, et al. (2001) Phase I clinical trial of curcumin a chemopreventive agent in patients with high-risk or pre-malignant lesions. Anticancer Res 21(4B): 2895-2900.
- Shoba G, Joy D, Joseph T, Majeed M, Rajendran R, et al. (1998) Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med 64(4): 353-356.
- Aggarwal BB, Sundaram C, Malani N, Ichikawa H (2007) Curcumin the Indian solid gold. AdV Exp Med Biol 595: 1-75.
- Aggarwal BB, Kumar A, Bharti AC (2003) Anticancer potential of curcumin preclinical and clinical studies. Anticancer Res 23(1A): 363- 398.
- Hsu CH, Cheng AL (2007) Clinical studies with curcumin. AdV Exp Med Biol 595: 471-480.
- Wahlstrom B, Blennow GA (1978) study on the fate of curcumin in the rat. Acta Pharmacol Toxicol (Copenhagen) 43(2): 86-92.
- Ravindranath V, Chandrasekhara N (1980) Absorption and tissue distribution of curcumin in rats. Toxicology 16(3): 259-265.
- Ravindranath V, Chandrasekhara N (1981) Metabolism of curcumin studies with [3H] curcumin. Toxicology 22(4): 337-344.
- Perkins S, Verschoyle RD, Hill K, Parveen I, Threadgill MD, et al. (2002) Chemopreventive efficacy and pharmacokinetics of curcumin in the min/+ mouse a model of familial adenomatous polyposis. Cancer Epidemiol. Biomarkers PreV 11(6): 535-540.
- Sharma RA, Euden SA, Platton SL, Cooke DN, Shafayat A, et al. (2004) Phase I clinical trial of oral curcumin: biomarkers of systemic activity and compliance. Clin Cancer Res 10(20): 6847-6854.
- Yang KY, Lin LC, Tseng TY, Wang SC, Tsai TH (2007) Oral bioavailability of curcumin in rat and the herbal analysis from Curcuma longa by LCMS/ MS. Biomed Life Sci 853(1-2): 183-189.
- Maiti K, Mukherjee K, Gantait A, Saha BP, Mukherjee PK (2007) Curcumin-phospholipid complex: Preparation, therapeutic evaluation and pharmacokinetic study in rats. Int J Pharm 330(1-2): 155-163.
- Marczylo TH, Verschoyle RD, Cooke DN, Morazzoni P, Steward WP, et al. (2007) Comparison of systemic availability of curcumin with that of curcumin formulated with phosphatidylcholine. Cancer Chemother Pharmacol 60(2): 171-177.
- BRavindranath V, Chandrasekhara N (1981) In vitro studies on the intestinal absorption of curcumin in rats. Toxicology 20(2-3): 251-257.
- Garcea G, Jones DJ, Singh R, Dennison AR, Farmer PB, et al. (2004) Detection of curcumin and its metabolites in hepatic tissue and portal blood of patients following oral administration. Br J Cancer 90(5): 1011-1015.
- Garcea G, Berry DP, Jones DJ, Singh R, Dennison AR, et al. (2005) Consumption of the putative chemopreventive agent curcumin by cancer patients: assessment of curcumin levels in the colorectum and their pharmacodynamic consequences. Cancer Epidemiol Biomarkers PreV 14(1): 120-125.
- Hoehle SI, Pfeiffer E, Solyom AM, Metzler M (2006) Metabolism of curcuminoids in tissue slices and subcellular fractions from rat liver. J Agric Food Chem 54(3): 756-764.
- Holder GM, Plummer JL, Ryan AJ (1978) The metabolism and excretion of curcumin (1,7-bis-(4-hydroxy-3-methoxyphenyl)-1,6- heptadiene- 3,5-dione) in the rat. Xenobiotica 8(12): 761-768.
- Ireson C, Orr S, Jones DJ, Verschoyle R, Lim CK, et al. (2001) Characterization of metabolites of the chemopreventive agent curcumin in human and rat hepatocytes and in the rat in vivo, and evaluation of their ability to inhibit phorbol ester-induced prostaglandin E2 production. Cancer Res 61(3): 1058-1064.
- Asai A, Miyazawa T (2000) Occurrence of orally administered curcuminoid as glucuronide and glucuronide/sulfate conjugates in rat plasma. Life Sci 67(23): 2785-2793.
- Nirmala C, Puvanakrishnan R (1996) Effect of curcumin on certain lysosomal hydrolases in isoproterenol-induced myocardial infarction in rats. Biochem Pharmacol 51(1): 47-51.
- Sandur SK, Pandey MK, Sung B, Ahn KS, Murakami A, et al. (2007) Curcumin, Demethoxycurcumin, Bisdemothoxycurcumin, Tetrahydrocurcumin, and Turmerones Differentially Regulate Antiinflammatory and Antiproliferative Responses Through a ROS Independent Mechanism. Carcinogenesis
- Pfeiffer E, Hoehle SI, Walch SG, Riess A, Solyom AM, et al. (2007) Curcuminoids form reactive glucuronides in vitro. J Agric Food Chem 55(2): 538-544.
- Kim JM, Araki S, Kim DJ, Park CB, Takasuka N, et al. (1998) Chemopreventive effects of carotenoids and curcumins on mouse colon carcinogenesis after 1,2-dimethylhydrazine initiation. Carcinogenesis 19(1): 81-85.
- Okada K, Wangpoengtrakul C, Tanaka T, Toyokuni S, Uchida K, et al. (2001) Curcumin and especially tetrahydrocurcumin ameliorate oxidative stress-induced renal injury in mice. J Nutr 131(8): 2090- 2095.
- Naito M, Wu X, Nomura H, Kodama M, Kato Y, et al. (2002) The protective effects of tetrahydrocurcumin on oxidative stress in cholesterol-fed rabbits. J Atheroscler Thromb 9(5): 243-250.
- Pari L, Amali DR (2005) Protective role of tetrahydrocurcumin (THC) an active principle of turmeric on chloroquine induced hepatotoxicity in rats. J Pharm Pharm Sci 8(1): 115-123.
- Murugan P, Pari L (2006) Effect of tetrahydrocurcumin on plasma antioxidants in streptozotocin-nicotinamide experimental diabetes. J Basic Clin Physiol Pharmacol 17(4): 231-244.
- Sharma RA, McLelland HR, Hill KA, Ireson CR, Euden SA, et al. (2001) Pharmacodynamic and pharmacokinetic study of oral Curcuma extract in patients with colorectal cancer. Clin Cancer Res 7(7): 1894-1900.
- Yallapu MM, Jaggi M, Chauhan SC (2012) Curcumin nanoformulations: a future nanomedicine for cancer. Drug Discov Today 17(1-2): 71-80.
- Liu A, Lou H, Zhao L, Fan P (2006) Validated LC/MS/MS assay for curcumin and tetrahydrocurcumin in rat plasma and application to pharmacokinetic study of phospholipid complex of curcumin. J Pharm Biomed Anal 40(3): 720-727.
- Yu H, Huang Q (2012) Improving the oral bioavailability of curcumin using novel organogel-based nanoemulsions. J Agric Food Chem. 60(21): 5373-5379.
- Hu L, Jia Y, Niu F, Jia Z, Yabg X, et al. (2012) Preparation and enhancement of oral bioavailability of curcumin using microemulsions vehicle. J Agric Food Chem. 60(29): 7137-7141.
- Khalil NM, Nascimento TC, Casa DM, Dalmolin LF, Mattos AC, et al. (2013) Pharmacokinetics of curcumin-loaded PLGA and PLGA-PEG blend nanoparticles after oral administration in rats. Colloids Surf B: Biointerfaces 101: 353-360.
- Antony B, Merina B, Iyer VS, Judy N, Lennertz K, et al. (2008) A pilot cross-over study to evaluate human oral bioavailability of BCM-95CG (Biocurcumax), a novel bio-enhanced preparation of curcumin. Indian J Pharm Sci 70(4): 445-449.
- Cuomo J, Appendino G, Dern AS, Schneider E, McKinnon TP, et al. (2011) Comparative absorption of a standardized curcuminoid mixture and its lecithin formulation. J Nat Prod 74(4): 664-669.
- Sasaki H, Sunagawa Y, Takahashi K, Imaizumi A, Fukuda H, et al. (2011) Innovative preparation of curcumin for improved oral bioavailability. Biol Pharm Bull 34(5): 660-665.
- Kulkarni SK, Akula KK, Deshpande J (2012) Evaluation of antidepressant-like activity of novel water-soluble curcumin formulations and St. John’s wort in behavioral paradigms of despair. Pharmacology 89(1-2): 83-90.
- Ralf Jäger, Ryan P Lowery, Allison V Calvanese, Jordan M Joy, Martin Purpura, et al. (2014) Comparative absorption of curcumin formulations 13: 11.
- Edakkadath RS, Korengath CP, Ramadasa NK (2010) Antioxidant activity of carotenoid lutein in vitro and in vivo. Indian Journal of Experimental Biology 48: 843-848.
- Kulkarni SK, Akula KK, Deshpande J (2012) Evaluation of antidepressant-like activity of novel water-soluble curcumin formulations and St. John’s wort in behavioral paradigms of despair. Pharmacology 89(1-20: 83-90.
- Wu L, Han H, Wang Q, Hou H, Tong H, et al. (2007) Mifepristone Repairs Region -Dependent Alteration of Synapsin I in Hippocampus in Rat Model of Depression. Neuropsychopharmacology 32: 2500-2510.
- Lister RG (1987) The use of a plus-maze to measure anxiety in the mouse, Psychopharmacology 92(2): 180-185.
- Lister J (1990) Ethologically based animal models of anxiety disorders. Pharmacol Ther 46(3): 321-340.
- Bancroft JD, Gamble M (2008) Theory and practice of histological techniques. 6th ed London Churchill Livingstone.
- Kakkar P, Das B, Viswanathan PN (1984) A modified spectrophotometric assay of superoxide dismutase. Indian Journal of Biochemistry & Biophysics 21(2): 130-132.
- Beers RF, Sizer IW (1952) A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. Journal of Biological Chemistry 195(1):133-140.
- Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Analytical Biochemistry 95(2): 351-358.
- Jollow DJ, Mitchell JR, Zampaglione N, Gillete JR (1974) Bromobenzene induced liver necrosis: protective role of glutathione and evidence for 3,4-bromobenzene as the hepatotoxic metabolite. Pharmacology 11(3): 151-169.
- Lowry OH, Rosenborough NJ, Farr AL, Randal RJ (1951) Protein measurement with Folin phenol reagent. The Journal of Biological Chemistry 193: 265-275.
- Ola Thomsson, Bodil Ström-Holst, Ylva Sjunnesson, Ann-Sofi Bergqvist (2014) Validation of an enzyme-linked immunosorbent assay developed for measuring cortisol concentration in human saliva and serum for its applicability to analyze cortisol in pig saliva. Acta Veterinaria Scandinavica 56:55.
- Poli G, Leonarduzzi G, Biasi F, Chiarpotto E (2004) Oxidative stress and cell signalling Current Medicinal Chemistry 11(9): 1163-1182.
- Sohal RS (2002) Role of oxidative stress and protein oxidation in the aging process Free Radical Biology and Medicine 33(1): 37-44.
- Lundgren K, Kalev K, Chuansi GAO, Ingvar H (2013) Effects of heat stress on working populations when facing climate change Industrial Health 51(1): 3-15.
- Kock MD, Jessup DA, Clark RK, Franti CE (1987) Effects of capture on biological parameters in free-ranging bighorn sheep (Ovis canadensis): evaluation of drop-net, drive-net, chemical immobilization and the net-gun. Journal of Wildlife Diseases 23(4): 641-651.
- VHoult JRS, Moroney MA, Paya M (1994) Actions of flavonoids and coumarins on lipoxygenase and cyclooxygenase. Methods in Enzymology 234: 443-454.
- Kock MD, Clark RK, Franti CE, Jessup DA, Wehausen JD (1987) Effects of capture on biological parameters in free-ranging bighorn sheep (Ovis canadensis): evaluation of normal, stressed and mortality outcomes and documentation of post capture survival. Journal of Wildlife Diseases 23(4): 652-662.
- Ziegler R (1991) Changes in lipid and carbohydrate metabolism during starvation in adult Manduca sexta. Journal of Comparative Physiology B 161(2): 125-131.
- Golikov N (1985) Adaptation in Farm Animals. Agropromizdat Sofia Bulgaria.
- Reed DJ (1995) Toxicity of oxygen in Molecular and Cellular Mechanisms of Toxicity. F De Matteis and L L Smith, Eds, pp. 35–68, CRC Press, Boca Raton, Fla, USA.
- Younes M (1999) Free radicals and reactive oxygen species, in toxicology By H. Marguardt, Mechanisms of antioxidant and prooxidant effects of lipoic acid in the diabetic and non-diabetic kidney. Kidney International 67(4): 1371-1380.
- Marks DB, Marks AD, and Smith CM (1996) Oxygen metabolism and toxicity in Basic Medical Biochemistry: A Clinical Approach. pp. 327– 340, Williams and Wilkins, Baltimore, Md, USA.
- Pan MH, Huang TM, Lin JK (1999) Biotransformation of curcumin through reduction and glucuronidation in mice. Drug Metab Dispos 27(4): 486-494.







