Bulk Metal Alloys as a Precursor of Dispersed Particles Catalyzing Growth of Carbon Nanofibers
Aleksey A Vedyagin1,2*, Ilya V Mishakov1,2 and Yurii I Bauman1
1Boreskov Institute of Catalysis SB RAS, Novosibirsk, Russia 2National Research Tomsk Polytechnic University, Russia
Submission: September 01, 2017; Published: September 11, 2017
*Corresponding author: Boreskov Institute of Catalysis SB RAS, Novosibirsk, Russia, National Research Tomsk Polytechnic University, Tomsk, Russian Federation, Russia, Email: vedyagin@catalysis.ru
How to cite this article: Vedyagin AA, Mishakov IV, Bauman YI. Bulk Metal Alloys as a Precursor of Dispersed Particles Catalyzing Growth of Carbon Nanofibers. JOJ Material Sci. 2017; 2(4): 555594. DOI:10.19080/JOJMS.2017.02.555594
Abstract
Bulk metal alloys can be considered as a precursor of self-organized catalysts consisting of dispersed metal particles fixed within the structure of carbon nanofibers, which were grown as a result of catalytic chemical vapor deposition of halogen-substituted hydrocarbons. Being subjected to interaction with aggressive reaction mixture, the bulk alloy precursors undergo relatively fast process of metal dusting, which ends with complete wastage of initial metal item. The catalysts thus obtained are characterized with excellent long-term stability of operation along with high activity in decomposition of hydrocarbons of different nature. The approach described opens new horizons for a synthesis of nanostructured carbon materials including carbon-carbon composites of bimodal structure.
Keywords: Bulk metal alloys; Metal dusting; Self-disintegration; Carbon nanofibers; Catalytic chemical vapor deposition
Introduction
Industrially manufactured bulk metal alloys mostly being the resistive materials are widely applied in the heating systems (as a heating element), in the explosives and fireworks industry (as a bridge wire in igniters), in ceramic (as an internal support structure), etc. These materials are quite stable under both oxidative and reductive atmospheres, but undergo fast self-disintegration if exposed to carbon-containing (carburizing) medium at elevated temperature. The process of self-disintegration of metal items, also known as a metal dusting, is considered as an actual problem of modern chemical industry, which results in a wastage of chemical reactors [1-9]. Thus, steel reactors operated at 400-800 °C during hundreds hours become almost completely disintegrated into a dust consisting of dispersed metal particles and carbon. The situation becomes more complicated if the aggressive medium of halogen-substituted hydrocarbons is used [10,11].
There are a lot of papers devoted to the study on the mechanism of the metal dusting [1,3,6,7,9]. According to Grabke [1], the process goes through the following stages:
- Decomposition of carbon-containing precursor with subsequent transfer of carbon into solid solution up to oversaturation.
- Nucleation of metal carbide phase at surface and grain boundaries and its growth with protrusions into the bulk metal phase.
- Nucleation of the graphite phase.
- Decomposition of carbide to metal and graphite. Nano particle formed plays a role of catalytic sites for carbon nanofibers (or nanotubes) growth.
Formed catalytic sites (dispersed metallic or alloyed particles), being stabilized within the structure of carbon nanofibers, catalyze further formation of nanostructured carbon via catalytic chemical vapor deposition. Thereby, the process of self-disintegration (metal dusting) can be proposed as a novel approach to prepare dispersed metal particles from bulk metal precursors for the synthesis of nanostructured carbon [12-17].
Discussion
During the last two decades the process of metal dusting as a way to obtain nanostructured carbon materials attracts more and more attention [12-24]. Carbon nanotubes and nanofibers of various morphologies were produced over stainless steel, iron- and nickel-containing alloys. In all these cases, the surfaceof bulk metals was intentionally subjected to controlled process of metal dusting yielding carbon deposits.
As it was already mentioned, the process of self-disintegration is characterized with prolong induction period (up to few hundred hours), when no visible changes take place. At the same time, if the halogenated hydrocarbon is used as a carbon source, the process accelerates significantly [10,11,25,26]. Thus, interaction of bulk nickel alloys (nichrome, chromel, alumel, etc.) with vapors of chlorinated hydrocarbons results in a fast metal dusting with induction period no longer than 3 hours at 550 °C. It should be noted that induction period shortens along with temperature increase. At the end of induction period, the weight of the sample sharply grows up, and the system became inverted into an ensemble of dispersed metal particles fixed within the structure of grown carbon nanofibers. These particles can be considered as self-organized catalyst, and can be applied for decomposition of any other carbon-containing substrates with formation of nanostructured carbon. Thereby it can be concluded that halogen contained in a molecule of substrate affects crucially the rate of metal dusting. HCl emerged in hydrogen-containing reaction mixture provides the occurrence of reversible chlorination-dechlorination process, which leads to quick chemical corrosion of bulk alloy surface. It should be also emphasize that the addition of hydrogen excess into reaction mixture is required for dechlorination of the alloy surface [10].
The duration of induction period of the self-disintegration process can be shortened by using the pretreatment procedures [25,26]. Among such procedures, the most efficient are etching in a mixture of mineral acids (HCl/HNO3=3/1, 2-3min) and redox activation (altering of oxidative and reductive treatment at 500 °C, 3 cycles, 90min).These methods make a rough reconstruction of the alloy surface that eases further formation of dispersed active particles.
While the considered alloys are resistive materials, they can be heated up to desired temperature by a direct supply of current. Decomposition of 1,2-dichloroethane in such regime is reported in Ref. [27]. In this case, besides the processes described above, hydrodechlorination and dechloro-coupling reactions take place leading to formation of ethane, ethylene and butenes within the gas phase products. At the resistive heating, the process of substituted hydrocarbon decomposition proceeds through the stages of C-H bond breakage, and hydrogenolysis and breakage of C-C and C-Cl bonds [28]. Unsaturated hydrocarbons and corresponding radical species formed as a result of hydrogenolysis undergo further pyrolysis over nickel crystallites.
Table 1 summaries the data on carbon yield resulted from catalytic chemical deposition of 1,2- dichloroethane during 2 hours over different industrially manufactured alloys. As seen, among the studied alloys, nichrome (Ni-80%, Cr-20%) occupies the leading position. It allows one to obtain the carbon yield of 67.5 gC/gAl, which surpasses the values for other samples in one order of magnitude. If the resistive heating is applied (Table 1, row #2), the carbon yield over the same type of nichrome decreases in approximately three times, which is connected, as it was mentioned above, with occurrence of parallel processes [29]. The lowest carbon yield (0.5gC/gAl) was observed in the case of stainless steel, thus indicating that 2 hours is not long enough time for such kind of bulk precursor.
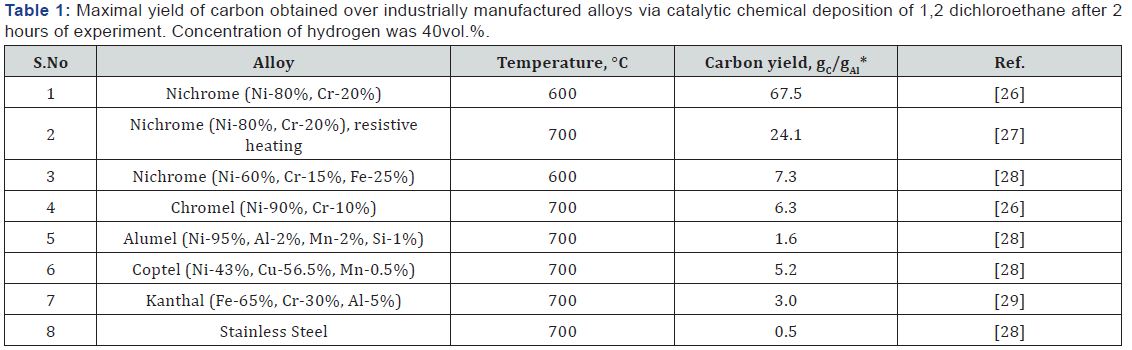
*gC -weight of carbon product; gAl -weight of alloy sample
Besides the industrially manufactured alloys, similar solid solutions of predefined composition can be especially prepared via multi-stage route involving coprecipitation of salt followed by high-temperature reduction in hydrogen, or via single-stage route of mechanical alloying of metal powders in a planetary mill [30-32]. An advantage of the first approach is a guaranteed formation of solid solution with desired ratio of the metals and homogeneous distribution. The formation of solid solutions was confirmed by precise powder X-ray diffraction analysis [33- 38]. Data on carbon yield over such alloys are shown in Table 2. As it follows, the nature and loading of an alloying metal affect noticeably the productivity of bulk precursor towards carbon. Thus, addition of iron worsens the catalytic behavior of the resulted alloy, while molybdenum, oppositely, significantly enhances it. It should be mentioned that these especially prepared alloy samples being subjected to complete self-disintegration process under aggressive medium of 1,2-dichloroethane can also be considered as self-organized catalyst for further usage in decomposition of various organic substrates [39].
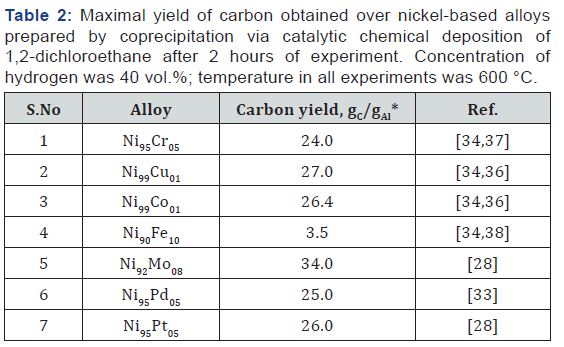
*gC–weight of carbon product; gAl–weight of alloy sample.
Self-organized catalysts obtained using both types of bulk precursors (industrially manufactured and especially prepared) were found to be quite active and extremely stable in catalytic decomposition of various hydrocarbons and their mixtures (C2H6, C2-C4 mix, C6H6) [40]. Additionally, it was shown that even chlorofluorocarbons can be efficiently decomposed, but in this case introduction of odd hydrogen into reaction feed is of great importance in order to bind the halogen atoms into HF and HCl. Surprisingly, the interaction of CF2Cl2 with submicron Ni crystals formed during the self-disintegration of bulk alloy precursor and fixed within the structure of carbon nanofibers causes their secondary disintegration which leads to formation of unique bimodal carbon nanomaterial.
All processes of catalytic chemical vapor deposition with formation of carbon nanofibers described above were considered within a concept of flow-through or ‘open’ systems, when the reagent(s) continuously inlets into the reaction volume and the gas phase products pass the volume away. On the other hand, application of so called ‘closed’ reaction volume system was recently shown to be perspective for synthesis of various nanostructured inorganic materials [41-46]. Regarding the decomposition of substituted hydrocarbons, the process has the same requirements as in the case of ‘open’ system: both halogen and hydrogen source substrates should present in a reaction volume. Thus, for example, the use of halogenated organics (hexafluorobenzene, hexachlorobenzene, 1-bromobutane 1-iodobutane, etc.) together with hexamethylbenzene (as an inner hydrogen source) was shown to initialize the metal dusting process of bulk NiCr alloy with formation of nanostructured product [46]. Depending on the nature of halogen-containing substrate, the starting temperature of active particles formation detected by means of a ferromagnetic resonance spectrometry was different. The approach additionally provides a possibility to study the metastable reaction intermediates, which is inaccessible in the flow-through regime.
Conclusion
Preparation of the catalysts for various catalytic processes remains to be a challenge task. At the same time, creation of the appropriate, in most cases aggressive, conditions for the catalyst’s precursor might shift the system towards state of self-organization. One of the most illustrative examples of this phenomenon is a nickel-based catalytic system for chemical vapor deposition of halogen-substituted hydrocarbons resulting in formation of nanostructured carbon fibers. Conventionally used for this process Ni-containing catalysts supported on oxide or carbonaceous supports are known to be rapidly deactivated under the reaction conditions. Oppositely, bulk metal alloys subjected to interaction with aggressive halogencontaining medium were shown to undergo the metal dusting process, which ends with complete wastage of initial bulk item. The dispersed metal particles formed as a result of such selfdisintegration were found to be quite stable and extremely active in decomposition of any other substituted and unsubstituted hydrocarbons that allows one to consider them as self-organized catalysts. The main regularities of the process are similar for the cases of external and direct (resistive) heating of the bulk alloy precursor, and if compare ‘open’ reaction volume systems with ‘closed’ one.
Acknowledgment
This work was supported by Russian Academy of Sciences and Federal Agency of Scientific Organizations (project number 0303-2016-0014).
References
- Grabke HJ (2003) Metal dusting. Mater Corros 54: 736-746.
- Aguero A, Gutierrez M, Korcakova L, Nguyen TTM, Hinnemann B, et al. (2011) Metal Dusting Protective Coatings. A Literature Review. Oxid Met 76(1-2): 23-42.
- Chun CM, Mumford JD, Ramanarayanan TA (2002) Mechanisms of metal dusting corrosion of iron. J Electro chem Soc 149(7): B348-B355.
- Zhang J, Young DJ (2007) Kinetics and mechanisms of nickel metal dusting I. Kinetics and morphology. Corros Sci 49(3): 1496-1512.
- Zhang Q, Han B, Heier K, Li JX, Hoffman J, et al. (2011) First principles study of steam carbon reaction on γ-Fe(111) surface. J Phys Chem C 115(24): 12068-12076.
- Szakalos P (2003) Mechanisms and driving forces of metal dusting. Mater Corros 54(10): 752-762.
- Young D (2008) High Temperature Oxidation and Corrosion of Metals. Elseviers. Corrosion Series. Amsterdam, Netherlands.
- Grabke HJ (1998) Thermodynamic Mechanisms and Kinetics of metal dusting. Mater Corros 49(5): 303-308.
- Szakalos P (2004) Mechanisms of metal dusting. KTH Doctoral thesis, Sweden, p. 81.
- Bauman YI, Mishakov IV, Buyanov RA, Vedyagin AA, Volodin AM (2011)Catalytic properties of massive iron-subgroup metals in dichloroethane decomposition into carbon products. Kinet Catal 52(4): 557-564.
- Mishakov IV, Bauman YI, Korneev DV, Vedyagin AA (2013) Metal dusting as a route to produce active catalyst for processing chlorinated hydrocarbons into carbon nanomaterials. Top Catal 56(11): 1026- 1032.
- Chang JK, Tsai HY, Tsai WT (2008) A metal dusting process for preparing nano-sized carbon materials and the effects of acid post-treatment on their hydrogen storage performance. Int J Hydrogen Energ 33: 6734- 6742.
- Chang CH, Tsai WT (2009) Carburization behavior under the pits induced by metal dusting in 304L and 347 stainless steels. Mater Chem Phys 116(2-3): 426-432.
- Chang JK, Tsai HY, Tsai WT (2008) Different types of nanosized carbon materials produced by a metal dusting process. J Phys Chem C 112(51): 20143-20148.
- Ghorbani H, Rashidi AM, Rastegari S, Mirdamadi S, Alaei M (2011) Mass production of multi-wall carbon nanotubes by metal dusting process with high yield. Mater Res Bull 46(5): 716-721.
- Nerushev OA, Novopashin SA, Smovzh DV (2008) Synthesis of carbon nanofibers on an austenitic stainless steel. Nanotechnol Russia 3(7-8): 464-469.
- Pacheco Benito S, Lefferts L (2010) The production of a homogeneous and well-attached layer of carbon nanofibers on metal foils. Carbon 48(10): 2862-2872.
- Camilli L, Scarselli M, Del Gobbo S, Castrucci P, Lamastra FR, et al. (2012) High coercivity of iron-filled carbon nanotubes synthesized on austenitic stainless steel. Carbon 50(2): 718-721.
- Hashempour M, Vicenzo A, Zhao F, Bestetti M (2013) Direct growth of MWCNTs on 316 stainless steel by chemical vapor deposition: Effect of surface nano-features on CNT growth and structure. Carbon 63: 330- 347.
- Latorre N, Cazana F, Sebastian V, Royo C, Romeo E, et al. (2016) Growth of carbonaceous nanomaterials over stainless steel foams. Effect of activation temperature. Catal Today 273: 41-49.
- He M, Fedotov PV, Obraztsova ED, Viitanen V, Sainio J, et al. (2012) Chiral-selective growth of single-walled carbon nanotubes on stainless steel wires. Carbon 50(11): 4291-4301.
- Romero P, Oro R, Campos M, Torralba JM, de Villoria RG (2015) Simultaneous synthesis of vertically aligned carbon nanotubes and amorphous carbon thin films on stainless steel. Carbon 82: 31-38.
- Vishwakarma R, Shinde SM, Rosmi MS, Takahashi C, Papon R, et al. (2016) Influence of oxygen on nitrogen-doped carbon nanofiber growth directly on nichrome foil. Nanotechnology 27(36): 365602.
- Buyanov RA, Mishakov IV, Vedyagin AA (2013) Carbon erosion of hardware made of iron subgroup metals and their alloys. Doklady Phys Chem 448(1): 11-13.
- Bauman YI, Kenzhin RM, Volodin AM, Mishakov IV, Vedyagin AA (2012) Formation of growth centres of carbon nanofibres during selfdispersing Ni-containing alloys: Studies by means of ferromagnetic resonance. Chem Sustain Develop 20: 119-127.
- Bauman YI, Mishakov IV, Vedyagin AA, Dmitriev SV, Mel’gunov MS, et al. (2012) Processing of organochlorine waste components on bulk metal catalysts. Catal Ind 4(4): 261-266.
- Bauman YI, Sigaeva SS, Mishakov IV, Vedyagin AA, Tsyrulnikov PG, et al. (2017) Pyrolysis of 1,2-dichloroethane over Ni–Cr catalyst at resistive heating. Reac Kinet Mech Catal 120(2): 691-701.
- Bauman YI, Vedyagin AA, Mishakov IV (2016) Carbon erosion of FeCrAl bulk alloy by chlorinated hydrocarbons. Protect Metal Phys Chem Surf 52(2): 309-315.
- Gholizadeh R, Shabestari SG (2011) Investigation of the effects of Ni, Fe, and Mn on the formation of complex intermetallic compounds in Al- Si-Cu-Mg-Ni alloys. Metall Mater Trans A 42(11): 3447-3458.
- Reddy BSB, Das K, Das S (2007) A review on the synthesis of in situ aluminum based composites by thermal, mechanical and mechanical– thermal activation of chemical reactions. J Mater Sci 42(22): 9366- 9378.
- Mulas G, Delogu F, Pistidda C, Cocco G (2008) Mechanochemical effects on hydrogen absorption in Mg2Ni alloys under mechanical processing conditions. J Mater Sci 43(15): 5193-5198.
- Bauman YI, Shorstkaya YV, Mishakov IV, Plyusnin PE, Shubin YV, et al. (2017) Catalytic Conversion of 1,2-dichloroethane over Ni-Pd System into Filamentous Carbon Material. Catal Today 293-294: 23-32.
- Bauman YI, Mishakov IV, Vedyagin AA, Rudnev AV, Plyusnin PE, et al. (2017) Promoting effect of Co, Cu, Cr and Fe on activity of ni-based alloys in catalytic processing of chlorinated hydrocarbons. Top Catal 60(1-2): 171-177.
- Bauman YI, Mishakov IV, Korneev DV, Shubin YV, Vedyagin AA, et al. (2017) Comparative study of 1,2-dichlorethane decomposition over Ni-based catalysts with formation of filamentous carbon. Catal Today doi: 10.1016/j.cattod.2017.05.015.
- Rudnev AV, Lysakova AS, Plyusnin PE, Bauman YI, Shubin YV, et al. (2014) Ni-Cu and Ni-Co alloys: Synthesis, structure, and catalytic activity for the decomposition of chlorinated hydrocarbons. Inorg Mater 50(6): 566-571.
- Bauman YI, Lysakova AS, Rudnev AV, Mishakov IV, Shubin YV, et al. (2014) Synthesis of nanostructured carbon fibers from chlorohydrocarbons over bulk Ni-Cr alloys. Nanotechnol Russia 9(7-8): 380-385.
- Bauman YI, Kutaev NV, Plyusnin PE, Mishakov IV, Shubin YV, et al. (2017) Catalytic behavior of bimetallic Ni–Fe systems in the decomposition of 1,2-dichloroethane. Effect of iron doping and preparation route. Reac Kinet Mech Catal 121(2):413-423.
- Bauman YI, Mishakov IV, Vedyagin AA, Ramakrishna S (2017) Metal dusting of Ni-based alloys: new opportunities for design of selforganizing catalysts and synthesis of carbon nanomaterials. Mater Lett 201: 70-73.
- Streltsov IA, Bauman YI, Mishakov IV, Vedyagin AA, Buyanov RA (2012) A method to produce carbon nanofibers. RU Patent 2456234.
- Pol SV, Pol VG, Gedanken A (2004) Reactions under autogenic pressure at elevated temperature (RAPET) of various alkoxides: formation of metals/metal oxides-carbon core-shell structures. Chem Eur J 10(18): 4467-4473.
- Pol VG, Pol SV, Moreno JMC, Gedanken A (2006) High yield one-step synthesis of carbon spheres produced by dissociating individual hydrocarbons at their autogenic pressure at low temperatures. Carbon 44: 3285-3292.
- Gershi H, Gedanken A, Keppner H, Cohen H (2011) One-step synthesis of prolate spheroidal-shaped carbon produced by the thermolysis of octene under its autogenic pressure. Carbon 49(4): 1067-1074.
- Butovsky E, Perelshtein I, Nissan I, Gedanken A (2013) Fabrication, characterization, and printing of conductive ink based on multi coreshell nanoparticles synthesized by RAPET. Adv Funct Mater 23(46): 5794-5799.
- Teller H, Krichevski O, Gur M, Gedanken A, Schechter A (2015) Ruthenium phosphide synthesis and electroactivity towards oxygen reduction in acid solutions. ACS Catal 5(7): 4260-4267.
- Pol VG, Thiyagarajan P (2010) Remediating plastic waste into carbon nanotubes. J Environ Monit 12(2): 455-459.
- Kenzhin RM, Bauman YI, Volodin AM, Mishakov IV, Vedyagin AA (2016) Structural self-organization of solid-state products formed in reaction of halogenated compounds with bulk Ni-Cr alloy. Mater Lett 179: 30- 33.






























