Retama Raetam Recrystallization using Supercritical Antisolvent Process
Asma Rejab and Hatem Ksibi*
University of Sfax, IPEIS, Tunusia
Submission: May 20, 2022; Published: June 20, 2022
*Corresponding author: Hatem Ksibi, University of Sfax, IPEIS, Tunusia
How to cite this article: Asma R, Hatem K. Retama Raetam Recrystallization using Supercritical Antisolvent Process. JOJ Wildl Biodivers. 2022: 4(3): 555640 DOI: 10.19080/JOJWB.2022.04.555640
Abstract
In this work, the Supercritical AntiSolvent (SAS) process has been used to generate micronized crystals of Retama raetam. The process was performed using ethanol and carbon dioxide as solvent and antisolvent, respectively. Recrystallization was made at various temperatures (30-50°C) and pressures (8-12MPa) using a low flow rate of supercritical carbon dioxide. Size and morphology were characterized by Scanning Electron Microscopy (SEM) and ImageJ software. The spraying of the supercritical solution through a nozzle directing the flow towards the bottom of the precipitator results in the deposition of very fine particles with nearly identical morphology at the bottom. Here, thermodynamic and hydrodynamic aspects are discussed so as to rationalize the powder and spongious film characteristics and provide a new way to control the SAS process applied to plant derivatives.
Keywords: Retama raetam; Supercritical antisolvent process; Scanning electron microscopy; copreciptiation proportions; Recrystallization process; Linoleic acids; Organic solvent; Spheroidal particles; Ethanol solution; Carbon dioxide
Introduction
Crystallization with fluids at high-pressure appears to be an attractive technical alternative. The use of fluids in the supercritical or near-critical state as solvents or anti-solvents in particle production has been demonstrated by several researchers over the last four decades, its importance as an agent for modifying particle properties such as size distribution, crystalline appearance, and morphology [1]. Carbon dioxide is attractive because it is readily available, inexpensive, environmentally friendly, and considered as safe with a rather low critical temperature which allows a mild operating temperature suitable for heat-sensitive materials with a rigorous adjusting of their precipitation or copreciptiation proportions [2].
SAS supercritical fluid nucleation is an attractive recrystallization method for many plant derived organic compounds, especially those that are difficult to fragment or recrystallize, such as very long-chain pharmaceuticals that require ultrafine particles that are uniform in morphology [3-4]. In fact, SAS technique allows an accurate control of the recrystallization process, capable of generating very small and uniform particles. Moreover, the separation of the anti-solvent from the particles after precipitation is easily obtained. Thus, large amounts of solvent by-products can be avoided and a potentially advantageous flow of solvent and anti-solvent can be provided. A relatively narrow size distribution and a different morphology can also be expected. Compared to the conventional process using liquid solvent, fewer processing steps are necessary and a potentially difficult and lengthy filtration step can be avoided. Also, the products can be free from organic solvent residues.
Methods and Materials
In this work Retama raetam is taken as vegetative material (Figure 1 & 2) which is shown as a very rich source in polyphenols and flavonoids among other Tunisian abundant steppe plants [5]. Dried samples are finely ground in a swing mill and stocked away from light at room temperature for further use. Experimental runs were established using the experimental setup [6]. In fact, the given cases revolve around variation of pressure, temperature, and different flow rates mainly those of Antisolvent and solution. The main interpretations are carried out according micrographic characterization techniques which make it possible to deduce particle size and distribution at such case.
Results and Discussions
Samples of the powder collected from the precipitators (SAS assays) were analyzed by a high-resolution field emission scanning electron microscope with focus ion beam FIB-SEM (Auriga Compact, Zeiss). Before SEM observation, samples were carefully collected in both regions of bottom and inner wall of the precipitator, see zoom in (Figure 1). Morphologies and particle size distributions, deduced from the given photomicrographs for each sample, were analyzed by ImageJ software. ImageJ can automatically recognize particle morphology using macros after adjusting thresholds and making in binary format. Finally data are processed to plot histograms showing particle size distribution. Each peak in the histogram presents one shape size in the considered image, so we can distinguish particles from the substrate which is in the case of Retama raetam fatty acids mainly linoleic acids (omega-3).

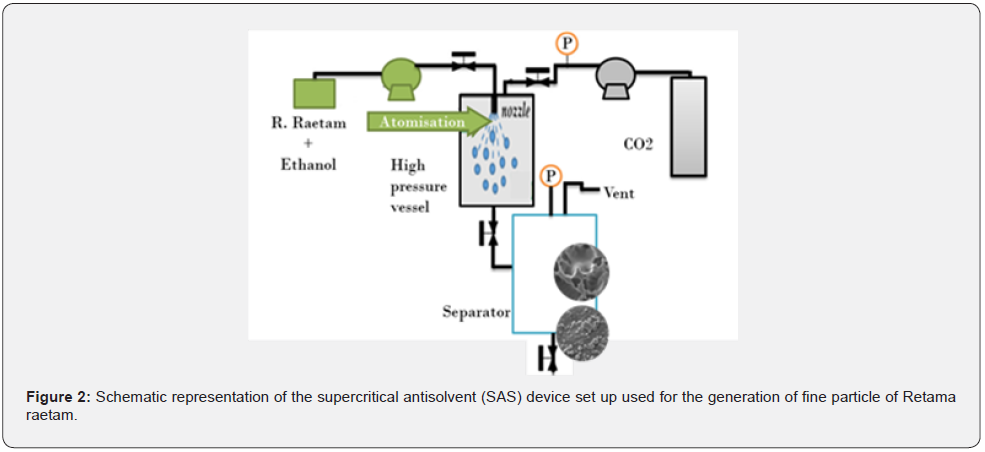

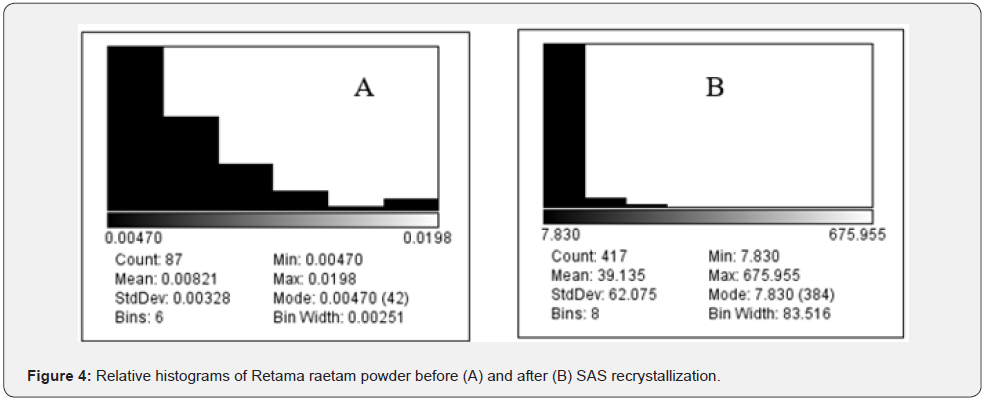
Photomicrographs were chosen among many exposures to illustrate the Retama raetam size disparity and the different obtained morphologies via SAS process. Measurements were performed in duplicate for each sample. By comparison between (Figures 3A and 3B) we can conclude easily that the largest Retama raetam particles present in the initial powder disappear after the SAS process. During conventional processes crystals can grow in size uncontrollably (Figure 3A), whereas SAS process can give likemonodisperse morphology particles, here spheroids. By the same way, we notice that an important reduction of size is remarked more than 210 times, (Figure 4). Moreover, the nucleation of particles and their growth are so important factors affecting the final particle size. There is a rapid mass transfer of antisolvent and therefore the solvent causes a high supersaturation which results in rapid nucleation and growth of more than one particle per primary droplet. This happens because at higher pressure, the deforming pressure forces must increase to break the droplets into smaller particles. The number of particles in a predetermined size range was plotted histogram of particle size distribution above. Both histograms show the percentage of particles found in each size range. It shows that the medium diameter size of the obtained particles via SAS process is about 7μm, [7].
Conclusion
Retama raetam particles were micronized from ethanol solution with a semi-continuous the SAS process by using supercritical carbon dioxide as an antisolvent. Spheroidal particles with mean area particle sizes down to 40μm² were obtained depending on process conditions. In particular, the mean particle size of particles was strongly influenced by temperature. Smaller particles with monodisperse morphology were obtained applying process near critical conditions that increased the supersaturation and therefore accelerated particle formation kinetics. Moreover, it is shown that a suitable selection of operating conditions such as temperature, solution flow rate, and pressure, can selectively produce monodisperse morphology with spheroidal shape, as well as other different polymorphic forms. Furthermore, SAS process did not cause degradation of the product; on the contrary, it allowed purification of Retama raetam from away fatty and lipidic acids. SAS process appeared stable against dissociation when it undergoes with carbon dioxide as an antisolvent which is abundant and environmentally caring.
References
- Ksibi H, Subra P, Garrabos Y (1995) Formation of fine powders of caffeine by RESS. Advanced Powder Technology 6(1): 25-33.
- Ksibi H, Subra P (1996) Powder coprecipitation by the RESS process. Advanced Powder Technology 7(1): 21-28.
- Schall JM, Capellades G, Myerson AS (2019) Methods for Estimating Supersaturation in Antisolvent Crystallization Systems. CrystEngComm 21: 5811-5817.
- McGinty J, Chong MWS, Manson A, Brown CJ, Nordon A, et al. (2020) Effect of Process Conditions on Particle Size and Shape in Continuous Antisolvent Crystallisation of Lovastatin. Crystals 10(10): 925.
- Saada M, Falleh H, Catarino M, Cardoso S, Ksouri R (2018) Plant Growth Modulates Metabolites and Biological Activities in Retama raetam (Forssk.) Webb. Molecules 23(9): 2177.
- Weber Brun G, Martín Á, Cassel E, Vargas RM F, Cocero MJ (2012) Crystallization of Caffeine by Supercritical Antisolvent (SAS) Process: Analysis of Process Parameters and Control of Polymorphism. Cryst Growth Des 12(4): 1943-1951.
- Ksibi H, Ben Moussa A, Baccar M (2006) Powder Structure Transition under the Recrystallization Conditions in the RESS Process. Chemical Engineering & Technology 29(7): 868-874.






























