- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
Targeting Cancer by siRNA; A Review on Current Strategies
Muhammad Irfan* and Iqra Rashid
Department of Biochemistry and Biotechnology,University of Gujrat,Pakistan
Submission: July 10, 2018;; Published: September 14, 2018
*Corresponding author: Muhammad Irfan, Department of Biochemistry and Biotechnology, University of Gujrat, Gujrat, Pakistan(50700), Email: Muhammad.irfan@uog.edu.pk
How to cite this article: Muhammad Irfan, Iqra Rashid. Targeting Cancer by siRNA; A Review on Current Strategies. JOJ scin. 2018; 1(5): 555573. DOI: 10.19080/JOJS.2018.01.555573
- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
Abstract
Recent development in RNA Interference (RNAi) brought enthusiasm in the scientific community and opened the new ways for many therapeutic applications. The RNAi is a phenomenon which is used to targets mRNA and chops them up. The most appealing work of RNAi is observed in cancer inhibition by silencing genome sequences. Ultimate level of specificity can be achieved through siRNA targeted therapeutics. Short interfering RNA(siRNA) belongs to dsRNA molecules that can be produced exogenously and endogenously. This review sums up the recent approaches for cancer inhibition by siRNA. The article also points out the current targets of siRNA in cancer therapy development including expression level of cancerous and non-cancerous cells, their genetic concerns and siRNA drug delivery system like lipid base Nano-vectors. Currently, many of RNA based drugs are in practice at clinical trials level to overcome this deadly disease. In view of the encouraging results of RNAi based therapy, it is obvious that in near future siRNA will prove itself as a major cancer inhibitor and can become a standard cancer treatment.
Keywords: siRNA; RNAi; Gene silencing; Cancer therapeutics
- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
Introduction
The accessibility to the human genome sequence revolutionized the strategy of utilizing nucleic acids with sequences complementary to specific target genes to improve drug discovery and target validation [1]. Development of sequence-specific DNA or RNA analogs that can block the activity of selected single-stranded genetic sequences offers the possibility of rational design with high specificity, lacking in many current drug treatments for various diseases including cancer, at relatively inexpensive costs [2]. RNA interference (RNAi) is one such example that is a simple and rapid method of targeted gene silencing in a range of organisms [3]. Gene silencing is done by degradation of RNA into short RNA strands that are involved in activation of certain ribonucleases, which target homologous mRNA [4]. The standard RNAi takes place at the post-transcriptional level and is initiated by the introduction of nucleic acidbased tools. It has been shown by several studies, conducted in-vivo and in-vitro, that RNAi- based therapy can be used for treating single-gene disorders and those with overexpression of proteins [5]. Currently, the three basic categories of RNAi tools used for this purpose are siRNA (small interfering RNA), shRNA (short hairpin RNA) and miRNA (microRNA) [6]. siRNA is the most commonly used RNAi tool for induction of short term gene silencing. It is a short, double-stranded RNA [dsRNA] duplex of 19-21 nucleotides in length,and is prepared from long dsRNA by the RNase III enzyme Dicer (endogenously), or introduced into a cell exogenously [7]. The siRNA does not require genome integration and is easy to synthesize, that’s why it is more suitable for drug use.
- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
Natural and Economic Conditions of Azerbaijan
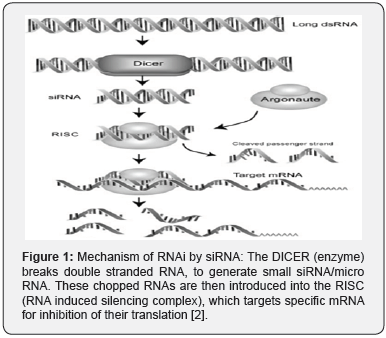
As illustrated in (Figure 1), on entering the cell, endogenously transcribed or exogenously introduced long dsRNA, triggers the RNAi process. It is first processed by the RNase III enzyme Dicer in an ATP-dependent reaction. The Dicer processes long dsRNA into small [21-23 nucleotide] siRNA strands, with 2-nucleotide 3’ overhangs [8]. siRNA can also be synthesized outside a cell and then be introduced into a cell through in-vitro delivery systems (transfection, electroporation etc) [9]. The siRNA is incorporated into the RISC RNAi effector complex, which consists of an Argonaute (Ago) protein as one of its main components [10]. Active RISC is formed when the passenger (sense) strand of the siRNA duplex is cleaved and discarded by Ago. The remaining (antisense) strand acts as the guide strand and escorts the RISC to its homologous mRNA, causing the endonucleolytic cleavage of the target mRNA [7].
Most cancers are caused by gene mutation or over expression of an oncogene, so it is possible to cure cancer by blocking disease-related gene in cancer cells with sequence specificity, through rational siRNA design [11]. Therefore, RNAi has rapidly established into one effective therapeutic technology for cancer, as it is an affordable technology for researchers [12]. Among the invivo and in-vitro clinical trials of RNAi based therapy of different diseases, administration of most siRNAs was done by local delivery, typically via the intravitreal or intranasal routes. But, local delivery may not be appropriate for all diseases [13]. Under some conditions, systemic drug administration by intravenous (IV) injection is needed, and other delivery systems are also required to administer the siRNA [14, 15].
- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
siRNA in current Cancer Therapeutics
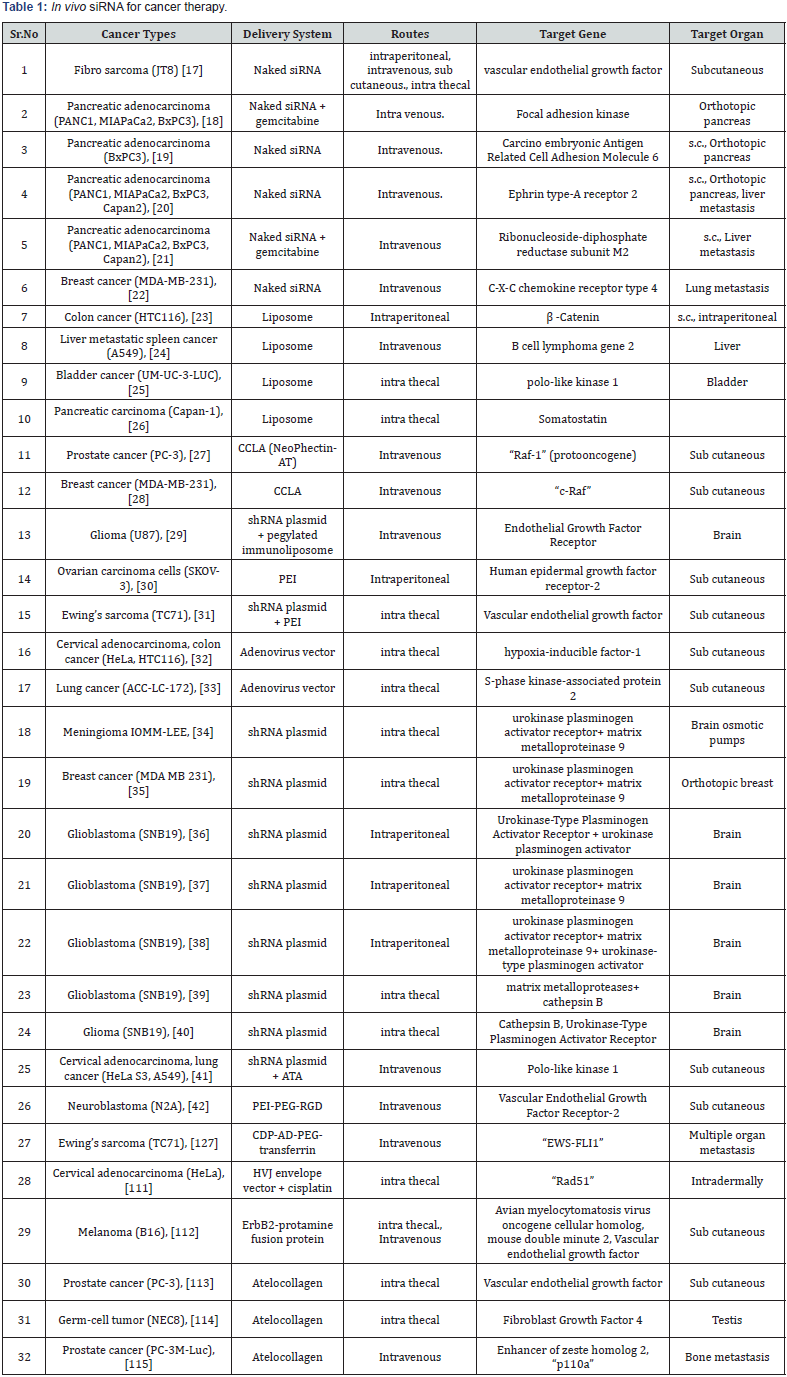
To target cancer, siRNA can be administered to whole organism or only to a specific tissue (eye, or brain tumor). In first case larger amount of siRNA is required to attain the desired down regulation while in second scenario, only small amount of siRNA is needed to target specific site with minimum side effects [14]. siRNA delivery, particularly with cholesterol conjugates, polymer-based nanoparticle stratagies and liposoms, has also been extensively discussed with reasonable success. Targeting via peptides, aptamers and antibodies have also been published [16- 42]. Different delivery methods to date are represented in Table 1. The table 1reveals in very elaborative way the delivery methods, routes of administration, target gene and target organ for siRNA in different cancer types.
In comparison to chemotherapeutic anti-cancer drugs, siRNA drugs have a lot of advantages. There are four significant benefits of siRNA as a potential cancer therapeutic strategy due to its special mechanism [16]. To start with, is its high level of safety. siRNA does not interact with DNA while acting on the posttranslational stage of gene expression, and in this way maintains a strategic distance from the mutation and teratogenicity dangers of gene therapy. The second advantage of siRNA is its high efficacy. With just several copies of siRNA, striking suppression of gene expression can be caused in a single cancer cell. The best advantages of siRNA as compared to other drugs (small molecule or antibody-based) are the unlimited targets and specificity, due to the complementary base pairing [43]. Basically, siRNA drugs are rationally designed to silence specific cancer-promoting genes to treat cancer, but of course they can also be designed to target any disease gene according to their mRNA sequence, effectively [44].Despite these advantages, there are still several barriers hindering the clinical use of siRNA for cancer therapy (Figure 2). The first is the instability of siRNA under physiological conditions. It is readily digested by the serum nucleases, during its passage through the blood [45].
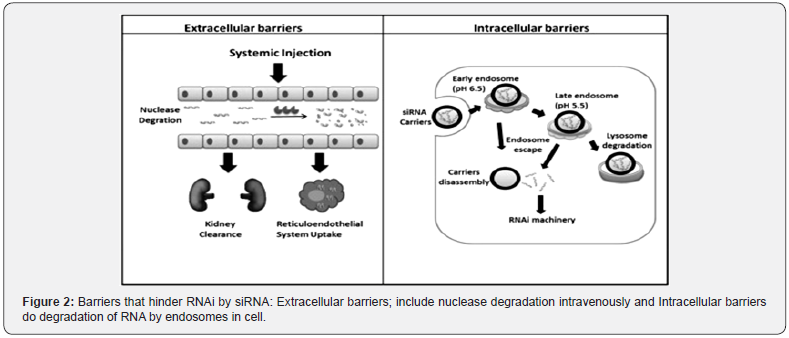
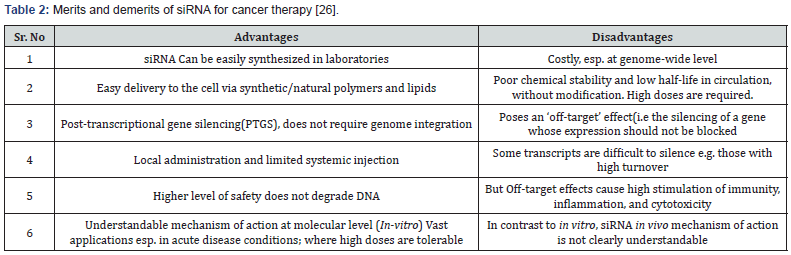
Among the biological barriers, firstly the siRNA, is encountered the nuclease activity in plasma and tissues, on administration [46]. Secondly, the role of kidney in siRNA clearance; as reported by the several studies in animals that the highest uptake of bio distribution of siRNA is shown by the kidney [47]. The third barrier is the effects of siRNA that are off-target and induce unexpected phenotypes that lead to complications in the interpretation of the therapeutic benefits offered by siRNA [48]. Lastly, the safety of siRNA is not up to the mark, and its high levels cause the activation of recipient’s innate immune response and cytokines production in vitro and in vivo [49,50]. Considering these barriers, certain chemical modifications and delivery methods must be required to transport siRNA to its target without detrimental effects. To accomplish that, variant materials are under exploration to confront the challenges of in vivo delivery. These materials include polymers, peptides, lipids, aptamers, antibodies, and small molecules [51]. With the help of rational designs and highthroughput screens successful systems have been developed or discovered. Table 2 shows the Merits and demerits of siRNA for cancer therapy [52].
- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
The Criteria for Designing siRNA Delivery System for Cancer Therapy
For systemic and safe administration of siRNA across physiological barriers to its site of action, such delivery systems should be synthesized that, dodge immune recognition, provide serum stability, prevent renal clearance, increase vascular permeability to reach cancer tissues, reduce interactions with serum proteins and non-cancer cells, contain low toxicity and allow cell entry and endosome escape to enter the RNAi machinery [52,53]. To avoid the problems, a common strategy is to incorporate 2′-O-methyl and 2′-deoxy-2′-fluoro groups, nucleic acids (locked or unlocked), or phosphorothioate linkages [54]. Despite the solution of some problems by chemical modifications, encapsulation of siRNA by nanoparticles is better option for protecting it from degradation and immune system [55]. Unfavorable aggregation with erythrocytes can be caused due to the high positive charges of nanoparticles [56], but such interaction between serum proteins and nanoparticles can also help in uptake by cancer cells [57,58]. The typical strategy to minimize this interaction is to shield the nanoparticle surface with polyethylene glycol (PEG) [59].
Ways by which siRNA leaves the bloodstream after systemic administration, are through the liver, spleen, kidney and lung. But the most common pathway is kidney clearance [60]. The glomeruli of kidney work as a natural filter which allows water and small molecules to pass into urine and larger molecules remain in the circulation [61]. The pore size of the glomerular filter is roughly 8nm [62], and typically kidney excretes molecules less than 50kDa in size [63]; the molecular weight of naked siRNA is about 13kDa [64]. That’s why siRNA easily passes through glomeruli and drifts into the urine. The size of the delivery nanoparticle can be increased, by addition of synthetic materials to siRNA, to keep it for alternative organ targets rather than glomerular filtration through the kidneys [65]. Mostly highly effective delivery systems are larger than 20nm [66]such as dynamic polyconjugates (DPCs; 10nm) [67]and triantennary N-acetylgalactosamine [GalNAc] conjugates.
Most siRNA delivery systems include cellular internalization by endocytosis. The rate of cellular uptake is improved by siRNA delivery systems, as they induce receptor-mediated endocytosis by incorporating targeting ligands that bind specifically to receptors on target cells [68]. But this ligand–receptor interaction is hindered by adsorption of serum proteins on the nanoparticle surface [69]. Cell uptake, through endocytosis or non-endocytic mechanisms, of cell-penetrating peptides is used by other systems [70]. Additionally, the most important part of the siRNA delivery systems is low toxicity. If an unacceptable toxicity is provoked by siRNA delivery on either a cellular or systemic level, even the most effective siRNA delivery system will be become useless. For example, among the first vehicles to be studied for siRNA delivery, the viral vectors induce toxicity by activating immune responses [71,72]. Thus, as alternatives to viral vectors, synthetic lipids and polymers have been for nucleic acid delivery applications. Their formulation has been done carefully, to avoid activation of the immune response [73]. For clearance of larger molecular mass materials, they must be biodegradable. That’s why high molecular mass polycations and polymers containing linkages that can be cleaved inside the cell are used. As they are biodegradable, they can help decrease cytotoxicity [74].
- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
siRNA Delivery Systems for Cancer Inhibition
Scientists have reported different strategies for cancer inhibition including siRNA [75]. The delivery of siRNA holds great importance. Different factors effect siRNA delivery including Biocompatibility, Serum stability, Biodegradability and Nonimmunogenicity [76]. There are different categories of siRNA delivery systems including Lipid-based Nano-vectors, Polymer mediated delivery, Chemical modification and conjugate delivery systems [77]. Advantages and Disadvantages of Different siRNA Delivery approaches has been discussed in Table 3. Both 3 and 5 terminals of siRNA and the sugar backbone can be chemically modified [78]. The most important chemical modifications include the addition of 2´-deoxy-2´-fluoro and 2´- O –methyl [79]. Furthermore, at the 3-terminal of RNA’s sugar backbone, phosphodiester group can be replaced with phosphothioate group. 2´-O-alkyl can be combined with 4´-thiolate for chemical modification [80]. The chemical modification should not have negative effects on serum stability and gene silencing effect of siRNA. Different metabolites used in chemical modification can cause a safety issue[81-83].

- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
siRNA deliverance Using Lipid Based Nano Vectors
In order to improve the efficacy of plasmid DNA or siRNA transfection, a lipid formulation called as Lipofectamine 2000 is used [84]. Liposomes transfection mechanism includes the interaction between lipids, which are positively charged, and nucleic acids which are negatively charged [85], which forms a lipoplexes. DOPC (1,2-dioleoylsn-glycero-3-phosphatidylcholine) is an important component in siRNA delivery using lipids [86]. 1,2-dioleoyl-3-trimethylamonium-propane [DOTAP] and dioleoylphosphatidylethanolamine are among the other important formulations for siRNA delivery [87]. SNALPs [stable nucleic acidlipid particle], the lipids nanoparticles, are thought to be important vectors for siRNA delivery [88]. In SNALPs, diffusible polyethylene glycol surrounds the fusogenic and cationic lipids [89]. SNALPs gives high bioavailability, so there are more chances of siRNA delivery to cancer cells [90]. Lipoid nanoparticles which include the PEG-modified lipids and cholesterol [91], also proved to be better vehicles for siRNA delivery e.g. 98N12-5, which reduced the FVII or APoB expression from 75 to 90 % in hepatocytes of mice [92].
- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
siRNA transfer by Polymers for Cancer Inhibition
Solid, biodegradable and colloidal systems; usually known as polymeric Nano particles can also be used for siRNA delivery e.g. poly (d, l-lactide) and poly (d, l-lactide-co-glycolide) [93]. Another type of polymer mediated siRNA delivery includes water soluble cationic polymers e.g. polyethyleneimine [PEI]. Cyclodextrin, first used in 1999 for siRNA delivery, proved to be better vehicle for siRNA delivery [94]. They are poly-cationic oligomers of di- amine bearing cyclodextrin monomers and di- methyl suberimidate[95]. Efficacy of this siRNA delivery system can be improved by using Adamantane- PEG-transferrin and Adamantane- PEG are used for both in-vivo and in-vitro experiments Polyethylenimine proved important. Polycaprolactane are micelleplex Nano particles; during experiments they suppressed the expression of green fluorescent protein from forty to seventy percent [96]. Another component, m[PEG]-b-[PCL]-b[PPEEA] proved to be successful during trials on mice [97]. Other components including Poly [d,l-lactide] [PLA] and poly [d,l-lactide-co-glycolide] were successful with spermidine especially for gene silencing in vaginal mucosa during experiments by Saltzman in 2009 [98]. All these components have been successful during initial trials and have great potential to be used in siRNA delivery [99].
- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
Experiments on siRNA delivery using conjugates have shown positive results. Different types of conjugates are used including peptides, lipids and aptamers (modified oligonucliotides) [100]. The early experiments were performed using lipid-based conjugates such as cholesterol [101]. A pyrrolidone linkage, joined cholesterol on the 3-terminal of siRNA’s sense strand. The pharmacokinetic behavior and transfection efficacy of siRNA was increased by cholesterol [102]. In the next step, high density lipoprotein was attached with the siRNA-cholesterol complex. It improved the gene silencing effect 8-15 times [103]. Transfection efficacy can also be improved by using cell-penetrated peptides such as trans-activator protein [TAT], which is taken from HIV- 1[104]. Hetero bi-functional cross linker [Sulfosuccinimidyl – 4 – p-maleimidophenyl butyrate] was used to link TAT with siRNA’s antisense strand [105]. Apart from its advantage of improving intracellular siRNA delivery, it can cause cyto-toxicity because of immunogenicity [106]. Insulin receptor substrate’s expression was sixty percent reduced by using receptor-ligand mediatory delivery, in which a carboxylic group from D-[Cys-Ser-Lys-Cys] which is the peptide mimetic of IGF1, was bound to amine group at 5-terminal of siRNA’s sense strand [107]. Better specificity and stability has made antibody-mediated delivery advantageous, in which a biotin-streptavidin linkage joined the siRNA with the monoclonal antibody; which targets the transferring receptor on blood-brain barrier [108]. During experiments on rat with intracranial transplanted brain tumors, the reporter gene expression was greatly reduced due to intravenous administration of siRNA-Antibody complex [109]. As aptamers contains selective affinity for proteins, so streptavidin-biotin linkage was used to conjugate it to siRNA. It does not use transfection agents and still gives positive results. Gal-Nac& DPCs, the advanced delivery systems are currently in focus[110].
- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
Conclusion
siRNA drugs are one of the most promising drugs for cancer treatment due to their great advantages, such as high efficacy, excellent safety, specificity and unrestricted choice of targets. Many highly effective delivery systems have been developed for solution of the delivery problems of siRNA and are quite different in terms of size, structure and chemistry. But still for optimal delivery systems, some guidelines should be followed e.g. Nano particle size should be about 20-200nm, PEG should be used as a shielding agent, chemical modifications should be made to avoid renal filtration and phagocytic filtration, to prevent non-specific interactions and immune recognition in circulation, to avoid nuclease digestion respectively. Additionally, for siRNA uptake by cancer cells, endogenous or exogenous targeting ligands should be used. Although, the great potential of siRNA in cancer treatment has been demonstrated by a number of reports but there are still few remaining challenges to meet the full clinical potential of siRNA. Highs and lows are being experienced by siRNA drug development in recent years. Big pharmaceutical companies have shown over-optimistic attitude towards RNAi drugs. Hence, the key to siRNA drug development is a good delivery system. Significant breakthrough of research in siRNA drug delivery systems will make siRNA to occupy a strong position in the drug market, especially the anti-cancer drug market.
- Review article
- Abstract
- Introduction
- Mechanism of Gene Silencing by siRNA
- siRNA in current Cancer Therapeutics
- The Criteria for Designing siRNA Delivery System for Cancer Therapy
- siRNA Delivery Systems for Cancer Inhibition
- siRNA deliverance Using Lipid Based Nano Vectors
- siRNA transfer by Polymers for Cancer Inhibition
- Delivery Systems for siRNA Using Conjugates for Cancer Inhibition
- Conclusion
- References
References
- Larson D, Jackson LN, Chen LA (2007) Effectiveness of siRNA uptake in target tissues by various delivery methods. Surgery 142(2): 262-269.
- Devi GR (2006) siRNA-based approaches in cancer therapy. Cancer Gene Therapy 13(9): 819-829.
- Schultz N, Marenstein DR, Angelis DAD (2011) Off-target effects dominate a large-scale RNAi screen for modulators of the TGF-pathway and reveal microRNA regulation of TGFBR2. Silence 2(3).
- Neema Agrawal, Dasaradhi PV, Mohmmed A, Malhotra P, Bhatnagar RK, et al. (2003) RNA Interference: Biology, Mechanism, and Applications. Microbiol Mol Biol Rev 2003; 67(4): 657-685.
- Dykxhoorn DM, Judy Lieberman (2006) Knocking down disease with siRNAs. Cell 126(2): 231-235.
- Friedman RC (2009) Most mammalian mRNAs are conserved targets of microRNAs. Genome Rese 19(1): 92-105.
- Irfan M, Mehmood S (2014) RNA Interference: Molecular mechanism and milestone therapeutic Applications. Journal of Cell and Molecular Biology 12(1-2): 11-17.
- Marques JT, Williams BR (2005) Activation of the mammalian immune system by siRNAs. Nature Biotechnology 23(11): 1399-1405.
- Heil F, Hemmi H, Hochrein H, Ampenberger F, Kirschning C, et al. (2004) Species-specific recognition of single-stranded RNA via toll-like receptor 7 and 8. Science 303(5663): 1526-1529.
- Karikó K, Bhuyan P, Capodici J (2004) Small interfering RNAs mediate sequence-independent gene suppression and induce immune activation by signaling through toll-like receptor 3. J Immunol 172(11): 6545-6549.
- Cong-fei Xu, Jun Wang (2015) Delivery systems for siRNA drug development in cancer therapy. Asian Journal of Pharmaceutical Sciences 10(1): 1-12.
- Irfan M, Anwer Z, Naveed M, Ayub H, Amman M (2016) HCV Genotypes and Risk factors; Current Scenario in Pakistan. PSM Biol Res 01(S1): S1-S5.
- Iwasaki A, Medzhitov R (2004) Toll-like receptor control of the adaptive immune responses. Nat Immunol 5(10): 987-995.
- Vaishnaw AK, Gollob J, Gamba-Vitalo C, Hutabarat R, Sah D (2010) A status report on RNAi therapeutics. Silence 1(14).
- Scanlon KJ (2004) Anti-genes: siRNA, ribozymes and antisense. Curr Pharm Biotechnol 5(5): 415-420.
- Hennink WE, Storm G (2008) Sheddable coatings for long-circulating nanoparticles. Pharma res 25(1): 55-71.
- Naveed M, Tehreem S, Mubeen F, Nadeem F, Zafa, et al. (2016) Insilico analysis of non-synonymous-SNPs of STEAP2. To provoke the progression of prostate cancer. Open Life Sciences 11(1): 402-416.
- Duxbury MS, Ito H, Zinner MJ, Ashley SW, Whang EE (2004) EphA2: a determinant of malignant cellular behavior and a potential therapeutic target in pancreatic adenocarcinoma. Oncogene 23(7): 1448-1456.
- Naveed M, Kazmi SK, Anwar F, Arshad F, Dar TZ, et al. (2016) Computational Analysis and Polymorphism study of Tumor Suppressor Candidate Gene-3 for Non Syndromic Autosomal Recessive Mental Retardation. Journal of Applied Bioinformatics & Computational Biology 5(2).
- Liang Z, Yoon Y, Votaw J, Goodman MM, Williams L, et al. (2005) Silencing of CXCR4 blocks breast cancer metastasis. Cancer Res 65(3): 967-971.
- Verma UN, Surabhi RM, Schmaltieg A, Becerra C, Gaynor RB (2003) Small interfering RNAs directed against β-catenin inhibit the in vitro and in vivo growth of colon cancer cells. Clin Cancer Res 9(4): 1291- 300.
- Yano J, Hirabayashi K, Nakagawa SI, Yamaguchi T, Nogawa M, et al. (2004) Antitumor activity of small interfering RNA/cationic liposome complex in mouse models of cancer. Clin Cancer Res 10(22): 7721- 7726.
- Nogawa M, Yuasa T, Kimura S, Tanaka M, Kuroda J, et al. (2005) Intravesical administration of small interfering RNA targeting PLK- 1 successfully prevents the growth of bladder cancer. J Clin Invest 115(4): 978-985.
- Carrere N, Vernejoul F, Souque A, Asnacios A, Vaysse N, et al (2005) Characterization of the bystander effect of somatostatin receptor sst2 after in vivo gene transfer into human pancreatic cancer cells. Human Gen Ther 16(10): 1175-1193.
- Pal A, Ahmad A, Khan S, Sakabe I, Zhang C, et al. (2005) Systemic delivery of RafsiRNA using cationic cardiolipin liposomes silences Raf- 1 expression and inhibits tumor growth in xenograft model of human prostate cancer. Int J Oncol 26: 1087-1091.
- Chien PY, Wang J, Carbonaro D, Lei S, Miller B, et al. (2005) Novel cationic cardiolipin analogue-based liposome for efficient DNA and small interfering RNA delivery in vitro and in vivo. Cancer Gen Ther 12(3): 321-328.
- Zender L, Hütker S, Liedtke C, Tillmann HL, Zender S, et al. (2003) Caspase 8 small interfering RNA prevents acute liver failure in mice. Proc Natl Acad Sci U S A 100(13): 7797-7802.
- Guan H, Zhou Z, Wang H, Jia SF, Liu W, et al. (2005) A small interfering RNA targeting vascular endothelial growth factor inhibits Ewing’s sarcoma growth in a xenograft mouse model. Clinical Cancer Research 11(7): 2662-2669.
- Sumimoto H, Yamagata S, Shimizu A, Miyoshi H, Mizuguchi H, et al. (2005) Gene therapy for human small-cell lung carcinoma by inactivation of Skp-2 with virally mediated RNA interference. Gene Ther 12(1): 95-100.
- Tummalapalli P, Gondi CS, Dinh DH, Gujrati M, Rao JS (2007) RNA interference-mediated targeting of urokinase plasminogen activator receptor and matrix metalloproteinase-9 gene expression in the IOMMlee malignant meningioma cell line inhibits tumor growth, tumor cell invasion and angiogenesis. Int J Oncol 31(1): 5-18.
- Kunigal S, Lakka SS, Gondi CS, Estes N, Rao JS (2007) RNAi‐mediated downregulation of urokinase plasminogen activator receptor and matrix metalloprotease‐9 in human breast cancer cells results in decreased tumor invasion, angiogenesis and growth. Int J Cancer 121(10): 2307-2316.
- Gondi CS, Lakka SS, Dinh DH, Olivero WC, Gujrati M, et al. (2007) Intraperitoneal injection of an hpRNA-expressing plasmid targeting uPAR and uPA retards angiogenesis and inhibits intracranial tumor growth in nude mice. Clin Cancer Res 13(14): 4051-4060.
- Lakka SS, Gondi CS, Dinh DH, Olivero WC, Gujrati M, et al. (2005) Specific interference of uPAR and MMP-9 gene expression induced by double-stranded RNA results in decreased invasion, tumor growth and angiogenesis in gliomas. J Biol Chem 280: 21882-21892.
- Gondi CS, Lakka SS, Dinh D, Olivero W, Gujrati M, et al. (2004) Downregulation of uPA, uPAR and MMP-9 using small, interfering, hairpin RNA (siRNA) inhibits glioma cell invasion, angiogenesis and tumor growth. Neuron Glia Biology 1(02): 165-176.
- Lakka SS, Gondi CS, Yanamandra N, Olivero WC, Dinh DH, et al. (2004) Inhibition of cathepsin B and MMP-9 gene expression in glioblastoma cell line via RNA interference reduces tumor cell invasion, tumor growth and angiogenesis. Oncogene 23: 4681-4689.
- Gondi CS, Lakka SS, Dinh DH, Olivero WC, Gujrati M, et al. (2004b) RNAi-mediated inhibition of cathepsin B and uPAR leads to decreased cell invasion, angiogenesis and tumor growth in gliomas. Oncogene 23: 8486–8496.
- Spankuch B, Matthess Y, Knecht R, Zimmer B, Kaufmann M, et al. (2004) Cancer inhibition in nude mice after systemic application of U6 promoter-driven short hairpin RNAs against PLK1. J Natl Cancer Inst 96: 862-872.
- Schiffelers RM, Ansari A, Xu J, Zhou Q, Tang Q, et al. (2004) Cancer siRNA therapy by tumor selective delivery with ligand-targeted sterically stabilized nanoparticle. Nucleic Acids Res 32: e149.
- Hu-Lieskovan S, Heidel JD, Bartlett DW, Davis ME, Triche TJ (2005) Sequence-specific knockdown of EWS-FLI1 by targeted, nonviral delivery of small interfering RNA inhibits tumor growth in a murine model of metastatic Ewing’s sarcoma. Cancer Res 65: 8984-8992.
- Ito M, Yamamoto S, Nimura K, Hiraoka K, Tamai K (2005) Rad51 siRNA delivered by HVJ envelope vector enhances the anti-cancer effect of cisplatin. J Gene Med 7: 1044-1052.
- Song E, Zhu P, Lee SK, Chowdhury D, Kussman S, et al. (2005) Antibody mediated in vivo delivery of small interfering RNAs via cell-surface receptors. Nat Biotechnol 23: 709-717.
- Takei Y, Kadomatsu K, Yuzawa Y, Matsuo S, Muramatsu T (2004) A small interfering RNA targeting vascular endothelial growth factor as cancer therapeutics. Cancer Res 64: 3365-3370.
- Irfan M, Sughra K, Iqbal NM, Mehmood S, Shaheen A (2016) Arresting Cancer Progression by VEGF Inhibitions: An Update. PSM Microbiology: 1(2)72-78.
- Xu Cf, Wang J (2015) Delivery systems for siRNA drug development in cancer therapy. Asian Journal of Pharmaceutical Sciences 10(1): 1-12.
- Ohkuma S, Poole B (1978) Fluorescence probe measurement of the intralysosomal pH in living cells and the perturbation of pH by various agents. Proc Natl Acad Sci 75(7): 3327-3331.
- Layzer JM, McCaffrey AP, Tanner AK, Huang Z, Kay MA (2004) In vivo activity of nuclease-resistant siRNAs. RNA 10(5): 766-771.
- van de Water FM, Boerman OC, Wouterse AC, Peters JG, Russel FG (2006) Intravenously administered short interfering RNA accumulates in the kidney and selectively suppresses gene function in renal proximal tubules. Drug Metab Dispos 34(8): 1393-1397.
- Jackson AL, Linsley PS (2010) Recognizing and avoiding siRNA offtarget effects for target identification and therapeutic application. Nat Rev Drug Discov 9(1): 57-67.
- Hornung V, Guenthner-Biller M, Bourquin C, Ablasser A, Schlee M (2005) Sequence-specific potent induction of IFN-α by short interfering RNA in plasmacytoid dendritic cells through TLR7. Nat Med 11(3): 263-270.
- Judge AD, Sood V, Shaw JR, Fang D, McClintock K (2005) Sequencedependent stimulation of the mammalian innate immune response by synthetic siRNA. Nature Biotechnology 23(4): 457-462.
- Martina MS, Nicolas V, Wilhelm C (2007) The in vitro kinetics of the interactions between PEG-ylated magnetic-fluid-loaded liposomes and macrophages. Biomaterials 28(28): 4143-4153.
- Alexis F, Pridgen E, Molnar LK, Farokhzad OC (2008) Factors affecting the clearance and biodistribution of polymeric nanoparticles. Mol Pharm 5(4): 505-515.
- Whitehead KA, Langer R, Anderson DG (2009) Knocking down barriers: advances in siRNA delivery. Nat Rev Drug Discov; 8(2): 129-138.
- Deleavey GF, Damha MJ (2012) Designing chemically modified oligonucleotides for targeted gene silencing. Chem Biol 19(8): 937-54.
- Wang AZ, Langer R, Farokhzad OC (2012) Nanoparticle delivery of cancer drugs. Annu Rev Med 63(1): 185-198.
- Malek A, Merkel O, Fink L, Czubayko F, Kissel T (2009) In vivo pharmacokinetics, tissue distribution and underlying mechanisms of various PEI (-PEG)/siRNA complexes. Toxicol Appl Toxicol 236(1): 97- 108.
- Wolfrum C, Shi S, Jayaprakash KN, Jayaraman M, Wang G (2007) Mechanisms and optimization of in vivo delivery of lipophilic siRNAs. Nature Biotechnology 25(10): 1149-1157.
- Akin A, William Q, Soma D, June Q, Maria F (2010) Targeted delivery of RNAi therapeutics with endogenous and exogenous ligand-based mechanisms. Mol Ther 18(7): 1357-1364.
- Bazile D, Prud’homme C, Bassoullet MT, Marlard M, Spenlehauer G (1995) Stealth Me. PEG-PLA nanoparticles avoid uptake by the mononuclear phagocytes system. J Pharm Sci 84(4): 493-498.
- Shim MS, Kwon YJ (2012) Stimuli-responsive polymers and nanomaterials for gene delivery and imaging applications. Adv Drug Deliv Rev 64(11): 1046-1059.
- Jarad G, Miner JH (2009) Update on the glomerular filtration barrier. Curr Opin Nephrol Hypertens 18(3): 226-232.
- Wartiovaara J, Ofverstedt LG, Khoshnoodi J, Zhang J, Mäkelä E (2004) Nephrin strands contribute to a porous slit diaphragm scaffold as revealed by electron tomography. J Clin Invest 114(10): 1475-1483.
- Rappaport J, Hanss B, Kopp JB, Copeland TD, Bruggeman LA (1995) Transport of phosphorothioate oligonucleotides in kidney: implications for molecular therapy. Kidney Int 47(5): 1462-1469.
- Whitehead KA, Langer R, Anderson DG (2009) Knocking down barriers: advances in siRNA delivery. Nat Rev Drug Discov 8(2): 129-138.
- Lee H, Lytton-Jean AK, Chen Y, Love KT, Park AI (2012) Molecularly self-assembled nucleic acid nanoparticles for targeted in vivo siRNA delivery. Nature Biotechnology 7(6): 389-393.
- Rozema DB, Lewis DL, Wakefield DH, Wong SC, Klein JJ (2007) Dynamic Poly Conjugates for targeted in vivo delivery of siRNA to hepatocytes. Proc Natl Acad Sci 104(32): 12982-12987.
- Yu B, Zhao X, Lee LJ, Lee RJ (2009) Targeted delivery systems for oligonucleotide therapeutics. AAPS J 11(1): 195-203.
- Salvati A, Pitek AS, Monopoli MP, Prapainop K, Bombelli FB (2013) Transferrin-functionalized nanoparticles lose their targeting capabilities when a biomolecule corona adsorbs on the surface. Nat Nanotechnol 8(2): 137-143.
- Bolhassani A (2011) Potential efficacy of cell-penetrating peptides for nucleic acid and drug delivery in cancer. Biochim Biophys Acta-Rev Cancer 1816(2): 232-246.
- Barquinero J, Eixarch H, Perez-Melgosa M (2004) Retroviral vectors: new applications for an old tool. Gene Ther 11(Suppl 1): S3-S9.
- Paul CP, Good PD, Winer I, Engelke DR (2002) Effective expression of small interfering RNA in human cells. Nat Biotechnol 20(5): 505-508.
- Judge AD, Sood V, Shaw JR, Fang D, McClintock K (2005) Sequencedependent stimulation of the mammalian innate immune response by synthetic siRNA. Nature Biotechnology 23(4): 457-462.
- Geest VGD, Bonne S, Vinken M, Haecke TV, Heimberg H (2008) Prolonged gene silencing in hepatoma cells and primary hepatocytes after small interfering RNA delivery with biodegradable poly (β-amino esters). J Gene Med 10(7): 783-794.
- Juliano R, Alam MR, Dixit V (2008) Mechanisms and strategies for effective delivery of antisense and siRNA oligonucleotides. Nucleic Acids Res 36(12): 4158-4171.
- Strumberg D, Schultheis B, Traugott U (2012) Phase I clinical development of Atu027, a siRNA formulation targeting PKN3 in patients with advanced solid tumors. Int J Clin Pharmacol Ther 50(1): 76-78.
- Kanasty R, Dorkin JR, Vegas A (2013) Delivery materials for siRNA therapeutics. Nat Mater 12(11): 967-977.
- Braasch DA, Jensen S, Liu Y (2003) RNA interference in mammalian cells by chemically-modified RNA. Biochemistry 42(26): 7967-7975.
- Chiu YL, Rana TM (2003) siRNA function in RNAi: a chemical modification analysis. RNA 9(9): 1034-1048.
- Aigner A (2007) Nonviral in vivo delivery of therapeutic small interfering RNAs. Curr Opin Mol Ther 9(4): 345-352.
- Dande P, Prakash TP, Sioufi N (2006) Improving RNA interference in mammalian cells by 4′-thio-modified small interfering RNA (siRNA): effect on siRNA activity and nuclease stability when used in combination with 2′-O-alkyl modifications. J Med Chem 49(5): 1624- 1634.
- Hall AH, Wan J, Shaughnessy EE (2004) RNA interference using boranophosphate siRNAs: structure–activity relationships. Nucleic Acids Res 32(20): 5991-6000.
- Santel A, Aleku M, Keil O (2006) A novel siRNA-lipoplex technology for RNA interference in the mouse vascular endothelium. Gene Ther 13(16): 1222-1234.
- Hayes ME, Drummond DC, K Hong (2006) Assembly of nucleic acidlipid nanoparticles from aqueous-organic monophases. Biochim Biophys Acta Biomembr 1758(4): 429-442.
- Khoury M, Louis-Plence P, Escriou V (2006) Efficient new cationic liposome formulation for systemic delivery of small interfering RNA silencing tumor necrosis factor α in experimental arthritis. Arthritis Rheum 54(6): 1867-1877.
- Zuhorn IS, Oberle V, Visser WH (2002) Phase behavior of cationic amphiphiles and their mixtures with helper lipid influences lipoplex shape, DNA translocation, and transfection efficiency. Biophys J 83(4): 2096-2108.
- Landen CN, Chavez-Reyes A, Bucana C (2005) Therapeutic EphA2 gene targeting in vivo using neutral liposomal small interfering RNA delivery. Cancer Res 65(15): 6910-6918.
- Wu SY, McMillan NA (2009) Lipidic systems for in vivo siRNA delivery. AAPS J 11(4): 639-652.
- Sørensen DR, Leirdal M, Sioud M (2003) Gene silencing by systemic delivery of synthetic siRNAs in adult mice. J Mol Biol 327(4): 761-766.
- Bao Y, Jin Y, Chivukula P (2013) Effect of PEGylation on biodistribution and gene silencing of siRNA/lipid nanoparticle complexes. Pharm Res 30(2): 342-351.
- Lin S, Zhao W, Tsai H (2012) Sterically polymer-based liposomal complexes with dual-shell structure for enhancing the siRNA delivery. Biomacromolecules 13(3): 664-675.
- Morrissey DV, Lockridge JA, Shaw L (2005) Potent and persistent in vivo anti-HBV activity of chemically modified siRNAs. Nat Biotechnol 23(8): 1002-1007.
- Judge AD, Robbins M, Tavakoli I (2009) Confirming the RNAi-mediated mechanism of action of siRNA-based cancer therapeutics in mice. J Clin Invest 119(3): 661-673.
- Shen H, Sun T, Ferrari M (2012) Nanovector delivery of siRNA for cancer therapy. Gene Ther 19(6): 367-373.
- Akinc A, Zumbuehl A, Goldberg MA (2008) combinatorial library of lipid-like materials for delivery of RNAi therapeutics. Nat Biotechnol 26(5): 561 - 569.
- Sahoo SK, Labhasetwar V (2003) Nanotech approaches to drug delivery and imaging. Drug Discov Today 8(24): 1112-1120.
- Wang X, Wang Y, Chen ZG (2009) Advances of cancer therapy by nanotechnology. Cancer Res Treat 41(1): 1-11.
- Davis ME (2009) The first targeted delivery of siRNA in humans via a self-assembling, cyclodextrin polymer-based nanoparticle: from concept to clinic. Mob Pharm 6(3): 659-668.
- Gonzalez H, Hwang SJ, Davis ME (1999) New class of polymers for the delivery of macromolecular therapeutics. Bioconjug Chem 10(6): 1068-1074.
- Bellocq NC, Pun SH, Jensen GS (2003) Transferrin-containing, cyclodextrin polymer-based particles for tumor-targeted gene delivery. Bioconjug Chem 14(6): 1122-1132.
- Bartlett DW, Davis ME (2006) Insights into the kinetics of siRNAmediated gene silencing from live-cell and live-animal bioluminescent imaging. Nucleic Acids Res 34(1): 322-333.
- Bartlett DW, Su H, Hildebrandt IJ (2007) Impact of tumor-specific targeting on the biodistribution and efficacy of siRNA nanoparticles measured by multimodality in vivo imaging. Proc Natl Acad Sci 104(39): 15549-15554.
- Urban-Klein B, Werth S, Abuharbeid S (2005) RNAi-mediated genetargeting through systemic application of polyethylenimine (PEI)- complexed siRNA in vivo. Gene Ther 12(5): 461-466.
- Philipp A, Zhao X, Tarcha P (2009) Hydrophobically modified oligoethylenimines as highly efficient transfection agents for siRNA delivery. Bioconjug Chem 20(11): 2055-2061.
- Urban-Klein B, Werth S, Abuharbeid S (2005) RNAi-mediated genetargeting through systemic application of polyethylenimine (PEI)- complexed siRNA in vivo. Gene Ther 12(5): 461-466.
- Grayson AC, Doody AM, Putnam D (2006) Biophysical and structural characterization of polyethylenimine-mediated siRNA delivery in vitro. Pharm Res 23(8): 1868-1876.
- Navarro G, Sawant RR, Essex S (2011) Phospholipid–polyethylenimine conjugate-based micelle-like nanoparticles for siRNA delivery. Drug Deliv Transl Res 1(1): 25-33.
- Navarro G, Sawant RR, Biswas S (2012) P-glycoprotein silencing with siRNA delivered by DOPE-modified PEI overcomes doxorubicin resistance in breast cancer cells. Nanomedicine 7(1): 65-78.
- Sun TM, Du JZ, Yan LF (2008) Self-assembled biodegradable micellar nanoparticles of amphiphilic and cationic block copolymer for siRNA delivery. Biomaterials 29(32): 4348-4355.
- Filleur S, Courtin A, Ait-Si-Ali S, Guglielmi J, Merle C, et al. (2003) SiRNA-mediated inhibition of vascular endothelial growth factor severely limits tumor resistance to antiangiogenic thrombospondin-1 and slows tumor vascularization and growth. Cancer Res 63(14): 3919-3922.
- Duxbury MS, Ito H, Benoit E, Zinner MJ, Ashley SW, et al. (2003) RNA interference targeting focal adhesion kinase enhances pancreatic adenocarcinoma gemcitabine chemosensitivity. Biochem Biophys Res Commun 311(3): 786-792.






























