Properties Enhancement of AISI 1045 Steel by Rapid Al and Si Codeposition Assisted by DCF
Lei Tang1,2, Fei Xie1,2, Xianbo Pan1 and Jing Hu1,2*
1NationaI Experimental Demonstration Center for Materials Science and Engineering, Changzhou University, China
2Jiangsu Key Laboratory of Materials Surface Science and Technology, Changzhou University, China
Submission: December 19, 2017; Published: January 08, 2018
*Corresponding author: Jing Hu, Jiangsu Key Laboratory of Materials Surface Science and Technology, Changzhou University, Changzhou 213164, China, Email: jinghoo@126.com
How to cite this article: Lei T, Fei X, Xianbo P, Jing H. Properties Enhancement of AISI 1045 Steel by Rapid Al and Si Codeposition Assisted by DCF . JOJ Material Sci. 2017; 3(4): 555619. DOI: 10.19080/JOJMS.2018.03.555619
Abstract
A rapid powder Al and Si codeposition was conducted for AISI 1045 steel by adding direct current field (DCF). The high temperature oxidation resistance of the samples was evaluated in air at 800 OC by measuring and calculating the weight change, and the corrosion resistance was evaluated by immersing specimens in 10% H2SO4 solution. The results demonstrated that the rapidly formed coating is thermally and chemically stable, and thus can provide effective protection against high temperature oxidation at 800 0C and against corrosion in H2SO4 solution.
Keywords: AISI 1045 steel; Codeposition; Corrosion; Oxidation
Introduction
Powder aluminizing and siliconizing cementation is a kind of powder chemical thermal-diffusion technology, which has been used for improving the oxidation and corrosion resistance of the base materials in the application [1-3]. With the improvement of surface science & technology, single powder aluminizing or powder siliconizing can't meet the advanced requirements in some status; therefore, Al and Si codeposition has been widely explored [4-6].
Generally, Al and Si codeposition was performed at temperatures above 900 0C due to slow diffusion of Al and Si at lower temperature. Unfortunately, the treatment at so high temperature inevitably brings out some side effects to the base materials [5-8]. Therefore, reducing the heating temperature is of significant value. Our previous research found that direct current field (DCF) has significant effect on reducing the heating temperature in powder-pack boriding, powder-pack aluminizing and siliconizing cementation [9-11]. In this study, DCF was applied in powder Al and Si codeposition, and the goal is to further investigate the properties of AISI 1045 steel modified by rapid Al and Si codeposition assisted by DCF.
Experimental
The material used in this study was AISI 1045 steel with composition given in Table 1. The specimens were cut with the size of ®10mm*5mm and then degreased and polished progressively by hand finishing, the specimens were then ultrasonically cleaned in an ethanol bath for 5 minutes, dried prior to pack cementation process. The pack powders (wt.%) were comprised of 30% FeAl as Al source, 20% FeSi as Si source, 48% quartzite as inert filler to prevent pack from sintering and 2% NH4Cl as halide activator. The pack powders were dried in the oven at 100 °C for 1h before packing.

Schematic diagram of the main part of the experimental apparatus was shown in Figure 1. In the process of powder Al and Si codeposition assisted by DCF, two electrodes with a distance of 10 mm were buried in pack powders charged into a pot, which was then sealed with a lid by fire clay mortar to prevent access gas from expelling when it was heated to high temperatures. The pot was cured in a furnace for about 1h at approximately 100 °C in order to take out the remaining moisture. The pot was then placed in an electric furnace and applying direct current field at 700 °C and 750 °C for 4h. After 4h heating, the pot was left in the furnace to cool to room temperature by switching off the power supply. For comparison, a reference sample which was not connected to the circuit was also placed in the container as shown in Figure 2.

CK-40M OLYMPUS optical microscope was employed for observing the cross-sectional microstructure and investigating the coatings thickness. High-temperature oxidation tests of the samples were carried out in air at 800 °C for 120h, the weights were measured to 0.0001 g every 10h. Corrosion resistance tests were carried out by immersing the specimens in 10% H2SO4 solution for 8 h, and the weights were measured to 0.0001g every 1h.
Results and Discussion
Cross-sectional microstructure and coatings thickness
The Cross-sectional microstructures of AISI 1045 steel after powder Al and Si codeposition at different conditions are shown in Figure 2. It can be clearly seen that the coating thickness increased almost twice assisted by DCF when the furnace temperature was the same as shown in (a) and (b), and comparing (b) with (c), it is clear that the coating treated at 700°C with DCF was obviously thicker than that treated at 750 °C without DCF, which means that DCF can significantly lower treating temperature or duration.
High-temperature oxidation resistance
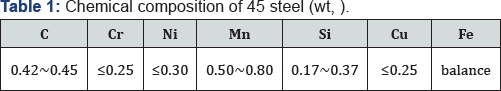
Figure 3 shows the high-temperature oxidation kinetics of AISI 1045 samples as-received and treated by powder Al and Si codeposition with DCF, and the oxidation temperature was 800 C. It illustrates that the rapidly obtained coatings assisted by DCF could greatly improve the high temperature oxidation resistance, and thus effectively protect the substrate against high-temperature oxidation.
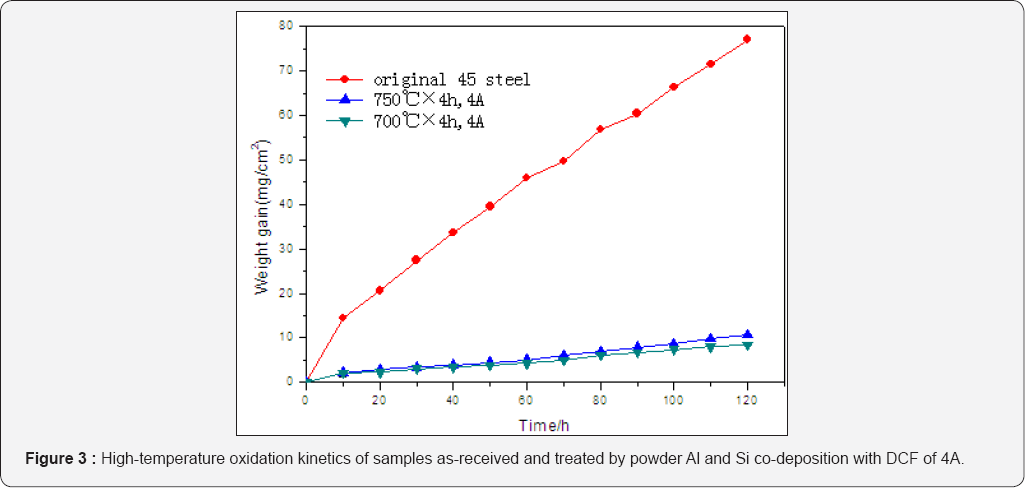
Corrosion resistance
Figure 4 shows the corrosion kinetics of specimens as- received and treated by powder Al and Si codeposition with DCF, which presents that the corrosion resistance of the treated specimen was greatly improved, in other words, the coating can effectively prevent AISI 1045 steel from corrosion in 10% H2SO4 solution.
Discussions
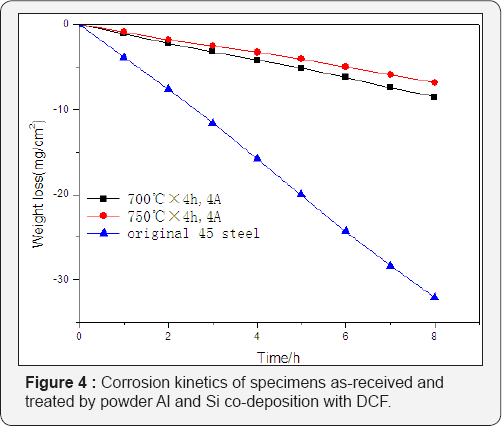
The coating thickness increases almost twice assisted by DCF when the furnace temperature, agents contents and holding time are the same as shown in Figure 1, which means that DCF has significant accelerating effect on the diffusion of active atoms. The schematic DCF enhancement mechanism in powder Al and Si codeposition process is shown in Figure 5 .
Firstly, DCF has thermal effect on agents, cathode and anode specimens which can promote the decomposition of FeAl and FeSi agents, thus increase the concentration of the active Al and Si atoms in the agents. More importantly, the active Al and Si atoms produced are positively charged by DCF and forced to diffuse directionally toward the cathode specimen. This directional diffusion is obviously faster than that in random, and it can also reduce the absorption of the active Al and Si atoms by the inner wall of crucible and the non-working surface of component.
Therefore, the concentrations of the active atoms around the surface of the specimen (cathode) can be much higher than that at any other positions, and thus the usage efficiency of the active atoms in the agents is significantly improved, hence promoting the formation of coating layer and resulting in thicker depth. Figure 5 also clearly presents the non-uniform distribution of the active atoms in the agents, especially with much higher concentration of Al atoms around the facing anode surface of the specimen due to the enhanced decomposition of agents and rapid directional diffusion to cathode assisted by DCF.
Conclusion
1. DCF can significantly lower treating temperature or duration comparing with the conventional Al and Si pack cementation processes.
2. The Al and Si codeposition coatings rapidly formed by DCF have excellent oxidation resistance at 800 °C
3. The Al and Si codeposition coatings rapidly formed by DCF have good corrosion resistance in 10% H2SO4 solution.
Acknowledgement
The research was supported by National Natural Science Foundation of China (51774052), Top-notch Academic Programs Project of Jiangsu Higher Education Institutions (TAPP), and Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
References
- Sakidja R, Rioult F, Werner J (2006) Aluminum pack cementation of Mo-Si-B alloys. Scripta Materialia 55(10): 903-906.
- Bai CY, Luo YJ, Koo CH (2004) Improvement of high temperature oxidation and corrosion resistance of superalloy IN-738LC by pack cementation. Surface and Coatings Technology 183(1): 74-88.
- Majumdar S, Sharma IG, Suri AK (2008) Develpoment of oxidation resistance coatings on Mo-30W alloy. International Journal of Refractory Metals & Hard Materials 26(6): 549-554.
- Xiang ZD, Datta PK (2003) Codeposition of Al and Si on nickel base superalloys by pack cementation process. Materials Science and Engineering: A 356(1-2):136-144.
- Majumdar S, Sharma IG, Raveendra S, Samajdar I, Bhargava P (2008) In situ chemical vapour co-deposition of Al and Si to form diffusion coatings on TZM. Materials Science and Engineering : A 492(1-2): 211217.
- Gruters J, Galetz MC (2015) Influence of thermodynamic activities of different masteralloys in pack powder mixtures to produce low activity aluminide coatings on TiAl alloys. Intermetallics 60: 19-27.
- Wang CJ, Chen SM (2006) The high-temperature oxidation behavior of hot-dipping Al-Si coating on low carbon steel. Surface & Coatings Technology 200(22-23): 6601-6605.
- Erdeniz D, Dun DC (2014) Microstructure development during pack aluminization of nickel and nickel-chromium wires. Intermetallics 50: 43-53.
- Zhan ZL, He YD, Wang DR, Gao W (2006) Low-temperature processin Fe-Al intermetallic coatings assisted by ball milling. Intermetallics 14(1): 75-81.
- Vishwanadh B, Majumdar S, Orsborn J, Banerjee R, Tewari R, et al. (2015) Characterization of silicide phases formed during pack siliconizing coating on the Nb-1Zr-0.1C alloy. Intermetallics 63: 59-66.
- Zhou Z H, Xie F, Hu J (2008) A novel powder aluminizing technology assisted by direct current field at low temperatures. Surface & Coatings Technology 203(1-2): 23-27.






























