- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
Genetic Engineering of Solanum tuberosum L. to Enhance Resistance Against Abiotic Stresses: A Review
Sarbesh Das Dangol1, Muhammad Naeem1, Mohammad Hussein Azimi1, Aneela Yasmeen1,2, Mehmet Emin Caliskan1 and Allah Bakhsh1*
1Department of Agricultural Genetic Engineering, Nigde Omer Halisdemir University, Turkey
2Centre of Excellence in Molecular Biology, University of the Punjab, Pakistan
Submission: July 23, 2018;; Published: August 27, 2018
*Corresponding author: Allah Bakhsh, Department of Agricultural Genetic Engineering. Faculty of Agricultural Sciences and Technologies, Nigde Omer Halisdemir University, 51240 Nigde, Turkey, Email: abthebest@gamil.com
How to cite this article: Sarbesh Das D, Muhammad N, Mohammad H A, Aneela Y, Mehmet Emin C, Allah B. Genetic Engineering of Solanum tuberosum L. to Enhance Resistance Against Abiotic Stresses: A Review JOJ scin. 2018; 1(4): 555571. DOI: 10.19080/JOJS.2018.01.555571
- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
Abstract
Potato (Solanum tuberosum L.) today is the fifth most significant crop worldwide after wheat, maize, rice and sugar cane. Conventionally intogressing agronomic traits in potato is considered laborious and time consuming task because of sexuality barriers between wild and cultivated potatoes. However, potato has gone through genetic manipulations with the advent of genetic engineering technologies. These technologies have helped the researchers to introduce traits of economic importance. Several studies for abiotic (i.e. drought, chilling, heat, salt) tolerances and improvement in nutrient quality have been documented. Modern day technologies have emerged as a necessary tool in potato breeding programs, strengthening classical strategies to improve yield and yield contributing factors. The present review article describes the genetic improvements in potato by scientists worldwide utilizing modern biotechnological approaches to enahnce abiotic tolerance in crop along with the future prospects of the transgenic potato.
Keywords: Genetic improvements; Abiotic stress; GMOs
Abbreviations: GNP: Gross National Agricultural Product; BV: Biological Value; IPM: Integrated Pest Management; ABA: Abscisic Acid; bZIP: Basic Leucine Zipper; TF: Transcription Factor; CE: GC-rich Coupling Elements; ABRE: ABA-responsive Element; CBP80: Cap-Binding Protein 80; ROS: Reactive Oxygen Species; BADH: Betaine Aldehyde Dehydrogenase; COD: Choline Oxidase; NT: Non-transgenic; TPS1: Trehalose-6-phosphate Synthase; amiRNAs: Artificial MicroRNAs; DREB; Dehydration-responsive Element-binding, DRE/CRT: Dehydration-responsive Element/C-repeat; EMSA: Electrophoretic Mobility Shift Assay; P5CS: 𝛿1-pyrroline-5-carboxylate Synthetase; CDPKs: Calcium-dependent Protein Kinases; GalUR: D-Galacturonic Acid Reductase; desA: Acyl-lipid 12-desaturase; SCOF-1: Soybean Cold Inducible Zinc Finger Transcription Factor; sHsps: Small Heat Shock Proteins; ATP: Adenosine Triphosphate; CuZnSOD: Copper–zinc Superoxide Dismutase; APX: Ascorbate Peroxidase; NDPK2: Plant Nucleoside Diphosphate Kinase 2; SSA: Transgenics with CuZnSOD genes and APX transgenes only; SN: Transgenics with NDPK2 only; 2-Cys Prx : 2-cysteine peroxiredoxin; MV: Methyl Viologen; DHN4: Dehydrin 4; miRNA: Micro RNA; CRISPR: Clustered Regularly Interspaced Short Palindromic Repeats; Cas9: CRISPR-associated protein9 nuclease
- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
Introduction
Potato is a family of the Solanaceae, an enormous plant family with more than 3000 varieties [1]. More than 374,463,885 million tones of potatoes are produced worldwide [2]. As the global population reaches to 9.7 billion by 2050, the potato will play important role in securing global food resources. In Turkey, potato accounts for 3% of the gross national agricultural product (GNP), whereas it is about 3.1% in the EU-27 countries, hence contributing significantly to Turkish economy. It was cultivated on an area of 172,000 ha with the production of 3.9 million tons of potatoes. Central Anatolia including Nigde shares more than 60% potato production in this regard [3]. Potato has high amount of protein after soybean; patatin is the dominant storage protein [4]. The tubers of potato contain important dietary origin of starch, protein, vitamins and antioxidants [5]. Potato protein contains great biological value (BV) with a BV of 90-100 in contrast with whole egg (100) [6].
Potato crop is exposed to various sorts of abiotic stresses including drought saline cold and heat stresses. In areas where there is erratic rain fall or inadequate water supply, the cultivation of potato remains challenging [7]. As the world is going to face global warming issues, water restriction would be a threat in coming decades [8,9]. Drought stress periods may vary yearly in terms of duration and severity [10]. As per Hijmans [11], the potato losses due to climate change are expected to be 18-32% in first quarter of this century. Therefore, the development of transgenic drought resistant potatoes is an important issue. Potato as a frost-sensitive species adopts cool environment, and its regular growth and improvement are prevented by high temperature. When the temperature increases above 25 °C, tuber’s growth is terminated, and when the temperature rises to 39 °C, growth of stems and leaves are terminated [12]. Therefore, efforts are going on to develop abiotic stress tolerant potatoes. The potato presents unique challenges and advantages to plant breeders. As it is propagated vegetatively by tuber cuttings, potato cultivars don’t need to be bred to produce homogenous plants from true seed. A major disadvantage of potatoes for breeders is that potato is tetraploid, making it difficult to transfer desirable traits between cultivars and to have them expressed in subsequent progeny. The several species of Solanum are cultivated in Peru and Central America. These species provide a rich source for potential traits to breed into S. tuberosum, including tuber qualities (e.g. colors ranging from white to deep purple skin and flesh) and resistance to insect pests and diseases. Unfortunately, many of these wild Solanum relatives are diploid that further complicates breeding process. Hence, insertion of candidate gene(s) encoding for desirable economic trait by genetic engineering is a particularly attractive and valuable process for developing new potato cultivars [13]. There are many success reports of development of transgenic crops with traits of tolerance to abiotic stress [14]. These transgenic plants are becoming important components of integrated pest management (IPM) worldwide [15].
- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
Improvement of Drought Stress Tolerance in Potatoes
S. tuberosum L. has been reported to be extremely sensitive to the drought stress, and with its leaves being more sensitive as this plant has less capability to absorb water from soil as compared to other crops [16]. The shallow root architectural system in potatoes is particularly detrimental to potatoes in that under drought, increased salinity and extreme temperature fluctuations, tuber yield and quality can be significantly plummeted [16-19]. Drought alone is expected to significantly decrease potato yield as much as 18 to 32% over the period of 2040-2069 [11]. Drought stress can bring about reduction in photosynthetic rate as well as reduction on biomass fresh weights of potato leaves and also it affects leaf number, leaf size and shoot length [16-18, 20-22]. One of the strategies that have been employed in ameliorating drought tolerance has been by introducing transgene that codes for bZIP (basic leucine zipper) TFs (transcription factors). On ABA (Abscisic acid) signal perception by specific receptors, signal transduction takes place when phosphorylation of bZIP TFs takes place via Ser/ Thr kinases which activate these bZIP TFs to bind to CE (GC-rich coupling elements) or cis-acting ABRE (ABA-responsive element) sequences to elicit various abiotic stresses [23]. Moon et al. [24] reported that the CaBZ1 gene isolated from hot pepper (Capsicum annuum) that encodes for bZIP transcription factor induced by ABA and under salinity and osmotic stresses when over-expressed in potatoes didn’t result in detrimental phenotypic traits in transgenic potatoes.
Under ABA treatment and drought condition, the transgenic plants showed a prompt stomatal closure with a decline in water loss rate and elevated yield under drought stress as compared to the wild-type. Kim et al. [25] generated improved drought tolerant transgenic potatoes based on decrease in water loss by overexpressing AtYUC6 gene associated with auxin biosynthesis in potato under CaMV 35S promoter using pCAMBIA1300pt–YUC6 construct. The transformed plants exhibited phenotypes associated with high-auxin like increment in the heights, erect stature, longevity and narrow-downward curled leaves. Pieczynski et al. [26] silenced the cap-binding protein 80 (CBP80) gene in potato cultivar Desiree using artificial miRNAs. CBP80 protein regulates miR159, MYB33 and MYB101 levels which are known to be the important regulators of ABA transduction pathway and drought tolerance. Downregulation of CBP80 was observed in silenced potato plants with decreased miR159 levels and increased levels of its target mRNAs, MYB33 and MYB101, which rendered higher drought tolerance in silenced plants. In addition, ABA-hypersensitive stomatal closing, elevation in thrichome and leaf stomata densities with decrease in number of michrochannels were reported, which correlated with increased tolerance to water stress. Similar pattern was reported in cbp80 mutant Arabidopsis. One of the major osmolytes, glycine betaine, accrues when encountered by abiotic stresses such as drought, high salinity and extreme temperature conditions. Not all plant species are capable of natural production or accretion of glycine beta. There has been extensive research with the ability to generate this compound in transgenics to ameliorate its tolerance to salt, cold, drought or high temperature stresses. Choline monoxygenase catalyzes conversion of choline to betaine aldehyde and ultimately to glycine beta under the catalysis of betaine aldehyde dehydrogenase [27-29]. Cheng et al. [30] cloned codA gene, obtained from Arthrobacter globiformis that directly converts choline to glycine beta, under the SWPA2 promoter to generate transgenic potatoes. The water stress was simulated with 20% PEG under the control of SWPA2 promoter. Accumulated glycine beta was reported in transgenic potatoes while non-transgenic potatoes exhibited no glycine beta accumulation as potato is not a glycine beta accumulating plant. Furthermore, reactive oxygen species (ROS) was shown to be accumulated and the transgenic potatoes exhibited strong drought resistance and recover ability. They further demonstrated that the glycine beta produced as a result of codA gene expression in transgenics could prevent membrane lipid peroxidation and degradation of chlorophyll caused by stress. Zhang et al. [31] reported that the cloning of transgene BADH gene (Betaine aldehyde dehydrogenase), under the control of drought- and NaCl- induced promoter rd29A from A. thaliana, into potato cultivar Gannongshu 2 increased the plant height of transgenic potatoes under NaCl and PEG stresses by 0.4 to 0.9cm, whereas fresh weight per plant increased from 17-29% as compared to non-transgenic controls indicating the drought and salt tolerances enhanced in the transgenics when BADH activity was upregulated.
Choline can be directly catalyzed to GB by choline oxidase (Cod) but it does not exist in higher plants. Different sources were adopted to transform codA gene: one by taking the gene from rhizobacterium and using promoter SWPA2 from sweet potato [32], and the other from transit peptide of Rubisco from tobacco [33]. The transgenic plants showed better tolerance to drought and salinity stress as compared to non-transgenic (NT) ones. There was also improvement in biomass of potted transgenic plants as compared to NTs when they were kept in water less conditions for 14 days to observe drought tolerance. Cheng et al. [30] further proceeded with these experiments by focusing on antioxidant system in drought conditions. The plants were kept in drought stress for 4 days and later were subject to rehydration conditions for 2 days. Transgenic plants were better to non-transgenic ones in many aspects including higher chlorophyll content, higher activity of antioxidant enzymes (caralase, peroxidases and superoxide dismutase), lower MDA and better recovery from water deficiency. Thus, tolerance against drought stress can be controlled in potato plants by increasing accumulation of GB. One of the elements involved in ROS signaling pathway in Arabidopsis is Arabidopsis annexin 1 (AtANN1). A family of calcium and membrane binding proteins, annexin, has also been found to confer stress tolerance to plants. ROS triggers boost in Ca2+ under salinity stress conditions and AtANN1 has been thought to be involved in activation of calcium conductance by NADPH oxidases in root epidermal cells. Annexin’s major role is to offset oxidative stress to maintain cell redox homeostasis and ameliorate tolerance against drought stress [34, 35]. Szalonek et al. [35] studied the role of StANN1 by overexpressing StANN1 in potatoes under CaMV 35S promoter and obtained more drought tolerant transgenics than the wild type with more water in green tissues, maintained chloroplast function with chlorophyll b and xanthophylls accretion. Also, the study inferred that the transgenic potatoes maintained effective photosynthesis during drought stress that increased its yield rate than non-transformed plants even under water stress. Trehalose overexpression has been shown to confer resistance against abiotic stress in plants such as rice and Arabidopsis [36-38]. However, in potatoes, it has been reported to be challenging to develop drought resistance transformants [39]. Nevertheless, improved drought tolerance in potatoes has been reported. Stiller et al. [40] cloned trehalose-6-phosphate synthase (TPS1) gene isolated from yeast with StDS2 drought inducible promoter and transformed the construct in White Lady potato cultivar. The resulting transgenics were drought tolerant and displayed higher stomatal conductance with increased net photosynthesis rates as compared to the wild types. There has also been a report on the use of artificial microRNAs (amiRNAs) in silencing CBP80/ABH1 gene in S. tuberosum to increase tolerance to water shortage conditions [41]. Myrothamnus falbellifolia (a terrestrial plant) and plants of Liliaceae family have been reported to synthesize glucosyl glycerol under drought stress conditions [42,43]. Transgenic potato plants were prepared by expressing ggpPS gene using 2 different types of promoters, i.e. CaMV35 and rd29A. Both type of transformed plants showed improvement in shoot length under both drought and saline stresses in green house conditions. Also, accumulation of GG was observed in leaves in both cases. But only rd29A transformants were able to accumulate GG in tuber [44]. Drought tolerance was introduced in potato plants by over expression of AtYUC6, as YUCCA family is known for its contribution in auxin biosynthesis. Potted transgenic plants of 4 months were evaluated, and they showed drought tolerance [25]. El-Banna et al. [45] used overexpression of gene regulating PR-10a (which is induced in osmotic/salinity stress) to control osmotic and salinity stress in potato callus. High proline accumulation and low oxidized glutathione was observed in transgenic callus in stress condition. Transgenic potatoes were developed by transferring sweet potato orange gene (ibOr). The transgenic plants were given drought stress in green house conditions. The transgenic lines showed improved resistances [46]. Thus, multiple researchers quoted above prove that there are a large number of genes which have important contribution for development of drought tolerance in potato plants.
- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
Improvement of Salt Stress Tolerance in Potatoes
There are various implications of excessive soil salinity such as reduced yield and water potential, toxicity, alteration in metabolism of plants, ion imbalances and a drop-in assimilation of CO2. By the mid 21st century, a rise in arable land salinization seems to result in a loss of 50% of the arable land [47,48]. Potato has been categorized under moderately salt-sensitive crop; nonetheless, different potato cultivars respond differentially to salinity [49,50]. It has been identified that the Dehydration-responsive elementbinding (DREB)-1 TFs have been strongly induced under heat, drought, and high salinity as well as low-temperature stress conditions [51]. It has been elucidated that DREB1A and DREB2A TFs recognize DRE core sequence G/ACCGAC [52,53]. Additionally, DREB1A/CBF3 and DREB2A TFs specifically interact with cisacting dehydration-responsive element/C-repeat (DRE/CRT) region which play the role in drought and cold stress–responsive gene expression in A. thaliana [54]. Also, DREB1A/CBF1, DREB1A/ CBF3, DREB1B/CBF1 and DREB1C/CBF2 TFs have been found to be induced under low temperature stress conditions [55-57]. Bouaziz et al. [19] isolated StDREB2 gene from potato (cv. Nicola) plants and overexpressed it in transgenic potatoes, bioinformatics analysis of which unraveled that the StDREB2 protein belongs to the A-5 group of DREB subfamily. The overexpression in transgenic plants resulted in increased tolerance against salt stress. With the help of electrophoretic mobility shift assay (EMSA), they further found that this transcription factor was bound specifically to the DRE core element (ACCGAGA) in vitro. 𝛿1-pyrroline-5-carboxylate synthetase (P5CS) was increased in transgenics under salinity stress with collateral upregulation in proline accumulation inferring that StDREB2 might be responsive to abiotic stresses via ABA signaling regulation and through proline synthesis mechanism. In another similar study conducted by Bouaziz et al. [58], StDREB1 was found to be induced by drought, sodium chloride, cold temperature, and ABA. StDREB1 cDNA overexpression using pMDC32:StDREB1 construction in transgenic potatoes showed enhanced salt and drought stress tolerance in comparison to the control plants. As described above for StDREB2, the authors indicated that this increased stress tolerance could be due to P5CS-RNA expression as well the resulting proline osmoprotectant accumulation which also induced calcium-dependent protein kinases (CDPKs) stress responsive genes in standard and salt-stressed transgenics (Table 1).
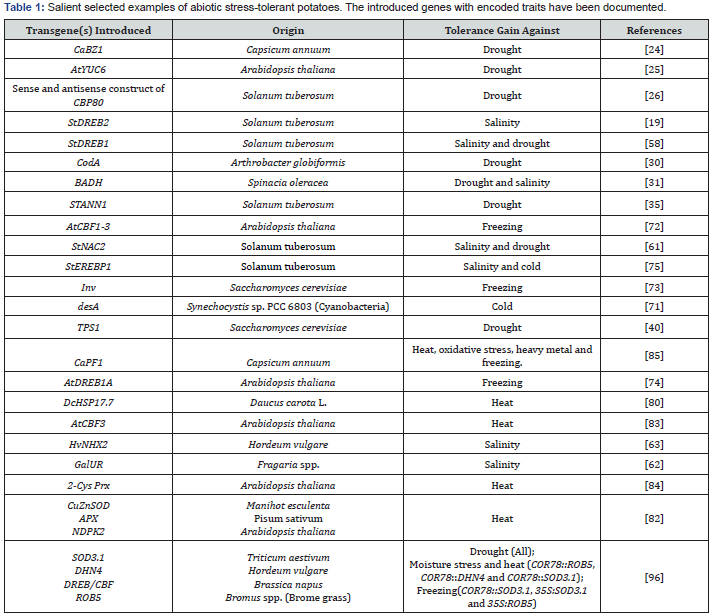
Agrobacterium mediated transformation was used to transform DREB1A of Arabidopsis in Longshu3 (L3) cultivar of potato. Positive gene integration and over expression of gene was confirmed using PCR, Southern blotting and semi quantitative RT-PCR Analysis. The transgenic lines showed very partial wilting when water was withheld for 14 days as compared to non-transgenic plants, thereby confirming that over expression of DREB1A improved drought tolerance in potato plants [59]. In plants, the other TF group, NAC, might be involved in regulating transcriptional reprogramming associated with plant stress responses and is involved in stress responses in plants [60]. StNAC2 overexpression under the control of CaMV 35S promoter in transgenic potato yielded ameliorated tolerance to salt in vitro and drought tolerance in pot growing condition. Phytophthora infestans also induced StNAC2 expression in addition to its induction by salt, drought and wounding stresses, indicating the possibility of cross-talking of signaling molecules involved in biotic and abiotic stresses [61].
Potato cultivar Taedong valley was overexpressed using Gal- UR gene (D-Galacturonic acid reductase) isolated from strawberry under the control of CaMV 35S promoter and exhibited enhanced ascorbate (AsA). The study identified the function of GSH (Reduced glutathione), its regulation via ascorbate pathway enzymes and its role in improving salt tolerance in plants. GalUR gene in transgenics enhanced ascorbic acid content (L-AsA), and ultimate< ly reduced oxidative stress-induced damage leading to salinity tolerance improvement with tub erization even at 200mM of NaCl in transformants as compared to wild types [62]. HvNHX2, an Na+/ H+ antiporter gene from barley, under the control of CaMV 35S was transformed to potato cultivars Skoroplodny and Jubilei Zhukova which conferred improved tolerance to NaCl. At 200mM NaCl, Jubilei Zhukova-derived transgenics survived, whereas Skoroplodny couldn’t. The transgenic plant had enhanced dry weight, root length, and suppressed cell explansion than the non-transformants. Postassium was found to be elevated in the transgenic roots instead of sodium [63]. Plant cells are prone to salinity damages in two perspectives i.e. ionic stress and osmotic stress. Osmoregulation in plants by using osmoregulator substances, e.g. Mannitol, has been used to confer salinity tolerance in plants [64- 66]. Potato is among the plant species which do not accumulate mannitol naturally. Rahnama et al. [67] produced transgenic potatoes by taking the mannitol-1-phosphate dehydrogenase (mtlD) gene from E. coli Mt1D gene was proved to confer salinity tolerance to potato.
- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
Improvement of cold stress tolerance in potatoes
The other stress that holds devastation for the cultivated potatoes is freezing temperature, and as potatoes being frost sensitive species, these plants are inept to thrive well under cold stress condition with maximum freezing tolerance threshold of -3 °C, both before and after exposure to freezing temperatures. Drop in temperature can bring about a plunge in various enzymatic activities with ice formation under low temperature, and ultimately dehydration in cytoplasm. There are some wild potatoes which are better frost tolerant than the cultivated species which could be potential genetic resource for its use in genetic improvement of cultivated potato crops [68,69]. It has been shown that the desaturase gene protein can be helpful in conferring cold resistant stress in plants [70]. Amiri et al. [71] demonstrated that the desA gene (acyl-lipid 12-desaturase) isolated from Synechocystis sp. PCC 6803 (cyanobacteria) transformed in potato lines could ameliorate the cold tolerance by varying lipid polyunsaturated fatty acid levels. Under cold stress, the study observed more sensitivity in control plants than those for desA gene introduced transgenic plants. The damage index in control plants was more at 7 °C in the control plants, whereas those for the three lines in transgenics were significantly reduced. Additionally, the study concluded that the desA protein has negative role on stem growth as the stem length in transformants reduced by nearly 60% than the control plants.
The S. tuberosum cv. Umatilla was overexpressed with AtCBF1-3 driven by CaMV 35S or a stress-inducible Arabidopsis promoter rd29A. Under CaMV 35S promoter, AtCBF1 and AtCBF3 increased freezing tolerance about 2 °C, whereas AtCBF2 transgenics couldn’t. Under rd29A promoter, transgenics exhibited same level of freezing tolerance in few hours, whereas CaMV 35S exhibited tolerance only under low temperature but not under freezing condition. Transgenics with AtCBF constitutive expression under freezing temperature exhibited smaller leaves, stunted growth, delay in flowering, and yield loss. Under the same freezing condition, transgenics driven by rd29A resulted in improvement of phenotype indicating use of stress inducible promoter to direct CBF transgene expression to ameliorate freezing tolerance in potatoes [72]. Pino et al. [69] further reported that the ectopic overexpression of AtCBF1 in frost-sensitive S. tuberosum and S. commersonii both induced COR gene expression devoid of cold stimulus and stimulated increase in tolerance against cold temperature of about 1-2 °C and 4 °C in transgenic S. tuberosum and S. commersonii respectively as compared to their wild types. The study highlighted that S. tuberosum has CBF regulated genes that can increase the freezing tolerance of plants grown at warm temperature. However, the authors suggest that there may be lack of additional cold-tolerant genes in potatoes that made it incapable for transgenic potatoes to increase chilling tolerance beyond those conferred by AtCBF1 overexpression as it was reported that the transgenic S. tuberosum showed no further gain in freezing stress tolerance under low temperature condition while S. commersonii could exhibit better acclimation to low temperature. Invertase gene was isolated from yeast and transformed in potato (cv. Desiree) under the control of B33 class 1 tuber-specific promoter. Invertase activity, content of sugar and cold tolerance were measured by MDA content in in vitro plants. Under controlled conditions in transgenic plants, invertase content and sugar content increased in leaves at 22 °C and MDA content enhanced as compared to the wild types. The authors have suggested that the invertase gene as a transgene might have conferred tolerance at chilling temperature apparently as a result of variation in sugar ratio [73].
Desiree cultivar of potato was transformed with the gene construct of A. thaliana derived AtDREB1A, the gene driven by the rd29A promoter from the same source, which showed enhanced freezing tolerance than the wild type. Also, in many of the transgenic lines, the authors observed recovery from freezing stress [74]. Improved salt tolerance at 75mM and cold tolerance was observed in transgenic potatoes (cv. Superior) when StEREBP1 was overexpressed under a constitutive CaMV 35S promoter inferring that StEREBP1 is another TF associated with abiotic stresses in plants. The yield in elite transformant improved by approximately 50% under cold stress. Also, it was observed that StEREBP1 binds to DRE/CRT and DCC cis-elements for its activity and microarray and RT-PCR techniques showed that many other stress responsive genes containing GCC box had been under the overexpression of StEREBP1 TF [75].
Increased cold stress resistance was induced in potato plants by transformation of soybean cold inducible zinc finger transcription factor (SCOF-1). The transgenic plants were kept under cold stress at 4 °C for 5 days. The expression of SCOF-1 correlated positively with the cold stress. The results showed that overexpression of SCOF-1 can efficiently increase the tolerance against freezing stress [76].
- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
Improvement of Heat Stress Tolerance in Potatoes
Potato yield, mainly in the warm tropical region, is narrowed down by high temperature by hindering synthesis of starch in tubers [77]. The potato tuber growth optimal temperature is approximately 20 °C as potato is a cool-season crop [78]. The high temperature has been reported to cause build-up of glycoalkaloid in potatoes that alters the carbohydrate metabolism in tuber tissue leading to heat-induced damage of potato tubers [79]. Increased temperatures can also lead to a drop-in tuber dry matter accumulation. The tetraploidy of the cultivated potatoes makes potato genome more byzantine and high degree of sterility has precluded development of conventional breeding. This makes genetic engineering of potatoes to improve thermotolerance in potatoes inevitable [80]. Most small heat shock proteins (sHsps) are perceived only under heat stress in vegetative tissues and not under normal growth conditions. With increment in temperature up to 10-15 °C above the normal growth condition could actually be lethal to the organisms and eventually can induce heat shock response and stress tolerance in plants [81]. sHsps are adenosine triphosphate (ATP)-independent molecular chaperones which preclude irreversible aggregation or initiates correct refolding of the incorrectly folded or partially damaged proteins. In addition, many studies have reported incorporation of sHSP genes in ameliorating thermotolerance in various organisms [80]. Enhanced thermotolerance in the transgenic Desiree potatoes, probably the first one in potato, were obtained when the carrot gene that codes for heat shock protein (DcHSP17.7 gene) was transformed, regulated by CaMV 35S promoter. Under normal condition without stress, DcHSP17.7 expressed constitutively, though not in abundant amount. Transgenics showed ameliorated stability of the cellular membrane at increased temperature and prompt high tuber yield even at constant 29 °C stress when compared with the non-transformants. The study found that there was a good increase in percentages and dry weight of microtubes [80].
Transgenic potato cv. Atlantic which expressed cassava CuZn- SOD (Copper–zinc superoxide dismutase), pea APX (Ascorbate peroxidase) and Arabidopsis NDPK2 (Plant nucleoside diphosphate kinase 2) genes together under the control of stress-inducible SWPA2 promoter, all of these genes were shown to be ameliorating tolerance to high temperature stress and methyl viologen- induced oxidative stress. This transgenics (SSAN) showed enhanced tolerance to methyl viologen than the non-transgenics, SSA (transgenics with CuZnSOD genes and APX transgenes only) and SN (transgenics with NDPK2 only) plants. SSAN sprayed with 40μM methyl viologen resulted 53% less visible damage than SSA and 83% less than SN. Furthermore, high temperature tolerance as high as 42 °C was achieved in SSAN transgenics with only 6.2% reduction in photosynthesis rate than those grown at 25 °C, while this drop in rate was 50% for SN and 18% for SSA transgenics [82]. Dou et al. [83] isolated CBF3 gene from Arabidopsis which can be induced under cold stress, cloned under CaMV 35S promoter control as well as rd29A promoter and transformed the construct into the ‘luyin NO. 1’ potato cultivar. AtCBF3 could be expressed under heat stress even at the temperature higher than 40 °C. The accretion of O2 • − and H2O2 was declined in the transformants as compared to the non-transformants with increased D1 protein accumulation, net phososynthetic rate and maximal PSII photochemical efficiency in transgenics. The results inferred that the amelioration in heat stress tolerance was exhibited as a result of ectopic expression of AtCBF3 gene which enhanced photosynthesis and antioxidant defense. Nevertheless, HSP70 accumulation was lesser in transgenics than the wild types indicating HSP70 role was uninvolved in the pathway.
In another study, Kim et al. [84] overexpressed potato cultivar Atlantic with antioxidative enzyme 2-cysteine peroxiredoxin (2-Cys Prx) gene, which aids in eliminating peroxides and shields the photosynthetic membrane from oxidative damage, regulated by stress-inducible SWPA2 promoter or 35S promoter. The treatment with 3μM methyl viologen (MV) on both promoter-driven transgenics exhibited approximately 33% and 15% less damage than the wild type. Photosynthetic activity for SWPA2-promoter driven transgenics declined by 25% when 300μM MV was sprayed onto whole plants, whereas for non-transgenics it dropped further to 60%. SWPA2-driven transgenics could tolerate up to 42 °C.
Multiple tolerance against stresses including heat, oxidative stress, heavy metal and freezing was achieved in transgenic potatoes when ERF/AP2-type TF CaPF1 gene isolated from Capsicum annuum was cloned under the control of CaMV 35S promoter and the construct vector was transformed to Atlantic potato cultivar. However, the tuber formation in transgenics in vitro was severely impeded as compared with the wild-type [85].
- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
In addition to the genes directly related to different stresses, transcription factors also play a vital role in plants’ natural defense against various abiotic stresses. So, transformation of better transcription factor can be a good strategy to enhance tolerance of plants against various abiotic stresses [72,86,87]. Transgenic potatoes were developed in independent researches via transforming AtDREB/CBF gene. Different promoters were used in different researches i.e. rd29A [72,74,88,89] and 35S promoters [72,90]. The transgenic potato plant showed increased resistance against cold stress, salinity and drought tolerance [91]. Youm et al. [85] transformed a CaPF1 gene from Capsicum annuum in potato plants, this gene encodes the transcription factor AP2/ERF which has role in tolerance against cold stress and pathogens. The results showed an increased tolerance against drought, freezing, heat, heavy metal ions and oxidative stress in transgenic lines but tuber formation was retarded in these transformants as compared to non-transgenic plants. R2R3-type Myb TF has been reported to be involved in secondary metabolism and responding to biotic and abiotic stresses [92,93]. It is encoded by IbMybb1 gene. Cheng et al. [94] transformed this gene in potato plants which ultimately showed higher level of secondary metabolites (anthocynins, flavonoids and total phenols). These secondary metabolites are released in drought and UV-B ray stresses, thus the transformants showed better response under these stresses. Shin et al. [95] developed transgenic potato plants by transformation of StMyb1R-1 gene (a stress inducible gene) which encodes R-1 type MYB-like TF. The transgenic plants showed better response under drought stress.
- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
Combination of Many Different Tolerances
Waterer et al. [96] developed potato cultivar Desiree by introducing transgenes under the control of 35S promoter or stress-inducible Arabidopsis COR78 promoter. Four types of transgenes were used: wheat mitochondrial MnSOD (SOD3.1), barley dehydrin 4 (DHN4), cold-inducible transcription factor DREB/ CBF from canola and stress-inducible brome grass derived ROB5 gene coding for LEA group 3-like protein. In total, six transgenics were used: COR78:: SOD3:1, COR78::DHN4, COR78::DREB/ CBF, COR78::ROB5, 35S::SOD and 35S::ROB5. Many of the transformed lines were reported to produce higher yield even at the significant drought stress. Under moisture stress, COR78::ROB5, COR78::DHN4 and COR78::SOD3.1 transgenics performed with higher yield with heat tolerance up to 44 °C. The tolerance was seen the highest for COR78::SOD3.1 transformants. At 10 °C, 35S::- SOD3.1 grew better than the non-transformants and COR78::- SOD3.1, 35S:SOD3.1 and 35S:ROB5 transgenics also showed improved tolerance against freezing stress.
StnsLTP1 is a potential gene from potato which was reported to be thermo-tolerant. Transgenic potato lines were developed which showed overexpression of this gene. The transgenic plants not only showed increased cell membrane integrity under stress conditions but also depicted increased antioxidant enzyme activity. The stress related genes (StAPX, StCAT, StSOD, StHsfA3, StHSP70 and StSHSP20) were also unregulated in transgenic lines [97].
- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
Future prospective
Micro RNAs as a prospective candidate in improving abiotic stress tolerance in potato
Micro RNAs (miRNA) have been known to play an important role in various abiotic stress tolerance and some of them have been elucidated for transgenic plants. miR156 overexpression in Arabidopsis revealed enhanced tolerance to heat stress [98], constitutive overexpression of miR169 in tomato ameliorated tolerance against drought [99], miR319 overexpression in transgenic rice enhanced cold stress tolerance [100] and miR402 overexpression in Arabidopsis increased tolerance against drought, salinity and cold stresses in the transformants [82]. Similar type of miRNA or other miRNAs that have been explored to be involved in various abiotic stress tolerances can be studied by overexpression, silencing or other manipulations in potatoes which could improve the abiotic stress tolerance in cultivated potatoes.
CRISPR/Cas9, an emerging technology for improving abiotic stress tolerance in potato
A newly introduced technology Clustered regularly interspaced short palindromic repeats (CRISPR) and CRISPR-associated protein9 nuclease (Cas9) is revolutionizing the genetic engineering concepts in plants. CRISPR has been successfully implemented in various organisms including plants. It was first utilized in model plants like Arabidopsis, tobacco, and with passage of time it has been used for making genetic transitions in crops like maize, soybean, sorghum, wheat, woody plants, etc. [101-108]. Osakabe et al. [109] have used CRISPR for dealing with drought stress in Arabidopsis plants and they have achieved some success. But authors suggested that more studies are needed in this respect. CRIPSR/Cas9 has been utilized to study genes related to abiotic tolerance in plants. Two glycosyltransferase genes, i.e. UGT79B2 and UBT79B3, are thought to be responsible for making Arabidopsis plants tolerant in case of drought cold and salt stress. When these genes were knocked down by using CRISPR/Cas9 the plants became more susceptible to these stresses [110]. Shi et al. [111] utilized CRISPR/ Cas9 for generating transgenic maize with improved drought tolerance. Thus, studies need to be done in potatoes by utilizing the efficacy of CRISPR/Cas9 to enhance abiotic stress resistance in crop.
- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
Acknowledgment
We thank TUBITAK 2215 (Turkey) for providing fully-funded PhD scholarship to Mr. Sarbesh das Dangol.
- Review article
- Abstract
- Introduction
- Improvement of Drought Stress Tolerance in Potatoes
- Improvement of Salt Stress Tolerance in Potatoes
- Improvement of cold stress tolerance in potatoes
- Improvement of Heat Stress Tolerance in Potatoes
- Use of Transcription Factor for Introducing Tolerance Against Abiotic Stress in Potatoes
- Combination of Many Different Tolerances
- Future prospective
- Acknowledgment
- References
References
- Jacobs MMJ, Smulders MJM, Van den berg RG, Vosman B (2011) What’s in a name; Genetic structure in Solanum section Petota studied using population-genetic tools. BMC Evol Biol 11: 42.
- FAO (2013) Food and Agricultural Organization of the United Nations Database. Rome, Italy.
- Caliskan ME, Onaran H, Arıoğlu H (2010) Overview of the Turkish potato sector: Challenges, achievements and expectations. Potato Res 53(4): 255-266.
- Liedl BE, Kosier T, Desborough SL (1987) HPLC isolation and nutritional value of a major tuber protein. Am J of Potato Res 64: 545-557.
- Burlingame B, Mouille B, Charrondiere R (2009) Nutrients, bioactive non-nutrients and anti-nutrients in potatoes. J Food Compos Anal 22(6): 494-502.
- Buckenhuskes HJ (2005) Chapter 1 Nutritionally relevant aspects of potatoes and potato constituents. In: Haverkort AJ, Struik PC (Eds.), Potato in Progress: Science Meets Practice. Wageningen Academic Pub, The Netherlands, pp. 17-45.
- Thiele G, Theisen K, Bonierbale M, Walker T (2010) Targeting the poor and hungry with potato science. Potato J 37(3-4): 75-86.
- FAO statistical databases (2012) FAOSTAT.
- Simelton E, Fraser EDG, Termansen M, Benton TG, Gosling Sn, et al. (2012) The socioeconomics of food crop production and climate change vulnerability: A global scale quantitative analysis of how grain crops are sensitive to drought. Food Secur 4(2): 163-179.
- Easterling W, Aggarwal P, Batima P, Brander K, Erda L (2007) Chapter 5 Food, fibre and forest products. In: Parry ML, Canziani OF, Palutikof JP, van der Linden PJ, Hanson CE (Eds.), Climate change 2007: Impacts, adaptation and vulnerability. Contribution of working group II to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, UK, pp. 273-313.
- Hijmans RJ (2003) The effect of climate change on global potato production. Am J Potato Res 80(4): 271-279.
- Krauss A, Marschner H (1984) Growth rate and carbohydrate metabolism of potato tubers exposed to high temperatues. Potato Res 27(3): 297-303.
- Kennedy GG (2008) Chapter 1 Integration of insect-resistant genetically modified crops within IPM programs. In: Romeis J, Shelton AM, Kennedy GG (Eds.), Integration of insect-resistant genetically modified crops within IPM programs. Springer Netherlands, The Netherlands, pp. 1-26.
- Bakhsh A, Hussain T (2015) Engineering crop plants against abiotic stress: current achievements and prospects. Emir J Food Agric 27(1): 24-39.
- Kos M, Van Loon JJ, Dicke M, Vet LE (2009) Transgenic plants as vital components of integrated pest management. Trends Biotechnol 27(11): 621-627.
- Weisz R, Kaminski J, Smilowitz Z (1994) Water-Deficit effects on Potato leaf growth and transpiration - Utilizing fraction extractable soil water for comparison with other crops. Potato J 71(12): 829-840.
- Deblonde P, Ledent J (2001) Effects of moderate drought conditions on green leaf number stem height, leaf length and tuber yield of potato cultivars. Eur J Agron 14(1): 31-41.
- Ekanayake I, Midmore D (1992) Genotypic variation for root pulling resistance associated with drought. Euphytica 61(1): 43-53.
- Bouaziz D, Pirrello J, Amor AHB, Hammami A, Charfeddine M, et al. (2012) Ectopic expression of dehydration responsive element binding proteins (StDREB2) confers higher tolerance to salt stress in potato. Plant Physiol Biochem 60: 98-108.
- Dalla Costa L, Vedove G, Gianquintoi G, Giovanardi R, Peressotti A (1997) Yield, water use efficiency and nitrogen uptake in potato: influence of drought stress. Potato Res 40(1): 19-34.
- Kiziloglu F, Sahin M, Tunc T, Diler S (2006) The effect of deficit irrigation on potato evapotranspiration and tuber yield under cool season and semiarid climatic condition. J Agronomy 5(2): 284-288.
- Li J, Cang Z, Jiao F, Bai X, Zhang D (2015) Influence of drought stress on photosynthetic characteristics and protective enzymes of potato at seedling stage. J Saudi Soc Agric Sci 16(1): 82-88.
- Banerjee A, Roychoudhury A (2015) Abscisic-acid-dependent basic leucine zipper (bZIP) transcription factors in plant abiotic stress. Protoplasma 254(1): 3-16.
- Moon S, Han S, Kim D, Yoon IS, Shin D (2015) Ectopic expression of a hot pepper bZIP-like transcription factor in potato enhances drought tolerance without decreasing tuber yield. Plant Mol Biol 89(4-5): 421- 431.
- Kim JI, Baek D, Park HC, Chun HJ, Oh D (2013) Overexpression of Arabidopsis YUCCA6 in potato results in high-auxin developmental phenotypes and enhanced resistance to water deficit. Mol Plant 6(2): 337-349.
- Pieczynski M, Marczewski W, Hennig J, Dolata J, Bielewicz D (2013) Down-regulation of CBP80 gene expression as a strategy to engineer a drought-tolerant potato. Plant Biotechol J 11(4): 459-469.
- Sakamoto A, Murata N (2000) Genetic engineering of glycinebetaine synthesis in plants: current status and implications for enhancement of stress tolerance. J Exp Bot 51(342): 81-88.
- Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59(2): 206- 216.
- Fu X, Khan EU, Hu S, Fan Q, Liu J (2011) Overexpression of the betaine aldehyde dehydrogenase gene from Atriplex hortensis enhances salt tolerance in the transgenic trifoliate orange (Poncirus trifoliata L. Raf.). Environ Exp Bot 74: 106-113.
- Cheng Y, Deng X, Kwak S, Chen W, Eneji A (2013) Enhanced tolerance of transgenic potato plants expressing choline oxidase in chloroplasts against water stress. Bot Stud 54(1): 30.
- Zhang N, Si H, Wen G, Du H, Liu B (2011) Enhanced drought and salinity tolerance in transgenic potato plants with a BADH gene from spinach. Plant Biotechnol Rep 5(1): 71-77.
- Kim KY, Kwon SY, Lee HS, Hur Y, Bang JW (2003) A novel oxidative stressinducible peroxidase promoter from sweetpotato: molecular cloning and characterization in transgenic tobacco plants and cultured cells. Plant Mol Biol 51(6): 831-838.
- Ahmad R, Kim M, Back K, Kim H, Lee H (2008) Stressinduced expression of choline oxidase in potato plant chloroplasts confers enhanced tolerance to oxidative, salt, and drought stresses. Plant Cell Rep 27(4): 687-698.
- Laohavisit A, Richards SL, Shabala L, Chen C, Colaco RD (2013) Salinityinduced calcium signaling and root adaptation in Arabidopsis require the calcium regulatory protein annexin 1. Plant Physiol 163(1): 253- 262.
- Szalonek M, Sierpien B, Rymaszewski W, Gieczewska K, Garstka M (2015) Potato annexin STANN1 promotes drought tolerance and mitigates light stress in transgenic Solanum tuberosum L plants. PLoS ONE 10(7): 1-38.
- Avonce N, Leyman B, Mascorro-Gallardo JO, Van DP, Thevelein JM, et al. (2004) The Arabidopsis trehalose-6- P synthase AtTPS1 gene is a regulator of glucose, abscisic acid, and stress signaling. Plant Physiol 136: 3649-3659.
- Garg AK, Kim JK, Owens TG, Ranwala AP, Choi YD (2002) Trehalose accumulation in rice plants confers high tolerance levels to different abiotic stresses. Proc Natl Acad Sci USA 99(25): 15898-15903.
- Jang IC, Oh SJ, Seo JS, Choi WB, Song SI, et al. (2003) Expression of a bifunctional fusion of the Escherichia coli genes for trehalose-6- phosphate synthase and trehalose-6-phosphate phosphatase in transgenic rice plants increases trehalose accumulation and abiotic stress tolerance without stunting growth. Plant Physiol 131(2): 516- 524.
- Kondrak M, Marnics F, Antal F, Juhasz Z, Banfalvi Z (2012) Effects of yeast trehalose-6-phosphate synthase 1 on gene expression and carbohydrate contents of potato leaves under drought stress conditions. BMC Plant Biol 12: 74.
- Stiller I, Dulai S, Kondrák M, Tarnai R, Szabó L (2008) Effects of drought on water content and photosynthetic parameters in potato plants expressing the trehalose-6-phosphate synthase gene of Saccharomyces cerevisiae. Planta 227(2): 299-308.
- Wyrzykowska A, Pieczynski M, Szweykowska-Kulinska Z (2016) Construction of artificial miRNAs to prevent drought stress in Solanum tuberosum. Methods Mol Biol 1398: 271-290.
- Kaneda M, Kobayashi K, Nishida K, Katsuta S (1984) Liliosides D and F, two glycerol glucosides from Lilium japonicum. Phytochemistry 23(4): 795-798.
- Bianchi G, Gamba A, Limiroli R, Pozzi N, Elster R (1993) The unusual sugar composition in leaves of the resurrection plant Myrothamnus flabellifolia. Physiol Plant 87: 223-226.
- Sievers N, Muders K, Henneberg M, Klhn S, Effmert M, et al. (2013) Establishing glucosylglycerol synthesis in potato (Solanum tuberosum L. cv. Albatros) by expression of the ggpPS gene from Azotobacter vinelandii. J Plant Sci Mol Bree 2: 1.
- El Banna A, Hajirezaei M, Wissing J, Ali Z, Vaas L (2010) Over expression of PR 10a leads to increased salt and osmotic tolerance in potato cell cultures. J Biotechnol 150(3): 277-287.
- Cho KS, Han EH, Kwak SS, Cho JH, Im JS (2016) Expressing the sweet potato orange gene in transgenic potato improves drought tolerance and marketable tuber production. C R Biol 339(5-6): 207-213.
- Bohnert HJ, Jensen RG (1996) Metabolic engineering for increased salt tolerance. Aust J Plant Physiol 23(5): 661-667.
- Sekmen AH, Turkana I, Takiob S (2007) Differential responses of antioxidative enzymes and lipid peroxidation to salt stress in salttolerant Plantago maritime and salt-sensitive Plantago media. Physiol Plant 131(3): 399-411.
- Martinez CA, Maestri M, Lani EG (1996) In vitro salt tolerance and proline 410 accumulation in Andean potato (Solanum spp.) differing in frost resistance. Plant Sci 116(2): 177-184.
- Ochat SJ, Marconi PL, Radice S, Arnozis PA, Caso OH (1999) In vitro recurrent 418 selection of potato: production and characterization of salt tolerant cell lines and 419 plants. Plant Cell Tiss Organ Cult 55: 1-8.
- Kidokoro S, Watanabe K, Ohori T, Moriwaki T, Maruyama K (2015) Soybean DREB1/CBF-type transcription factors function in heat and drought as well as cold stress-responsive gene expression. Plant J 81(3): 505-518.
- Liu Q, Kasuga M, Sakuma Y, Abe H, Miura S (1998) Two transcription factors, DREB1 and DREB2, with an EREBP/AP2 DNA binding domain separate two cellular signal transduction pathways in droughtand low-temperature-responsive gene expression, respectively, in Arabidopsis. Plant Cell 10(8): 1391-1406.
- Sakuma Y, Liu Q, Dubouzet JG, Abe H, Shinozaki K (2002) DNA-binding specificity of the ERF/AP2 domain of Arabidopsis DREBs, transcription factors involved in dehydration- and cold-inducible gene expression. Biochem Biophys Res Commun 290(3): 998-1009.
- Sakuma Y, Maruyama K, Osakabe Y, Qin F, Seki M (2006) Functional analysis of an Arabidopsis transcription factor, DREB2A, involved in drought-responsive gene expression. Plant Cell 18(5): 1292-1309.
- Gilmour SJ, Zarka DG, Stockinger EJ, Salazar MP, Houghton JM (1998) Low temperature regulation of the Arabidopsis CBF family of AP2 transcriptional activators as an early step in cold induced COR gene expression. Plant J 16(4): 433-442.
- Shinwari ZK, Nakashima K, Miura S, Kasuga M, Seki M (1998) An Arabidopsis gene family encoding DRE/CRT binding proteins involved in low-temperature-responsive gene expression. Biochem Biophys Res Commun 250(1): 161-170.
- Fowler S, Thomashow MF (2002) Arabidopsis transcriptome profiling indicates that multiple regulatory pathways are activated during cold acclimation in addition to the CBF cold response pathway. Plant Cell 14(8): 1675-1690.
- Bouaziz D, Julien P, Charfeddine M, Hammami A, Jbir R (2013) Overexpression of StDREB1 transcription factor increases tolerance to salt in transgenic potato plants. Mol Biotechnol 54(3): 803-817.
- Jia XX, Li YT, Qi EF, Ma S, Hu XY (2016) Overexpression of the Arabidopsis DREB1A gene enhances potato drought-resistance. Russ J Plant Physiol 63(4): 523-531.
- Nuruzzaman M, Sharoni AM, Kikuchi S (2013) Roles of NAC transcription factors in the regulation of biotic and abiotic stress responses in plants. Front Microbiol 4: 248.
- Xu Q, He Q, Li S, Tian Z (2014) Molecular characterization of StNAC2 in potato and its overexpression confers drought and salt tolerance. Acta Physiol Plant 36(7): 1841-1851.
- Upadhyaya CP, Venkatesh J, Gururani MA, Asnin L, Sharma K (2011) Transgenic potato overproducing L-ascorbic acid resisted an increase in methylglyoxal under salinity stress via maintaining higher reduced glutathione level and glyoxalase enzyme activity. Biotechnol Lett 33(1): 2297-2307.
- Bayat F, Shiran B, Belyaev DV, Yur’eva NO, Sobol’kova GI (2010) Potato plants bearing a vacuolar Na+/H+ antiporter HvNHX2 from barley are characterized by improved salt tolerance. Russ J Plant Physiol 57(5): 696-706.
- Patonnier M, Peltier J, Marigo G (1999) Droughtinduced increase in xylem malate and mannitol concentrations and closure of Fraxinus excelsior L stomata J Exp Bot 50(336): 1223-1229.
- Prabhavathi V, Rajam M (2007) Mannitolaccumulating transgenic eggplants exhibit enhanced resistance to fungal wilts. Plant Sci 173(1): 50-54.
- Shen B, Jensen R, Bohner H (1997) Increased resistance to oxidative stress in transgenic plants by targeting mannitol biosynthesis to chloroplasts. Plant Physiol 113(4): 1177-1183.
- Rahnama H, Vakilian H, Fahimi H, Ghareyazie B (2011) Enhanced salt stress tolerance in transgenic potato plants (Solanum tuberosum L.) expressing a bacterial mtlD gene. Acta Physiol Plant 33(4): 1521-1532.
- Chen HH, Li PH (1980) Characteristic of cold acclimation and deacclimation in tuber-bearing Solanum species. Plant Physiol. 65(6): 1146-1148.
- Pino M, Skinner J, Jeknic Z, Hayes PM, Soeldner AH (2008) Ectopic AtCBF1 over-expression enhances freezing tolerance and induces cold acclimation-associated physiological modifications in potato. Plant Cell Environ 31(4): 393-406.
- Craig W, Lenzi P, Scotti N, Palma MD, Saggese P (2008) Transplastomic tobacco plants expressing a fatty acid desaturase gene exhibit altered fatty acid profiles and improved cold tolerance. Transgenic Res 17(5): 769-782.
- Amiri R, Yur’eva R, Shimshilashvili K, Goldenkova-Pavlova I, Pchelkin V (2010) Expression of acyl-lipid 12-desaturase gene in prokaryotic and eukaryotic cells and its effect on cold stress tolerance of potato. J Integr Plant Biol 52(3): 289-297.
- Pino M, Skinner J, Park E, Jeknic Z, Hayes P (2007) Use of a stress inducible promoter to drive ectopic AtCBF expression improves potato freezing tolerance while minimizing negative effects on tuber yield. Plant Biotechol J 5(5): 591- 604.
- Deryabin AN, Trunova TI, Dubinina IM, Burakhanova EA, Sabel’nikova EP (2003) Chilling tolerance of potato plants transformed with a yeastderived invertase gene under the control of the B33 patatin promoter. Russ J Plant Physiol 50(4): 449-454.
- Behnam B, Kikuchi A, CelebiToprak F, Kasuga M, YamaguchiShinozaki K (2007) Arabidopsis rd29A::DREB1A enhances freezing tolerance in transgenic potato. Plant Cell Rep 26(8): 1275-1282.
- Lee HE, Shin D, Park SR, Han SE, Jeong MJ (2007) Ethylene responsive element binding protein 1 (StEREBP1) from Solanum tuberosum increases tolerance to abiotic stress in transgenic potato plants. Biochem Biophys Res Commun 353(4): 863-868.
- Kim YH, Kim MD, Park SC, Jeong JC, Kwak SS, et al. (2016) Transgenic potato plants expressing the cold-inducible transcription factor SCOF 1 display enhanced tolerance to freezing stress. Plant Breeding 135(4): 513-518
- Basu PS, Minhas JS (1991) Heat tolerance and assimilate transport in different potato genotypes. J Exp Bot 42(7): 861-866.
- Ewing EE (1981) Heat stress and the tuberization stimulus. Am J Potato Res 58: 31-49.
- Coria NA, Sarquis JI, Penalosa I, Urzua M (1998) Heat-induced damage in potato (Solanum tuberosum) tubers: membrane stability, tissue viability, and accumulation of glycoalkaloids. J Agric Food Chem 46(11): 4524-4528.
- Ahn Y, Zimmerman JL (2006) Introduction of the carrot HSP17.7 into potato (Solanum tuberosum L.) enhances cellular membrane stability and tuberization in vitro. Plant Cell Environ 29(1): 95-104.
- Sun W, Montagu MV, Verbruggen N, Moleculaire V (2002) Small heat shock proteins and stress tolerance in plants. Biochim Biophys Acta 1577(1): 1-9.
- Kim JY, Kwak KJ, Jung HJ, Lee HJ, Kang H (2010) MicroRNA402 affects seed germination of Arabidopsis thaliana under stress conditions via targeting DEMETER-LIKE Protein3 mRNA. Plant Cell Physiol 51(6): 1079-1083.
- Dou H, Xv K, Meng Q, Li G, Yang X (2015) Potato plants ectopically expressing Arabidopsis thaliana CBF3 exhibit enhanced tolerance to high-temperature stress. Plant Cell Environ 38(1): 61-72.
- Kim MD, Kim YH, Kwon SY, Jang BY, Lee SY (2011) Overexpression of 2-cysteine peroxiredoxin enhances tolerance to methyl viologenmediated oxidative stress and high temperature in potato plants. Plant Physiol Biochem 49(8): 891-897.
- Youm J, Jeon J, Choi D, Yi S, Joung H (2008) Ectopic expression of pepper CaPF1 in potato enhances multiple stresses tolerance and delays initiation of in vitro tuberization. Planta 228(4): 701-708.
- Lee,H, Xiong L, Ishitani M, Stevenson B, Zhu J (1999) Cold regulated gene expression and freezing tolerance in an Arabidopsis thaliana mutant. Plant J 17(3): 301-308.
- Ramanjulu S, Bartels D (2002) Droughtand desiccation induced modulation of gene expression in plants. Plant Cell Environ 25: 141- 151.
- Behnam B, Kikuchi A, CelebiToprak F, Yamanaka S, Kasuga M (2006) The Arabidopsis DREB1A gene driven by the stress inducible rd29A promoter increases salt stress tolerance in tetrasomic tetraploid potato (Solanum tuberosum) in proportion to its copy number. Plant Biotechnol 23: 169-177.
- Celebi Toprak F, Behnam B, Serrano G, Kasuga M, YamaguchiShinozaki K (2005) Tolerance to salt stress of the transgenic tetrasomic tetraploid potato, Solanum tuberosum cv. Desiree appears to be induced by the DREB1A gene and rd29A promoter of Arabidopsis thaliana. Breed Sci 55: 311-319.
- Movahedi S, Tabatabaei B, Alizade H, Ghobadi C, Yamchi A, et al. (2012) Constitutive expression of Arabidopsis DREB1B in transgenic potato enhances drought and freezing tolerance. Biol Plant 56(1): 37-42.
- Huynh D, Shimazaki T, Kasuga M, YamaguchiShinozaki K, Kikuchi A, et al. (2014) In vitro evaluation of dehydration tolerance in AtDREB1A transgenic potatoes. Plant Biotechnol 31(1): 77-82.
- Dubos C, Stracke R, Grotewold E, Weisshaar B, Martin C, et al. (2010) MYB transcription factors in Arabidopsis. Trends Plant Sci 15(10): 573- 581.
- Stracke R, Ishihara H, Huep G, Barsch A, Mehrtens F (2007) Differential regulation of closely related R2R3MYB transcription factors controls flavonol accumulation in different parts of the Arabidopsis thaliana seedling. Plant J 50(4): 660-667.
- Cheng YJ, Kim MD, Deng XP, Kwak SS, Chen W (2013) Enhanced salt stress tolerance in transgenic potato plants expressing IbMYB1, a sweet potato transcription factor. J Microbiol Biotechnol 23(12): 1737- 1746.
- Shin D, Moon S, Han S, Kim B, Park K, et al. (2011) Expression of StMYB1R-1, a novel potato single MYBlike domain transcription factor, increases drought tolerance. Plant Physiol 155(1): 421-432.
- Waterer D, Benning N, Wu G, Luo X, Liu X (2010) Evaluation of abiotic stress tolerance of genetically modified potatoes (Solanum tuberosum cv. Desiree). Mol Breed 25(3): 527-540.
- Gangadhar BH, Sajeesh K, Venkatesh J, Baskar V, Abhinandan K (2016) Enhanced tolerance of transgenic potato plants over-Expressing nonspecific lipid transfer protein-1 (StnsLTP1) against multiple abiotic stresses. Front Plant Sci 7: 1228.
- Stief A, Altmann S, Hoffmann K, Pant BD, Scheible WR (2014) Arabidopsis miR156 regulates tolerance to recurring environmental stress through SPL transcription factors. Plant Cell 26(4): 1792-1807.
- Zhang X, Zou Z, Gong P, Zhang J, Ziaf K, et al. (2011) Over-expression of microRNA169 confers enhanced drought tolerance to tomato. Biotechnol Lett 33(2): 403-409.
- Yang C, Li D, Mao D, Liu XUE, Ji C (2013) Overexpression of microRNA319 impacts leaf morphogenesis and leads to enhanced cold tolerance in rice (Oryza sativa L). Plant Cell Environ 36: 2207- 2218.
- Li JF, Aach J, Norville JE, McCormack M, Zhang D (2013) Multiplex and homologous recombination–mediated genome editing in Arabidopsis and Nicotiana benthamiana using guide RNA and Cas9. Nat Biotechnol 31(8): 688-691.
- Jiang W, Zhou H, Bi H, Fromm M, Yang B (2013) Demonstration of CRISPR/Cas9/sgRNA-mediated targeted gene modification in Arabidopsis, tobacco, sorghum and rice. Nucleic Acids Res 41(20): e188.
- Shan Q, Wang Y, Li J, Zhang Y, Chen K (2013) Targeted genome modification of crop plants using a CRISPR-Cas system. Nat Biotechnol 31(8): 686-688.
- Upadhyay SK, Kumar J, Alok A, Tuli R (2013) RNA-guided genome editing for target gene mutations in wheat. G3 (Bethesda) 3(12): 2233-2238.
- Liang Z, Zhang K, Chen K, Gao C (2014) Targeted mutagenesis in Zea mays using TALENs and the CRISPR/Cas9 system. J Genet Genomics 41(2): 63-68.
- Jia H, Wang N (2014) Targeted genome editing of sweet orange using Cas9/sgRNA. Plos ONE 9(4): e93806.
- Cai Y, Chen L, Liu X, Sun S, Wu C (2015) CRISPR/Cas9-mediated genome editing in soybean hairy roots. Plos ONE 10(8): e0136064.
- Jacobs TB, LaFayette PR, Schmitz RJ, Parrott WA (2015) Targeted genome modifications in soybean with CRISPR/Cas9. BMC Biotechnol 15: 16.
- Osakabe Y, Watanabe T, Sugano SS, Ueta R, Ishihata R, et al. (2016) Optimization of CRISPR/Cas9 genome editing to modify abiotic stress responses in plants. Sci Rep 6: 26685.
- Li P, Li YJ, Zhang FJ, Zhang GZ, Jiang XY (2017) The Arabidopsis UDP-glycosyltransferases UGT79B2 and UGT79B3, contribute to cold, salt and drought stress tolerance via modulating anthocyanin accumulation. Plant J 89(1): 85-103.
- Shi J, Gao H, Wang H, Lafitte HR, Archibald RL (2017) ARGOS8 variants generated by CRISPR-Cas9 improve maize grain yield under field drought stress conditions. Plant Biotechnol J 15(2): 207-216.






























