Abstract
Background: Often, we encounter patients with chest pain and left bundle branch block (LBBB) on ECG with a diagnostic dilemma of CAD.
Most of the time we presume it as new onset LBBB and end up having insignificant coronary disease on CAG. Hence, we wanted to analyze the
angiographic profile of symptomatic patients presenting with LBBB.
Methods: It is a prospective observational study done in 100 symptomatic LBBB in-patients aged 18 years or above with chest pain/
breathlessness admitted to King George Hospital, Visakhapatnam from 1st July 2021 to 30th June 2023. Clinical and angiographic data was
analysed and interpreted. Patients with pacemaker-insitu, chronic kidney disease (CKD), iatrogenic, valvular causes and asymptomatic LBBB
patients were excluded.
Results: Majority of them presented with chest pain (85%) and breathlessness (85%). Out of them 15 patients had acute myocardial infarction
and thrombolysed in 10 patients. Most of them had unstable angina (44 patients), chronic stable angina (9 patients) or dilated cardiomyopathy
(28 patients). LAD was most affected vessel followed by right coronary artery on CAG. Significant CAD was noted in 41 patients (41%), nonsignificant
CAD in 37 patients (37%) and normal coronaries in 22 patients (22%).
Conclusion: Hypertensive heart disease, coronary artery disease and dilated cardiomyopathy are the most common causes of LBBB. Asymptomatic
LBBB patients need regular follow-up for any worsening of LV function. Symptomatic LBBB patients need extensive evaluation to look for
structural heart disease. New onset LBBB with typical angina in LBBB patients may require CAG to look for CAD.
Keywords: Coronary artery disease; Heart failure; Left bundle branch block; Cardiomyopathy; Hypertension
Abbreviations:HTN: Hypertension; CAD: Coronary Artery Disease; LBBB: Left bundle branch block; PTCA: Percutaneous Transluminal Coronary Angioplasty; CKD: Chronic Kidney Disease; EF: Ejection Fraction
Introduction
Background: Left bundle branch block (LBBB) is caused by hypertension (HTN), coronary artery disease (CAD), valvular heart disease, cardiomyopathies, myocarditis, left ventricular hypertrophy, rate related ST-T changes, iatrogenic (TAVR related complication) and at times idiopathic [1]. LBBB is associated with increased risk of heart failure and coronary artery disease [1]. Often times we encounter patients with chest pain and LBBB on ECG with a diagnostic dilemma of CAD. Most of the time we don’t have old records and may have to presume it as new onset LBBB and CAD if pretest probability is high and end up having insignificant coronary disease in many of them. Hence, we wanted to analyze the angiographic profile of symptomatic patients presenting with LBBB.
Methods: It is a prospective observational study done in 100 symptomatic LBBB in-patients aged 18 years or above with chest pain/breathlessness admitted to King George Hospital, Visakhapatnam from 1st July 2021 to 30th June 2023. Clinical and angiographic data was analysed and interpreted. Patients with pacemaker-insitu, chronic kidney disease (CKD), iatrogenic, valvular causes and asymptomatic LBBB patients were excluded.
Results: (Figures 1,2,3) (Tables 1,2): Out of 100 symptomatic in-patients, mean age was 58.53 years with 80% of them above 50 years. Majority of them were presented with chest pain (85%) and breathlessness (85%). Out of them 15 patients had acute myocardial infarction and thrombolysed in 10 patients. Most of them had unstable angina (44 patients), chronic stable angina (9 patients) or dilated cardiomyopathy (28 patients). 3 patients (3%) presented with hypertensive emergency and 1 patient (1%) with supraventricular tachycardia. Asymptomatic LBBB patients were not included in the study. Risk factors seen were hypertension in 75 (75%), diabetes in 39 patients (39%), dyslipidemia in 38 patients (38%) and family history of coronary artery disease in 27 patients (27%).
Risk factors like smoking were seen in 22 patients (22%), alcohol intake in 22 patients (22%), obesity in 24 patients (24%), history of COVID in 3 patients (3%) and drug induced (beta-blocker) LBBB in 2 patients (2%). LAD was most affected vessel (31 patients out of 41 patients with significant CAD, 76%), followed by right coronary artery in 14 patients (36%) and left circumflex artery in 10 patients (24%). Significant CAD was noted in 41 patients (41%), non-significant CAD in 37 patients (37%) and normal coronaries in 22 patients (22%). 20 (20%) patients underwent percutaneous transluminal coronary angioplasty (PTCA), 8 (8%) underwent coronary artery bypass graft (CABG) and 72 (72%) were advised medical management.
Discussion
Left bundle branch block (LBBB) may be detected incidentally during routine check-ups in some while others can develop acutely. LBBB is caused by hypertension (HTN), coronary artery disease (CAD), valvular heart disease, cardiomyopathies, myocarditis, left ventricular hypertrophy, rate related ST-T changes, iatrogenic (TAVR related complication) and at times idiopathic [1]. When the active impulse falls in refractory phase of conducting nerve cells (3rd phase), tachycardia induced block occurs, which can also occur at slow rate when impulse occurs during spontaneous depolarization (4th phase) [1,2]. Genetic link to 22 loci has been associated with conduction abnormalities and variations in connexin 40 (expressed in atria, proximal conduction system) / connexin 43 (expressed in Purkinje cells and cardiomyocytes) associated with cardiomyopathies causing LBBB in some [1,3,4] As per ECG criteria (ACC/AHA/HRS) LBBB is defined by QRS duration > 120ms with QS or rS pattern in lead V1, monophasic R & absent Q in lead-V6, lead-I, broad notched/slurred R in lead- I,aVL,V5&V6, QS in lead-aVR & discordant ST/T wave [1,5].
Pathophysiology
LBBB results in blockage of electrical impulse transmission via purkinjee fibres. This leads to impulse transmission via myocardium to rest of the left ventricle (LV) delaying the conduction time. This altered activation leads to dyssynchronous contraction of LV with right ventricle (RV) and interventricular septum (IVS) contracting earlier than the lateral wall of LV [1,6]. Due to unopposed early contraction of RV and IVS blood is displaced into the lateral wall causing a pre-stretch and flattening in the lateral wall during the early systole. Early septal contraction and inward motion terminate LV filling by closing the mitral valve but does not lead to a relevant LV pressure rise, as the contracting myocardial volume is too small [1,7].
Pre-stretch of lateral wall in pre-ejection phase causes a powerful contraction of lateral wall of LV starting the ejection phase and IVS is pushed back into RV with a rebound stretch in IVS [1,7]. These uncoordinated contractions cause compensatory lateral wall hypertrophy with increased blood flow compared to septum [1,8]. Two possible mechanisms of such perfusion imbalance include: (i) physiologically altered autoregulation in response to reduced septal and increased lateral wall strain (ii) the impairment of diastolic coronary blood flow, decreased diastolic filling and endothelial dysfunction [1,9]. Over a period, left ventricle remodels by dilating or asymmetrically hypertrophying leading to impaired contractility [1]. This can lead to coaptation defects of mitral valve and mitral regurgitation further worsens LV function [1,10].
Clinical Significance
LBBB is associated with increased risk of heart failure and coronary artery disease [1,11,12] Framingham study showed 50% cardiac deaths in LBBB patients, although role of other comorbidities cannot be ruled out [1,12] Young LBBB patients (<45 years) without other cardiovascular risk factors had better prognosis compared with those who developed LBBB during or after their fifth decade and had associated risk factors [1,13] In iatrogenic/TAVR-induced LBBB LV ejection fraction worsens when compared to uncomplicated cases [1,14].
Echocardiography
1. ‘Septal flash/septal beak’: Unopposed early systolic
contraction of RV and IVS towards lateral wall followed by delayed
lateral wall contraction forms a beak like appearance on 2D-M
mode [1,15].
2. Apical Rocking: Early systolic contraction of septum
pre-stretches lateral wall and late systolic contraction of lateral
wall stretch back the septum, thereby forming a rocking motion
of septum.
3. Compensatory hypertrophying of lateral wall to
overcome the increased strain [1].
4. Shorter diastolic filling and ejection with decreased
cardiac output [1].
Diagnostic Challenges in LBBB
Often, we encounter patients with chest pain and LBBB on ECG with a diagnostic dilemma of CAD [1]. New onset LBBB in patients with chest pain can be taken as acute coronary event, but most of the time we don’t have old records and may have to presume it as new onset LBBB if pretest probability is high [1,16]. As per Sgarbossa’s criteria 5 points are given to concordant ST elevation of 1mm or more in one or more leads; concordant ST depression of > 1mm in V1-V3 is given 3 points; excessively discordant ST elevation > 5mm in leads with negative QRS is given 2 points. Total score of 3 or more has 20% sensitivity and 98% specificity in diagnosing acute myocardial infarction [1,17,18].
Other criteria like Smith modified sgarbossa criteria, Philips QRS area criteria and Selvester criteria have low sensitivity and high specificity [17,19]. Hence, any doubtful case if pretest probability is high, may need CAG evaluation to rule out CAD [20]. Detecting wall motion abnormality in LBBB is difficult due to septal dyskinesia on 2D-ECHO. Even stress echocardiography with specificity (64%) and positive predictive value (40%) can have diagnostic challenges in LBBB patients [1,21,22,23]. Speckle tracking echocardiography and myocardial perfusion studies can be tried in such cases [1,24]. However, even perfusion studies can give false positive results due to septal hypoperfusion in LBBB patients [1,25]. ECG- gated SPECT can give better results in LBBB patients with CAD [1,26].
Clinical Profile of Lbbb Patients
Age Distribution of Lbbb Patients:
Often patients with LBBB remain asymptomatic for decades before becoming symptomatic and are detected incidentally. Prevalence of LBBB in general population is 0.2% to 1.1% and it increases with age [1,27]. Bharath et al. study group of 116 LBBB patients had a mean age of 62.25 years with 85% of them above 50 years age [28]. In current study group, patients’ age ranged from 27 years to 80 years with a mean age of 58.53 years. 29 patients (29%) were aged between 50-59 years, 31 patients (31%) were between 60-69 years, and 20 patients (20%) were 70 years and above showing predilection in adults and elderly with 80 patients (80%) above 50 years age. Eriksson et al. in a prospective study of 855 Swedish men in the general population who were 50 years of age and followed for 30 years: the incidence of LBBB was 0.4% at age of 50, 2.3% at age of 75, and 5.7% at age of 80 [28]. LBBB is infrequent in young healthy participants. Prognosis of isolated LBBB in young is generally benign [29].
Sex Distribution of LBBB Patients:
Sex distribution of LBBB was insignificant regardless of age in current study. In the current study group males and females were equal in number (50 males and 50 females out of total 100 patients). This could be due to small sample size and incidental findings. Bharath et al. study group of 116 patients had 62 males (53.5%) and 54 females (46.5%) [28]. Shehata et al. [] study showed male preponderance with 52 male patients (65%) and 28 female patients (35%) out of the total eighty patients [29].
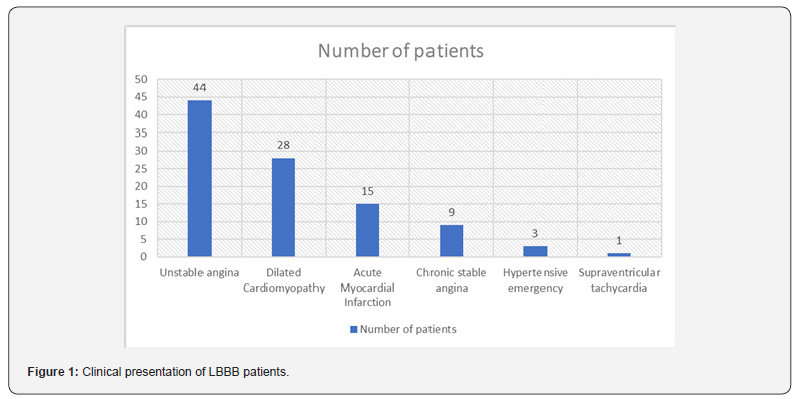
Clinical Presentation: (Figure 1)
Among current study group, most common presenting complaint was chest pain (85%) and breathlessness (85%). Syncope was noted in 4 patients (4%) and palpitations in 13 patients (13%). Bharath et al. study group had dyspnea in 41 patients (35.34%), chest pain in 37 patients (31.89%), syncope in 11 patients (10.54%), palpitations in 3-patients (2.58%) and asymptomatic in 24 patients (20.68%) [28]. Asymptomatic LBBB patients were not included in this study group. Yousif S et al. study group of 150-patients had chest pain in 96 patients (64%) and dyspnea in 41 patients (27.3%) [30].
Out of 100 patients in the current study, 15 patients (15%) had acute myocardial infarction, and thrombolysis was done in 10 patients (10%). 44 patients (44%) were diagnosed with unstable angina, 9 patients with chronic stable angina (9%), 28 patients (28%) with dilated cardiomyopathy, 3 patients (3%) presented with hypertensive emergency and one patient (1%) with supraventricular tachycardia. Bharath et al. [28] study group had acute myocardial infarction in 8 patients (6.8%), unstable angina in 22 patients (18.9%) and dilated cardiomyopathy in 26 patients (22.4%) [28].
Risk Factors: (Figure 2) (Table 3)
Bharath MS et al. reported 59 patients (51%) had hypertension and 35 patients (30%) had DM among the study group of 116 LBBB patients [28]. Current study group of 100 patients had hypertension in 75 (75%) and diabetes in 39 patients (39%). Hypertension is an independent risk factor for LBBB and John Aalen et al. []. States have shown that patients with LBBB are more sensitive to increased afterload/ hypertension [31]. With increase in blood pressure, there is worsening of ejection fraction in patients with LBBB due to loss of septal contribution to the ejection of blood [31]. sLIFE study (Losartan intervention for endpoint reduction in hypertension study) in 500 patients over a 5 year follow up showed a two-fold risk of hospitalization with heart failure in uncontrolled hypertensive LBBB patients compared to HTN controlled group [31].
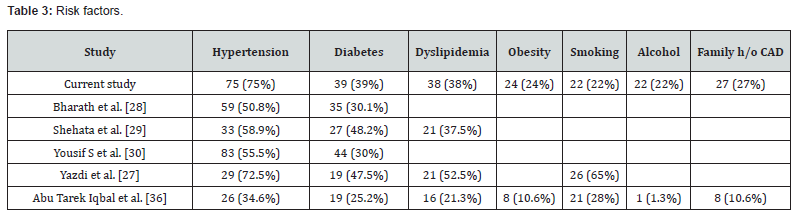
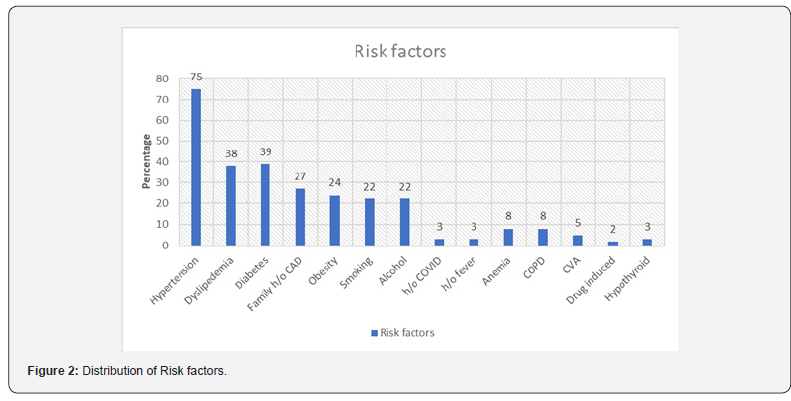
In current study group, patients were either taking antihypertensives previously or newly treated and hence significant relation between hypertension and ejection fraction (EF) could not be noted. Future studies can study if aggressive treatment goals of hypertension can improve EF better in patients with LBBB. Hypertension was the most common risk factor noted in 75 patients (75%) in the current study group. Diabetes was noted in 39 patients (39%), dyslipidemia in 38 patients (38%) and family history of coronary artery disease in 27 patients (27%) in the current study group. Risk factors like smoking were seen in 22 patients (22%), alcohol intake in 22 patients (22%), obesity in 24 patients (24%), history of COVID in 3 patients (3%) and drug induced (beta-blocker) LBBB in 2 patients (2%).
We had few cases who had new onset LBBB with significant CAD on coronary angiogram. Most other significant CAD cases in the study group had no previous records and hence had to be presumed as new onset LBBB when suspicion of CAD is high. In current study, 6 out of 15 patients (40%) with acute myocardial infarction met the sgarbossa criteria for STEMI on ECG. We didn’t include any asymptomatic LBBB patients for the study. Causes of exercise induced LBBB can be CAD, microvascular coronary artery disease, coronary vasospasm, coronary slow flow or without any cardiac disease like rate dependent bundle branch block [32].
Coronary vasospasm causing intermittent LBBB have been reported in past [32]. Patients with exercise induced LBBB are also at increased risk of developing permanent LBBB and hence ventricular dysynchrony and LV dysfunction [32]. Rate-dependent intermittent LBBB occurs due to prolongation of the refractory period of the main left bundle branch or both anterior and posterior fascicles together. At slower heart rates RR interval is longer than refractory period of conduction pathway normalizing the conduction [33]. In such cases, all the routine causes of LBBB must be looked for further evaluation although there were no such cases in the current study group.
Cardiac Risk in LBBB patients:
LBBB has independent cardiac risk compared to those without in patients with structural heart disease [29]. LBBB with CAD worsens the prognosis compared to those without LBBB [34]. Heart failure patients with LBBB are more prone to LV dilatation, worsening EF and increased morbidity compared to those without LBBB [1]. Mean ejection fraction (EF) is 43±0.13% (S.D) in the current study group with slightly lesser EF in females. In the current study group of 100 patients, 96 patients (96%) had diastolic dysfunction, 37 patients (37%) had EF 50% or more, 8 patients (8%) had EF 41% - 49% and 54 patients (54%) had EF 40% or less. Only 1 out of 100 patients had normal LV function. Azadani et al. reported 4.8% of asymptomatic LBBB patients developing heart failure newly over a 6 year follow up [35]. Lee et al. reported a 7.3% to 12% per year worsening of EF over a 45 to 52 months follow up in LBBB patients [27]. LBBB patients were at risk of LV systolic and diastolic dysfunction regardless of age and sex.
2D-ECHO:
On 2D ECHO, 34 patients (34%) had LV hypertrophy, 28 patients (28%) had dilated LV, 6 patients (6%) had RV dysfunction, 96 patients (96%) had LV diastolic dysfunction, and 62 patients (62%) had LV systolic dysfunction. 32 patients (32%) had global hypokinesia of LV, 43 patients (43%) had regional hypokinesia of LV, 61 patients (61%) had septal flash, 61 patients (61%) had apical rocking, and 74 patients (74%) had mitral regurgitation. LV hypertrophy was seen in 39 patients (33.6%) and LV dilatation in 26 patients (22.4%) in Bharath et al. [28] study group [28]. Bharath et al. [28] study group had systolic dysfunction in 62 patients (53.5%) and diastolic dysfunction in 29 patients (25%) [28]. This disparity in numbers could be because they included even asymptomatic LBBB patients in their study [28]. It also depends on the risk profile of the study group. Mitral regurgitation was seen in majority of patients in current study group, probably secondary to coaptation defects of mitral valves due to LV desynchrony in LBBB.
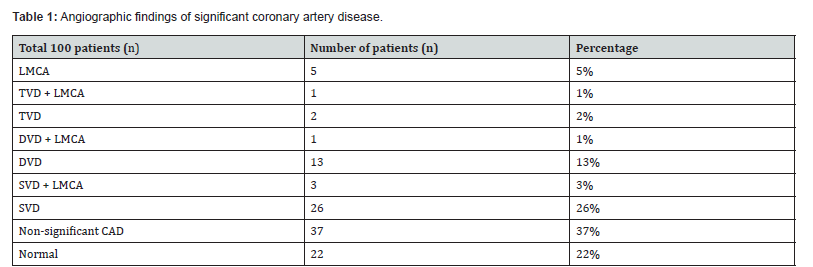
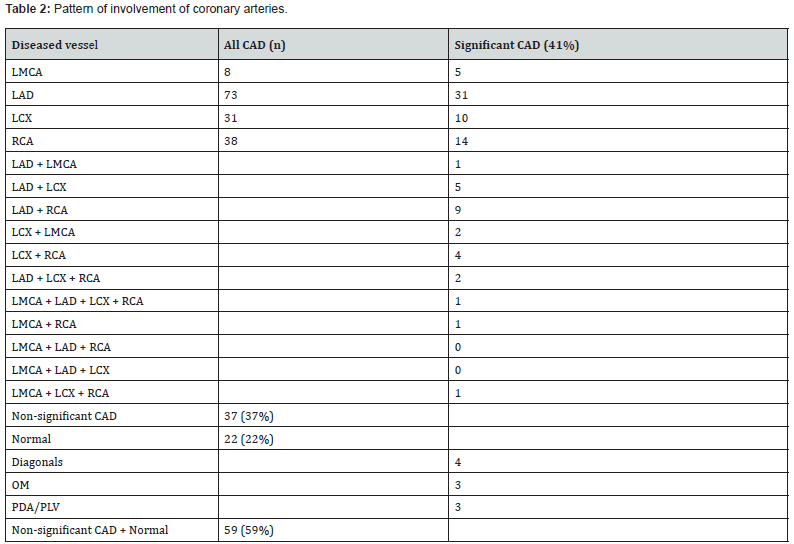
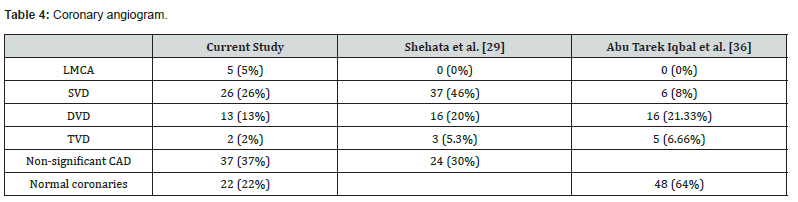
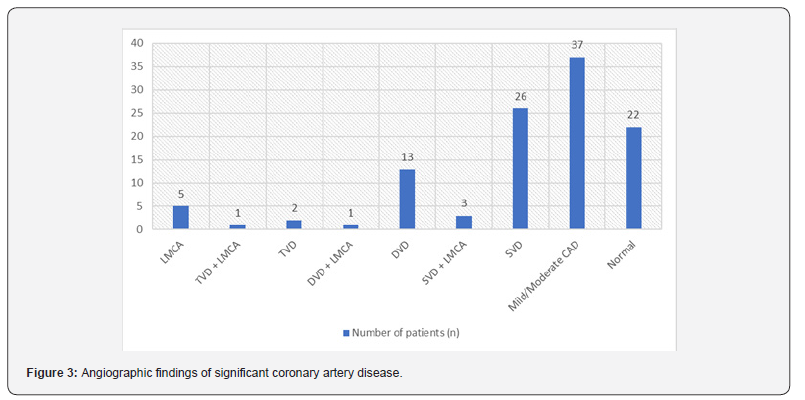
Coronary Angiogram: (Figure 3) (Table 1,2,4)
Yousif S et al. study showed significant coronary artery disease in 70 out of 150 LBBB patients (46.7%) and Rajjit Abrol et al. showed significant CAD in 54% of 336 LBBB patients [30]. Framingham study showed significant CAD in 48% of LBBB patients over an 18 year follow up study.(27) In current study, significant CAD was seen in 41 out of 100 symptomatic LBBB patients (41%). Shehata et al. study in 80 LBBB patients showed single vessel disease (SVD) in 37 patients (46.2%), double vessel disease in 16 patients (20%), three vessel disease in 3 patients (3.8%) and normal coronaries in 24 patients (30%) [29]. 61.3% of the patients had LAD coronary artery involvement. In current study, significant CAD involving LMCA was seen in 5 patients (5%), single vessel disease in 26 patients (26%), double vessel disease in 13 patients (13%) and triple vessel disease in 2 patients (2%).
Left anterior descending artery was most affected vessel (31 patients out of 41 patients with significant CAD, 76%), followed by right coronary artery in 14 patients (36%) and left circumflex artery in 10 patients (24%). Significant CAD was noted in 41 patients (41%), non-significant CAD in 37 patients (37%) and normal coronaries in 22 patients (22%). Abu Tareq Iqbal et al. [36] study group had higher percentage of normal coronaries (64%) compared to current study group (22%) [36]. This could be because of lesser comorbidity burden in their group. They had DM in 19 patients (25%), dyslipedemia in 16 patients (21.33%), obesity in 8 patients (10.6%) and hypertension in 26 patients (34.66%) as against 39 patients (39%), 38 patients (38%), 24 patients (24%) and 75 patients (75%) in current study group [36].
There was no significant difference in LV systolic dysfunction among those with significant and non-significant CAD. Either this could be due to small sample size or due to additional risk of LBBB causing LV systolic dysfunction in both the groups. While 29 patients (69%) with regional hypokinesia of LV had significant CAD, 13 patients (31%) didn’t have significant CAD. 16 patients (40%) with global hypokinesia of LV had significant CAD and 24 patients (60%) didn’t have significant CAD. 9 patients had global hypokinesia with more regional hypokinesia of LV mostly in LAD territory. However, presence of selective regional wall motion was significantly higher in those with significant CAD (p<0.05). No patient died in current study group, however Royal Canadian air force reported a 10 times greater risk of sudden death in patients who had LBBB than those who didn’t [27].
Azadani et al. reported in follow up of 1688 individuals without cardiovascular disease or heart failure for 6 years out of which 2.5% had LBBB on baseline ECG and those with LBBB had 2.85 times more likelihood of developing CHF and 4.34 times greater odds of dying from cardiovascular disease [28]. Heart outcome prevention evaluation study (HOPE) and the Coronary artery surgery study (CASS) also reported higher risk of major cardiovascular events in LBBB patients [27]. Framingham study reported a 50% mortality within 10 years of LBBB detection and only 11% remained free of cardiovascular abnormalities at 18 year follow up [35]. Hence, it is prudent to follow up patients with normal coronaries and those without heart failure at regular basis for early detection of worsening. After coronary angiogram as per disease burden, 20 (20%) patients underwent transluminal coronary angioplasty (PTCA), 8 (8%) underwent coronary artery bypass graft (CABG) and 72 (72%) were advised medical management in the current study group.
Conclusion
1. LBBB is most common in adults and elderly with
increasing prevalence with age irrespective of sex. Hypertension
is the most common risk factor in LBBB patients. Control of
hypertension is of paramount importance in LBBB patients.
Diabetes, dyslipidemia, obesity, smoking and alcohol were major
modifiable risk factors. Lifestyle modification with risk factor
management can go a long way in preventing or delaying major
cardiovascular adverse events apart from addressing the acute
decompensatory episodes.
2. Hypertensive heart disease, coronary artery disease and
dilated cardiomyopathy are the most common causes of LBBB and
most of them had LV systolic and diastolic dysfunction.
3. Even when patients had normal coronaries and good
LV systolic function, most of the LBBB patients had diastolic
dysfunction (99%), indicating that LBBB is at risk of worsening LV
function over time. Most patients are incidentally detected to have
LBBB with prolonged periods before onset and detection as was
in our study where many didn’t have previous records to suggest
acute onset LBBB. However, LBBB can cause cardiomyopathy
due to asynchronous LV contractions over a period. Hence,
asymptomatic LBBB patients need regular follow-up for any
worsening of LV function as they are at risk of developing
cardiomyopathy due to mechanical dyssynchrony secondary to
electrical dyssynchrony.
4. Symptomatic LBBB patients need extensive evaluation
to look for structural heart disease. Majority of symptomatic LBBB
patients presented with chest pain and breathlessness. Although
22% of patients had normal coronaries, significant number of
them (41%) had significant CAD on CAG. Hence, any new onset
LBBB with typical angina in LBBB patient may require CAG to look
for CAD.
Limitations
This being a small sample size from a single center, results may not be generalized to people of other ethnicities and different geographic locations. Large observational studies, retrospective studies and randomized trials may be needed for better insights.
Future Perspective
While coronary angiograms in symptomatic LBBB patients may be useful in ruling-in CAD, its role in asymptomatic LBBB patients’ needs further studies to understand the risk profile better in them.
Declarations
Informed Consent/ Ethics Clearance: Taken
Conflict Of Interest
None
Funding
None
Registry and registration number of the study: N/A
Animal studies
N/A
References
- Surkova E, Badano LP, Bellu R, Aruta P, Sambugaro F, Romeo G, et al. (2017) Left bundle branch block: From cardiac mechanics to clinical and diagnostic challenges. Europace 19(8): 1251-1271.
- Kumar V, Venkataraman R, Aljaroudi W, Osorio J, Heo J, Iskandrian AE et al. (2013) Implications of left bundle branch block in patient treatment. Am J Cardiol 111(2): 291-300.
- Ladenvall P, Andersson B, Dellborg M, Hansson P-O, Eriksson H, et al. (2015) Genetic variation atthe humanconnexin43locus but not at thecon- nexin 40 locus is associated with left bundle branch block. Open Heart 2(1): e000187.
- Fontes MSC, van Veen TAB, de Bakker JMT, van Rijen HVM (2012) Functional conse- quences of abnormal Cx43 expression in the heart. Biochim Biophys Acta 1818(8): 2020-2029.
- De Luna A (2006) The morphology of the electrocardiogram. In: AJ Camm, ed. The ESC Textbook of Cardiovascular Medicine. 1st ed. UK: Blackwell Publishing Ltd p1-34.
- Auricchio A, Fantoni C, Regoli F, Carbucicchio C, Goette A, et al. (2004) Characterization of left ventricular activation in patients with heart failure and left bundle-branch block. Circulation 109(9): 1133-1139.
- Walmsley J, Huntjens PR, Prinzen FW, Delhaas T, Lumens J (2016) Septal flash and septal rebound stretch have different underlying mechanisms. Am J Physiol Heart Circ Physiol 310(3): 394-403.
- Voigt JU (2016) Cardiac resynchronization therapy responders can be better identified by specific signatures in myocardial function. Eur Heart J Cardiovasc Imaging 17(2): 132-133.
- Claridge S, Chen Z, Jackson T, Sammut E, Sohal M, et al. (2015) Current concepts relating coronary flow, myocardial perfusion and metabolism in left bundle branch block and cardiac resynchronisation therapy. Int J Cardiol 181: 65-72.
- Aiba T, Hesketh GG, Barth AS, Liu T, Daya S, et al. (2009) Electrophysiological consequences of dyssynchronous heart failure and its res- toration by resynchronization therapy. Circulation 119: 1220-1230.
- Smith S, Hayes WL (1965) The prognosis of complete left bundle branch block. Am Heart J 70: 157-159.
- Schneider JF, Thomas HEJ, Kreger BE, McNamara PM, Kannel WB (1979) Newly acquired left bundle-branch block: the Framingham study. Ann Intern Med 90(3): 303-310.
- Rabkin SW, Mathewson FA, Tate RB (1980) Natural history of left bundle-branch block. Br Heart J 43(2): 164-149.
- Nazif TM, Williams MR, Hahn RT, Kapadia S, Babaliaros V, et al. (2014) Clinical implications of new-onset left bundle branch block after transcatheter aortic valve replacement: analysis of the PARTNER experience. Eur Heart J 35: 1599-1607.
- Walmsley J, Huntjens PR, Prinzen FW, Delhaas T, Lumens J (2016) Septal flash and septal rebound stretch have different underlying mechanisms. Am J Physiol Heart Circ Physiol 310(3): 394-403.
- Steg PG, James SK, Atar D, Badano LP, Lundqvist CB, et al. (2012) ESC Guidelines for the management of acute myocardial infarction in patients pre- senting with ST-segment elevation. Eur Heart J 33(20): 2569-2619.
- Gregg RE, Helfenbein ED, Babaeizadeh S (2013) New ST-segment elevation myocardial infarction criteria for left bundle branch block based on QRS area. J Electrocardiol 46(6): 528-534.
- Tabas JA, Rodriguez RM, Seligman HK, Goldschlager NF (2008) Electrocardiographic criteria for detecting acute myocardial infarction in patients with left bundle branch block: a meta-analysis. Ann Emerg Med 52(4): 329-336.
- Smith SW, Dodd KW, Henry TD, Dvorak DM, Pearce LA (2012) Diagnosis of ST-elevation myocardial infarction in the presence of leftbundlebranchblock with theST-elevation to S-wave ratio in a modified Sgarbossa rule. Ann Emerg Med 60(6): 766-776.
- Steg PG, James SK, Atar D, Badano LP, Lundqvist CB, et al. (2012) ESC Guidelines for the management of acute myocardial infarction in patients pre- senting with ST-segment elevation. Eur Heart J 33(20): 2569-2619.
- Mordi I, Tzemos N (2015) Non-invasive assessment of coronary artery disease in pa- tients with left bundle branch block. Int J Cardiol 184: 47-55.
- Lewis WR, Ganim R, Sabapathy R (2007) Utility of stress echocardiography in identify- ing significant coronary artery disease in patients with left bundle-branch block. Crit Pathw Cardiol 6: 127-130.
- Tandogan I, Yetkin E, Yanik A, Ulusoy F V, Temizhan A, et al. (2001) Comparison of thallium-201 exercise SPECT and dobutamine stress echocardi- ography for diagnosis of coronary artery disease in patients with left bundle branch block. Int J Cardiovasc Imaging 17(5): 339-345.
- Karavidas AI, Matsakas EP, Lazaros GA, Brestas PS, Avramidis DA, et al. (2006) Comparison of myocardial contrast echocardiography with SPECT in the evaluation of coronary artery disease in asymptomatic patients with LBBB. Int J Cardiol 112(3): 334-440.
- Koepfli P, Wyss CA, Gaemperli O, Siegrist PT, Klainguti M, et al. (2009) Left bundle branch block causes relative but not absolute septal underperfusion dur- ing exercise. Eur Heart J 30(24): 2993-2999.
- Demir H, Erbay G, Kir KM, Omurlu K, Berk F, et al. (2003) Clinical validation of tech- netium-99m MIBI-gated single-photon emission computed tomography (SPECT) for avoiding false positive results in patients with left bundle-branch block: comparison with stress-rest nongated SPECT. Clin Cardiol 26(4): 182-187.
- Yazdi AH, Khalilipur E, Taghdiri MH, Seifrabiee MA, Mehralizadeh N (2017) Structural heart disease in patients with left bundle branch block: State of knowledge. Iran Hear J 18(3): 6-12.
- Bharath MS, Sunayana NS, (2017) Diagnostic and prognostic value of left bundle branch block and its correlation with left ventricular functions: a prospective observational study. Int J Adv Med 4(3): 713.
- Jariwala P, Reddy A, Super Y, Hospitals S (2020) Percutaneous Coronary Intervention of the Large Right Coronary Artery Using a Renal Stent – An Off ‑ label Device for an On ‑ label Indication 51-5
- Yousif S, Alwan M (2013) Coronary angiographic findings in patients with complete left bundle branch block in Erbil city /Iraq. Zanco J Med Sci 17(1): 305-310.
- Aalen J, Storsten P, Remme EW, Sirnes PA, Gjesdal O, et al. (2019) Afterload Hypersensitivity in Patients With Left Bundle Branch Block. JACC Cardiovasc Imaging 12(6): 967-977.
- Chowdhury S, Chakraborty P pratim (2017) Universal health coverage ‑ There is more to it than meets the eye. J Fam Med Prim Care 6(2): 169-1
- Chakrabarti D, Bhattacharjee AK, Bhattacharyya AK (2013) Intermittent left bundle branch block - A diagnostic dilemma. Journal, Indian Acad Clin Med 14(3-4): 278-279.
- Martinez-Selles M, Bramlage P, Thoenes M, Schymik G (2015) Clinical significance of conduction disturbances after aortic valve intervention: current evidence. Clin Res Cardiol 104(1): 1-12.
- Sheikh Jan M, Mohammad Tahir G, Samiera H, Nusrat B (2020) Incidence of Left Ventricular Systolic Dysfunction in Asymptomatic, Incidently Detected Left Bundle Branch Block (LBBB) in Apparently Healthy Individuals: A Prospective Observational Study at a Tertiary Care Cardiac Center in North India. Int J Clin Cardiol 7(6): 1-7.
- Iqbal AT, Ayub M, Salehuddin M, Ahmed K (2018) Coronary Angiographic Findings Among the Left Bundle Branch Block Patients. Univ Hear J 12(2): 88-90.






























