Abstract
Coronary artery calcium; Coronary artery bypass graft; Complex percutaneous coronary intervention; Fluoroscopy; Coronary angiogram
Abbreviations: CAC: Coronary Artery Calcium; MESA: Multi-Ethnic Study of Atherosclerosis; PROSPECT: Providing Regional Observations to Study Predictors of Events in the Coronary Tree; IVUS: Intravascular Ultrasound; MACE: Major Adverse Cardiac Events; NIRS: Near Infrared Spectroscopy
Introduction
Background: Often, we come across calcific coronary vessels with specks of calcification noted on fluoroscopy. However, it is rare to see entire coronary vessels on fluoroscopy without contrast injection due to extensive calcification. Here, we report one such case and discuss pathophysiology and challenges in the management of calcific coronaries.
Case Details: 69-year-old male patient presented to us with a recent worsening of anginal pain and exertional dyspnea progressing from NYHA grade II to grade III over 3 days. He was admitted and treated as unstable angina. He was diabetic, hypertensive and CKD. CAG showed LMCA disease + Triple vessel disease with dense calcific coronaries. All coronaries were seen even before contrast injection which is not so common. He was advised CABG/Medical management (SYNTAX score > 22).
Investigations: Hb 11.4 g/dl, total count 8,600 cells/mm3, Platelet count 2.36 lakh/mm3, Creatinine 1.4mg/dl, eGFR – 45ml/min/1.73m2, viral markers were negative, TSH 6.2mU/ml, Serum Calcium 9.8mg/dl, phosphate 6.9mg/dl, Liver function tests were normal, Sodium 138mEq/L, Potassium 4.95mEq/L, RBS 208mg/dl, Lipid profile: Total Cholesterol 89mg/dl, Triglycerides 177mg/dl, HDL 23mg/dl, VLDL 35mg/dl, LDL 31mg/dl. Ultrasound abdomen showed bilateral grade III renal parenchymal changes. Chest rays was normal. ECG showed sinus rhythm with no specific ST-T changes. 2D Echo showed no regional motion abnormalities. He had grade I diastolic dysfunction with good LV systolic function of EF 60%.
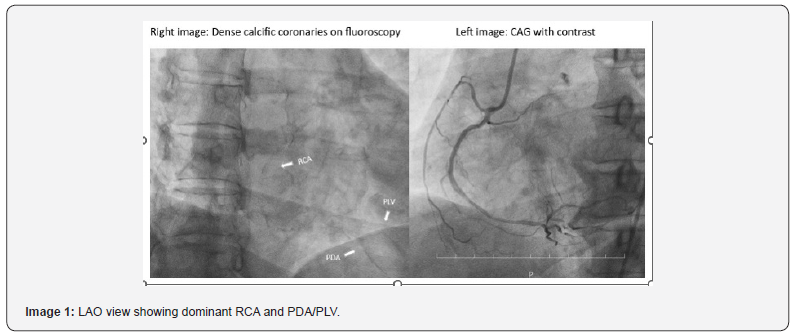
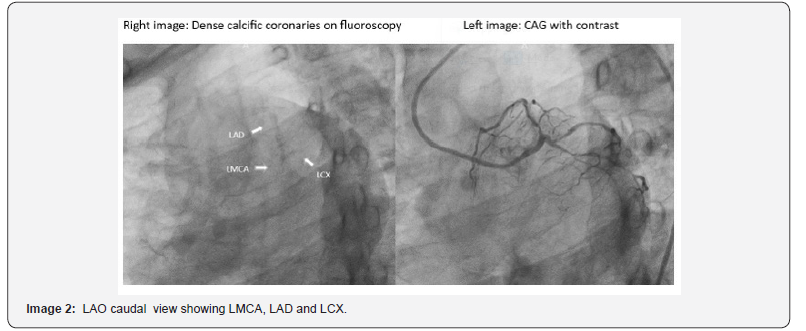
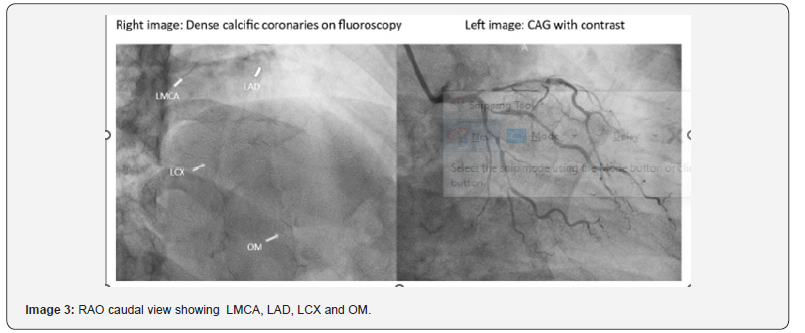
Coronary Angiogram: Suggestive of LMCA + Triple vessel disease (Images 1,2,3)
LMCA - Thick calcific vessel with 50% stenosis
LAD – (Type 3) Thick calcific vessel, ostio-proximal 90% stenosis, proximal to mid long segment 70-80% stenosis. Diagonal-1 large vessel, ostial disease, proximal 50% stenosis, distal 80-90% stenosis
LCX – (Non-Dominant) calcific vessel, proximal to mid two discrete lesions 80-90% stenosis. OM-1 large vessel, ostio-proximal 80-90% stenosis, mid 50% stenosis; OM-3 moderate size, proximal to mid two lesions 70-80% stenosis.
RCA – (Dominant) Thick calcific vessel, proximal mild disease, mid long segment 60% stenosis, distal 60-70% stenosis PDA/PLV diseased
Management: Advised CABG/Medical management (SYNTAX score > 22)
Discussion
While atherosclerotic plaques in coronaries determine the cardiovascular risk, recent interest in coronary artery calcium (CAC) burden in risk assessment by electron-beam/multidetector- CT has gained traction along with coronary CT angiography. Density, pattern, amount of calcium and its progression in atheroma determines the future cardiac risk in most cases rather than just the coronary artery calcium score [1]. In Multi-Ethnic Study of Atherosclerosis (MESA) study, while CAC area, volume and Agatston score had positive correlation with cardiac risk, CAC density was inversely proportional [1]. This is because of lower risk of plaque rupture in dense calcific lesions with less lipid core, less inflammation and absence of high-risk thin cap fibrous atheroma in them [1]. Thin cap fibrous atheroma with high necrotic lipid core are more prone for plaque rupture with subsequent thrombus causing luminal obstruction [1].
Amount of atheroma, degree of luminal obstruction and plaque contents determine the major cardiac outcomes as was shown with positive correlation in Providing Regional Observations to Study Predictors of Events in the Coronary Tree (PROSPECT) study by Xu et al1 using intravascular ultrasound (IVUS) to determine future major adverse cardiac events (MACE) [1]. Spotty calcium with < 90degree coronary circumference in vulnerable plaques were at high risk of acute myocardial infarction/unstable angina [1,2]. Mizukoshi et al. [1] showed similar results using intracoronary optical coherence tomography (OCT) with spotty calcium near lumen in unstable plaques and deeper and denser calcium in stable plaques [1].
While Ferencik et al. [1]. found spotty calcium in most patients of acute coronary syndromes with significant obstructive lesions, Leber et al. [1] saw dense calcific plaques in most patients with chronic stable angina and more number/area of non-calcific lesions in acute coronary events [1]. Ahmadi et al. [1] found three times more mortality risk in patients with non-calcific plaques compared to those with CAC Agatston scores > 400 with dense calcific plaques as did Hou et al1 in their 3-year follow-up study which showed lower risk with calcific plaques compared to mixed/non-calcific plaques [1].
Patients with high coronary calcium (CAC) scores are at risk of coronary artery stenosis when compared to those with low CAC scores and CKD patients have much higher calcification burden [3]. However, Thijis T. Jansz et al3 study in a comparative study between asymptomatic CKD patients on dialysis and those with normal renal function showed much lower risk of significant coronary stenosis even with high coronary artery calcium scores (CAC >400) among CKD patients on CT angiography [3]. They found a higher risk of significant coronary stenosis in patients with normal renal function if they had high CAC scores > 400 [3].
In our patient as well, despite such significant calcium burden, he hardly had any ECG or 2D-ECHO abnormalities suggesting a more stable plaque with chronic obstruction. Our patient had an eGFR – 45ml/min/1.73m2 and Calcium*Phosphate product of 67. In CKD patients, it’s mainly secondary to metabolic dysregulation, high calcium and phosphate product with subsequent deposition of calcium in the tissues and blood vessels [3]. Osteoblastic transformation of myocytes of blood vessels due to cytokine stimulation is another pathway of calcification of the atherosclerotic plaques. While intimal calcification lead to significant stenosis in patients with normal renal function, its mainly medial calcification in CKD patients that can be nonobstructive in spite of extensive calcification until late in the course [3].
CAC can be used in cardiac risk assessment with increasing risk at higher values; however, it’s the CAC progression that’s more important in predicting future risk. Budoff et al. suggested progressive CAC values to be more definitive in risk interpretation than a single baseline CAC (>15% yearly increase) [4]. Multi- Ethnic Study of Atherosclerosis (MESA) study showed a 3-to-6- fold higher risk when the progression was >300units increase of Agatston score [4]. Statin therapy helps in plaque reduction by decreasing lipid levels, inflammatory cells and stabilizing the endothelial function, but calcifications become dense as a consequence.
This is said to stabilize the plaque and decease the future vulnerability which is in contrast to our prior understanding of higher risk with higher CAC levels [4]. Hence, interpretation of CAC levels has to be done with focus on natural history as well as plaque modifications by various treatment modalities before risk assessment [4]. Major risk factors for coronary calcification are male gender, age, smoking, diabetes, dyslipidemia, renal failure, high calcium-phosphate product, dialysis, high intake of calcium salts as phosphate binders, chronic inflammation, oxidative stress, malnutrition and hyperparathyroidism [5,6].
Extensive calcification was seen in post-CABG patients suggesting rapid progression of atherosclerosis in grafted coronary segments due to competitive flow causing blood stasis and reduced shear stress causing severe calcification [7]. Annalisa Noce et al [5] study showed no significant difference in atherogenic indices or lipoprotein ratio in males and females among those with Agatston scores < or > than 400 among nondiabetic patients without evidence of cardiovascular disease [5]. They noticed significantly higher parathormone levels among those with scores > 400 and lesser 7year survival in patients with Agatston score > 400 and iPTH levels > 300pg/L with less than 10% survival in patients with extensive calcification with Agatston score > 1000 [5]. Medial calcification is more in patients on maintenance hemodialysis/uremia and diabetics [5]. Lifestyle modification is most important modifiable factor in the long run.
It’s the thin fibrocalcific plaques with spotty/micro calcifications (1-4mm length and < one quadrant of vessel on IVUS; <3mm on MDCT) that are more vulnerable than dense calcifications [4,2]. Hence, lower CAC has high negative predictive value and higher CAC (>400) has modest specificity/ positive predictive value for obstructive coronary artery disease [4]. Again, this has to be taken with a pinch of salt as even zero CAC doesn’t rule out presence of atheromatous plaques as most vulnerable plaques have microcalcifications (<3mm) which can be missed and most acute plaque ruptures with thrombosis may even lack calcium in acute coronary events [4]. These can be detected on IVUS, OCT or MDCT coronary angiography.
Sodium fluoride (NaF) positron emission tomography imaging can be used to detect spotty calcification (<1mm) or microcalcification (<50 mm) in early stages as they may be associated with plaque rupture. NaF preferentially binds to micro- calcification beyond the resolution of the CT scan in the vasculature [2]. Cardiac magnetic resonance angiography is superior to contrast‐enhanced coronary CT angiography for the evaluation of luminal narrowing in heavily calcified coronary segments [7]. Coronary angiogram is an invasive method to determine CAC.
It has low-moderate sensitivity with high specificity to detect CAC. Radio-opacities on cardiac fluoroscopy (still image) suggest severe CAC; similarly, if the opacities are visible only during cardiac motion, it suggests intermediate CAC. Most accurate method to determine the presence and extent of CAC is IVUS with sensitivity and specificity reaching as high as 90% to 100% and 99% to 100%, respectively [7]. In IVUS, the CAC appears as hyperechogenic mass with acoustic shadow behind. Van der Giessen et al. showed that 53% of the calcifications seen on IVUS images cannot be detected on contrast-enhanced 64-slice multislice CT angiography images because of their size [7].
In OCT, the CAC appears as well delineated, low-signal region with sharp borders with 10times more resolution than IVUS [7]. Pre-intervention OCT imaging delineates plaque constituents and provides accurate measurements of the minimum lumen area (MLA), lesion length, and vessel diameters, as well as visualization of the projected stent landing zones. These parameters can be used for procedural planning, including proper lesion preparation and stent sizing. As CAC obstructs the detailed evaluation of a plaque by IVUS, newer methods in combination with IVUS such as near infrared spectroscopy (NIRS) imaging just before PCI helps in diagnosing necrotic-core and lipid-rich plaques, that is, lipid content in CAC can be detected easily. This also reduces periprocedural MI [8].
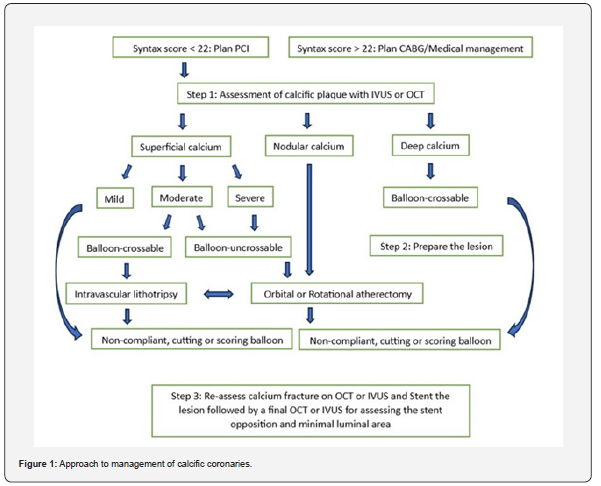
Planning PCI for calcific coronaries has lot of challenges like difficult wire crossing, advancing balloons and deploying stents. These procedural complexities with submaximal stent expansion and stent/polymer damage can lead to stent thrombosis and restenosis with poor long-term outcomes [6,9,10]. Longer procedural and fluoroscopic times, higher radiation and contrast medium volume, procedural complications like pericardial tamponade/perforation, dissection are other major issues with PCI of calcific coronaries [7,9,10]. PCI has favorable outcome in patients with SYNTAX score < 22 and CABG is preferred if SYNTAX score is > 22 with a brief approach delineated in Figure 1 [9-11]. SYNTAX trial in triple vessel disease on 5year follow-up showed better results in CABG with less MACE compared to PCI although in patients with lower SYNTAX score (<22) PCI is acceptable option with a higher risk of repeat revascularization [11].
References
- Thomas IC, Forbang NI, Criqui MH (2018) The evolving view of coronary artery calcium and cardiovascular disease risk. Clin Cardiol 41(1):144-150.
- Nakahara T, Dweck MR, Narula N, Pisapia D, Narula J, et al. (2017) Coronary Artery Calcification: From Mechanism to Molecular Imaging. JACC Cardiovasc Imaging 10(5): 582-593.
- Jansz TT, Go MHY, Hartkamp NS, Stöger JL, Celeng C, Leiner T, et al. (2021) Coronary Artery Calcification as a Marker for Coronary Artery Stenosis: Comparing Kidney Failure to the General Population. Kidney Med 3(3): 386-394.e1.
- Rodriguez-Granillo GA, Carrascosa P, Bruining N (2016) Progression of coronary artery calcification at the crossroads: Sign of progression or stabilization of coronary atherosclerosis? Cardiovasc Diagn Ther 6(3): 250-258.
- Noce A, Canale MP, Capria A, Rovella V, Tesauro M, et al. (2015) Coronary artery calcifications predict long term cardiovascular events in non diabetic Caucasian hemodialysis patients. Aging (Albany NY) 7(4): 269-279.
- Iqbal A, Hussain A, Iqbal A (2018) Coronary artery calcium–a review article. Int J Radiol Radiat Ther 5(6): 318-324.
- Mashayekhi KA, Pyxaras SA, Werner GS, Galassi AR, Garbo R, et al. (2023) Contemporary issues of percutaneous coronary intervention in heavily calcified chronic total occlusions: an expert review from the European CTO Club. EuroIntervention 19(2): e113-e122.
- Jadhav KP, Kavalipatu KNR, Kuchulakanti PK, Reddy RP, Athuluri R, et al. (2021) Coronary Artery Calcification: From Cell to Stent-A Review. Indian J Clin Cardiol 2(2): 97-109.
- Andò G, Alagna G, De Rosa S, Pelliccia F, Gragnano F, et al. (2023) Severely calcified coronary artery lesions: focus on interventional management. Vessel Plus 7.
- Hennessey B, Pareek N, Macaya F, Yeoh J, Shlofmitz E, et al. (2023) Contemporary percutaneous management of coronary calcification: Current status and future directions. Open Hear 10(1): e002182.
- Head SJ, Davierwala PM, Serruys PW, Redwood SR, Colombo A, et al. (2014) Coronary artery bypass grafting vs. percutaneous coronary intervention for patients with three-vessel disease: Final five-year follow-up of the SYNTAX trial. Eur Heart J 35(40): 2821-2830.






























