CdS Nanocrystalline Structured deposited on Al substrates
AMA Jafarov*, EF Nasirov and SA Jahangirova
Baku State University, Baku, Azerbaijan
Submitted: April 04, 2018; Published: April 30, 2018
*Corresponding author: MA Jafarov, Baku State University, Baku, Azerbaijan, Email: maarif.jafarov@mail.ru
How to cite this article: MA Jafarov, EF Nasirov ,SA Jahangirova.CdS Nanocrystalline Structured deposited on Al substrates. JOJ Mater Sci. 2018; 4(4): 555641. DOI: 10.19080/JOJMS.2018.04.555641.
Abbreviations
Abbreviations: DI: Digital Instruments; AFM: Atomic Force Microscopy
Mini Review
II-VI type of nano Crystal is extensively studied for their optoelectronic, photochemical, and nonlinear optical properties [1]. Semiconductor nano crystals, like CdS, show size dependent optical properties and are very important in basic nano science research, colloidal science, biomedical labeling, light emitting diodes, solar cells, and lasers [2]. In general, semiconductor nano crystals show novel optical and electronic properties when they have size comparable to, or smaller than, the dimensions of the exciton within their corresponding bulk material. This allows us to create unique properties for the nano crystal by engineering the size and composition and the chemical functionality of their surrounding medium. The techniques and capability of this manipulation of the size is an important way to create desired nano crystals [3]. The most common semiconductor materials grown by precipitation technique are CdS of which CdTe has special technological importance because it is the only known II-VI material that can form conventional p-n junctions. The chemical nature of the initial substances aswell as the preparation conditions determines the properties of the films. By chemical deposition, the nucleation of seeds and their growth in a solution lead to the formation of nano crystallites of almost spherical shape, while the deposition of films by molecular epitaxy or electrochemical deposition give nano crystallites of a non spherical shape.
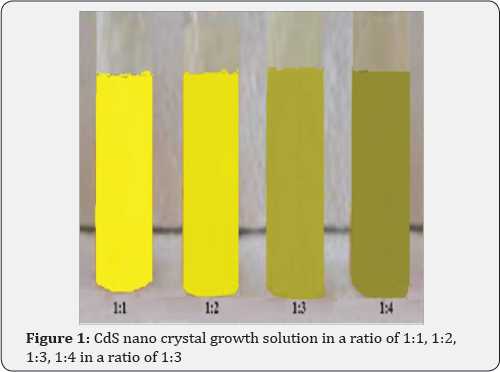
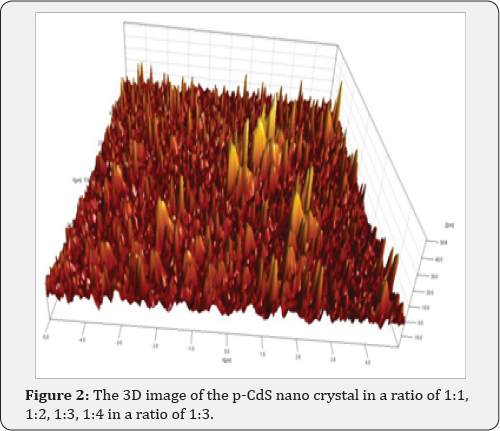
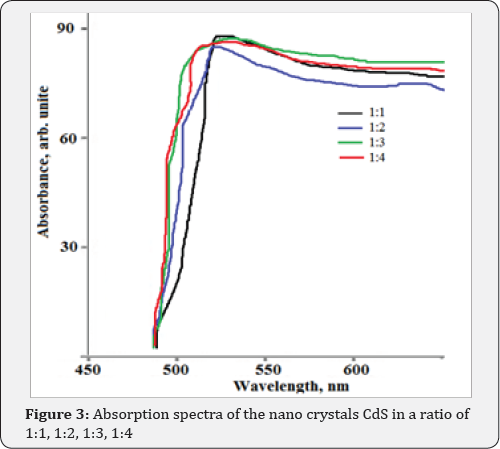
The deposition of a film from a solution allows us to control the process of film growth. Depending on the conditions under which the process is carried out, we can obtain polycrystalline or smooth solid films. Films and nanocrystalline samples of p-CdS were obtained on aluminum substrates by chemical deposition from a solution containing cadmium chloride (0.05M CdCl2), thiourea (0.15M (NH2)2CS) in a ratio of 1:3, and a complexing agent (20mL NH4OH) at a temperature of 80 °C for 20-45 min. The supernatant liquid - p-CdS nano crystals dispersed was separated and characterized for absorption, photoluminescence, substrate surface and particle size determination using AFM. CdS nano crystal solutions in a ratio of 1:1, 1:2, 1:3, 1:4 shown in Figure 1, were used to obtain the absorption spectra using a Perkin Elmer UV-visible Lambda 19 spectrometer. All luminescence measurements were acquired by using a Renishaw in Via Reflex confocal AFM scanning of the nano crystals on silicon was done with Digital Instruments (DI) tapping mode technique. 3D image of the p-CdSnano crystal growth solution is presented in Figure 2. One can clearly see the change in color indicative of the difference in the size of particles from left to right. In our case, the size difference was the primary concern. Immediately after injection, the color of the solution started changing from greenish yellow to light orange then to deep orange and finally grayish black to black over a period of one hour. This is a clear indication of the gradual growth of different size (and different band gap) CdS nano crystals as a function of the growth time. The grayish black and black solutions are not included in the picture. As the time changed, the particle size changed and correspondingly the band gap also changed. The size change continued for an hour where it reached the saturation point.
Surface morphology of CdS nano crystals was determined by atomic force microscopy. Sample preparation with drop casting always resulted in piling up of the nano crystals and did not result in uniform thin film. This is usually the case with drop casting and to avoid piling up and to obtain uniform film spin casting method needed to be adopted. We tried extensively to produce close packed continuous nano crystalline thin film. Spin casting was tried using several dilutions of nano crystals in chloroform with different rotation speeds and time. In many cases we ended up with islands of mono layer of nano crystalline film with spots of 3-D aggregates over the entire substrate surface. This type of deposition occurs always when nano crystalline film is deposited on solid substrate surfaces like silicon. But for our AFM study both drop casted and spun samples were used. CdS nano crystalline film was formed on Alsubstrate either by spin casting or drop casting. In a special case, the nano crystals grown at 45 minutes were initially drop casted on Al substrate and studied by Atomic Force Microscopy (AFM) and the AFM pictures are presented in Figure 3. As can be seen, the AFM image the particle height is 4.2nm and the particles are distributed unevenly. The following is the bearing chart which characterizes the relative roughness of the surface in terms of high and low areas.
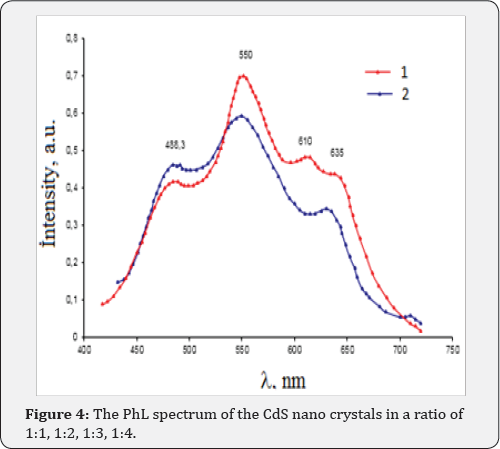
Figure 2 shows the absorption spectra of the nano crystals grown at various times. As can be seen, there is a shift of absorption peak as well as the band gap as the time of growth changes. Clearly the sixty minute growth sample indicates the saturation in terms of the peak position as well as the band gap. The absorption maxima are at 521, 480, 430, and 403nm respectively corresponding to band gaps of 2.58, 2.61, 2.67 and 2.72. It appears that nano crystal growth size saturates out between 45 and 60 minutes indicating no further shift in the peak neither height nor band gap change. (Figure 3) The luminescence spectrum of the CdS nano crystals are presented in Figure 4. In the case of the luminescence of the nano crystals CdS, in a ratio of 1:3 (1) and 1:4 (2) growth sample. Since this is similar to the previous results, the time dependent growth also becomes an attractive way of growth for the large-scale synthesis of nano crystals.
References
- Castillo Alvarado FL, Inoue Chávez JA, Vigil Gálan O, Sánchez Meza E, Löpez Chávezb E, et al. (2010) Thin Solid Films 518: 1796.
- Rios Flores A, Pena JL, Castro Pena V, Ares O, Castro Rodriguez R, et al. (2010) Sol Energy 84: 1020.
- J Han, C Spanheimer, G Haind, G Fu, V Krishnakumar, et al. (2011) Sol Energ Mat Sol C 95: 816.






























