Ventricular Tachycardia in Hypertrophic Cardiomyopathy Complicated with Apical Aneurysm: About two cases
Sarr SA1, Niang Mboup W2, Diouf Y1, Aw F1, Ndiaye M1, Dia K2, Mingou JS1, Diop KR1, Bodian M1, Ndiaye MB1 and Diao M1
1 Aristide Le Dantec Hospital Dakar, Senegal
2 Principal Hospital, Dakar, Senegal
Submission: June 12, 2023; Published: June 21, 2023
*Corresponding author: Sarr Simon Antoine, Cardiology Unit, Aristide Le Dantec Hospital, Dakar, Senegal
How to cite this article: Sarr SA, Niang Mboup W, Diouf Y, Aw F, Ndiaye M, et al. Ventricular Tachycardia in Hypertrophic Cardiomyopathy Complicated with Apical 002 Aneurysm: About two cases. J Cardiol & Cardiovasc Ther. 2023; 18(4): 555994. DOI: 10.19080/JOCCT.2023.18.555994
Abstract
Introduction: Apical aneurysm is a complication of hypertrophic cardiomyopathy which increases an already high rhythmic risk.
Case presentation: We report the cases of two subjects aged 50 and 58 with no family history of sudden death. One was hypertensive and the other an active weaned smoker. They were admitted for intense palpitations associated with vertigo in one. Clinical examination on admission revealed regular tachycardia in both cases. One of the patients was in cardiovascular collapse. The EKG showed regular tachycardia with wide and monomorphic QRS in both cases. Heart rate was 300 cycles per minute in one case and 215 cycles per minute in the other. Electrical cardioversion at 300 joules was performed, and sinus rhythm was achieved in both cases. An early recurrence of the arrhythmia was noted in one. This led to the administration of amiodarone and a second 300 joules shock. The EKG after cardioversion revealed polymorphic premature ventricular beats associated with a deep negative T wave in the septo-apico-lateral in one patient. In the other case, there was a septo-apico-lateral Q wave of necrosis. Biological tests were normal. Echocardiography Doppler revealed Maron's III type hypertrophic cardiomyopathy with mid-ventricular obstruction, an apical aneurysm and thrombus in both cases. Beta-blocker therapy combined with anti-vitamin K was subsequently initiated in both cases. A double-chamber defibrillator was implanted in one of them. The other patient suffered sudden death before the implantation.
Conclusion: Apical aneurysms should be systematically investigated in HCM. It increases rhythmic risk and is included in the risk score proposed by the American Society of Cardiology.
Keywords: Hypertrophic cardiomyopathy; Aneurysm; Ventricular tachycardia
Introduction
Hypertrophic cardiomyopathy (HCM) is one of the main causes of sudden death in young people, particularly in athletes under 35. Its incidence has recently been estimated at between 0.02 and 0.2% of the general population. It is encountered in around 0.5% of patients referred to an echocardiography laboratory in the absence of any prior screening [1].
Resting obstruction is noted in 25% to 35% of cases. The rate may exceed 75% of affected subjects during exercise [2]. Medioventricular obstruction can lead to an apical aneurysm, aggravating the rhythmic risk and increasing the risk of heart failure and thrombo-embolic complications [3]. This is an under-diagnosed complication, but one that is increasingly well described, thanks in particular to the frequent use of MRI to assess HCM [3].
The increased rhythmic risk associated with this aneurysm makes it one of the main parameters in the decision to implant an ICD in primary prevention according to American guidelines.
We report the cases of two subjects with HCM complicated by apical aneurysm who presented with severe arrhythmias.
Case Presentation
We report the cases of two subjects aged 50 and 58 with no family history of sudden death. One was hypertensive and the other an active smoker. They were admitted for intense palpitations associated with vertigo in one.
Clinical examination on admission revealed regular tachycardia in both cases. One of the patients was in cardiovascular collapse. There was no sign of heart failure. The electrocardiogram showed regular tachycardia with wide QRS, monomorphic in both cases. Heart rate was 300 cycles per minute in one case and 215 cycles per minute in the other. Electrical cardioversion at 300 joules was performed, and sinus activity was obtained on the EKG in both cases. An early recurrence of the arrhythmia was noted in one. This led to the administration of amiodarone and a second 300 joules shock. The EKG after cardioversion revealed polymorphic premature ventricular beats associated with a deep negative T wave in the septo-apico-lateral position in one patient. In the other, there was a septo-apico-lateral Q wave of necrosis associated with an exclusive R wave in V1.
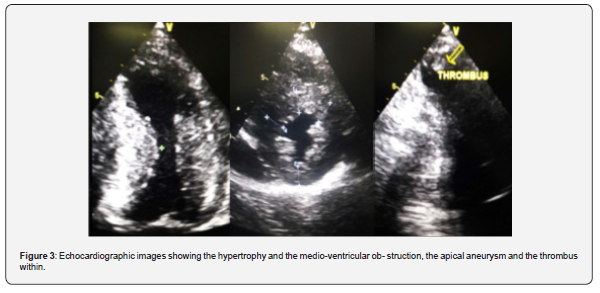
Biological tests were normal. Echogradiography Doppler revealed in one case asymmetrical left ventricular parietal hypertrophy defining Maron's III type hypertrophic cardiomyopathy. The hypertrophy was greatest in the midline (measured at a maximum of 26 mm). There was a significant acceleration of flow at the mid-ventricular level suggesting an obstruction, which was confirmed by continuous Doppler. The maximum gradient was 45 mm Hg. There was an aneurysmal deformity of the apex associated with an enclosed, poorly mobile thrombus. There was a moderate mitral regurgitation (RV=32 ml). The left atrium was enlarged, there was significant diastolic dysfunction with a restrictive mitral profile. In the second, The Echocardiography showed an asymmetrical medio-ventricular hypertrophy with a maximum intra-ventricular gradient at 10 mm Hg at rest. We also noticed an apical aneurysmal with an enclosed and poorly mobile thrombus. Both atria were dilated with a diastolic dysfunction and a restrictive mitral profile.
Because of the necrosis on the EKG, coronarography was performed in one case and was normal. The evolution was marked by an early recurrence of nonsustained ventricular tachycardia in one. In the second, there was a recurrence of the ventricular tachycardia after a week. In both cases, amiodarone combined with beta-blockers was administered.
An automatic implantable defibrillator was offered in both cases. One patient was implanted with a dual-chamber defibrillator. The other patient suffered sudden death before the implantation.
Comments
Apical aneurysm is currently considered as a severe complication of HCM and is an arrythmia substrate exposing to potentially fatal arrythmias. These aneurysms are seen in 2% of HCM [4] and can lead to prognostic implications. Indeed, Marron et al, in a study of 1299 subjects, reported an incidence of 2.2% [4,5].
In a more recent study, the incidence was higher, estimated at 4.8% [3]. On the other hand, the incidence is higher in patients with mid-ventricular obstruction, reaching 10-25% [6,7].
The existence of this aneurysm is linked to several factors, the first of which is mid-ventricular hypertrophy. This will delimit two adjacent cavities on either side of the systolic obstruction: one apical and the other basal. The distal apical cavity, a zone of hyperpressure, will be subject to necrosis in connection with chronic sub-endocardial ischemia. This is followed by fibrotic scarring, with thinning of the wall and aneurysmal deformation [8].
The EKG and Echocardiography Doppler are two very useful tools for diagnosing HCM. However, echocardiography may be limited by the difficulty of visualizing the apex of the left ventricle, or by its shortening [6,7]. The use of contrast may improve the diagnostic yield of echocardiography (Figures 1-4).


Magnetic resonance imaging is very useful and appears to be more sensitive than echocardiography for the detection of smaller apical aneurysms. In addition, it enables more accurate localization and quantification of hypertrophy, provides information on the mitral valve as a whole, and enables rhythmic risk to be assessed [9].

Patients with HCM and apical aneurysms are exposed to a very high embolic risk in connection with apical thrombus formation, as in our cases. The risk of sudden death from ventricular arrhythmia is a major concern for these patients [10].
Serious ventricular arrhythmias are of apical origin and the large areas of fibrosis at this level would constitue the substrate. The latter would explain the need for more aggressive therapy, as drugs alone are only marginally effective [11].
In a recent study of the risk in this sub-group of patients, the authors reported several complications: sudden death (4.7%/year), tachycardia requiring at least two shocks (13 patients/18 ICDs), non-fatal thromboembolic events (1.1%/year) versus none in previously anticoagulated patients (for apical thrombus) [3].
Beta-blockers and calcium channel blockers, which are negative inotropes, are very important drugs for reducing the gradient. Septal myomectomy can have the same effect, but is not applicable to the mid-ventricular form.
The management of mid-ventricular HCM associated with apical aneurysms is controversial. Combining medical treatment with an implantable automatic defibrillator appears to be a sensible approach [12]. In our case, the rhythm disorder dictated implantation of a defibrillator as part of secondary prevention.
Conclusion
Apical aneurysm is a complication of HCM. It increases the risk of serious rhythm disorders, as well as thombo-embolic events. The defibrillator is an alternative but is not always available in our african context.
References
- Maron BJ (2002) Hypertrophic cardiomyopathy: a systematic review. JAMA 287(10): 1308-1320.
- Maron MS, Olivotto I, Betocchi S, Casey SA, Lesser JR, et al. (2003) Effect of left ventricular outflow tract obstruction on clinical outcome in hypertrophic cardiomyopathy. N Engl J Med 348(4): 295-303.
- Rowin EJ, Maron BJ, Haas TS, Garberich RF, Wang W, et al. (2017) Hypertrophic Cardiomyopathy With Left Ventricular Apical Aneurysm: Implications for Risk Stratification and Management. J Am Coll Cardiol 69(7): 761-773.
- Maron MS, Finley JJ, Martijn Bos J, Hauser TH, Manning WJ, et al. (2008) Prevalence, Clinical Significance, and Natural History of Left Ventricular Apical Aneurysms in Hypertrophic Cardiomyopathy. Circulation 118(15): 1541-1549.
- Binder J, Attenhofer Jost CH, Klarich KW (2011) Apical hypertrophic cardiomyopathy: prevalence and correlates of apical outpouching. J Am Soc Echocardiogr Off Publ Am Soc Echocardiogr 24(7): 775-781.
- Matsubara K, Nakamura T, Kuribayashi T, Azuma A, Nakagawa M (2003) Sustained cavity obliteration and apical aneurysm formation in apical hypertrophic cardiomyopathy. J Am Coll Cardiol 42(2): 288-295.
- Minami Y, Kajimoto K, Terajima Y, Yashiro B, Okayama D, et al. (2011) Clinical implications of midventricular obstruction in patients with hypertrophic cardiomyopathy. J Am Coll Cardiol 57(23): 2346-2355.
- Anantha subramaniam K (2014) Hypertrophic cardiomyopathy. In: Crawford MH. Current Diagnosis & Treatment: Cardiology. 4th New York: McGraw-Hill Education pp. 301-313.
- Maron MS, Maron BJ (2015) Clinical impact of contemporary cardiovascular magnetic resonance imaging in hypertrophic cardiomyopathy. Circulation 132(4): 292-298.
- Aoki M, Uekita K, Obata H, Makiguchi N, Mitsuoka T, et al. (2007) Assessment of pathophysiology based on the left ventricular shape in five patients with midventricular obstructive hypertrophic cardiomyopathy. J Cardiol 50(1): 29-38.
- Furushima H, Chinushi M, Iijima K, Sanada A, Izumi D, et al. (2010) Ventricular tachyarrhythmia associated with hypertrophic cardiomyopathy: incidence, prognosis, and relation to type of hypertrophy. J Cardiovasc Electrophysiol 21(9): 991-999.
- Sivanandam A, Ananthasubramaniam K (2016) Mid Ventricular Hypertrophic Cardiomyopathy with Apical Aneurysm: Potential for Underdiagnosis and Value of Multimodality Imaging. Case Rep Cardiol 2016: 9717948.






























