Microfluidic Preparation of Microencapsulated Phase Change Materials with Polyurea/Polymethyl Methacrylate Shell for Thermal Energy Storage
Hui Chang Wang, Ying Chen, Jun Li*, Lisi Jia* and Songping Mo
School of Materials and Energy, Guangdong University of Technology, China
Submitted: February 09, 2022; Published: February 17, 2022
*Corresponding author: Jun Li and Lisi Jia, School of Materials and Energy, Guangdong University of Technology, China
How to cite this article: Hui Chang W, Ying C, Jun L, Lisi J, Songping M. Microfluidic Preparation of Microencapsulated Phase Change Materials with Polyurea/Polymethyl Methacrylate Shell for Thermal Energy Storage. JOJ Material Sci. 2022; 6(5): 555699. DOI:10.19080/JOJMS.2022.06.555699
Abstract
A facile three-step approach was presented to prepare microencapsulated phase change materials (MEPCMs) with n-octadecane as core and polyurea/polymethyl methacrylate (PUA/PMMA) as shell. First, a droplet-based microfluidic technique was introduced to generate microdroplets of n-octadecane with controllable size. Then, an interfacial polymerization technique was employed to form a protective shell of PUA on microdroplets to prevent them from collapse and coalescence. Finally, an in-situ polymerization technique was adopted to form a compact shell of PMMA inside the PUA shell to improve the thermal performance of MEPCMs. The MEPCMs with PUA/PMMA double-layered shell exhibited high monodispersed in size and sphericity, good phase-change performance, and enhanced thermal stability and durability for thermal energy storage applications.
Keywords: Microencapsulation; Phase change material; Double-layered shell; Microfluidics; Thermal energy storage
Abbreviations: PCM: Phase Change Material; TES: Thermal Energy Storage; MMA: Methyl Methacrylate; DSC: Differential Scanning Calorimeter; TGA: Thermogravimetric Analyzer
Introduction
MEPCMs are small composite particles of phase change material (PCM) with stable core–shell structure [1]. The shell of MEPCMs can isolate the PCM core from the outside environment and ensure the safety and reliability of MEPCMs. MEPCMs as thermal energy storage (TES) material can modulate the temperature by cyclically storing and releasing latent heat through phase transition, and thus have potentially wide-ranging applications in smart thermo-regulated textiles [2], electronic and electrical appliances [3], battery-electrified vehicles [4], and intelligent buildings [5]. It is noted that MEPCMs fabricated by conventional methods, such as in-situ polymerization, interfacial polymerization, and mini emulsion polymerization, often feature nonuniform sizes and irregular shapes because the emulsion procedure is usually triggered by mechanical stirring or ultrasonic vibration [1,6,7]. The polydispersity of MEPCMs is probably the reason for inconsistency and unrepeatability of current studies on TES applications [8-10]. Considering that controllable size and morphology is an important prerequisite for stabilizing the performance of MEPCMs, a novel droplet-based microfluidic technique was employed in the study instead of mechanical emulsification. Droplet-based microfluidics can realize the massive production of monodisperse droplets and easy control of each droplet [11,12]. Interfacial and in-situ polymerization techniques were combined to from a double-layer polymer shell on PCM droplets. The former was adopted for forming a thin layer (PUA) instantly on PCM droplets to prevent collision and coalesce, and the latter for synthesizing a compact and stiff shell (PMMA) by using the stable droplets as templates to enhance thermal performance for TES. The resultant MEPCMs with PUA/PMMA shell were expected to have good morphology and stable performance.
Materials and Methods
The co-flow microfluidic setup used to generate the MEPCMs with controllable structure and size is displayed in Figure 1. The dispersed and continuous phases were injected into the glass capillaries using the syringe pumps, and the corresponding flow rates were set at 500 μL/min and 20 μL/min, respectively. The dispersed PCM phase was a mixture solution consisting of 2,4-toluene diisocyanate (TDI, 2.5 wt.%), methyl methacrylate (MMA), and 2,2-azobisisobutyronitrile (AIBN) in n-octadecane. The continuous phase was an aqueous solution consisting of tetraethylenepentamine (TEPA, 5.0 wt.%) and sodium dodecyl sulfate (SDS, 5.0 wt.%) in deionized water. All materials were purchased from Sigma-Aldrich and used as received, Singapore and used as received. The mass concentration of MAA monomers ranged from 30 wt.% to 45 wt.%, and the mass ratio of MMA to AIBN used to induce the polyreaction of MMA monomers was set at 20.

The MEPCM preparation involved three steps: the formation of PCM microdroplets, the formation of MEPCMs with PUA shell, and the formation of MEPCMs with PUA/PMMA shell. Firstly, the dispersed phase traveled to the capillary orifice (40μm) and broke into the spherical PCM microdroplets by a shear force of continuous phase. The precise flow control of the continuous and dispersed phases facilitated the production of monodisperse microdroplets. Secondly, the shell of PUA was formed on the PCM microdroplets when they migrated in the direction of the continuous phase. The TDI in the microdroplets could react with the TEPA in the continuous phase to form PUA through interfacial polymerization. The reaction was terminated once the PUA polymer completely encapsulated the PCM microdroplets. Finally, the MEPCMs with PUA shell were collected in a vial of deionized water and placed in a warm-box for 8 h at 80°C to form the second layer of PMMA inside the PUA shell through the in situ polymerization of MMA monomers. The resultant MEPCMs with double-layered shell were washed with deionized water and dried at 45°C for 12 h to remove the water for performance testing.
The morphology of the MEPCMs with PUA/PMMA shell was measured using a field-emission scanning electron microscopy (SEM, Sigma 300, ZEISS, Germany), the chemical structure was measured using a fourier transformation infrared spectroscope (FTIR, 8400S, Shimadzu Corp., Japan), and the thermal stability was evaluated using a thermogravimetric analyzer (TGA Q50, TA Instruments, USA). The phase change performance of the MEPCMs was measured using a differential scanning calorimeter (DSC 3500, Netzsch, Germany). The measurements were carried out in the temperature range of 0°C-50°C at a heating/cooling rate of 5oC/min. The encapsulation efficiency (Een=(ΔHm,MEPCM/ΔHm,PCM)×100%), the energy storage efficiency (Ees=(ΔHm,MEPCM+ΔHf,MEPCM)/ (ΔHm,PCM+ΔHf,PCM)×100%), and the thermal storage capability (Ces=(Ees/Een)×100%) of the MEPCMs could be calculated from the melting (ΔHm) and freezing(ΔHf) enthalpies obtained from DSC scans [13,14]. The prepared MEPCMs were also subjected to 200 cycles of heating and cooling in DSC to evaluate thermal durability.
Results and Discussion
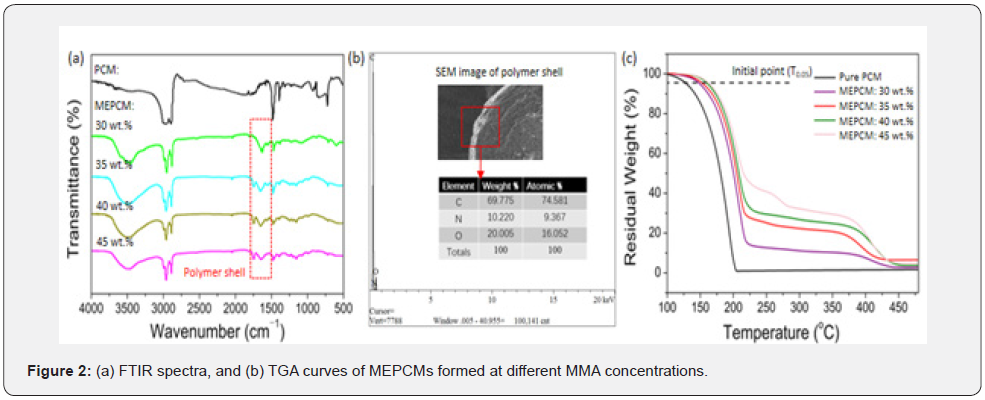
Figure 2a shows the FTIR spectra of the n-octadecane and the MEPCMs with PUA/PMMA shell. The MEPCMs presented three additional peaks compared to the n-octadecane. The peaks at 1649 and 1558 cm-1 were assigned to the carbamide group of PUA, and the peak at 1732cm-1 was assigned to the ester carbonyl group of PMMA, suggesting that the n-octadecane was successfully encapsulated by the PUA/PMMA shell. A typical elemental spectrum of the MEPCMs was given in Figure 2b, and the presence of carbon, oxygen and nitrogen elements also confirmed the microencapsulation of n-octadecane with PUA and PMMA as shells. Figure 2c shows the TGA curves of the pure n-octadecane and the MEPCMs formed at different MMA concentrations. The thermal degradation of n-octadecane exhibited one stage with the initial weight-loss temperature (T0.05) of around 131°C. The decomposition of n-octadecane went quickly and stopped at around 206oC. The prepared MEPCMs degraded in two stages: the first stage was the weight-loss of n-octadecane in the temperature range of 150-225oC, and the second stage showed the decomposition of polymer shell in the temperature range of 225- 450oC. Apparently, the decomposition of n-octadecane core was successfully delayed by the PUA/PMMA shell, indicating that the MEPCMs achieved good thermal stability.
The SEM micrographs of the MEPCMs prepared at different MMA concentrations were displayed in Figure 3a. The MEPCMs had a regular spherical shape and a uniform size distribution around 100μm, demonstrating that the first shell of PUA could stabilize and prevent the PCM microdroplets from breakup in forming the second shell of PMMA. The polydispersity index (PDI) was used to characterize the size distribution of the MEPCMs. PDI is a number calculated from σ/da, where da is the average size, and σ is the standard deviation. In the study, the maximum value of PDI did not exceed 0.03, indicating that highly monodispersed MEPCMs was achieved via the droplet-based microfluidic approach. Figure 3b shows the SEM micrographs of the MEPCMs subjected to thermal cycling test consisting of 200 cycles between 0oC and 50oC. The MEPCMs formed at 40 wt.% and 45 wt.% maintained good spherical shape and compact surface, whereas for those formed at 30 wt.% and 35 wt.%, broken MEPCMs with irregular shapes were observed, namely, the thin shell couldn’t protect the n-octadecane from leakage anymore. In short, the MEPCMs with PUA/PMMA shell could achieve long-term thermal durability when the MMA concentration was higher than 40 wt.%.

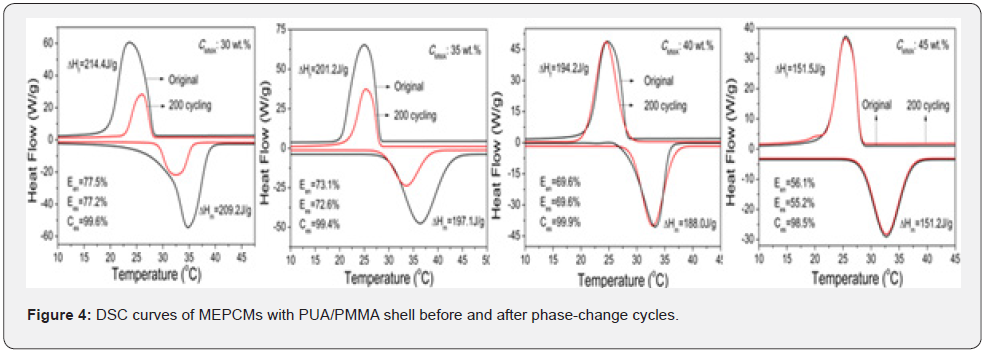
Figure 4 presents the DSC curves of MEPCMs formed at different MMA concentrations. For all MEPCMs, the onset temperatures of freezing (Tf) were approximately equal to those of melting (Tm), implying that the PMMA shell could serve as nucleation substrate to promote phase change of n-octadecane without supercooling (ΔT=Tm-Tf). Supercooling elimination of MEPCMs is beneficial for reducing the energy consumption and making the TES system efficient [15-17]. With increasing MMA concentration, the phasechange enthalpies of MEPCMs decreased, and the encapsulation efficiency (Een) reduced from 77.5% to 56.1%. The energy storage efficiencies (Ees) of MEPCMs were close to the encapsulation efficiencies, suggesting that n-octadecane core in MEPCMs could freeze completely and release almost all of latent heat in the subsequent melting. The calculation of thermal storage capability (Ces≥98.5%) further verified that n-octadecane was well encapsulated by polymer shell and could fully exert its function during the phase-change cycle [14]. Figure 4 also shows the DSC curves of MEPCMs after 200 phase-change cycles. The MEPMCs formed at 40 wt.% and 45 wt.% exhibited little temperature and enthalpy changes in exothermic and endothermic peaks, whereas significant changes appeared in those formed at 30 wt.% and 35 wt.%. It is indicated that the MEPCMs with encapsulation efficiencies of 69.6% and 56.1% possessed good energy storage and release performance for TES.
Conclusion
Microencapsulated n-octadecane with PUA/PMMA doublelayered shell were successfully fabricated by combing the microfluidic emulsification, interfacial and in-situ polymerization techniques. SEM results verified that highly monodisperse MEPCMs with regular spherical shape were attained, the polydispersity index of which was lower than 0.03. TGA measurements showed that the compact PUA/PMMA shell could protect the MEPCMs from thermal decomposition. DSC analysis confirmed that no supercooling was required for the phase change of MEPCMs, and good thermal storage capacities higher than 98.5% were achieved. Thermal cycling tests indicated that the MEPCMs with encapsulation efficiency up to 70% had long-term thermal durability. The advanced MEPCMs with PUA/PMMA shell possess good potentials for TES purposes, such as intelligent textiles, solar cells, and building materials.
Acknowledgment
The work was supported by the National Natural Science Foundation of China [grant number 51876045], and the authors acknowledge the support from Guangdong Special Support Program (Grant No. 2017TX04N371).
References
- Chang Z, Wang K, Wu X, Lei G, Wang Q, et al. (2022) Review on the preparation and performance of paraffin-based phase change microcapsules for heat storage. J Energy Storage 46: 103840.
- Zhao L, Luo J, Wang H, Song G, Tang G (2016) Self-assembly fabrication of microencapsulated n-octadecane with natural silk fibroin shell for thermal-regulating textiles. Appl Therm Eng 99: 495-501.
- Rostamian F, Etesami N, Haghgoo M (2021) Management of electronic board temperature using heat sink containing pure and microencapsulated phase change materials. Int Commun Heat Mass 126: 105407.
- Zhang X, Li Z, Luo L, Fan Y, Du Z (2022) A review on thermal management of lithium-ion batteries for electric vehicles. Energy 238: 121652.
- Chen X, Kong X, Wang S, Fu X, Yu B, et al. (2022) Microencapsulated phase change materials: Facile preparation and application in building energy conservation. J Energy Storage 48: 104025.
- Zhao Y, Zhang X, Hua W (2021) Review of preparation technologies of organic composite phase change materials in energy storage. J Mol Liq 336: 115923.
- Tao J, Luan J, Liu Ye, Qu Da, Yan Z, et al. (2022) Technology development and application prospects of organic-based phase change materials: An overview. Renew Sust Energ Rev 159: 112175.
- Ran F, Chen Y, Cong R, Fang G (2020) Flow and heat transfer characteristics of microencapsulated phase change slurry in thermal energy systems: A review. Renew Sust Energ Rev 134: 110101.
- Chai L, Shaukat R, Wang L, Wang H (2018) A review on heat transfer and hydrodynamic characteristics of nano/microencapsulated phase change slurry (N/MPCS) in mini/microchannel heat sinks. Appl Therm Eng 135: 334-349.
- Jurkowska M, Szczygieł I (2016) Review on properties of microencapsulated phase change materials slurries (mPCMS). Appl Therm Eng 98: 365-373.
- Sattari A, Hanafizadeh P, Hoorfar M (2020) Multiphase flow in microfluidics: From droplets and bubbles to the encapsulated structures. Adv Colloid Interfac 282: 102208.
- Zhu P, Wang L (2017) Passive and active droplet generation with microfluidics: a review. Lab on a Chip 17(1): 34-75.
- Zhao L, Luo J, Wang H, Song G, Tang G (2016) Self-assembly fabrication of microencapsulated n-octadecane with natural silk fibroin shell for thermal-regulating textiles. Appl Therm Eng 99: 495-501.
- Zhao J, An J, Yang J, Yang E (2018) Microencapsulated phase change materials with composite titania-polyurea (TiO2-PUA) shell. Appl Energ 215: 468-478.
- Zahir M, Mohamed S, Saidur R, Al Sulaiman F (2019) Supercooling of phase-change materials and the techniques used to mitigate the phenomenon. Appl Ener 240: 793-817.
- R Al Shannaq J, Kurdi S, Al Muhtaseb, Dickinson M, Farid M (2015) Supercooling elimination of phase change materials (PCMs) microcapsules. Energy 87: 654-662.
- Shamseddine I, Pennec F, Biwole P, Fardoun F (2022) Supercooling of phase change materials: A review. Renew Sust Energ Rev 158: 112172.






























