Enhanced Antiangiogenic Activity of Silver Nano-Particles Grafted on Graphene Oxide
Hafiz Amir Nadeem1, Muhammad Imran1*, Kashif Mehmood2, Ammarah Batool1 and Zahra Rafiq1
1Department of Chemistry, Government College University, Lahore, Pakistan
2School of Chemical and Materials Engineering (SCME), National University of Science and Technology, Islamabad, Pakistan
Submitted: March 25, 2022; Published: April 18, 2022
*Corresponding author: Muhammad Imran, Department of Chemistry, Government College University, Lahore, Pakistan
How to cite this article: Hafiz Amir N, Muhammad I, Kashif M, Ammarah B, Zahra R. Enhanced Antiangiogenic Activity of Silver Nano-Particles Grafted on Graphene Oxide. JOJ Material Sci. 2022; 7(1): 555703. DOI:10.19080/JOJMS.2022.07.555703
Abstract
In the study, the silver nanoparticle were grafted on graphene oxide with and without use of Allium sativum as stabilizing or reducing agent and ultrasonic assistant have been successfully prepared and then their comparative anti-angiogenesis and hemolytic studies have been carried out. The synthesized particles have been characterized by using modern techniques including P-XRD, AT-FTIR, UV-Visible spectroscopy, SEM analysis, and BT-90 Nano size analyzer (BT-90). The results revealed uniform distribution of Ag on the GO sheet in both cases with mean particle size of 32.96 nm with d-spacing 2.3634 nm and 11.65 nm with d-spacing 2.3594 nm for GO-Ag-G NPs and GO-Ag NPs, respectively. The maximum absorbance was obtained at 448.08 nm of GO-Ag-G NPs and 443.29 of GO-Ag NPs. Both types of NPs showed dose-dependent anti-angiogenesis activity analyzed via CAM assay and showed % hemolysis of range (0.0971-.1246) for GO-Ag-G NPs and (0.14-0.5797) for GO-Ag NPs.
Keywords: Graphene oxide; Graphene oxide silver nanoparticles; Anti-angiogenesis; Hemolytic; Allium sativum
Abbreviations: SERS: Surface-Enhanced Raman Scattering; CAM: Chick Chorioallantoic Membrane; ETCs: Endothelial Cells; AAG: Anti-Angiogenesis; ROS: Reactive Oxygen Species; GO: Graphene oxide
Introduction
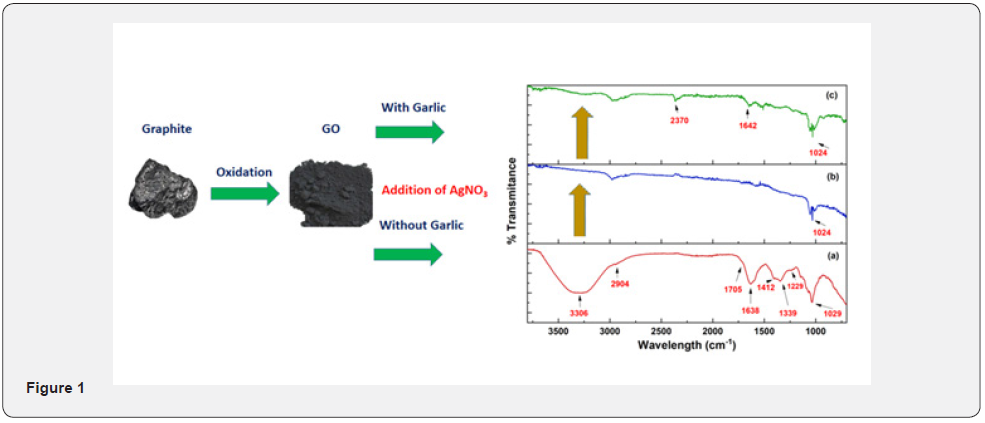
A huge effort has been made in search of new materials possessing biological applications in the field of medicine [1]. Nanomaterials usage as a replacement for antibiotics and medicine is becoming popular [2]. AgNPs out of all transition elements are of great interest owing to their biocompatibility and bactericidal ability [3]. They found use in every field including optics, catalysis, biosensors, electronics, biomedicine, and medication [4,5]. AgNPs and their hybrid are proved to be reliable antimicrobial agents as many research articles mentioned them as a disinfectant, antiseptics, and pharmaceutical applications [6-8]. These exhibit activity against several bacterial species, the size of AgNPs is a basic factor in its activity, more the surface area, easier will be the cell penetration, and chances of its contact with bacteria [3,9,10] (Figure 1).
Recent studies on nanomaterials suggest graphene and its derived compounds as a unique biocompatible entity due to its structure as well as functionalities [11,12]. Graphene oxide (GO) is a highly oxidized graphite sheet that consists of one layer, contain carbons arranged in hexagonal structure possess functional group alkoxy (C-O-C), hydroxyl (-OH), carbonyl (C=O), carboxylic acid (-COOH), carbonyl group (C-O) along with other oxygen-carrying groups. These functional units give graphene oxide hydrophilic nature making it a suitable water-soluble nano substance [13-15]. Graphene has exhibited numerous applications in diverse fields including sensors [16], electronics, photocatalytic activity [17], biosensors, anticancer [18], biomedicine coatings, water decontamination [19], antibacterial activity [20] and drug delivery [21]. The hybrids of graphene with metal-based nanoparticles including Au, Pt, and Ag have revealed exceptional biological ability. The large surface area containing oxygen base functional units provides proper anchoring sites for the attachment of metals and helps in its dispersion. The sheet of graphene oxide provides support in terms of the growth and stabilization of metal nanoparticles [22]. Various research articles supported Ag coupling with graphene oxide as an appropriate material with potential biocompatibility and being used as microbicides, catalysis, cancer nano therapy, supercapacitors, and surface-enhanced Raman scattering (SERS) [23,24]. A report by Shen et al. [25] reported Ag nanoparticles combined with graphene sheets showed antibacterial activity [25]. The combined antimicrobial action results from the trapping of bacteria by graphene oxide while Ag plays the role of killing bacteria [26]. Another report by Das et al. [27] discusses the effect of size and shape factors of Ag nanoparticles combined with graphene oxide on the antiseptic activity [27]. The collaborative upshot of GO/Ag nanoparticles for the in situ preparation of GO/Ag NPs discussed in detail in an article by Ma et al. [28].
Several synthetic methods for GO-Ag composites are available in the literature, for instance, a single step synthesis process that involves a solution phase of GO to reduce silver ions on graphene oxide sheets [29]. Though this method is bound with some drawbacks as morphology and size of Ag could not be controlled as Ag nanoparticles developed directly on GO sheets through silver ion reduction. One of the reports adopted dry decoration of Ag nanoparticles on GO surface by an arc plasma source utilizing electrostatic force [30]. An eco-friendly approach was implemented by Dinh et al. [31] to prepare GO-Ag composites through in situ ultrasonication using citric acid and reported its enhanced conductivity by 40 times in comparison to reduced graphene oxide [31].
Presently, the liquid-phase reduction method is made of use to place Ag nanoparticles on the surface of graphene, but it also led to the generation of biotoxic and hazardous agents [32]. The sonochemical method combined with shock freezing was also reported in preparing graphene oxide-Ag nanocomposite [33]. The preparation of graphene oxide-AgNP has been done utilizing different nature of reducing agents and stabilizing agents to avoid agglomeration and regulate structure. Hydroquinone is used as a reductant whereas citrate is used as a stabilizer by Bao et al. [34]. Das et al made use of NaBH4 for reduction along with trisodium citrate as a stabilizer [27,35]. NaBH4 coupled with ethylene glycol was used as a reductant by Shen et al. [25]. In another report, Shen et al prepared graphene oxide-Ag composites using ascorbic acid as a reductant at 160°C while ionic liquid acts as a dispersing agent. These synthetic methods also hold the disadvantage of toxic reducing agents [36].
Allium sativum (Garlic) is eminent specie of onion genius allium and known for its potential biological activities including anti-mutagenic, anti-carcinogenic, free-radical scavenging, and as detoxifying agent [37]. In the current work, eco-friendly reducing agent (Allium sativum) was utilized to incorporate Ag NPs onto Graphene oxide. The Allium sativum would both act as stabilizing as well as reducing agent to minimize the toxicity using ultrasonication. The resultant particles were characterized via modern techniques including P-XRD, AT-FT-IR, UV-Visible spectroscopy, SEM, and BT- 90 nano-size analyzer. The antiangiogenesis activity using CAM assay and % hemolytic activity using human erythrocytes was investigated for GO-Ag NPs obtained via both methods.
Experimental
Materials
Natural graphite powder (99%, 325 mesh) was sourced through Alfa Aesar (China). Sodium nitrate [(NaNO3) 99%], potassium permanganate [(KMnO4) 99.9%], and silver nitrate [(AgNO3) 99.7%] were brought from the Sigma-Aldrich (St. Louis, USA)), Sulfuric acid [(H2SO4) 98%], hydrogen peroxide [(H2O2) 30% W/V], hydrochloric acid [(HCl) 37%] were sourced through Merck KGaA (Darmstadt, Germany). Deionized water (Millipore) was used for the preparation of all aqueous solutions. All the reagents used in the synthesis were of analytical grade and they were used as received without any further purification.
Preparation of Graphene oxide (GO)
The Hummer’s method with few modifications was utilized to prepare GO [38]. Firstly, 1g of graphite powder and 1g of sodium nitrate were poured to 80 mL of H2SO4 (conc.) along with continuous stirring at 0°C. After 3 hours of a continuous stir at 600 rpm, 9g of KMnO4 was put in the mixture. Kept the temperature < 00 C and pour dropwise H2O (150mL), the fumes continuously evolved during this, followed by vigorously agitation of mixture for 2 hours at room temperature. Now, mixture was refluxed in two sets; in first set, refluxed for 20 min at 90oC followed by for 10 min at 25oC in second set until the mixture turned dark brown. Oxidation was completed by the addition of 30mL of H2O2 and colour turned to light brown. The contents were centrifuged with 10% HCl and deionized water. Graphene oxide gel was later, vacuumed at 50oC to get GO powder.
Preparation of Allium sativum (Reducing Agent)
Garlic extract was prepared from fresh cloves of Allium sativum. The garlic bulb was peeled and separated cloves were cleaned with deionized water. Weighed 5g of garlic cloves and were mechanically crushed in porcelain pestle and mortar using 50mL deionized water. The homogenate was left overnight and later sieved over a 0.2 μm filter. The concentration of freshly prepared pale-yellow garlic extract was determined by evaporation of 2mL aqueous extract and subsequently weighing the residual left behind. The concentration of prepared garlic extract was found to be 20 mg/mL. Garlic extract was stored at 20oC for experimental work.
Grafting of Ag-NPs on GO
For the production of GO-Ag nanoparticles, two preparatory methods were employed for comparison. Firstly, the silver nitrate solution was prepared by adding 42.4 mg of AgNO3 in 50mL of deionized water to prepare a 5 mM solution. Three colloidal suspensions of graphene oxide having different concentrations were made by dispersing 25mg, 50mg, and 75mg of GO in 50mL of deionized water. One batch was prepared by using garlic extract as a reducing agent while the other was synthesized in the absence of a reducing agent. GO suspension was subjected to sonication for 15 minutes followed by the addition of 50 mL AgNO3 (5 mM) in each suspension. The reaction mixture was further sonicated for 30 minutes to ensure sufficient contact between silver ions and functional groups of GO. The same process was repeated for the other batch and garlic extract was also added as a reducing agent after the addition of AgNO3. Finally, the reaction contents were centrifuged at 4000 rpm and the precipitates were collected, dried, and stored for further analysis.
Characterizing techniques of GO-Ag NPs
UV-Vis spectroscopy was used for the confirmation of Ag nanoparticles by detecting plasmon absorption bands over the range between 200–800 nm. FT-IR spectra were obtained using Cary-630 FTIR spectrophotometer over a frequency range of 4000 – 400 cm-1 to analyze the structure of nanoparticles. X-ray diffraction pattern of the synthesized nanoparticles was determined using a 6100 X-ray diffractometer (Shimadzu, Japan) at 30 mA current and 40 kV voltage. SEM scan was performed on a scanning electron microscope (JSM-6490 LV, USA) at 5 kV voltage and lower vacuum. The samples were placed on carbon tape joined with the sample tab and mounted for the scan. The images were recorded, and magnified images were analyzed to examine the cross-section of nanoparticles. BT-90 nano-size analyzer was used to record the particle size of prepared GO-Ag nanoparticles. Samples were diluted enough to get a 20-40 K photon number.
Hemolytic activity
The EDTA was first diluted with saline (0.9% NaCl), and then it was centrifuged at 1000 rpm for 10 minutes, after it the samples of blood were collected in EDTA. The separation of erythrocytes was done and was diluted in phosphate buffer saline of pH 7.4 to make its suspension. The plant extract was added and incubated for 5 min at normal room temperature. It was induced with an oxidative breakdown of membrane lipids. Quercetin was engaged in the form of a reference compound. The absorbance was noticed at 540 nm by spectrophotometer [36].
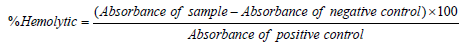
Anti-angiogenesis activity of nanoparticles
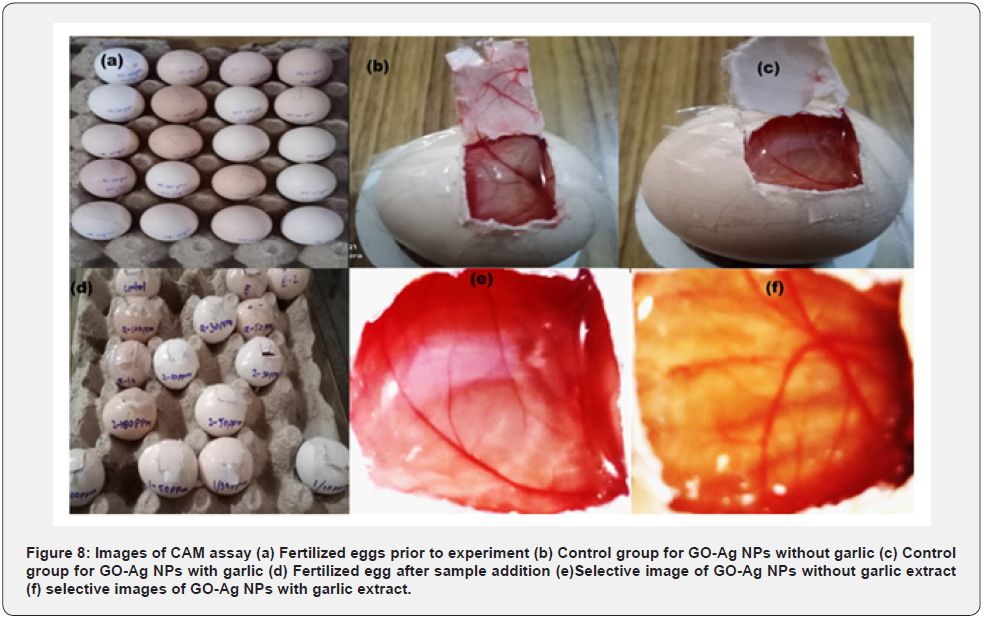
Chick chorioallantoic membrane (CAM) assay was done to examine the angiogenesis inhibitory potential of the synthesized nanoparticles. Twelve fertile chicken eggs were taken after 5 to 6 days of incubation from an incubation center and divided into two groups where each group contained six eggs. The eggs were thoroughly cleaned with ethanol and incubated at 37oC and 70 – 80 % humidity for the next 24 hours [39]. One group was taken as control while the other group was treated with doses of synthesized GO-Ag nanoparticles. A window was cut on the wide side of the incubated eggs on the 6th day and sterile filter paper loaded with 20 μg/mL ethanolic dose of GO-Ag nanoparticles was placed and the window was sealed using parafilm. The window was de-sealed after the next 48 hours of incubation to examine the effect of nanoparticles on angiogenesis [40]. (which composite is used reduced with Allium sativum or simple Ag-GO composite???).
Result and Discussion
Characterization with UV-Vis spectroscopy
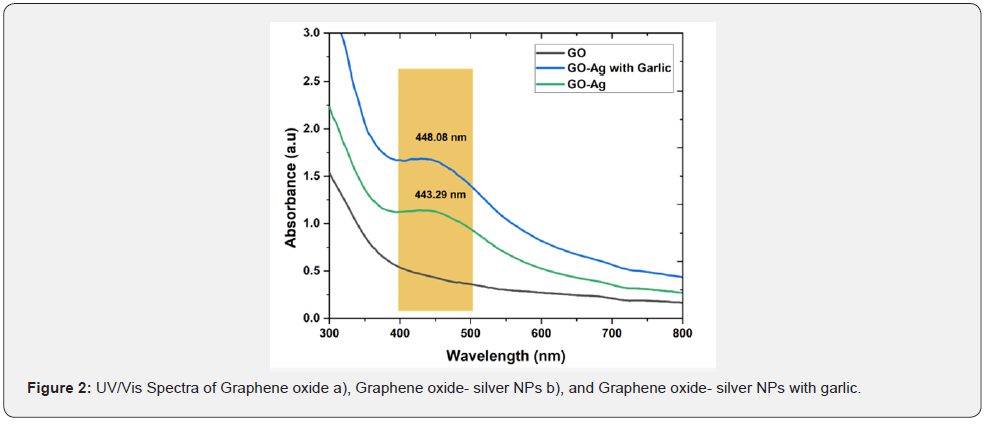
The UV/Vis absorbance spectroscopy helps to confirm the formation of GO-Ag NPs with or without reducing agents as shown in Figure 2. The typical characteristics peaks of GO were shown λmax at 230nm and ~300nm. These peaks are accredited to π to π* transition of the Carbon-carbon bonds and n to π* transitions of Carbon-oxygen (C-O) groups respectively [39]. In the case of GO-Ag NPs without reducing agent, the absorbance spectrum has shown λmax at 443.29 nm that depicted typical surface plasmon resonance (SPR) absorption of Ag NPs [41]. In the case of GO-Ag NPs with garlic extract, the characteristic absorbance spectrum of AgNPs has shown λmax at 448.08 nm [40]. The peak of UV-Vis spectra was symmetrical that reflects the uniform and small size of the impregnated Ag-NPs on the surface of GO. However, during the formation of AgNPs, the GO did not experience chemical change as a band at 230 nm appear in all the cases. The garlic extract act as stabilizing as well as reducing agent during the reaction. The absorption peak at 300 nm disappeared in both cases showed the completion of the reaction [41].
ATR-FT-IR analysis
ATR-FTIR spectrum is used to investigate the different functional groups the appeared or disappeared before and after the formation of GO-Ag NPs with or without reducing agent and shown in Figure 3. In the case of GO, a peak appeared at 3306 cm-1 the hydroxyl group of water molecules which adsorbed on GO are responsible for this broad peak. The absorption peaks for symmetric and anti-symmetric stretching vibrations of the C-H bond appeared at 2886 and 2904 cm-1. The peak appeared at 1638 cm-1 which is attributed to C=C and 1704 cm-1 that is appeared owing to the carboxylic group [41]. The three absorption peaks that appeared between 1412 cm-1, 1039 cm-1, and 1229 are attributed to stretching vibrations of C=C of aromatic groups and C-O group of alcohol and carboxylic acid respectively. The formation of oxygen-containing functional groups confirmed the formation of GO from oxidation of graphite [42].
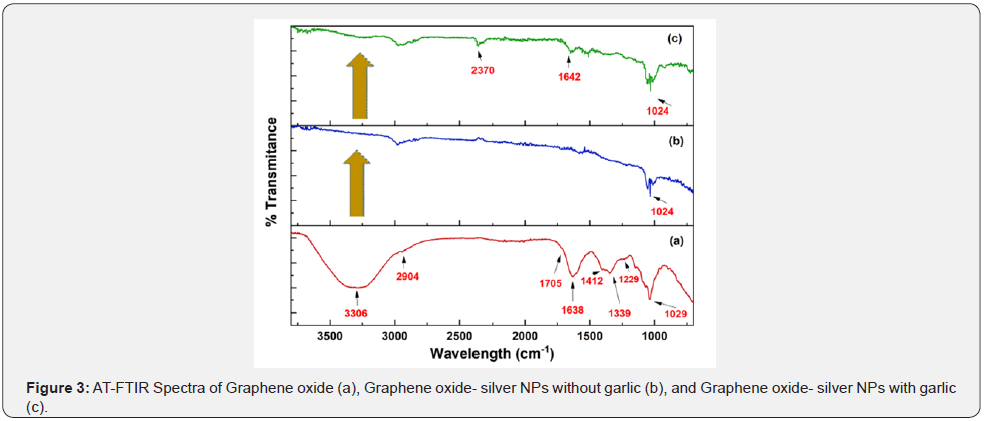
In the case of GO-Ag NPs with and without reducing agent, the major difference appeared in GO-Ag NPs with garlic peaks at 1618 cm-1 that is attributed to peptide linkage of the carboxylic group and C-O stretching vibration of in carbonyl. The deformation of bands that appeared at various places like 910 cm-1 and 1015 cm-1 can be attributed to the deformation of C-H and C-N (stretching vibrations). The weak band was observed at 3342 cm-1 and 1914 cm-1 reflected the asymmetrical stretching of O-H and C-H [43]. It has been observed that the intensity of OH in the case of GOAg NPs with garlic is more than without, it can be owing to garlic extract components.
PXRD analysis of GO, GO-NPs with and without garlic
The powder X-rays diffraction analysis of GO, GO-Ag NPs with and without garlic are present in Figure 4. In the case of GO, the diffraction peak appeared at 10.780 with the corresponding plane (001). While the diffraction peaks in the case of GO-Ag NPs without garlic extract appeared at 38.090 and 44.430 which can be attributed to the planes (111) and (200) respectively of silver with face-centered cubic structure (JCPDS card, No. 04-0783). The peaks of NPs are narrow which is attributed to their large size. The diffraction peaks of GO disappeared, the possible reasons might be the effect of the ratio of GO and silver on XRD pattern or exfoliation of Go via sonication evolve for the synthesis of GO-Ag NPs. Two diffraction peaks of Ag appeared means preferred orientation growth of Ag NPs. In the case of GO-Ag NPs with garlic extract as stabilizing agent four different peaks appeared at 38.140, 44.400, 64.340, and 77.210 which can be attributed to (111), (200), (220), and (311) planes of metallic Ag respectively according to JCPDS No.89-3722 [41].
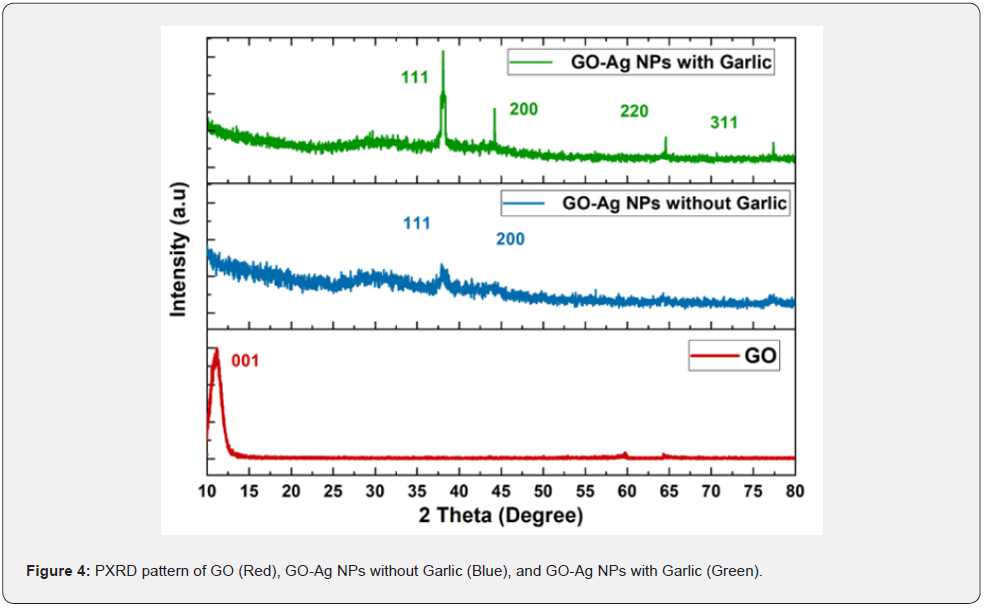
The Bragg equation is used to calculate the interlayer distance of GO, GO-Ag NPs with and without garlic extract.
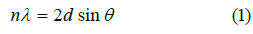
In equation (1), λ represents X-ray wavelength its value is 1.54178 A0, Ɵ is the angle between the incident and reflected rays. In the case of GO, the d-spacing is calculated from the major peak (Table 1) is 0.82nm which is higher than the graphite reported value of d-spacing (0.34nm). This might be owing to exfoliation, the addition of oxygen-containing functional groups as a result of chemical oxidation of graphite, hence completion of the reaction. In the case of GO-Ag NPs without garlic and with garlic extract the calculated d-spacing from major peaks is 2.3634 nm and 2.3594 nm respectively, which is approximately the same, which means NPs prepared via both methods equally increase the interlayer distance [42,44].
To calculate the average layers of GO and average particle size of NPs the Debye-Scherrer’s equation (2) was used
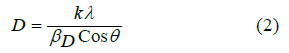
Where, K is the shape factor its value is 0.94, lambda (λ) is the wavelength of X-ray source (0.154178 nm), βD is FWHM in radian, D is crystalline size, and Ɵ is peak position. The average size of GO was 10.62nm. In the case of GO, the average layers of GO per nm obtained by dividing average size with d-spacing were ~ 13. The average sizes obtained for GO-NPs with and without garlic extract were 32.96 nm and 11.65 nm respectively.
SEM analysis
The SEM images at different resolutions and magnification of GO, GO-Ag NPs without garlic and with garlic are shown in Figure 5. The layered structure with wrinkled edges, surfaces, and folding appeared in all the cases and with a clear decoration of GO with Ag NPs in the case of GO-Ag NPs.

BT-90 nanoparticle size analysis
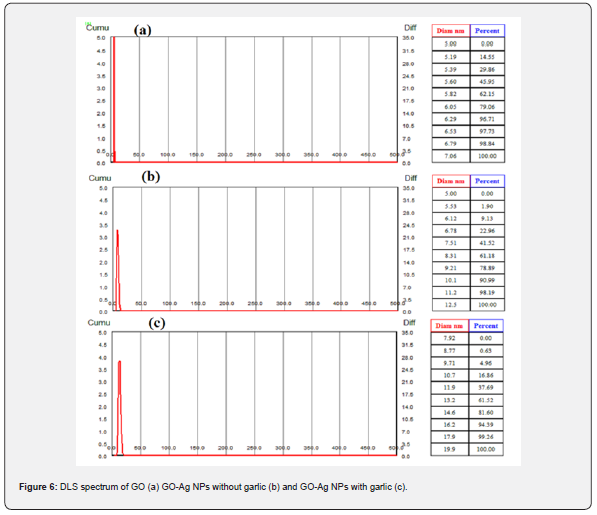
The average size and hydrodynamic diameter of the GO-Ag NPs prepared with and without Allium sativum were determined via a BT-90 nanoparticle size analyzer. The scattering angle was adjusted 900 at room temperature, with laser wavelength 635nm, and analysis mode was selected 2.0 CONTIN. The Brownian motion caused, the scattering of the light i.e. Dynamic Light Scattering Principle [43]. The Stokes-Einstein equation was used to determine the hydrodynamic diameter of GO, GO-Ag NPs [44]. For every analysis, freshly prepared solutions were utilized (dissolve 1mg of each NPs in 5mL deionized water subsequently sonication to get homogenized mixture). In the case of GO, the D50, D (4,3), and D (2,3) were 5.65, 5.59, and 5.57 nm respectively with polydispersity index (PDI) value 0.011 which reflected the homogenous distribution of GO sheets. In the case of GO-Ag NPs without garlic extract the values D50 (7.82 nm), D(4,3) (7.25 nm), and D(2,3) (7.04 nm) having PDI value 0.085 while for GO-Ag NPs with garlic extract 12.5 nm, 11.8 nm and 11.5 values obtained for D50, D(4,3), and D(2,3) respectively with PDI value 0.068. All the results are complimentary to PXRD and SEM [45] (Figure 6).
Hemolytic studies results
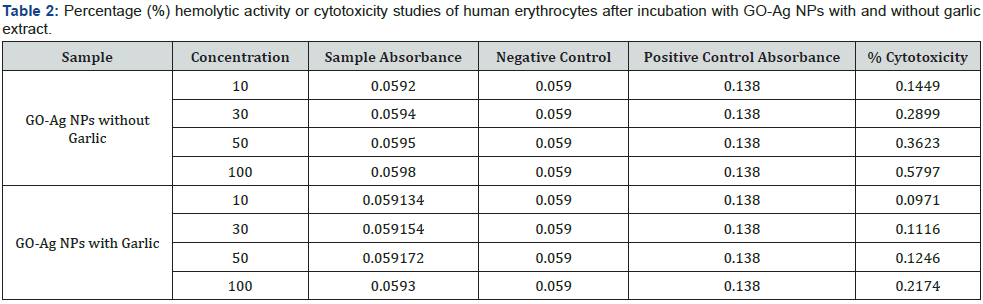
The hemolytic activity of GO and GO-Ag NPs with and without garlic extract was studied, and results are given in (Table 2) In hemolysis erythrocytes (red blood cells) destroy with the release of hemoglobin to blood plasma from red blood cells. One of the principal causes of hemolysis is antibiotics especially peptide antibiotics, which damage the lipid layer as a result its structural integrity is lost. Therefore, while developing a novel antibiotic it is unavoidable to check its hemolytic activity simultaneously to lessen the side effects. All three types of samples investigated showed low hemolytic activity over the entire range of concentrations [46].
Anti-angiogenesis activity of GO-Ag NPs with and without Allium sativum
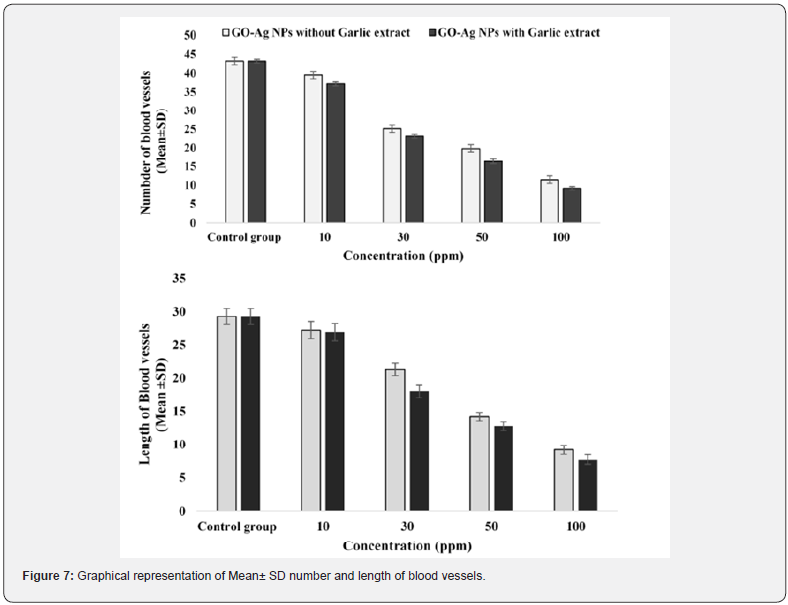
A physiological process, Angiogenesis means the development of new blood vessels from existing vasculature [47]. The process of angiogenesis involves proliferation, stimulation, migration of endothelial cells (ETCs), and formation of the capillary tube [48]. A body maintains the balance of angiogenesis via pro- and anti-angiogenesis signals [47] to avoid several disorders that result in an imbalance of angiogenesis including distortion, ischemic, immunity problems, and infections [49]. The process of suppressing angiogenesis with the help of drugs is termed anti-angiogenesis (AAG) [50] and employed as a potential cancer treatment strategy [51]. In vivo model, CAM assay has been employed to investigate the pro/anti-angiogenesis potential of NPs [52] The magnified images of CAM membranes were examined, and average branch points were calculated. The results showed that nanoparticles inhibit angiogenesis by depriving the embryo cells of oxygen and nutrients. All the control eggs had developed a well-arborized active vascular system and all the vessels were clear and filled with blood. While in the test group, the blood vessels were very thin and had a lesser extent of vascularization. The mean of the number (43.21 ± 0.45) and length (29.28 ± 1.2) of the control group. GO-Ag NPs with and without Allium sativum showed a significant decrease in number and length as compared to control group in dose dependent manner. In case of GO-Ag NPs without Allium sativum, the mean of number and length at a concentration 10 ppm: (39.53 ± 0.49), (29.28 ± 0.67); 30 ppm: (25.12 ± 1.2), (21.29± 0.89); 50 ppm: (19.87± 1.1), (14.21 ± 2.3; 100 ppm: (11.42 ± 0.76), (9.19± 2.3) showed a significant decrease (p < 0.05). Similarly, in case of GO-Ag NPs with Allium sativum, the mean number and length at concentration 10 ppm: (37.23 ± 0.33), (26.89 ± 0.11); 30 ppm: (23.10 ± 1.9), (17.99 ± 0.45); 50 ppm: (16.32 ± 2.1), (12.81 ± 0.42); 100 ppm: (9.16 ±1.09), (7.69 ± 2.1) showed a significant decrease (p < 0.005). The GO-Ag NPs with Allium sativum showed a better result as compared to without Allium sativum.
The anti-angiogenesis activity of silver nanoparticles is most probably due to the blockage of VEFG (vascular endothelial growth factor) mediated signal pathway and thus inhibiting the formation of blood vessels. The inhibitory activity of GO-Ag nanoparticles is attributed to the interaction of Ag nanoparticles with enzymes that contain thiol substituents, thioredoxin peroxide, and super dioxide dismutase which reduce the oxidative stress of Reactive Oxygen Species (ROS) produced within mitochondrial metabolism [53]. ROS accumulation causes deterioration of mitochondria to liberate cytochrome C which eventually results in cell death. It is observed that this activity is more prominent in the case of GO-Ag NPs prepared using garlic extract [54] (Figures 7 & 8).
Conclusion
GO-Ag NPs were successfully prepared using ultrasonic assistance and with/without freshly prepared garlic extract. The resultant particles were characterized using modern techniques including AT-FT-IR, P-XRD, UV-visible spectroscopy, BT-90 nanosize analyzer, and SEM. In the case of GO-Ag NPs without garlic extract the maximum absorption was obtained at 443.29nm, the average size of particles was 11.65nm with 2.3594 nm d-spacing which is comparatively larger than for GO (0.82nm). In the case of GO-Ag-G NPs with garlic extract, the maximum absorbance was obtained at 448.08 nm, the average size of particles was 32.96 nm with d-spacing 2.3634. The % hemolytic activity revealed that both show less toxicity i.e. of range (0.0971-0.1246) for GO-Ag-G NPs and (0.14-0.5797) for GO-Ag NPs. The CAM assay was used to investigate the anti-angiogenesis activity, the results revealed that GO-Ag-G NPs showed better performance than GO-Ag NPs. Both showed dose-dependent activity, low concentration of both shows lessen the destruction of vascular organization and inhibit blood vessel formation.
References
- Marin S, Vlasceanu GM, Tiplea RE, Bucur IR, Lemnaru M, et al. (2015) Applications and toxicity of silver nanoparticles: a recent review. Curr Top Med Chem 15(16): 1596-1604.
- Huh AJ, Kwon YJ (2011) “Nanoantibiotics”: a new paradigm for treating infectious diseases using nanomaterials in the antibiotics resistant era. J Control Release 156(2): 128-145.
- Marambio JC, Hoek EMJ (2010) A review of the antibacterial effects of silver nanomaterials and potential implications for human health and the environment. J Nanopart Res 12(5): 1531-1551.
- Li J, Wang M, Liu G, Zhang L, He Y, et al. (2018) Enhanced iodide removal from water by nano-silver modified anion exchanger. Ind Eng Chem Res 57(51): 17401-17408.
- Khatami M, Alijani HQ, Sharifi I (2018) Biosynthesis of bimetallic and core-shell nanoparticles: their biomedical applications-a review. IET Nanobiotechnol 12(7): 879-887.
- Oyanedel C, Smith JA (2008) Sustainable colloidal-silver-impregnated ceramic filter for point-of-use water treatment. Environ Sci Technol 42(3): 927-933.
- Pal S, Yoon EJ, Tak YK, Choi EC, Song JM (2009) Synthesis of highly antibacterial nanocrystalline trivalent silver polydiguanide. J Am Chem Soc 131(44): 16147-16155.
- Nasrollahzadeh M, Atarod M, Jaleh B, Gandomirouzbahani M (2016) In situ green synthesis of Ag nanoparticles on graphene oxide/TiO2 nanocomposite and their catalytic activity for the reduction of 4-nitrophenol, congo red and methylene blue. Ceram. Int 42(7): 8587-8596.
- Liu YK, Lee MT (2014) Laser direct synthesis and patterning of silver nano/microstructures on a polymer substrate. ACS Appl Mater Interfaces 6(16): 14576-14582.
- Raza MA, Kanwal Z, Rauf A, Sabri AN, Riaz S, et al. (2016) Size-and shape-dependent antibacterial studies of silver nanoparticles synthesized by wet chemical routes. Nanomater 6(4): 74.
- Minitha CR, Rajendrakumar RT (2013) Synthesis and characterization of reduced graphene oxide. Adv Mat Res. Trans Tech Publ 678: 56-60.
- Jagiełło J, Chlanda A, Baran M, Gwiazda M, Lipińska L (2020) Synthesis and Characterization of Graphene Oxide and Reduced Graphene Oxide Composites with Inorganic Nanoparticles for Biomedical Applications. Nanomater 10(9): 1846.
- Smith AT, LaChance AN, Zeng S, Liu B, Sun L (2019) Synthesis, properties, and applications of graphene oxide/reduced graphene oxide and their nanocomposites. NMS 1(1): 31-47.
- Moussavi G, Hossaini Z, Pourakbar M (2016) High-rate adsorption of acetaminophen from the contaminated water onto double-oxidized graphene oxide. Chem Eng Technol. 287: 665-673.
- Zhang L, Ni C, Jiu H, Xie C, Yan J, et al. (2017) One-pot synthesis of Ag-TiO2/reduced graphene oxide nanocomposite for high performance of adsorption and photocatalysis. Ceram Int 43(7): 5450-5456.
- Liu Y, Dong X, Chen P (2012) Biological and chemical sensors based on graphene materials. Chem Soc Rev 41(6): 2283-2307.
- Singh P, Shandilya P, Raizada P, Sudhaik A, Sani AR, et al. (2020) Review on various strategies for enhancing photocatalytic activity of graphene-based nanocomposites for water purification. Arab J Chem 13(1): 3498-3520.
- Ghosh D, Dhibar S, Dey A, Mukherjee S, Joardar N, et al. (2019) Graphene oxide dispersed supramolecular hydrogel capped benign green silver nanoparticles for anticancer, antimicrobial, cell attachment and intracellular imaging applications. J Mol Liq 282: 1-12.
- Safarpour M, Khataee A (2019) Graphene-based materials for water purification, in Nanoscale Materials in Water Purification. Elsevier p. 383-430.
- Hu W, Peng C, Luo W, Lv M, Li X, et al. (2010) Graphene-based antibacterial paper. ACS nano 4(7): 4317-4323.
- Zhang L, Xia J, Zhao Q, Liu L, Zhang Z, et al. (2010) Functional graphene oxide as a nanocarrier for controlled loading and targeted delivery of mixed anticancer drugs. Small 6(4): 537-544.
- Cobos M, Pinta IDL, Quindós G, Fernández MJ, Fernández MD (2020) Graphene oxide–silver nanoparticle nanohybrids: Synthesis, characterization, and antimicrobial properties. Nanomater 10(2): 376.
- Cheng Y, Li H, Fang C, Ai L, Chen J, et al. (2019) Facile synthesis of reduced graphene oxide/silver nanoparticles composites and their application for detecting heavy metal ions. J Alloys Compd 787: 683-693.
- Pasricha R, Gupta S, AK Srivastava (2009) A facile and novel synthesis of Ag–graphene‐based nanocomposites. Small 5(20): 2253-2259.
- Shen J, Shi M, Li N, Yan B, Ma H, et al. (2010) Facile synthesis and application of Ag-chemically converted graphene nanocomposite. Nano Res 3(5): 339-349.
- Vi TTT, Lue S (2016) Preparation of silver nanoparticles loaded graphene oxide nanosheets for antibacterial activity. IOP Conf Ser Mater Sci Eng IOP Publishing.
- Das MR, Sarma RK, Saikia R, Kale VS, Shelke MV, et al. (2011) Synthesis of silver nanoparticles in an aqueous suspension of graphene oxide sheets and its antimicrobial activity. Colloids Surf B Biointerfaces 83(1): 16-22.
- Ma J, Zhang J, Xiong Z, Yong Y, Zhao XS (2011) Preparation, characterization and antibacterial properties of silver-modified graphene oxide. J Mater Chem 21(10): 3350-3352.
- Zhou X, Huang X, Qi X, Wu S, Xue C, FYC Boey, et al. (2009) In situ synthesis of metal nanoparticles on single-layer graphene oxide and reduced graphene oxide surfaces. J Phys Chem C 113(25): 10842-10846.
- Lu G, Mao S, Park S, Ruoff RS, Chen J, et al. (2009) Facile, noncovalent decoration of graphene oxide sheets with nanocrystals. Nano Res 2(3): 192-200.
- Dinh D, Hui KS, Hui KN, Cho YR, Zhou W, et al. (2014) Green synthesis of high conductivity silver nanoparticle-reduced graphene oxide composite films. Appl Surf Sci 298: 62-67.
- Zhongchun L (2010) Research Progress on Controllable Synthesis of Bimetallic Nanomaterials. Rare Metal Mat Eng 10.
- Kumari S, Sharma P, Yadav S, Kumar J, Vij A, et al. (2020) A Novel Synthesis of the Graphene Oxide-Silver (GO-Ag) Nanocomposite for Unique Physiochemical Applications. ACS Omega 5(10): 5041-5047.
- Bao Q, Zhang D, Qi P (2011) Synthesis and characterization of silver nanoparticle and graphene oxide nanosheet composites as a bactericidal agent for water disinfection. J Colloid Interface Sci 360(2): 463-470.
- Das MR, Sarma RK, Borah SC, Kumari R, Saikia R, et al. (2013) The synthesis of citrate-modified silver nanoparticles in an aqueous suspension of graphene oxide nanosheets and their antibacterial activity. Colloids Surf B 105: 128-136.
- Shen J, Shi M, Yan B, Hongwei M, Lia N, et al. (2011) One-pot hydrothermal synthesis of Ag-reduced graphene oxide composite with ionic liquid. J Mater Chem 21(21): 7795-7801.
- Das B, Chaudhuri KJ (2014) Amelioration of sodium arsenite induced toxicity by diallyl disulfide, a bioactive component of garlic: the involvement of antioxidants and the chelate effect. RSC Adv 4(40): 20964-20973.
- Hummers WS, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80(6): 1339-1339.
- Nambiar DK, Kujur PK, Singh RP (2016) Angiogenesis assays, in Cancer Chemoprevention. Springer pp. 107-115.
- Avram S, Ghiulai R, Pavel IZ, Mioc M, Babuta R, et al. (2017) Phytocompounds targeting cancer angiogenesis using the chorioallantoic membrane assay. In: Badria FA (Eds.), Natural Products and cancer drug discovery, Tech: p. 45-66.
- Imran M, Hussain S, Mehmood K, Saeed Z, Parvaiz M, et al. (2021) Optimization of ecofriendly synthesis of Ag Nanoparticles by Linum usitatissimum Hydrogel using Response Surface Methodology and its biological applications. Mater Today Commun 29: 102789.
- Imran M, Saeed Z, Pervaiz M, Mehmood K, Ejaz R, et al. (2021) Enhanced visible light photocatalytic activity of TiO2 co-doped with Fe, Co, and S for degradation of Cango red. Spectrochim Acta A pp. 119644.
- Fan D, You F, Zhang Y, Huang Z (2018) Flame Retardant Effects of Fabrics Finished by Hybrid Nano-Micro Silica-based Sols. Procedia Eng 211: 160-168.
- Murdock RC, Stolle LB, Schrand AM, Schlager JJ, Hussain SM (2008) Characterization of nanomaterial dispersion in solution prior to in vitro exposure using dynamic light scattering technique. Toxicol Sci 101(2): 239-253.
- Priya GS, Kanneganti A, Kumar KA, Rao KV, Bykkam S (2014) Biosynthesis of Cerium oxide nanoparticles using Aloe barbadensis miller gel. Int J Sci Res Publ 4(6): 199-224.
- Asharani P, Sethu S, Vadukumpully S, Zhong S, Lim CT, et al., (2010) Investigations on the structural damage in human erythrocytes exposed to silver, gold, and platinum nanoparticles. Adv Funct Mater 20(8): 1233-1242.
- Carmeliet P (2005) Angiogenesis in life, disease and medicine. Nature 438(7070): 932-936.
- Yancopoulos GD, Davis S, Gale NW, Rudge JS, Wiegand S, et al. (2000) Vascular-specific growth factors and blood vessel formation. Nature 407(6801): 242-248.
- Folkman J (2006) Angiogenesis. Annu Rev Med 57: 1-18.
- Folkman J (1984) Angiogenesis. Trends Cardiovasc Med pp. 412-428.
- Shojaei F (2012) Anti-angiogenesis therapy in cancer: current challenges and future perspectives. Cancer Lett 320(2): 130-137.
- Ribatti D (2010) The chick embryo chorioallantoic membrane as an in vivo assay to study antiangiogenesis. Pharmaceuticals 3(3): 482-513.
- Mohammadzadeh R (2011) Hypothesis: Silver nanoparticles as an adjuvant for cancertherapy. Adv Pharm Bull 2(1): 133.
- Chen X, Schluesener HJ (2008) Nanosilver: a nanoproduct in medical application. Toxicol Lett 176(1): 1-12.






























