Abstract
Atherosclerotic cardiovascular disease remains the leading cause of global morbidity and mortality despite major advances in lipid-lowering therapies. Although aggressive reduction of low-density lipoprotein cholesterol has substantially improved clinical outcomes, a significant proportion of patients continue to experience recurrent cardiovascular events. This persistent risk has led to the recognition of residual inflammatory risk, a concept describing ongoing vascular inflammation that is not adequately addressed by lipid-focused strategies alone. Atherosclerosis is now understood as a chronic inflammatory disease driven by complex interactions between lipids, immune cells, and inflammatory mediators. Innate and adaptive immune responses contribute to plaque initiation, progression, and destabilization, while inflammatory biomarkers such as high-sensitivity C-reactive protein and interleukin-6 provide clinically relevant measures of residual risk.
Importantly, recent clinical trials have demonstrated that selective targeting of inflammatory pathways can reduce cardiovascular events independently of lipid lowering, providing causal evidence for inflammation as a therapeutic target. This narrative review summarizes current understanding of the inflammatory pathophysiology of atherosclerosis, discusses established and emerging biomarkers of residual inflammatory risk, and critically evaluates pharmacological anti-inflammatory therapies, including colchicine and cytokine-targeted interventions. Finally, future directions and challenges related to patient selection, safety, and clinical integration of inflammation-targeted strategies are discussed, highlighting the potential role of precision medicine in optimizing cardiovascular risk reduction.
Keywords: Residual inflammatory risk; Atherosclerosis; Inflammation; Anti-inflammatory therapy; Cardiovascular disease
Abbreviations: ASCVD: Atherosclerotic Cardiovascular Disease; LDL-C: Low-Density Lipoprotein Cholesterol; OXLDL: Oxidized Low-Density Lipoprotein; HS-CRP: High-Sensitivity C-Reactive Protein; NLR: Neutrophil-To-Lymphocyte Ratio; SII: Systemic Immune-Inflammation Index; PCSK9: Proprotein Convertase Subtilisin/Kexin Type 9
Introduction
Atherosclerotic cardiovascular disease (ASCVD) remains the leading cause of morbidity and mortality worldwide despite major advances in lipid-lowering therapies and preventive strategies. For decades, atherosclerosis was primarily viewed as a disorder of cholesterol accumulation within the arterial wall. However, accumulating experimental and clinical evidence has firmly established atherosclerosis as a chronic inflammatory disease of the arterial intima, involving complex interactions between lipids, immune cells, and inflammatory mediators [1]. Endothelial dysfunction represents the earliest detectable stage of atherogenesis, promoting leukocyte adhesion, transendothelial migration, and subsequent activation of innate and adaptive immune responses. Monocyte-derived macrophages, T lymphocytes, and inflammatory cytokines play a central role in plaque initiation, progression, and destabilization [2].
These inflammatory processes contribute not only to plaque growth but also to thinning of the fibrous cap, rendering plaques vulnerable to rupture and thrombosis. The widespread use of statins and other lipid-lowering agents has led to substantial reductions in low-density lipoprotein cholesterol (LDL-C) levels and cardiovascular events. Nevertheless, a considerable proportion of patients continue to experience recurrent ischemic events despite achieving guideline-recommended LDL-C targets [3]. This phenomenon has led to the recognition of “residual risk,” which is increasingly understood to include a substantial inflammatory component independent of lipid burden. Highsensitivity C-reactive protein (hs-CRP) has emerged as the most extensively studied biomarker of systemic inflammation in ASCVD. Elevated hs-CRP levels are consistently associated with increased cardiovascular risk, even among individuals with well-controlled LDL-C levels [4].
Importantly, randomized clinical trials have demonstrated that selective inhibition of inflammatory pathways can reduce cardiovascular events without directly affecting lipid concentrations, thereby providing causal evidence for the inflammatory hypothesis of atherosclerosis [5]. These observations have shifted contemporary cardiovascular prevention paradigms toward a dual-target approach addressing both lipid-driven and inflammation-driven pathways. The present review aims to summarize the biological basis of inflammation in atherosclerosis, evaluate current evidence for inflammationtargeted therapies, and discuss future directions for integrating anti-inflammatory strategies into clinical cardiovascular care.
Inflammatory Pathophysiology of Atherosclerosis
Atherosclerosis is now recognized as a chronic, maladaptive inflammatory disease of the arterial wall that evolves over decades under the influence of both systemic risk factors and local vascular responses. Traditional cardiovascular risk factors, including hypercholesterolemia, diabetes mellitus, smoking, and hypertension, initiate endothelial dysfunction, which represents the earliest and most critical step in atherogenesis.
Dysfunctional endothelial cells lose their vasoprotective properties and acquire a pro-inflammatory phenotype characterized by increased permeability, reduced nitric oxide bioavailability, and enhanced expression of adhesion molecules and chemokines [6]. These endothelial changes facilitate the recruitment of circulating leukocytes into the subendothelial space, setting the stage for sustained vascular inflammation. Importantly, endothelial dysfunction is not merely a passive consequence of risk factor exposure but actively drives immune cell activation and retention within the arterial intima. The major inflammatory mechanisms, key cellular components, and their pathophysiological consequences throughout different stages of atherosclerosis are summarized in Table 1.

Abbreviations: ICAM-1, intercellular adhesion molecule-1; IFN-γ, interferon gamma; IL, interleukin; MCP-1, monocyte chemoattractant protein-1; M-CSF, macrophage colony-stimulating factor; NLRP3, NOD-like receptor family pyrin domain-containing 3; NO, nitric oxide; oxLDL, oxidized lowdensity lipoprotein; VCAM-1, vascular cell adhesion molecule-1.
Innate Immune Activation
The innate immune system constitutes the primary driver of inflammatory amplification in atherosclerosis. Circulating monocytes adhere to activated endothelial cells and migrate into the intima, where they differentiate into macrophages under the influence of macrophage colony-stimulating factors and other local signals. These macrophages avidly internalize modified lipoproteins, particularly oxidized low-density lipoprotein (oxLDL), through scavenger receptors, leading to the formation of foam cells, a hallmark of early atherosclerotic lesions [7]. As atherosclerosis progresses, macrophages exhibit remarkable functional heterogeneity.
Pro-inflammatory macrophage subsets produce cytokines, proteases, and reactive oxygen species that intensify local inflammation and promote extracellular matrix degradation. In contrast, impaired clearance of apoptotic cells (defective efferocytosis) results in secondary necrosis, expansion of the necrotic core, and further inflammatory activation. These processes contribute directly to plaque growth and vulnerability. A central mechanism linking lipid accumulation to innate immune activation is the engagement of pattern recognition receptors. Among these, the NLRP3 inflammatory cytokine production [8] has emerged as a key molecular platform integrating metabolic danger signals with inflammatory cytokine production [8].
Adaptive Immune Response
While innate immunity initiates and sustains inflammation, adaptive immune responses critically modulate plaque evolution and stability. CD4⁺ T lymphocytes are abundant within atherosclerotic plaques and predominantly display a pro-inflammatory phenotype. T helper 1 (TH1) cells secrete interferon-γ, which enhances macrophage activation, suppresses collagen synthesis by vascular smooth muscle cells, and thereby weakens the fibrous cap [9]. These effects link adaptive immunity directly to plaque destabilization and acute coronary events.
Conversely, regulatory T cells exert anti-inflammatory and plaque-stabilizing effects through the release of immunosuppressive cytokines and inhibition of effector T-cell responses. A relative deficiency or functional impairment of regulatory T cells has been associated with accelerated atherosclerosis, highlighting the importance of immune balance rather than absolute immune activation. B lymphocytes also contribute to atherosclerosis through both protective and pathogenic mechanisms. Certain B-cell subsets produce atheroprotective antibodies, whereas others promote inflammation via cytokine secretion and antigen presentation. This dual role underscores the complexity of adaptive immune regulation in vascular disease [10].
NLRP3 Inflammasome and Cytokine Cascade
Cholesterol crystals, which accumulate within advanced atherosclerotic plaques, act as potent activators of the NLRP3 inflammasome. Upon activation, this intracellular multiprotein complex triggers caspase-1–mediated cleavage of pro– interleukin-1β into its biologically active form. Interleukin-1β subsequently induces downstream production of interleukin-6, which stimulates hepatic synthesis of C-reactive protein, thereby linking local plaque inflammation to systemic inflammatory markers measurable in clinical practice [11].
This IL-1β–IL-6–CRP axis represents a central inflammatory pathway in atherosclerosis and provides a mechanistic explanation for the strong association between circulating inflammatory biomarkers and cardiovascular risk. Beyond biomarker elevation, activation of this cytokine cascade promotes endothelial dysfunction, enhances thrombogenicity, and accelerates fibrous cap degradation, ultimately increasing susceptibility to plaque rupture and thrombosis (Figure 1).
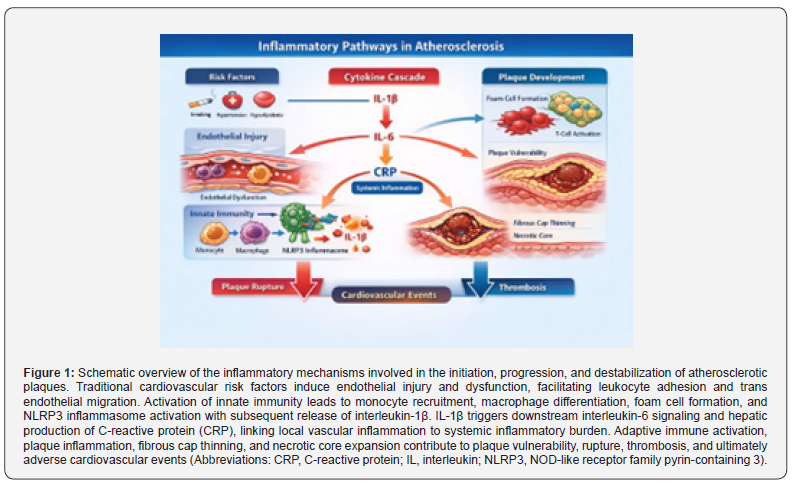
Biomarkers of Residual Inflammatory Risk
Despite optimal lipid-lowering therapy, a substantial proportion of patients with atherosclerotic cardiovascular disease continue to experience recurrent ischemic events. This observation has led to the concept of residual inflammatory risk, which reflects persistent vascular inflammation that is not adequately addressed by lipid-focused interventions alone. Biomarkers of inflammation provide a practical means of identifying this high-risk phenotype and offer insights into the underlying biological activity driving disease progression [12].
High-Sensitivity C-Reactive Protein
High-sensitivity C-reactive protein (HS-CRP) remains the most extensively validated biomarker of residual inflammatory risk in clinical practice. Synthesized in the liver in response to interleukin-6 signaling, hs-CRP reflects systemic inflammatory burden and correlates with both plaque activity and cardiovascular outcomes. Large observational studies and post hoc analyses of randomized trials have consistently demonstrated that elevated hs-CRP levels predict adverse cardiovascular events independently of low-density lipoprotein cholesterol levels [13].
Importantly, hs-CRP is not merely a passive marker of inflammation but a clinically actionable biomarker. Patients achieving low LDL-C but persistently elevated HS-CRP levels represent a subgroup with disproportionately high residual risk. This discordance between lipid and inflammatory risk has been repeatedly observed in secondary prevention cohorts, underscoring the limitations of lipid-centric risk assessment alone [14].
Interleukin-6 Pathway as an Upstream Biomarker
Interleukin-6 occupies a central position in the inflammatory cascade of atherosclerosis and represents an upstream driver of hepatic acute-phase reactant production. Genetic studies have provided compelling evidence for a causal relationship between IL-6 signaling and atherosclerotic cardiovascular disease, supporting its role not only as a biomarker but also as a potential therapeutic target [15]. Compared with hs-CRP, IL-6 may offer greater specificity for vascular inflammation, as it reflects upstream immune activation rather than downstream hepatic response. Elevated IL-6 levels have been associated with plaque vulnerability, adverse remodeling, and increased risk of cardiovascular mortality across diverse patient populations [16].
Composite and Emerging Inflammatory Indices
Beyond single biomarkers, composite inflammatory indices derived from routine laboratory parameters have gained increasing attention. The neutrophil-to-lymphocyte ratio (NLR), systemic immune-inflammation index (SII), and C-reactive protein–to–albumin ratio integrate multiple dimensions of immune activation and systemic stress. These indices have demonstrated prognostic value in patients with stable coronary artery disease, acute coronary syndromes, and those undergoing percutaneous coronary intervention [17].
However, despite their accessibility and low cost, these emerging biomarkers are influenced by comorbid conditions, acute illness, and non-cardiovascular inflammatory states. As such, their role in routine risk stratification remains complementary rather than definitive, highlighting the need for standardized thresholds and prospective validation before widespread clinical adoption [18]. Established and emerging biomarkers used to identify residual inflammatory risk, along with their clinical significance and limitations, are outlined in Table 2.

Abbreviations: CRP, C-reactive protein; HS-CRP, high-sensitivity C-reactive protein; IL, interleukin.
Pharmacological Anti-Inflammatory Therapies
The recognition of inflammation as a causal driver of atherosclerotic cardiovascular disease has prompted growing interest in pharmacological strategies that specifically target inflammatory pathways, independent of lipid lowering. Unlike stains, whose anti-inflammatory effects are largely secondary to lipid reduction, these therapies aim to directly modulate immune activation, cytokine signaling, and plaque inflammation.
Colchicine
Colchicine has emerged as the most extensively studied and clinically feasible anti-inflammatory agent in the context of atherosclerotic cardiovascular disease. Unlike biologic therapies targeting specific cytokines, colchicine exerts broad anti-inflammatory effects by disrupting microtubule assembly, a process essential for leukocyte migration, inflammasome activation, and intracellular trafficking of inflammatory mediators. Through these mechanisms, colchicine interferes with neutrophil chemotaxis, adhesion, and activation, thereby attenuating early inflammatory responses within the vascular wall [19]. At the molecular level, colchicine suppresses activation of the NLRP3 inflammasome, resulting in reduced production of interleukin-1β and downstream cytokines involved in atherothrombosis.
This effect is particularly relevant in the setting of plaque inflammation, where neutrophil-driven immune activation contributes to endothelial dysfunction, oxidative stress, and plaque instability. By dampening these processes, colchicine promotes a more quiescent plaque phenotype without directly influencing lipid metabolism. Clinical interest in colchicine has been reinforced by randomized controlled trials demonstrating significant reductions in recurrent cardiovascular events when administered at low doses. In patients with recent myocardial infarction, low-dose colchicine reduced the incidence of ischemic events, supporting the hypothesis that inflammation represents an independent and modifiable driver of cardiovascular risk [20].
Importantly, these benefits were observed on top of contemporary guideline-directed medical therapy, including statins and antiplatelet agents, as recommended by current secondary prevention guidelines, emphasizing the additive value of inflammation-targeted intervention [21]. Despite its favorable efficacy profile, colchicine therapy is not without limitations. Gastrointestinal intolerance remains the most reported adverse effect and represents a frequent cause of treatment discontinuation. In addition, concerns have been raised regarding infection risk and drug–drug interactions, particularly in patients with renal impairment or those receiving concomitant therapies that affect colchicine metabolism.
These considerations underscore the importance of careful patient selection and dose optimization in clinical practice. Overall, colchicine represents a pragmatic and cost-effective approach to inflammation modulation in atherosclerotic disease. Its oral administration, low cost, and robust clinical evidence distinguish it from other anti-inflammatory strategies. However, long-term safety, optimal duration of therapy, and identification of patient subgroups most likely to benefit areas of active investigation, positioning colchicine as both a current therapeutic option and a foundation for future inflammation-targeted cardiovascular strategies.
Interleukin-1 Pathway Inhibition
Interleukin-1β (IL-1β) represents a pivotal mediator linking innate immune activation to vascular inflammation in atherosclerosis. As a downstream effector of NLRP3 inflammasome activation, IL-1β amplifies local inflammatory signaling within the arterial wall by promoting endothelial activation, leukocyte recruitment, and smooth muscle cell dysfunction. This cytokinedriven inflammatory amplification contributes to plaque progression, destabilization, and thrombotic complications.
Pharmacological inhibition of IL-1β provided the first direct clinical proof that targeting inflammation—independent of lipid lowering—can reduce cardiovascular events. Suppression of IL-1β signaling leads to downstream attenuation of the IL- 6–C-reactive protein axis, resulting in substantial reductions in systemic inflammatory burden without altering low-density lipoprotein cholesterol levels [22]. Importantly, the magnitude of cardiovascular benefit has been shown to correlate with the degree of achieved inflammatory suppression, underscoring inflammation as a causal rather than associative factor in atherothrombosis.
Despite these encouraging findings, IL-1β inhibition is associated with several practical limitations. Chronic suppression of IL-1–mediated immune responses increase susceptibility to infections, reflecting the central role of IL-1β in host defense mechanisms. In addition, the requirement for parenteral administration and high treatment costs restricts widespread clinical applicability. Consequently, IL-1β inhibitors are currently viewed as proof-of-concept agents rather than broadly implementable therapies, highlighting the need for safer and more accessible strategies to modulate this pathway [23].
Interleukin-6 Pathway Inhibition
Interleukin-6 (IL-6) occupies an upstream and integrative position within the inflammatory cascade of atherosclerosis, serving as a critical link between innate immune activation and systemic inflammatory responses. Unlike downstream biomarkers such as C-reactive protein, IL-6 directly reflects ongoing immune activity within atherosclerotic plaques and has been consistently associated with plaque vulnerability, adverse remodeling, and cardiovascular mortality. Targeting IL-6 signaling offers several theoretical advantages over downstream cytokine inhibition. By intervening earlier in the inflammatory cascade, IL-6 pathway modulation may achieve broader suppression of pro-atherogenic signaling while preserving aspects of immune competence. Experimental and translational studies have demonstrated that inhibition of IL-6 signaling leads to marked reductions in inflammatory biomarkers, improvement in endothelial function, and attenuation of vascular inflammation [24].
Genetic evidence further supports a causal role for IL-6 signaling in atherosclerotic cardiovascular disease, strengthening its candidacy as a therapeutic target. Compared with IL-1β inhibition, IL-6 blockade may offer a more favorable balance between efficacy and safety, although definitive outcome data are still emerging. Ongoing and future clinical trials are expected to clarify whether biomarker reductions associated with IL-6 inhibition translate into sustained reductions in major adverse cardiovascular events [25]. Collectively, IL-6 pathway inhibition represents a promising evolution of inflammation-targeted therapy, shifting the focus from downstream inflammatory markers toward upstream immune modulation with potential implications for precision cardiovascular prevention.
Non-Specific Anti-Inflammatory Strategies
Not all anti-inflammatory approaches have proven effective in cardiovascular disease. Broad immunosuppressive agents, such as low-dose methotrexate, failed to reduce inflammatory markers or cardiovascular events in high-risk populations. These negative findings emphasize that effective cardiovascular risk reduction requires targeted modulation of specific inflammatory pathways rather than generalized immune suppression [26]. Collectively, current evidence supports a paradigm in which selective antiinflammatory therapies, particularly those targeting innate immune activation and cytokine signaling, represent promising adjuncts to lipid-lowering strategies in carefully selected patients with high residual inflammatory risk. A comparative overview of major anti-inflammatory therapies targeting residual inflammatory risk, including their mechanisms, evidence base, and limitations, is provided in Table 3.

Abbreviations: CRP, C-reactive protein; IL, interleukin; LDL-C, low-density lipoprotein cholesterol; NLRP3, NOD-like receptor family pyrin domaincontaining 3; RCT, randomized controlled trial.
Discussion
Inflammation-Targeted Therapy Versus Lipid-Lowering Strategies
Lipid-lowering therapy remains the cornerstone of atherosclerotic cardiovascular disease prevention, with robust evidence demonstrating that reductions in low-density lipoprotein cholesterol translate into proportional reductions in cardiovascular events. Statins, ezetimibe, and proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors have substantially improved outcomes across a wide spectrum of risk categories. However, even in patients achieving very low LDL cholesterol levels, a considerable burden of cardiovascular risk persists, highlighting the limitations of a lipid-centric therapeutic paradigm [27].
Statins exert modest anti-inflammatory effects in addition to lipid lowering, as reflected by reductions in circulating inflammatory biomarkers. Nevertheless, these effects are largely secondary to LDL cholesterol reduction and are insufficient to fully suppress vascular inflammation in many high-risk individuals. This dissociation between lipid control and inflammatory activity has been consistently observed in contemporary clinical trials and real-world cohorts, reinforcing the concept that lipid-related and inflammation-related risks represent partially independent biological pathways [28].
PCSK9 inhibitors provide a compelling illustration of this divergence. While these agents achieve profound LDL cholesterol lowering and robust reductions in cardiovascular events, they exert minimal effects on systemic inflammatory markers. Patients with persistently elevated inflammatory biomarkers despite optimal lipid lowering remain at increased risk, suggesting that inflammation-targeted interventions may offer additive benefit when used alongside intensive lipid-lowering therapy [29].
From a mechanistic perspective, lipid accumulation and inflammation are tightly interconnected but not interchangeable processes. Lipid lowering primarily reduces substrate availability for plaque growth, whereas anti-inflammatory therapies modulate immune activation, cytokine signaling, and plaque stability. As such, the therapeutic goals of these strategies are complementary rather than competitive. A dual-target approach addressing both lipid burden and inflammatory activity may therefore provide a more comprehensive framework for cardiovascular risk reduction [30].
Current clinical guidelines strongly emphasize aggressive lipid lowering as first-line therapy, reflecting the depth and consistency of evidence supporting this approach. In contrast, inflammationtargeted therapies are not yet routinely incorporated into guideline-directed care, largely due to concerns regarding safety, cost, and limited long-term outcome data. Nonetheless, emerging evidence supports the selective use of anti-inflammatory strategies in carefully phenotyped patients with high residual inflammatory risk, particularly in secondary prevention settings [31]. Taken together, available data suggest that lipid-lowering therapy remains necessary but may not be sufficient for optimal cardiovascular risk reduction in all patients. The integration of inflammation-targeted therapies represents an evolving paradigm that moves beyond cholesterol alone toward a more nuanced, biology-driven approach to atherosclerotic disease management (Figure 2).
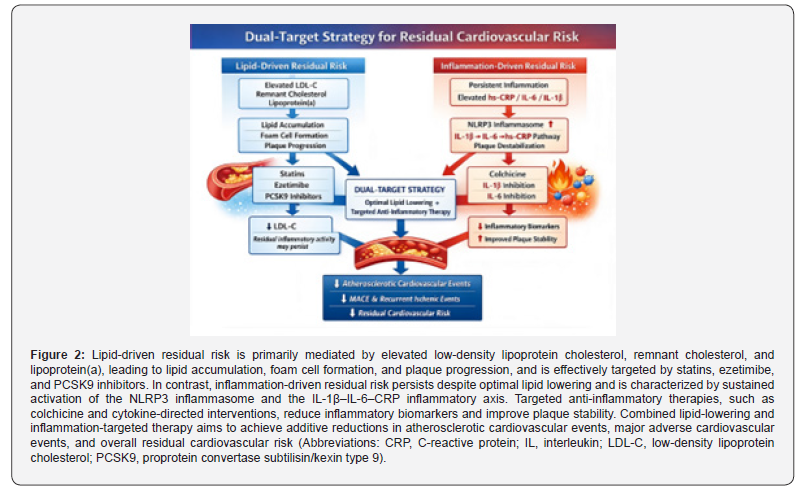
Clinical Integration and Patient Selection
While inflammation-targeted therapies have demonstrated proof of concept in reducing cardiovascular events, their integration into routine clinical practice requires careful patient selection and contextual interpretation of risk. Unlike lipidlowering therapy, which is broadly applicable across risk strata, anti-inflammatory strategies appear to confer benefit primarily in selected populations characterized by persistent inflammatory activity despite optimal guideline-directed medical therapy [13]. A key principle in clinical integration is the identification of patients with residual inflammatory risk.
This phenotype is typically defined by persistently elevated inflammatory biomarkers, most commonly high-sensitivity C-reactive protein, in the presence of well-controlled lowdensity lipoprotein cholesterol. Such patients frequently present in secondary prevention settings, including those with prior myocardial infarction or established chronic coronary syndromes, and exhibit disproportionately high rates of recurrent ischemic events [12]. Evidence supporting patient selection based on inflammatory risk has been reinforced by trials evaluating lowdose colchicine in stable coronary artery disease.
In these studies, cardiovascular benefit was observed predominantly in patients receiving contemporary background therapy, suggesting that inflammation-targeted interventions should be viewed as adjunctive rather than substitutive strategies [32]. Importantly, the magnitude of benefit appeared independent of baseline lipid levels, further supporting inflammation as a parallel therapeutic axis. Clinical integration must also account for safety considerations and competing risks. Anti-inflammatory therapies may increase susceptibility to infection or interact with commonly prescribed cardiovascular medications, particularly in elderly patients and those with renal dysfunction. As a result, risk–benefit assessment should incorporate comorbid conditions, frailty status, and anticipated treatment duration. These considerations partially explain why inflammation-targeted therapies have not yet been universally adopted into guidelinedirected care.
Current European guidelines continue to emphasize aggressive lipid lowering as the foundation of secondary prevention, while acknowledging inflammation as an emerging but not yet fully integrated therapeutic target [21,27]. In this context, inflammation-targeted therapy may be most appropriately considered in patients who remain at high risk despite achieving recommended lipid targets and who demonstrate objective evidence of persistent inflammatory activation. From a practical standpoint, clinical integration of anti-inflammatory strategies will likely evolve toward a precision medicine framework. This approach prioritizes biomarker-guided therapy, careful phenotyping, and shared decision-making, balancing potential cardiovascular benefit against safety and cost considerations. Ongoing trials and real-world data are expected to further refine patient selection criteria and inform future guideline recommendations [33].
Future Directions and Research Perspectives
The growing recognition of inflammation as a causal contributor to atherosclerotic cardiovascular disease has shifted research efforts toward more refined and personalized therapeutic strategies. Rather than broad immune suppression, future approaches are increasingly focused on selectively targeting specific inflammatory pathways while preserving essential host defense mechanisms. This paradigm reflects lessons learned from earlier trials, where non-specific anti-inflammatory therapies failed to improve cardiovascular outcomes despite strong biological rationale [34].
One of the most promising future directions is precision medicine–based risk stratification. In this framework, inflammatory biomarkers such as high-sensitivity C-reactive protein or upstream cytokines are used not merely for risk prediction but to guide therapeutic decision-making. Identifying patients with true residual inflammatory risk may allow clinicians to selectively apply anti-inflammatory therapies to those most likely to benefit, thereby improving the risk–benefit ratio of treatment [35]. Another important research avenue involves upstream targeting inflammatory signaling pathways. Genetic and translational studies increasingly support interleukin-6 signaling as a key mediator of atherogenesis, suggesting that upstream inhibition may offer broader and more durable suppression of vascular inflammation than downstream cytokine blockades.
Ongoing investigations aim to determine whether modulation of this pathway can achieve meaningful reductions in cardiovascular events with acceptable long-term safety profiles [25]. The integration of combination strategies targeting both lipid burden and inflammatory activity also represents a critical future direction. Rather than viewing lipid-lowering and antiinflammatory therapies as competing approaches, emerging evidence supports their complementary roles in addressing distinct components of residual cardiovascular risk. Such dualtarget strategies may be particularly relevant in high-risk secondary prevention populations with recurrent events despite optimal lipid control [36].
Finally, long-term safety and feasibility remain central challenges for the field. Chronic inflammation suppression raises concerns regarding infection risk, malignancy, and immune dysregulation, necessitating extended follow-up and real-world data. Future trials will need to incorporate longer observation periods, pragmatic designs, and biomarker-guided enrollment to better define the clinical boundaries of inflammation-targeted cardiovascular prevention [37]. Collectively, these evolving research directions suggest that inflammation-targeted therapy is transitioning from proof of concept toward selective clinical application. The success of this transition will depend on precise patient phenotyping, careful pathway selection, and integration with established guideline-directed therapies.
Limitations of Current Evidence
Despite substantial advances in understanding the inflammatory mechanisms underlying atherosclerotic cardiovascular disease, several important limitations constrain the interpretation and clinical translation of current evidence. First, most data supporting inflammation-targeted therapies originates from a limited number of large, randomized trials or post hoc analyses. These studies often involve highly selected populations, which may not fully represent the heterogeneity encountered in routine clinical practice [38]. Second, inflammatory biomarkers used to define residual inflammatory risk lack universal standardization.
While high-sensitivity C-reactive protein is the most widely validated marker, optimal thresholds for treatment initiation remain uncertain, and intra-individual variability may influence risk classification. Moreover, upstream cytokines such as interleukin-6 are not routinely measured in clinical settings, limiting their immediate applicability for patient selection [39]. Safety considerations represent another major limitation. Chronic modulation of inflammatory pathways may impair host defense mechanisms, increasing susceptibility to infections or other immune-related adverse effects. Signals of increased infectionrelated mortality observed in certain anti-cytokine trials underscore the need for cautious interpretation and long-term safety surveillance before widespread adoption of these therapies [22].
Additionally, the long-term effects of sustained inflammation suppression on non-cardiovascular outcomes, including malignancy and immune dysregulation, remain incompletely understood. Most cardiovascular outcome trials have relatively short follow-up periods and are not powered to detect rare but clinically meaningful adverse events, creating uncertainty regarding the risk–benefit balance of prolonged therapy [40]. Finally, current evidence does not clearly define how inflammationtargeted therapies should be integrated with existing guidelinedirected medical treatments. Questions regarding optimal timing, duration of therapy, and combination with lipid-lowering agents remain unresolved. Addressing these gaps will require pragmatic trials with biomarker-guided enrollment, longer follow-up, and real-world validation to inform future guideline recommendations.
Conclusion
A growing body of evidence supports inflammation as a central and causal component of atherosclerotic cardiovascular disease. While lipid-lowering therapies remain the cornerstone of cardiovascular prevention, they do not fully address the residual risk observed in many patients, particularly those with persistent inflammatory activity despite optimal lipid control. The concept of residual inflammatory risk provides a clinically meaningful framework to understand this unmet need and to identify patients who may benefit from additional therapeutic strategies.
Advances in the understanding of inflammatory pathophysiology have clarified the roles of innate and adaptive immune responses, cytokine signaling, and inflammasome activation in plaque progression and destabilization. In parallel, inflammatory biomarkers have emerged as practical tools for risk stratification beyond traditional lipid parameters. Pharmacological interventions targeting inflammation, including colchicine and cytokine-directed therapies, have demonstrated that cardiovascular risk can be reduced independently of lipid lowering, reinforcing inflammation as a valid therapeutic target.
Despite these advances, important challenges remain. Optimal patient selection, long-term safety, treatment duration, and integration with established guideline-directed therapies are not yet fully defined. Future progress will likely depend on precision medicine approaches that combine biomarker-guided risk assessment with selective targeting of specific inflammatory pathways. Such strategies may enable more individualized cardiovascular prevention and improve outcomes in patients with high residual inflammatory risk.
Contributorship:
All the authors contributed planning, conduct, and reporting of the work. All authors had full access to all data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis.
Funding:
No financial funding was received for this study.
Competing Interests:
All the authors have no conflict of interest.
References
- Mohammadnia N, Opstal TSJ, El Messaoudi S, Bax WA, Cornel JH (2023) An Update on Inflammation in Atherosclerosis: How to Effectively Treat Residual Risk. Clin Ther 45(11): 1055-1059.
- Arnold N, Koenig W (2025) Inflammation in atherosclerotic cardiovascular disease: From diagnosis to treatment. Eur J Clin Invest 55(7): e70020.
- Paul Guedeney, Bimmer E Claessen, Deborah N Kalkman, Melissa Aquino, Sabato Sorrentino, et al. (2019) Residual Inflammatory Risk in Patients With Low LDL Cholesterol Levels Undergoing Percutaneous Coronary Intervention. J Am Coll Cardiol 73(19): 2401-2409.
- Angela P Moissl, Graciela E Delgado, Hubert Scharnagl, Rüdiger Siekmeier, Bernhard K Krämer, et al. (2025) Comparing Inflammatory Biomarkers in Cardiovascular Disease: Insights from the LURIC Study. Int J Mol Sci 26(15): 7335.
- Zubirán R, Neufeld EB, Dasseux A, Remaley AT, Sorokin AV (2024) Recent Advances in Targeted Management of Inflammation In Atherosclerosis: A Narrative Review. Cardiol Ther 13(3): 465-491.
- Gimbrone MA Jr, García-Cardeña G (2016) Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ Res 118(4): 620-636.
- Tabas I, Bornfeldt KE (2016) Macrophage Phenotype and Function in Different Stages of Atherosclerosis. Circ Res 118(4): 653-667.
- Peter Duewell, Hajime Kono, Katey J Rayner, Cherilyn M Sirois, Gregory Vladimer, et al. (2010) NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464(7293): 1357-1361.
- Libby P, Hansson GK (2015) Inflammation and immunity in diseases of the arterial tree: players and layers. Circ Res 116(2): 307-311.
- Sage AP, Tsiantoulas D, Binder CJ, Mallat Z (2019) The role of B cells in atherosclerosis. Nat Rev Cardiol 16(3): 180-196.
- Ridker PM (2016) From C-Reactive Protein to Interleukin-6 to Interleukin-1: Moving Upstream To Identify Novel Targets for Atheroprotection. Circ Res 118(1): 145-156.
- Ridker PM (2016) Residual inflammatory risk: addressing the obverse side of the atherosclerosis prevention coin. Eur Heart J 37(22): 1720-1722.
- Paul M Ridker, Deepak L Bhatt, Aruna D Pradhan, Robert J Glynn, Jean G MacFadyen, et al. (2023) Inflammation and cholesterol as predictors of cardiovascular events among patients receiving statin therapy: a collaborative analysis of three randomised trials. Lancet 401(10384): 1293-1301.
- Bohula EA, Giugliano RP, Leiter LA, et al. (2018) Inflammatory and Cholesterol Risk in the FOURIER Trial. Circulation 138(2): 131-140.
- IL6R Genetics Consortium Emerging Risk Factors Collaboration; Nadeem Sarwar, Adam S Butterworth, Daniel F Freitag, John Gregson, et al. (2012) Interleukin-6 receptor pathways in coronary heart disease: a collaborative meta-analysis of 82 studies. Lancet 379(9822): 1205-1213.
- Ridker PM, Rifai N, Stampfer MJ, Hennekens CH (2000) Plasma concentration of interleukin-6 and the risk of future myocardial infarction among apparently healthy men. Circulation 101(15): 1767-1772.
- Julio Núñez, Eduardo Núñez, Vicent Bodí, Juan Sanchis, Gema Miñana, et al. (2008) Usefulness of the neutrophil to lymphocyte ratio in predicting long-term mortality in ST segment elevation myocardial infarction. Am J Cardiol 101(6): 747-752.
- Sager HB, Nahrendorf M (2016) Inflammation: a trigger for acute coronary syndrome. Q J Nucl Med Mol Imaging 60(3): 185-193.
- Gonzalo J Martínez, Stacy Robertson, Jennifer Barraclough, Qiong Xia, Ziad Mallat, et al. (2015) Colchicine Acutely Suppresses Local Cardiac Production of Inflammatory Cytokines in Patients with an Acute Coronary Syndrome. J Am Heart Assoc 4(8): e002128.
- Jean-Claude Tardif, Simon Kouz, David D Waters, Olivier F Bertrand, Rafael Diaz, et al. (2019) Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N Engl J Med 381(26): 2497-2505.
- Robert A Byrne, Xavier Rossello, J J Coughlan, Emanuele Barbato, Colin Berry, et al. (2023) 2023 ESC Guidelines for the management of acute coronary syndromes. Eur Heart J 44(38): 3720-3826.
- Paul M Ridker, Jean G MacFadyen, Brendan M Everett, Peter Libby, Tom Thuren, et al. (2018) Relationship of C-reactive protein reduction to cardiovascular event reduction following treatment with canakinumab: a secondary analysis from the CANTOS randomised controlled trial. Lancet 391(10118): 319-328.
- Libby P (2017) Interleukin-1 Beta as a Target for Atherosclerosis Therapy: Biological Basis of CANTOS and Beyond. J Am Coll Cardiol 70(18): 2278-2289.
- Espen Holte, Ola Kleveland, Thor Ueland, Gabor Kunszt, Marte Bratlie, et al. (2017) Effect of interleukin-6 inhibition on coronary microvascular and endothelial function in myocardial infarction. Heart 103(19): 1521-1527.
- Ridker PM, Rane M (2021) Interleukin-6 Signaling and Anti-Interleukin-6 Therapeutics in Cardiovascular Disease. Circ Res.128(11): 1728-1746.
- Paul M Ridker, Brendan M Everett, Aruna Pradhan, Jean G MacFadyen, Daniel H Solomon, et al. (2019) Low-Dose Methotrexate for the Prevention of Atherosclerotic Events. N Engl J Med 380(8): 752-762.
- François Mach, Colin Baigent, Alberico L Catapano, Konstantinos C Koskinas, Manuela Casula, et al. (2020) 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J 41(1): 111-188.
- Catapano AL, Pirillo A, Norata GD (2017) Vascular inflammation and low-density lipoproteins: is cholesterol the link? A lesson from the clinical trials. Br J Pharmacol 174(22): 3973-3985.
- Gregory G Schwartz, P Gabriel Steg, Michael Szarek, Deepak L Bhatt, Vera A Bittner, et al. (2018) Alirocumab and Cardiovascular Outcomes after Acute Coronary Syndrome. N Engl J Med 379(22): 2097-2107.
- Libby P (2021) The changing landscape of atherosclerosis. Nature 592(7855): 524-533.
- Kosmas CE, Silverio D, Sourlas A, Montan PD, Guzman E, et al. (2019) Anti-inflammatory therapy for cardiovascular disease. Ann Transl Med 7(7): 147.
- Stefan M Nidorf, Aernoud TL Fiolet, Arend Mosterd, John W Eikelboom, Astrid Schut, et al. (2020) Colchicine in Patients with Chronic Coronary Disease. N Engl J Med 383(19): 1838-1847.
- Frank LJ Visseren, François Mach, Yvo M Smulders, David Carballo, Konstantinos C Koskinas, et al. (2021) ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur Heart J 42(34): 3227-3337.
- Soehnlein O, Libby P (2021) Targeting inflammation in atherosclerosis - from experimental insights to the clinic. Nat Rev Drug Discov 20(8): 589-610.
- Libby P, Everett BM (2019) Novel Antiatherosclerotic Therapies. Arterioscler Thromb Vasc Biol 39(4): 538-545.
- Ridker PM (2019) Anticytokine Agents: Targeting Interleukin Signaling Pathways for the Treatment of Atherothrombosis. Circ Res 124(3): 437-450.
- Libby P (2021) Inflammation in Atherosclerosis-No Longer a Theory. Clin Chem 67(1): 131-142.
- Paul M Ridker, Brendan M Everett, Tom Thuren, Jean G MacFadyen, William H Chang, et al. (2017) Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N Engl J Med 377(12): 1119-1131.
- Thomas A Pearson, George A Mensah, R Wayne Alexander, Jeffrey L Anderson, Richard O Cannon 3rd, Michael Criqui, et al. (2003) Markers of inflammation and cardiovascular disease: application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation 107(3): 499-511.
- Kong P, Cui ZY, Huang XF, Zhang DD, Guo RJ, et al. (2022) Inflammation and atherosclerosis: signaling pathways and therapeutic intervention. Signal Transduct Target Ther 7(1): 131.






























