Development of Diffusion Models of Solid-Phase Transformations in Iron-Carbon Alloys
Sergij Bobyr*
Doctor of Engineering Sciences, Ukraine
Submission: March 30, 2018; Published: April 10, 2018
*Corresponding author: Sergij Bobyr, Doctor of Engineering Sciences, Ukraine, Email: svbobyr07@gmail.com
How to cite this article: Sergij B.Development of Diffusion Models of Solid-Phase Transformations in Iron-Carbon Alloys. JOJ Mater Sci. 2018; 4(3): 555636. DOI: 10.19080/JOJMS.2018.04.555636
Introduction
The development of metal physics and physical materials science in the coming decades is one of the main driving forces of the new scientific and technological revolution, which will cause significant changes in many sectors of industry and energy, construction and agriculture, military equipment and everyday life. The aim of the article is to describing new diffusion models of solid-phase transformations in iron-carbon alloys using the principles of non-equilibrium thermodynamics and their application to the solution of practical problems of metal science and heat treatment of alloys.
Diffusion Models Eutectoid Transformation of Austenite
One of the most important diffusion transformations in iron-carbon alloys is the eutectoid transformation of austenite. Development of theoretical bases eutectoid transformation of austenite has been made in the work of C. Zener, Hillert and Kahn, R. F. Mehl and W. C. Hagele, B. J. Ljubov in the 40th - 60th years of the 20th century and the experimental results given in the classical works of L. I. Mirkin, R. F. Mehl, Turnbull, J. W. Christian and G. V. Kurdyumov [1-11]. These studies have made an important contribution to the development of a new branch of knowledge - physics of metals. Zener suggested ratio for between - plates distance as a function of the steel overcooling value S0 ~ ΔT-1 [2]. Experimental results show, however, that the measured values of the distance between the plates of perlite significantly more of those values, which gives the Zener model [4,5,9]. An important kinetic characteristic of the process of transformation of austenite is the rate of growth of perlite v. The value of growth rate was obtained by Mail in the form: v = KS0-1, where S0 is the between - plates distance; K - coefficient of proportionality, for which location you need to solve a differential equation of the stationary growth of perlite [4,7,12].
Kahn found that in such models it is impossible to reach an equilibrium redistribution of the components in the perlite, however it is possible to calculate the growth rate and degree of redistribution of the components if the distance between the plates. This requires another physical principle, to lock the distance. In the author's works as this principle was used the law of energy conservation (balance of heat) during the transformation of austenite and the diffusion of the carbon balance [13-15]. The result is a characteristic equation system second degree relative thickness of perlite Δ=S0/2, whose solution is immediately possible to find the dependence for A and for the growth rate of perlite:
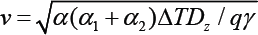
It is significantly different from that obtained by the Zener expression for the growth rate of perlite based on considerations of dimensionality v~ ΔT2exp(-Q/RT) Pirinciples:
The author found theoretically the temperature dependence between - plates distance of perlite on the magnitude of
supercooling of the alloy in the form of 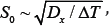 the corresponding experimental data [15].
the corresponding experimental data [15].
In works [16,17] have developed a diffusion model of transformation of austenite in iron-carbon alloy of eutectoid composition that takes into account the amount of energy supplies-under study on the formation of interfacial boundaries between the α -phase - cementite. As a result of the theoretical analysis of the obtained characteristic equation of the system of the third degree relative to the thickness of the perlite. Solving the author found that in this system the occurrence of two diffusional transformations of austenite: one with between - plates distance close to Δo - pearlite transformation. The second transformation is of the diffusion paths of diffusion close to the Δm (the characteristic size of the interfacial diffusion distance], and can be treated as the bainite transformation. In [16,17] has calculated the dependence of the number of the pearlite and bainitic structure in the alloy on the degree of overcooling, the calculated data with a sufficient degree of accuracy correspond to the experimental results.
Based on these results, the authors [20] concluded that the perlite and upper bainite formed as a result of intermittent diffusion of austenite. Lower bainite is a structural component in which the intermittent transformation of austenite is suppressed, and the diffusion of carbon occurs at a distance of less than Am is the characteristic size of the interfacial diffusion distances, the value of which the authors found theoretically. Further development of this model allowed the author to describe theoretically the transformation of austenite in iron-carbon alloys, the composition of which differs from the eutectoid. In [18] were found theoretical dependence of the kinetic parameters of the velocity-velocity of growth v ferrite and ferrite particle size DO on the magi v~ ΔT2exp(-Q/RT) steel
In [19], the authors theoretically estimated the amount and size of the structural components of ferrite and bainitic (acicular) ferrite formed during super cooling of low-carbon steel have shown their conformity with experimental data. The authors developed a diffusion model of discontinuous transformation of austenite is made possible theoretically to explain joint formation and bainitic ferrite in low carbon steel in the same temperature range [20].
New Diffusion Models of Solid-Phase Transformations in Iron-Carbon Alloys with the Use of the Principle of Non-Equilibrium Thermodynamics
When we consider the processes in the solid state should be taken into account that transfer of atoms of alloying elements and impurities cannot be considered as independent (stand-alone). Diffusion of the atom of the alloying element or impurities in the matrix alloy causes changes in the arrangement of the other atoms of the crystal lattice and their corresponding impact on the movement of the atom itself
In conventional thermodynamic theory, we study the conditions of equilibrium of different phases that can exist in this system, each phase is considered a homogeneous composition mutual location and energy state of atoms. However, the actual alloy is polycrystalline and contain defects of various types. One of the main mechanisms of diffusion in solids is a vacancy mechanism, taking into account the presence of lattice defects -vacancies [21,22]. Nonequilibrium thermodynamics provides equations of motion (diffusion) of atoms based on their interaction and in the presence of other gradients, in addition to the chemical potential [23-25]. The complexity of using these equations is that, first, it is often difficult to define thermodynamic driving forces; second, cross the unknown coefficients in the equations of motion.
However, for multicomponent diffusion systems there is the possibility of finding as the values of the thermodynamic forces - also as discontinuous [26-29]; and cross-ratios through the use of special variational procedure [28, 29]. It opens the way for the solution of diffusion problems in multi component system, which are alloyed iron-carbon alloys. Developed a diffusion model of solid-phase transformations was used to control the structure formation of iron-carbon alloys [30] and optimization of their thermal treatment; detection of conditions of formation of dispersed (nano) particles of carbides in some of the investigated alloys and possibilities of their transformation [29]. Application of the developed models of phase transformations for the analysis of the structure and properties of rolling rolls is given in the works [31,32].
Further Research Directions
With using the principles of nonequilibrium thermodynamics can be developed new non-equilibrium model of solid-phase transformations iron-carbon alloys: annealing, tempering, martensitic transformation of austenite; will be developed a technique for calculating values of the thermodynamic forces in iron-carbon alloys; will set the conditions for the formation of dispersed (nano) particles of carbides in some of the investigated alloys and possibilities of their transformation.
References
- GV Kurdiumov, LM Utevskyi, RY Entyn (1977) Prevrashchenyia v zheleze y staly.
- C Zener (1946) Trans. AIME 167.
- B Ia Liubov (1969) Kynetycheskaia teoryia fazovykh prevrashchenyi.
- RF Meil, UK Hagel (1960) Uspekhy fyzyky metallov T III.
- Y L Myrkyn (1941) Trudy Moskovskoho ynstytuta staly, sb Kh VIII.
- FC Hull, RF Mehl (1942) Trans. Amer Soc Metals 30.
- RF Mehl (1938) Hardenability of Alloy Steels. Amer Soc Metals, Cleveland, Ohio.
- M. Hillert (1957) Jerncontorets Ann 141.
- JU Krystyan (1968) Fyzycheskoe metallovedenye. Fazovye prevrashchenyia, Vyp 2.
- D Turnball (1955) Acta Met 3:43.
- JE Burke, D. Tarnball (1956) Uspekhy fyzyky metallov, Vyp 1.
- WS Hagel, GM Pound a (1956) RF Mehl. Acta Metallurgica 4(1):37.
- SV Bobyr, Stroytelstvo (2003) materyalovedenye y mashynostroenye: Sb nauch tr 22(2): 60.
- SV Bobyr, Stroytelstvo (2004) materyalovedenye y mashynostroenye: Sb nauch tr 26(1): 363.
- VI Bolshakov, SV Bobyr (2004) MyTOM, p. 8.
- VI Bolshakov, SV Bobyr (2005) Metaloznavstvo ta termichna obrobka metaliv: Naukov. ta inform biul PDABA 2:27.
- SV Bobyr (2006) Fundamentalnye y prykladnye problemy chernoi metallurhyy. Sb nauchnykh trudov 13: 241.
- SV Bobyr (2QQ8) Fundamentalnye y prykladnye problemy chernoi metallurhyy. Sb nauchnykh trudov 18: 257.
- SV Bobyr, VI Bolshakov (2012) Novi materialy i tekhnolohii v metalurhii ta mashynobuduvanni 1:21.
- SV Bobyr, VY Bolshakov (2014) Uspekhy fyzyky metallov 15(3): 123.
- BS Bokshteyn (1978) Diffuziya v metallah.
- MA Krishtal (1972) Mehanizm diffuzii v zheleznyh splavah.
- L Onsager (1931) Phys. Rev 37: 405.
- I Prigozhin (1960) Vvedenie v termodinamiku neobratimyh processov.
- S de Groot, P Mazur (1964) Neravnovesnaya termodinamika.
- AA Zhukov, RL Snezhnoy (1966) Diffuzionnye processy v metallah. Naukova dum ka, Kiev.
- MA Krishtal, AA Zhukov, RL Snezhnoy, EG Titenskiy (1971) Termodinamika, fizicheskaya kinetika strukturoobrazovaniya i svoystva chuguna i stali. Vyp 4.
- SV Bobyr(2013) Metallofiz. noveyshie tehnol 35(2): 199.
- SV Bobyr (2014) Metallofiz. noveyshie tehnol 36(10): 1385.
- SV Bobyr, VI Bolshakov, SA Sharfnadel (2016) Material science, Nonequilibrium phase transformations. Sofia, Bulgaria 4: 30.
- SV Bobyr, PV Krot, DV Loschkarev, MO Dedik, SA Sharfnadel (2017) Materials Science. Non-equilibrium phase transformations. Sofia, Bulgaria Is 4: 112.
- PV Krot, SV Bobyr, MO Dedik, NV Biba (2017) Int J of Microstructure and Materials Properties.






























