Colloidal Apatite Particles : A Multifunctional Platform in (Nano)Medicine
Christophe Drouet1*, Maëla Choimet1,2, Marina Simon3,4, Guillaume Devès3,4, Philippe Barberet3,4, Hervé Seznec3,4, Giovanna Rassu5, Olivier Marsan1, Audrey Tourrette1
1Phosphates, Pharmacotechnics, Biomaterials group, CIRIMAT, Université de Toulouse, France
2 Department of Material Science, SEPPIC- Air Liquide Healthcare, France
3Department of Biomedical Sciences, Université de Bordeaux, France
4Department of Nanobiotechnology, Centre d’Etudes Nucléaires Bordeaux Gradignan (CENBG), France
5Department of Chemistry and Pharmacy, University of Sassari, Italy
Submitted: December 18, 2019; Published: February 07, 2020
*Corresponding author: Christophe Drouet, Phosphates, Pharmacotechnics, Biomaterials group, CIRIMAT-Ensiacet, Université de Toulouse, CNRS, UPS, INP, 31030 Toulouse cedex 4, France
How to cite this article: Drouet C, Choimet M, Simon M, Devès G, Barberet P, Seznec H, Rassu G, Marsan O, Tourrette A, Colloidal Apatite Particles : A Multifunctional Platform in (Nano)Medicine. JOJ Material Sci. 2020; 6(1): 555676. DOI: o10.19080/JOJMS.2020.06.555676
Abstract
Calcium phosphate apatites are naturally present in all vertebrates, as a biomineral in teeth and bones. The possibility to prepare synthetic analogs in close-to-physiological conditions allows the development of bio-inspired materials for medical use. While apatite-based biomaterials are extensively developed for bone regeneration, their intrinsic biocompatibility also allows designing original applications as in nanomedicine. In this paper, we will illustrate why colloidal apatite particles can be seen as a multifunctional platform for (nano)medical uses, well beyond bone repair. Retrospective data will be summarized in this mini-review, especially relating to oncology and hematology applications of apatite colloidal particles. Then novel data will be presented, in the field of dermatology and of particle tracking, using Raman confocal microscopy and μPIXE elemental mapping on substituted apatites. By exploiting their exceptional surface and bulk reactivities, colloidal apatite particles appear as robust new comers to nanomedicine, with the capacity to exhibit tailorable functionalities.
Keywords: Apatite; Biomimetism; Nanoparticles; Nanomedicine; Colloid; Oncology; Hematology; Dermatology; Platform; Multifunctional
Introduction
The mineral component of bones and teeth is composed of a calcium phosphate apatitic phase [1,2]. While enamel contains nearly stoichiometric, well-crystallized hydroxyapatite Ca10(PO4)6(OH)2, exhibiting a high thermodynamic stability well suited to face bacterial acidic attacks, bone apatite is in contrast composed of nonstoichiometric (calcium-and hydroxide-deficient) nanocrystalline apatite more prone to undergo remodeling in vivo upon the controlled local acidic environment provided by resorptive osteoclastic cells [1]. Indeed, apatite solubility is directly related to its degree of stoichiometry [3]. The nonstoichiometry and nanocrystalline characters of bone apatite imply a high reactivity that can be used by the organism for maintaining the appropriate ionic concentrations in body fluids (homeostasis). This is all the more facilitated by the high mobility of ions from the surface of apatite nanocrystals, which are included in an amorphous-like ionic hydrated layer on the surface of the nanocrystals [4-9]. Thanks to these features, it becomes possible to modulate the reactivity of biomimetic nanocrystalline apatite crystals by appropriate ionic substitutions and/or surface ion exchanges (e.g. [10]) and also via the adsorption of molecular species like antibiotics [11], enzymes [12], bisphosphonates [13] and anticancer drugs [14-16], among others. Indeed, the sorption of several types of drugs or biomolecules is facilitated on this type of apatitic compounds exhibiting high surface reactivity/ionic mobility/surface area versus other compounds like stoichiometric, well-crystallized hydroxyapatite.
This possibility is especially appealing as it is possible to prepare synthetic analogs to bone apatite via precipitation methods (e.g. [17-20]), e.g. using close-to-physiological conditions of precipitation (temperature well below the water boiling point, pH close to 7.4). The bone-inspired character of such apatite compounds and their associated high reactivity has led to numerous studies, alone or combined with active agents/drugs, in the field of bone repair. More recently, the relevance of using such mineral phases in other domains than bone regeneration has however emerged and is now expanding by appropriation of the intrinsic qualities of bio-inspired apatites, namely their high biocompatibility, ability to modify ionic compositions and to adsorb various types of (bio)molecules and drugs. The present contribution will illustrate their potential multifunctionality by summarizing some of the discoveries made in the use of apatite particles in nanomedicine, well beyond bone repair, as in oncology and hematology; and it will also present some new data in the field of dermatology and apatite particle tracking.
Results and discussion
In the field of nanomedicine, particles are expected to play a role at the tissue or cellular levels. Therefore, the control of the particle size is often a prerequisite to optimize the targeted effects and limit drawbacks such as clogging. As apatite compounds have a strong natural tendency to aggregate–which contributes to decrease the impact of surface energy – the first aspect that needs to be addressed is the control of the agglomeration state. Successful results were obtained by using stabilizing agents, forming an “organic corona” around the (mineral) apatite particles and contributing to colloidal stabilization [21,22]. By exploiting a concept of steric hindrance and/or electrostatic repulsion between adjacent particles, it was indeed possible to obtain colloidal, individually- dispersed, apatite particles. Typical examples of relevant biocompatible stabilizing agents were tabulated in ref. [21]. Phosphonated polyethyleneglycol (PEGp) was shown to exert agglomeration control over the apatite particles in formation, and this effect was found to depend directly on the PEGp molecular weight. Typical particle sizes around 180 nm were for example obtained reproducibly. Other agents were found to impede more efficiently apatite agglomeration, such as phosphocholine, e.g. leading to apatite particles centered on 70 nm in size, or AEP (2-aminoethylphosphate). The latter was especially found to be effective to prepare nanometric apatite particles of sizes lower than 100 nm, and typically down to 30–40 nm depending on the relative proportions used in the precipitation medium [22].
Replacement of the phosphate end group in AEP–interacting with the apatite surface – by a phosphonate group was shown in this case to give very similar sizes. While 30–40nm particles are well-suited to interact with cells, e.g. in view of internalization, larger dimensions may be more adapted to other applications, for example in dermatology. The variety of sizes listed above allows envisioning various different medical uses depending on clinical needs. Large macromolecules such as DNA were also tested, leading to submicron particles (200–300 nm in length), and this approach is especially relevant in the scope of transfection applications where DNA delivery inside cells is needed [23]. Figure 1 gives a schematic view of one individual particle, which can be considered as hybrid as it involves a mineral core (apatite) and an organic corona. This corona may, as mentioned above, involve stabilizing/dispersing agent(s) to allow colloidal dispersion; and as such, it represents a first way to modulate the particle behavior when contacted with a liquid medium. Other ways to modulate the particles behavior can be combined to this colloidal stabilization, so as to seek additional functionalities, as also illustrated on Figure 1. One appealing ability is the possibility to interact more specifically with certain type(s) of cells. This issue can be addressed by adsorbing, besides the dispersing agent, another molecular species exhibiting cell-targeting capabilities. Such targeting agents can for example be in the form of antibodies [24,25] or other molecular groups that can be recognized by cell membrane receptors. For example, folic acid (FA) is known to interact strongly with folate receptors (FR) that are over expressed on the surface of some breast or ovary cancer cells [26,27].

Preliminary adsorption of FA on AEP-stabilized apatite particles prior to contact with breast cancer cells was indeed shown to target more specifically FR+ cells (here ZR-75-2) compared to FR– cells (here T-47-D) [21]. The adsorption of drugs is also one appealing capability of such engineered particles that may be used as drug carriers. Adsorption of a chemotherapeutic anti-folate drug, namely methotrexate (MTX), was for example shown to be feasible onto apatite crystals with an adsorption behavior very similar to FA [14]. A property of the apatite phase, especially in a highly-divided state, is its capacity to adsorb a very large variety of molecules, especially exhibiting charged end-groups. For instance, adsorption of other chemotherapeutics such as doxorubicin (DOX) has also been investigated, along with the consequential effect on cancer cell viability [15]. It may be added that the sorption of a combination of different drugs, as illustrated on Figure 1 as “drugs “A” and “B”, can also be considered as a way to allow local co-delivery of various active agents in the objective of synergistic effects at the relevant clinical site. Drug adsorption may also involve a “linker” (as represented in the case of drug “B” on Figure 1), which is a portion of the adsorbed molecule that may be cleaved on contact with some external stimulus like a drop in pH or the presence of specific circulating species, for example. The large possibilities offered by apatite particles in terms of versatile adsorption can also be appropriately exploited to modulate the local behavior of cell membranes.
This was recently demonstrated in the field of hematology, and more specifically for the cryoprotection of Red Blood Cells (RBC) [28]. The exposure of an organic corona exhibiting charged end-groups around the apatite colloidal particles may indeed modify the electrical properties of RBC membranes, in a reversible non-destructive way, making it more permeable to small molecules like the glucose dimer “trehalose”, a cryoprotecting agent. By increasing drastically trehalose permeation across RBC membranes, cell viability greater than 90% was reached after a cycle of freezing down to liquid nitrogen temperature and subsequent rapid thawing to ambient conditions, which parallels what is now a days obtained using toxic glycerol as cryoprotecting molecule [28,29]. This modified permeation was also shown on synthetic lipid bilayers as simplified view of cell membranes, using FITC as “diffusing model drug” [28]. All of the above shows how the organic corona around apatite particles can modify their properties in terms of particle dimensions, cell targeting, local (co)delivery of drugs, smart linker-associated drug release, or else concerning their interaction with synthetic or biological membranes. In addition, the modification of the chemical composition of the crystalline core of the particles is also a way to convey new functionalities to colloidal apatite particles (see Figure 1). One possibility is for example to confer to the particles some intrinsic luminescence features, as this can be used either in view of medical diagnosis (e.g. for tracking diseased cells having internalized the particles) or for the development of medical strategies using these particles, as in dermatology.

The luminescence property is then typically conveyed by substituting part of the Ca2+ ions from the calcium phosphate apatite phase by luminescent lanthanides like Eu3+ (red emission) or Tb3+ (green emission) [23,30, 31]. Such ionic substitutions were found to be feasible at least up to a few at.% [32], even in the colloidal state [30, 31], and the luminescence features are characteristic of these lanthanide ions which exhibit a low toxicity, a long luminescence lifetime (of the order of the millisecond compared to the autofluorescence of biological media of few nanoseconds) and a non-bleaching effect as opposed to organic dyes. The luminescent apatite particles can be used for example to follow the localization of the particles after application on skin. This approach was studied here in the case of porcine ear skin explants, and using the photoluminescence of Eu3+ under a Raman microscope. Figure 2 displays an example of 3D mapping obtained after applying onto fresh (non-frozen) ear skin a colloid composed of apatite particles stabilized with PEGp (MW 5200 g/mol) with an average size of 180 nm. As seen on this figure, coupled histological, Raman and luminescence features allowed us to point out the superficial localization of the particles on top the skin. This, in turn, allows envisioning dermatological uses of such colloidal particles, in view of topical (skin surface) applications. Besides luminescent ions, other ionic species can also be exploited as dopants of apatite particles in view of following their localization within tissues or inside cells. The use of techniques other than luminescence detection is then required.
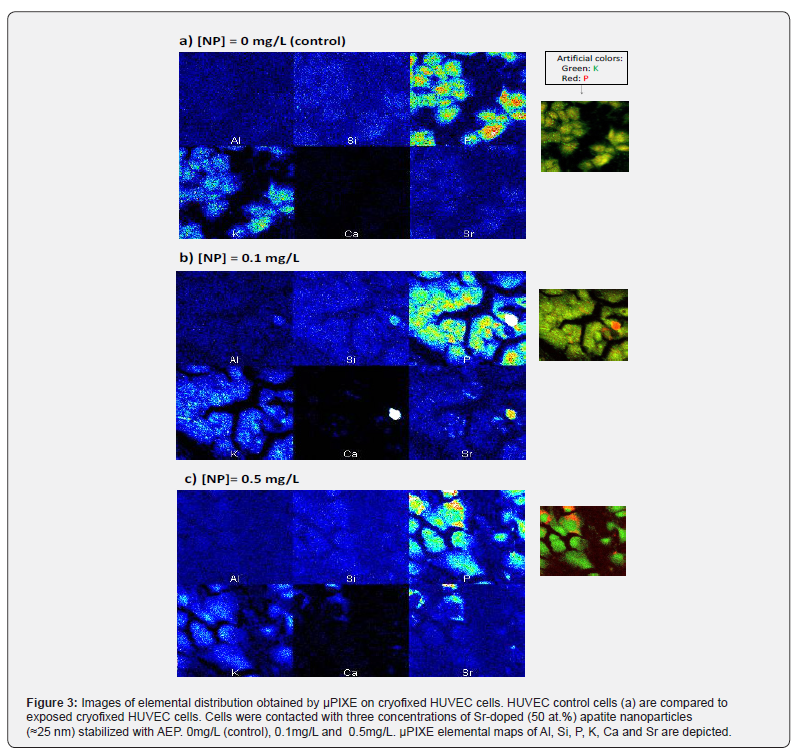
One appealing approach is the combination of μPIXE (Particle- induced X-ray emission) and RBS (Rutherford Backscattering Spectrometry) for elemental mapping and quantification purposes. μPIXE provides note only the chemical composition of the sample but also their elemental mapping (Figure 3). During the interaction with the exciting beam, the chemical elements undergo atomic excitation-de-excitation processes that eventually lead to the emission of a photon which exhibits the characteristic energy of the atomic number of the excited element. Standard experimental setups and detectors used for μPIXE allow simultaneous quantification of all elements heavier that Na with a 1 to 10 μg.g- 1 dry masse detection limit. In this work, AEP-stabilized apatite particles doped with Sr2+ ions – via partial substitution of Ca2+ ions – were contacted with HUVEC cells and analyzed by μPIXE (Figure 3). As may be seen, phosphorus is homogeneously distributed in the cell with a much higher concentration in the nucleus area (Figure 3, red), and potassium is homogeneously distributed in the cell volume (Figure 3, green). While P is overly present even in the control due to biochemical molecular contents (nucleic acids, proteins), an increase in Sr detection can be directly related to the internalization of the particles by HUVEC cells. Concomitantly, an increase in Ca can also be pointed out. The corresponding quantitative data are reported on Figure 4.
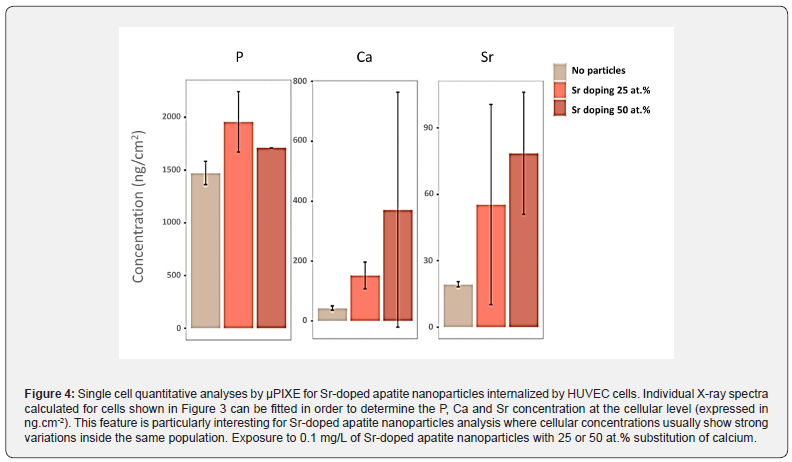
Besides particle tracking, the incorporated ions can also play a biological role, which may be relevant to the clinic. For example, ions such as Cu2+ or Zn2+ can be added as dopants to the apatite [33] so as to provide antibacterial and/or anti-inflammatory effects, among other properties. One interest in this approach can be a delayed therapeutic action: while active molecules/drugs grafted on the particle within the organic corona are prone to play their therapeutic role first, the retarded release of such doping ions in the apatitic structure will essentially occur in a subsequent step upon the apatite biodegradation process. Therefore, a dual-kinetic release may be expected with prolonged biological effects. Other properties from substituting ions or ionic clusters within the apatite structure can also be exploited. For example, magnetic properties can be provided by using iron-bearing apatite-based systems-that may also contain iron oxides clusters [14,15,34,35]. Conferring magnetic properties to the particles can be interesting from various viewpoints, whether for hyperthermia cancer therapy or for helping the displacement of the particles to some clinically relevant sites from a patient’s body via the application of an external magnetic field. Also, the release of drugs attached to the particles can be activated in an external magnetic field was shown for example for DOX-loaded magnetic apatite particles [15].
Finally, let us mention that in view of pharmaceutical use, the inclusion of apatite (nano)particles within a galenic formulation will often be necessary. The formulation can for example modulate the hydrophilic/hydrophobic behavior which may have an incidence on tissue penetration and other processes in vivo. In this work, to illustrate this statement, an alginate gel was formulated containing a model drug, folic acid (FA) in two conditions:
a) using a simple FA solution dispersed in the gel and
b) using PEGp-stabilized apatite particles with pre-adsorbed FA, dispersed in the gel.
Then the permeation of the gels across synthetic membranes supposed to mimic the cutaneous barrier was followed in dynamic conditions using permeation test cells: modified Franz diffusion system incorporating three in-line flow-through diffusion cells. In a preliminary step, for easier comparison, the viscosity of the gel containing FA in the dissolved form (without apatite particles) was increased by adding 7.5 mmol/L of Ca2+ ions in the form of calcium chloride so as to reach the viscosity of the gel containing the colloidal apatite particles, around 1.85Pa.s for a shear rate of 100s-1. Then the permeation of the gels, including or not the particles, was followed with two synthetic (hydrophilic) membranes with 0.05 and 0.2 μm porosities (Figure 5). As may be seen by comparing Figures 5a & 5b, a clear decrease of the particles permeation can be detected when they are included in the gel (e.g. 27% permeation at 100min, Figure 5b) compared to the particles only suspended in water (e.g. 90% permeation at 100min, Figure 5a), and whatever the membrane porosity studied.
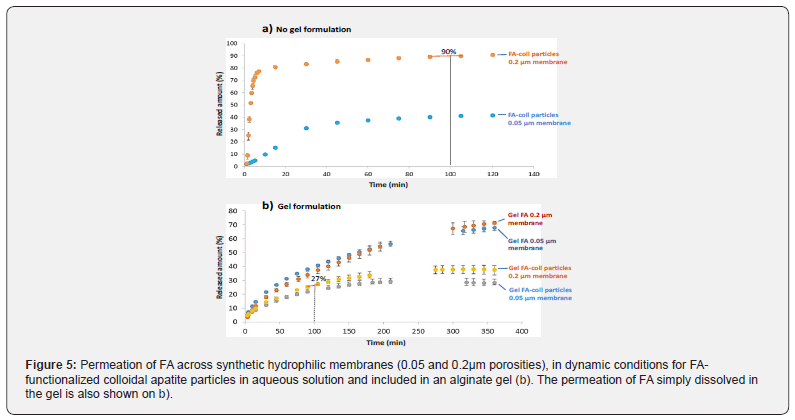
This result points out the clear effect on permeation played by the formulation of colloidal apatite particles. Also, it may be noted on Figure 5b that, in the gel state, adsorption of the drug-modeled here by FA-onto the apatite particles limits its diffusion across the membrane compared to the drug simply dissolved in the gel formulation. This may be relevant for favoring surface topical therapeutic effects for example in dermatology.
Concluding Remarks
This paper was intended as a mini-review pointing to the very versatile (nano)medical platform represented by bio-inspired apatite particles. Key reference works have been cited throughout the text to illustrate this, and allow the reader to obtain further details. In addition to their intrinsic biocompatibility due to similarity to bone apatite, the exceptional surface and bulk reactivity of colloidal apatite particles can be exploited to provide “à la carte” functionalities such as cell targeting, smart delivery of drugs/active agents, tailored interaction with cell membranes, luminescence and other tracking features, as well as magnetism. Plus, their pharmaceutical formulation may allow modulating even further their behavior relatively to relevant tissues or cells. Although “non-bone” applications are quite recent, as was illustrated here in the fields of oncology, hematology and dermatology, there is now doubt that engineered (colloidal) apatite particles should, from now on, be considered as major systems in (nano)medicine.
Experimental Details
The colloidal apatite particles used in the novel data reported in this work were synthesized using either AEP (2-aminoethylphosphate) or PEGp (phosphonated polyethyleneglycol, MW 5200g/mol) as stabilizing agents, as previously published [21,31], and the colloids were purified by dialysis as already reported [36]. Raman spectra were recorded on a Labram HR800 Horiba Jobin Yvon confocal microspectrometer using a green laser (wavelength 532 nm, 13 mW, 600 lines/mm), and processed using the LabSpec6 software. A x100 lens with 0.9 numerical aperture led to lateral resolution of 0.7μm and axial resolution of 2.6μm. The Raman 3D mapping of skin explant after colloid application was obtained with a red 532 nm laser (13 mW) with a 3μm pitch and 3s acquisition time in 1 accumulation. The porcine ear skin explant was used after biopsy: after cutting a relevant section, it was placed in resin molds (Tissue-Tek® OCT) and frozen at -80°C in isopentane. Sections 5 μm thick were sliced using a cryostat (Leica CM 1950), for analysis. Histological observations were carried out at the CPTP laboratory, Toulouse, France. The tissues were dehydrated in two successive absolute ethanol baths, followed by 2 xylene baths, and mounted on slats with Eukitt resin. μPIXE analyses were carried out on the AIFIRA facility (CENBG, France). Cells (HUVEC) were cultured directly onto ion beam microprobe sample holders as adapted from previous studies [37]. Briefly, cells were directly grown at high confluence on 2μm-thick polycarbonate foil for 24 hours in appropriate culture medium, and then exposed to 0.1 or 0.5 mg/L (as indicated in the text) of colloidal apatite particles doped with 25 or 50 at.% of strontium (replacing calcium in the apatite formula).
Cells exposed to only the aqueous medium containing hexametaphosphate 0.1 or 0.5 mg/L; HMP being used as redispersing agent in the formulation of the AEP-stabilized particles [21], were used as a control. Cells were rinsed once in culture medium, and very briefly rinsed twice in ultrapure water to remove excess of extracellular salts from culture medium. Finally, cells were plunge-freezed at -150°C into liquid nitrogen chilled 2-methylbutan (ReagentPlus, ≥99%, Sigma-Aldrich) and freeze-dried using a freeze-dryer (Christ alpha, ThermoFischer Scientific) in two phases: A phase of primary desiccation of 12 to 24 hours (T°C = -99°C, P = 0.001mbar) followed by a phase of secondary desiccation of 24 hours (T°C = +40°C, P = 0.001mbar). With no contamination and no ionic diffusion, freeze-drying was found to be the most adapted process. The samples were then kept at room temperature in a desiccator prior to PIXE analyses. The Sr, Ca, P, K, Al and Si elements were followed by PIXE using an incident proton beam of 1.5MeV, a beam diameter of 1 μm and an intensity of 150 pA. The scanned typical size was of 14μm, with an acquisition time of about 1h45 per zone. Protocols are described in previous works [37-39]. For the study on the effect of formulation, an alginate gel was used, incorporating the colloidal apatite particles stabilized by PEGp 5200 g/mol. To form the gel, 3% m/v of sodium alginate was added to the colloid using an Ultra-turrax® mixer at a speed of 6000 rpm.
For the gel containing only dissolved folic acid (FA), a solution of FA at 200 mg/L, pH 7.4 was prepared and calcium ions were added in the form of calcium chloride (7.5 mmol/L) to reach similar viscosity of the colloids, and sodium alginate was added as previously (3% m/v, Ultra-turrax® 6000 rpm). The permeation of FA (or FA-loaded particles) across two synthetic hydrophilic membranes with different porosities (polycarbonate: 0.05 μm and cellulose acetate: 0.2 μm) was monitored by UV spectroscopy (Shimadzu UV-160A, λ = 280 nm). Adsorption of FA onto the surface of the colloidal apatite particles was obtained by the addition of FA in the calcium salt solution prior to colloid formation (0.011mmol of FA for 4.87 mmol of Ca(NO3)2 . 4H2O). Permeation test cells developed at the Department of Chemistry and Pharmacy of the University of Sassari, Italy, [Italian Patent No. 102017000089514, granted on 10/17/2019, International Application No. PCT/IB2018/055726»PERMEATION TEST CELL», were used in this work as permeation system in dynamic conditions. It consists in three parallel thermostated permeation cells of vertical- diffusion type (32.0±0.5°C, approaching normal skin temperature) characterized by a diffusion area of 3.14 cm². Each cell is composed of an upper (donor) compartment on which the colloid/ gel is applied and of a lower (receiver) compartment where the FA concentration is measured. The recipient compartments were initially filled with buffer solution (250 mL of KH2PO4 0.2 M, 195.5 mL of NaOH 0.2M, deionized water up to 1L, pH close to the physiological) and magnetically stirred. The cells were connected to a peristaltic pump for providing dynamic circulation (total volume 50 mL, flowrate 6 mL/min) and to the UV-visible spectrophotometer (Shimadzu UV-160 A) as in-line drug monitoring system. An initial volume of 1ml colloid/gel was placed on the donor compartments at the start of the experiments. All experiments were done in triplicate.
Acknowledgements
M. Choimet wishes to thank the CIRIMAT Carnot Institute (now belonging to the Chimie Balard CIRIMAT Carnot Institute) for PhD funding and Toulouse INP for the travel grant to Sardinia, Italy. The AIFIRA facility, used for the μPIXE analyses, is financially supported by CNRS, Université de Bordeaux and the Région Nouvelle Aquitaine. We thank the technical staff members of the AIFIRA facility, P. Alfaurt and S. Sorieul. The authors also thank Dr. Talal Al Saati and Dr. Florence Capilla from the Centre de Physiopathologie de Toulouse-Purpan (CPTP), Toulouse, France, for support on the histology preparation.
Conflict of Interest
There is no conflict of interest to declare.
References
- Gomez-Morales J, Iafisco M, Delgado-Lopez JM, Sarda S, Drouet C (2013) Progress on the preparation of nanocrystalline apatites and surface characterization: Overview of fundamental and applied aspects. Progress in Crystal Growth and Characterization of Materials 59(1) : 1-46.
- Le Geros Z (1981) Apatites in biological systems, Progress in Crystal Growth and Characterization 4(1-2): 1-45.
- Rollin-Martinet S, Navrotsky A, Champion E, Grossin D, Drouet C (2013) Thermodynamic basis for evolution of apatite in calcified tissues. The American Mineralogist 98: 2037-2045.
- Von Euw S, Wang Y, Laurent G, Drouet C, Babonneau F, et al. (2019) Bone mineral: new insights into its chemical composition. Scientific Reports 9(1): 1-11.
- Drouet C, Aufray M, Rollin-Martinet S, Vandecandelaère N, Grossin D, et al. (2018) Nanocrystalline apatites: The fundamental role of water. The American Mineralogist 103(4): 550-564.
- Rey C, Combes C, Drouet C, Cazalbou S, Grossin D, et al. (2014) Surface properties of biomimetic nanocrystalline apatites; applications in biomaterials. Progress in Crystal Growth and Characterization of Materials 60(3-4) : 63-73.
- Vandecandelaere N, Rey C, Drouet C (2012) Biomimetic apatite-based biomaterials : on the critical impact of synthesis and post-synthesis parameters. J Mater Sci Mater Med 23(11): 2593-2606.
- Rey C, Lian J, Grynpas M, Shapiro F, Zylberberg L, et al. (1989) Non-apatitic environments in bone mineral: FT-IR detection, biological properties and changes in several disease states. Connective Tissue Research 21(1-4): 267-273.
- Rey C, Strawich E, Glimcher MJ (1994) Non-apatitic environments in Ca-P biominerals; Implications in reactivity of the mineral phase and its interactions with the organic matrix constituents. Bulletin de l'Institut Oceanographique (Monaco) 55-64.
- Drouet C, Carayon MT, Combes C, Rey C (2008) Surface enrichment of biomimetic apatites with biologically-active ions Mg2+ and Sr2+ : A preamble to the activation of bone repair materials. Materials Science & Engineering C 28(8): 1544-1550.
- Cazalbou S, Bertrand G, Drouet C (2015) Tetracycline-loaded biomimetic apatite: an adsorption study. The journal of physical chemistry. B 119(7): 3014-3024.
- Weber CG, Mueller M, Vandecandelaere N, Trick I, Burger-Kentischer A, et al. (2014) Enzyme-functionalized biomimetic apatites: concept and perspectives in view of innovative medical approaches. Journal of Materials Science-Materials in Medicine 25(3): 595-606.
- Pascaud P, Errassifi F, Brouillet F, Sarda S, Barroug A, et al. (2014) Adsorption on apatitic calcium phosphates for drug delivery: interaction with bisphosphonate molecules. Journal of Materials Science-Materials in Medicine 25: 2373-2381.
- Sarda S, Iafisco M, Pascaud-Mathieu P, Adamiano A, Montesi M, et al. (2018) Interaction of Folic Acid with Nanocrystalline Apatites and Extension to Methotrexate (Antifolate) in View of Anticancer Applications. Langmuir 34(40): 12036-12048.
- Iafisco M, Drouet C, Adamiano A, Pascaud P, Montesi M, et al. (2016) Superparamagnetic iron-doped nanocrystalline apatite as a delivery system for doxorubicin. Journal of Materials Chemistry B 4(1): 57-70.
- Iafisco M, Palazzo B, Marchetti M, Margiotta N, Ostuni R (2009) Smart delivery of antitumoral platinum complexes from biomimetic hydroxyapatite nanocrystals. Journal of Materials Chemistry 19(44): 8385-8392.
- Vandecandelaere N, Rey C, Drouet C (2012) Biomimetic apatite-based biomaterials: on the critical impact of synthesis and post-synthesis parameters. Journal of Materials Science: Materials in Medicine 23(11): 2593-2606.
- Iafisco M, Morales JG, Hernández-Hernández MA, García-Ruiz JM, Roveri N (2010) Biomimetic Carbonate–Hydroxyapatite Nanocrystals Prepared by Vapor Diffusion. Advanced Engineering Materials 12(7): B218-B223.
- Nassif N, Martineau F, Syzgantseva O, Gobeaux F, Willinger M, et al. (2010) In Vivo Inspired Conditions to Synthesize Biomimetic Hydroxyapatite, Chemistry of Materials 22(12): 3653-3663.
- Tas AC (2014) The use of physiological solutions or media in calcium phosphate synthesis and processing. Acta Biomaterialia 10(5): 1771-1792.
- Drouet C, Al-Kattan A, Choimet M, Tourrette A, Santran V (2015) Biomimetic Apatite-Based Functional Nanoparticles as Promising Newcomers in Nanomedicine: Overview of 10 Years of Initiatory Research. Journal of General Practice and Medical Diagnosis 1(1): 1-9.
- Bouladjine A, Al-Kattan A, Dufour P, Drouet C (2009) New advances in nanocrystalline apatite colloids intended for cellular drug delivery, Langmuir 25(20): 12256-12265.
- Mondéjar SP, Kovtun A, Epple M (2007) Lanthanide-doped calcium phosphate nanoparticles with high internal crystallinity and with a shell of DNA as fluorescent probes in cell experiments. Journal of Materials Chemistry 17(39) : 4153-4153.
- Iafisco M, Delgado-Lopez JM, Varoni EM, Tampieri A, Rimondini L (2013) Cell surface receptor targeted biomimetic apatite nanocrystals for cancer therapy. Small 9(22): 3834-3844.
- Iafisco M, Varoni E, Di Foggia M, Pietronave S, Fini M (2012) Conjugation of hydroxyapatite nanocrystals with human immunoglobulin G for nanomedical applications. Colloids and Surfaces B: Biointerfaces 90(1): 1-7.
- Antony AC (1992) The biological chemistry of folate receptors. Blood 79(11): 2807-2820.
- Spinella MJ, Brigle KE, Sierra EE, Goldman ID (1995) Distinguishing between folate receptor-alpha-mediated transport and reduced folate carrier-mediated transport in L1210 leukemia cells. J Biol Chem 270(14): 7842-7849.
- Stefanic M, Ward K, Tawfik H, Seemann R, Baulin V, et al. (2017) Apatite nanoparticles strongly improve red blood cell cryopreservation by mediating trehalose delivery via enhanced membrane permeation. Biomaterials 140:138-149.
- Scott KL, Lecak J, Acker JP (2005) Biopreservation of Red Blood Cells: Past, Present, and Future, Transfusion Medicine Reviews 19(2): 127-142.
- Al-Kattan A, Santran V, Dufour P, Dexpert-Ghys J, Drouet C (2014) Novel contributions on luminescent apatite-based colloids intended for medical imaging. Journal of Biomaterials Applications 28(5): 697-707.
- Al-Kattan A, Dufour P, Dexpert-Ghys J, Drouet C (2010) Preparation and physicochemical characteristics of luminescent apatite-based colloids. Journal of Physical Chemistry C 114(7): 2918-2924.
- Hosseini SM, Drouet C, Al-Kattan A, Navrotsky A (2014) Energetics of lanthanide-doped calcium phosphate apatite. American Mineralogist 99(11-12): 2320-2327.
- Vandecandelaère N (2012) Élaboration et caractérisation de biomatériaux osseux innovants à base d'apatites phospho-calciques dopé France, pp. 1-400.
- Adamiano A, Iafisco M, Sandri M, Basini M, Arosio P (2018) On the use of superparamagnetic hydroxyapatite nanoparticles as an agent for magnetic and nuclear in vivo Acta Biomaterialia 73: 458-469.
- Iannotti V, Adamiano A, Ausanio G, Lanotte L, Aquilanti G (2017) Fe-Doping-Induced Magnetism in Nano-Hydroxyapatites. Inorganic Chemistry 56(8): 4446-4458.
- Al-kattan A, Dufour P, Drouet C (2011) Purification of biomimetic apatite-based hybrid colloids intended for biomedical applications: A dialysis study. Colloids and Surfaces B: Biointerfaces 82(2): 378-384.
- Le Trequesser Q, Deves G, Saez G, Daudin L (2014) Single cell in situ detection and quantification of metal oxide nanoparticles using multimodal correlative microscopy. Anal Chem 86(15): 7311-7319.
- Simon M, Saez G, Muggiolu G, Lavenas M, Le Trequesser Q (2017) In situ quantification of diverse titanium dioxide nanoparticles unveils selective endoplasmic reticulum stress-dependent toxicity. Nanotoxicology 11(1): 134-145.
- Muggiolu G, Simon M, Lampe N, Deves G, Barberet P (2018) In Situ Detection and Single Cell Quantification of Metal Oxide Nanoparticles Using Nuclear Microprobe Analysis. J Vis Exp (132).






























