An Overview of Takotsubo Cardiomyopathy, a Potential Mimic of Acute Myocardial Infarction
Allan Roberto Bueso Pineda1, Thulasi Ram Gudi2, Ronald Mauricio Blanco Montecino3, Claudia Liliana Gutierrez Cornejo4, Felix Ricardo Bonilla Bonilla3, Jhon Navarro Gonzalez5, Pushan Aggarwal6, Andrea Galecio Chao7, Bijaya Pariyar8, Peggie Crisalida Mendoza Robles9, Rossy I Valecillos Paez10, Walter Manuel Robles Negrillo11, Jose Luis Hernandez Aparicio3,12 and Maria Isabel Gomez13
1Universidad Tecnologica Centroamericana, Honduras
2Merit Health River Region, Vicksburg, MS, USA
3Universidad de El Salvador, El Salvador
4Universidad Evangélica de El Salvador, El Salvador
5Universidad del Zulia, Venezuela
6Kasturba Medical College Manipal, India
7Universidad de Guayaquil, Ecuador
8Kist Medical College and Teaching Hospital, Nepal
9Universidad de San Martín de Porres, Peru
10Universidad Central de Venezuela, Venezuela
11Universidad San Juan Bautista, Peru
12Larkin Community Hospital, Florida, USA
13Universidad del Valle de Mexico, Mexico
Submission: June 12, 2023; Published: June 21, 2023
*Corresponding author: Maria Isabel Gomez Coral, Universidad del Valle de Mexico, 154 Samson Rd, Frisco, TX, 76081, Mexico
How to cite this article: Allan Roberto Bueso Pineda, Thulasi Ram Gudi, Ronald Mauricio Blanco Montecino, Claudia Liliana Gutierrez Cornejo, Felix Ricardo Bonilla Bonilla, et al. An Overview of Takotsubo Cardiomyopathy, a Potential Mimic of Acute Myocardial Infarction. J Cardiol & Cardiovasc Ther. 2023; 18(4): 555993. DOI: 10.19080/JOCCT.2023.18.555993
Abstract
Takotsubo syndrome, also known as stress-induced cardiomyopathy or "broken heart syndrome," is a transient cardiac condition characterized by sudden and reversible left ventricular dysfunction. It predominantly affects postmenopausal women, although it can occur in men and individuals of all ages. Various risk factors have been associated, including emotional or physical stressors. The clinical presentation of Takotsubo often resembles acute myocardial infarction, with symptoms such as chest pain or discomfort, shortness of breath, palpitations, and fatigue. The absence of significant coronary artery disease on angiography and the characteristic apical ballooning or other regional wall motion abnormalities seen on echocardiography help differentiate it from myocardial infarction. The diagnosis of Takotsubo relies on clinical findings, electrocardiographic changes, elevated cardiac biomarkers, and characteristic findings in imaging studies. The management of Takotsubo syndrome involves supportive care, including addressing the underlying triggers or stressors, symptomatic treatment, and close monitoring of cardiac function and hemodynamics. In most cases, the left ventricular dysfunction resolves spontaneously within days to weeks, and the prognosis is generally favorable, with a low risk of recurrence or long-term complications. However, cardiogenic shock or fatal arrhythmias may also occur as a complication of this condition. An overview of this common cardiomyopathy is provided in this article, highlighting its differences from acute myocardial infarction.
Abbreviations: TC: Takotsubo cardiomyopathy, TTS/TS: Takotsubo syndrome, MRI: Magnetic resonance imaging, PTSD: Post-traumatic stress disorder, CI: Confidence interval, VF: Ventricular fibrillation, AMI: Acute myocardial infarction, ECG/EKG: Electrocardiogram, LV: Left ventricular, ACS: Acute coronary syndrome, CAG: Coronary angiography, CAD: Coronary artery disease, TTE: Transthoracic echocardiography, LVOTO: Left ventricular outflow tract obstruction, CCTA: Computed tomography angiography, CMR: Cardiac magnetic resonance, STEMI: ST elevation myocardial infarction, NSTEMI: non-ST-elevation myocardial infarction, HF: Heart failure, ACEIs: Angiotensin-converting-enzyme inhibitors, ARB: Angiotensin receptor blockers
Keywords: Takotsubo syndrome; Stress cardiomyopathy; Broken heart syndrome; Diagnosis; Treatment; Acute Myocardial Infarction; Differentials
Introduction
Takotsubo syndrome, also known as stress-induced cardiomyopathy or "broken heart syndrome," is a transient cardiac condition characterized by sudden and reversible left ventricular dysfunction [1]. It often presents symptoms similar to acute myocardial infarction. However, it is typically triggered by emotional or physical stress rather than coronary artery disease [1,2]. Statistically, takotsubo cardiomyopathy accounts for approximately 1-2% of patients presenting with symptoms of acute coronary syndrome. It predominantly affects postmenopausal women, with a female-to-male ratio of around 9:1. However, it can also occur in men and individuals of different age groups, albeit less frequently [1,3]. The exact etiology of this cardiac condition is not fully understood, but it is believed to involve a complex interplay between catecholamine excess, microvascular dysfunction, and myocardial stunning [2,4]. In addition, emotional or physical stressors, such as the loss of a loved one, a severe medical illness, or a traumatic event, are thought to trigger a surge of catecholamines, leading to the characteristic transient dysfunction of the left ventricle [4].
Clinical presentation of takotsubo syndrome may mimic acute myocardial infarction, with symptoms such as chest pain, shortness of breath, and electrocardiographic changes. However, unlike myocardial infarction, there is typically no evidence of coronary artery occlusion during imaging studies. In addition, other features, such as transient wall motion abnormalities in the left ventricle, often extending beyond a single coronary artery territory, can aid in distinguishing takotsubo from other cardiac conditions. An accurate diagnosis of takotsubo syndrome involves a comprehensive evaluation, including clinical history, physical examination, electrocardiography, cardiac biomarkers, and imaging techniques such as echocardiography or cardiac magnetic resonance imaging (MRI) [5]. In addition, the diagnostic criteria, as outlined by the International Takotsubo Diagnostic Criteria (2018), help differentiate this syndrome from other cardiac disorders [3]. Management of takotsubo is mainly based on supportive care, including close monitoring of vital signs and hemodynamics. Treatment aims to alleviate symptoms, manage complications, and address underlying triggers [2,5,6]. The prognosis is generally favorable, with most patients experiencing complete recovery of left ventricular function within weeks to months. However, some individuals may develop complications such as heart failure or arrhythmias [6]. This narrative review aims to provide an overview of Takotsubo cardiomyopathy, a relatively common syndrome that can be challenging to diagnose due to its resemblance to acute myocardial infarction. By improving recognition and understanding of this condition, clinicians can enhance diagnostic accuracy and optimize patient care.
Physiopathogenesis
While there are many postulated theories, there is no consensus over the established causes underlying the development of Takotsubo or stress cardiomyopathy. There is also a lack of understanding of why some parts of the heart, particularly the apical region, and mid cavity, are affected more than others. Some commonly assigned underlying mechanisms are catecholamine excess, underlying coronary artery diseases, and possible underlying predispositions. Catecholamines have been demonstrated to induce microvascular spasm that results in subsequent microvascular stunning or even direct myocardial toxicity due to the molecular effects of catecholamines, as demonstrated by elevated clinical levels at presentation [7-10]. A study by Wittstein et al. [11] revealed that the plasma levels of epinephrine were critically elevated in TC patients, with emotional stress as its major precipitating factor. The same study also indicated that serum catecholamine concentration was two to three folds higher in Takotsubo than in myocardial infarction patients [11]. Excessive catecholamine released by the sympathetic nervous system could result in intracellular calcium overload and cardiac dysfunction through the b1 adrenoceptor signal transduction pathway. Calcium overload in myocardial cells leads to ventricular dysfunction and catecholamine cardiotoxicity [12,13].
In most cases of stress cardiomyopathy, angiography does not reveal any obstructive lesions, and only a minimal number of cases show even a small amount of medication-inducible vasospasm[14]. However, this warrants further exploration due to a growing body of clinical evidence that points towards a possible vasogenic/coronary vascular component- incidents of finding single and multifocal coronary vasospasm on angiography, improvement in coronary perfusion with the resolution of myopathy, and studies that found plaque rupture/thrombosis in patients with stress cardiomyopathy [15-17]. Patients with TC constantly demonstrate microvascular dysfunction features, including impairment of endothelium-dependent vasodilation, excessive vasoconstriction, and myocardial perfusion abnormality [14,16].
Concerning underlying factors, many studies have indicated the possible presence of an underlying genetic component, with the possibility that it may be inherited and even dependent on more than one gene (polygenic) [18-21]. Recently, a growing body of data has also indicated that stress cardiomyopathy has been more prevalent in people with diagnosed neurological and stress disorders and those experiencing prolonged stress in their daily lives [22,9].
Epidemiology & Risk factors
Takotsubo syndrome was initially considered a rare condition. However, recent studies have shown it is more prevalent than previously believed. It is estimated that Takotsubo cardiomyopathy accounts for approximately 1-2% of all cases of suspected acute coronary syndrome [23]. The condition primarily affects postmenopausal women, with over 90% of reported cases occurring in females. However, it can also occur in men, albeit less frequently. The prevalence of Takotsubo varies across different populations. Several studies have reported higher prevalence rates in countries such as Japan, where it was initially described [23,24]. In Japan, the prevalence has been reported to be as high as 1.7-2.2% among patients with suspected acute coronary syndrome. Prevalence rates in other regions, such as Europe and the United States, have been estimated to be around 1% [25,26]. The incidence of Takotsubo syndrome has been reported to range from 10 to 70 cases per 100,000 person-years, depending on the population studied. The condition is often considered to be more common in older individuals. Incidence rates may also vary depending on geographic location, socioeconomic status, and access to healthcare [27].
While the exact causes of Takotsubo syndrome remain unclear, several risk factors have been identified. Emotional or physical stress is a common trigger for the condition. Emotional stressors include grief, loss, relationship problems, or intense fear. Physical stressors include severe illness, surgery, and physical trauma [27,28]. Individuals with a history of psychiatric disorders, such as anxiety, depression, or post-traumatic stress disorder (PTSD), may have an increased risk of developing Takotsubo syndrome [23,29]. These conditions may contribute to altered stress response and increased vulnerability to the syndrome. Moreover, certain neurological conditions, such as seizures, stroke, or traumatic brain injury, have been associated with an increased risk of Takotsubo syndrome [24,28,29]. The precise mechanisms linking neurological disorders to the development of Takotsubo syndrome are not yet fully understood. Also, individuals with a history of cardiovascular disease, including hypertension, coronary artery disease, or heart failure, may have an increased risk of Takotsubo syndrome. It is thought that underlying structural heart abnormalities or impaired heart function may contribute to the development of the syndrome. Finally, hormonal factors, such as postmenopausal status, may also contribute to the increased risk observed in women [3,26]. Hormonal changes during and after menopause may influence the stress response and contribute to this population's higher prevalence of the syndrome.
Takotsubo generally has a favorable prognosis, with most individuals recovering entirely within days to weeks. However, the condition can sometimes lead to significant morbidity and mortality. Complications associated with Takotsubo syndrome include acute heart failure, arrhythmias, and, rarely, cardiogenic shock. The mortality rate of Takotsubo is estimated to be around 1-4%, with some studies reporting higher rates in specific populations, such as older individuals or those with underlying comorbidities [23,25,28].
Clinical Presentation
The clinical presentation of Takotsubo cardiomyopathy involves a range of symptoms, signs, and other findings. Patients often experience chest pain similar to a heart attack due to acute coronary syndrome. The pain may be severe and radiate to the neck, arm, or back. Also, palpitations, syncope, breathlessness, or difficulty breathing are common symptoms at rest or with exertion. Frequently, patients also exhibit profound fatigue or weakness due to compromised cardiac function. Moreover, hypotension may be observed due to impaired left ventricular function, pulmonary congestion signs (crackles or wheezing), and jugular venous distension. The latter signs will indicate severe disease with fluid overload [30].
The most frequently observed clinical features in Takotsubo cardiomyopathy (TC) patients are chest pain and difficulty breathing. According to a study, chest pain was reported in 185 out of 273 patients (67.8%, 95% CI: 62.0-73.0%; range: 20-94.7%), while dyspnea was present in 40 out of 225 patients (17.8%, 95% CI: 13.3-23.3%; range: 4.5-55.5%) [31]. More severe clinical presentations, such as cardiogenic shock (4.2%, 95% CI: 2.4-7.4%) and ventricular fibrillation (VF) (1.5%, 95% CI: 0.65-3.9%), can also occur [32]. Atypical Takotsubo syndrome (TS) exhibits distinct features compared to typical TS. These differences include an earlier onset in younger individuals, a higher occurrence of ST-segment depression on electrocardiograms, a greater prevalence of neurological disorders, a less significant reduction in left ventricular ejection fraction, and lower levels of brain natriuretic peptide upon admission [33,34].
It should be noted that Takotsubo's clinical presentation may resemble that of acute myocardial infarction (AMI), which presents symptoms such as severe, crushing, or squeezing chest pain that may radiate to the left arm, jaw, or back. Shortness of breath, sweating, nausea, and pain in other areas, such as the jaw, neck, shoulder, arm, or upper abdomen, are also commonly reported. Given the similar presentation of both cardiac conditions, the only definite way to differentiate between them is during imaging studies. In AMI, coronary angiography typically reveals significant blockages in the coronary arteries, while in TC, the coronary arteries usually appear normal or may show minor abnormalities. Also, in Takotsubo, an echocardiogram will exhibit the typical imaging finding of left ventricle dysfunction [30,35].
It is worth noting that the clinical presentation of Takotsubo cardiomyopathy can vary between individuals, and not all symptoms and findings may be present in every case. Diagnosing Takotsubo cardiomyopathy requires careful evaluation considering the clinical presentation, imaging studies, and exclusion of other possible causes [30].
Diagnosis& Differentials
Diagnosing Takotsubo syndrome is challenging because its clinical phenotype can closely resemble acute myocardial infarction concerning electrocardiogram (ECG) abnormalities and biomarkers. There is currently a lack of a widely established non-invasive tool that allows a rapid and reliable diagnosis of TTS, coronary angiography with left ventriculography is the gold standard for diagnosis to exclude or confirm TTS. Recently, research groups proposed new criteria for TTS and developed international diagnostic criteria (InterTAK Diagnostic Criteria) [34,36]. Criteria are described as follows:
1. Patients show transient left ventricular (LV) dysfunction (hypokinesia, akinesia, or dyskinesia) presenting as apical distension or focal, basal, or mid ventricular wall motion abnormalities. Right ventricular involvement may be present. In addition to these regional patterns of wall movement, there may be transitions between all types. The regional wall motion abnormality usually extends beyond a single epicardial vascular distribution; however, there may be rare cases in which the regional wall motion abnormality is present in the subtended myocardial territory of a single coronary artery (focal TTS) [34].
2. An emotional, physical, or combined trigger may precede the Takotsubo syndrome event, but this is not required [34].
3. Neurological disorders (e.g., subarachnoid hemorrhage, stroke/transient ischemic attack, or seizures) and pheochromocytoma may trigger takotsubo syndrome [34].
4. There are new ECG abnormalities (ST-segment elevation, ST segment depression, T wave inversion, and QTc interval prolongation); however, there are rare cases without ECG changes.
5. In most cases, cardiac biomarkers (troponin and creatine kinase) are moderately elevated; a significant elevation of brain natriuretic peptide is common [34].
6. Significant coronary artery disease does not contradict Takotsubo syndrome [34].
7. Patients have no evidence of infective myocarditis [34].
8. Postmenopausal women are predominantly affected [34].
The most common and widely recognized type of TTS is the apical bulging type, also known as the typical form of TTS, which occurs in the majority of cases. Atypical TTS types have been recognized more frequently, with wall motion patterns: midventricular, basal, and focal [37]. The International Takotsubo Registry's first results indicate that most 1750 patients analyzed are female (female-to-male ratio, 9:1) and, interestingly, more prone to emotional stressors [36]. The International Takotsubo Registry developed InterTAK Diagnostic Score to provide a model to estimate the probability of TTS's presence and can distinguish TTS from SCA with high sensitivity and specificity [36-37]. Points were assigned to each criterion, according to their diagnostic importance: female sex 25 points, emotional trigger 24 points, physical trigger 13 points, absence of ST-segment depression (except in aVR shunt) 12 points, psychiatric disorders 11 points; neurological disorders 9 points and QTc prolongation 6 points. Points were then added to a given patient to result in a score value ranging from 0 to 100 [38].
Electrocardiogram: ECG is abnormal in most patients with TTS, usually demonstrating ischemic ST-segment elevation, T-wave inversion, or both [39]. In the InterTAK Registry, ST-segment elevation was present in 44%, ST-segment depression in 8%, T-wave inversion in 41%, and left bundle branch block in 5% [39]. In both acute coronary syndrome (ACS) and TTS, the ECG demonstrates a time course typically with the resolution of initial ST-segment elevation (if present), followed by progressive T-wave inversion and QT interval prolongation for several days, with subsequent gradual resolution of T-wave inversion and QT prolongation over days to weeks [40]. Patients with ST-segment elevation should undergo urgent coronary angiography (CAG) with left ventriculography to exclude acute myocardial infarction (AMI). In patients with non-ST-segment elevation, the InterTAK Diagnostic Score can be considered. While an InterTAK Score ≤70 points suggest a low to intermediate probability of TTS, a score ≥70 indicates a high probability of TTS [41].
Imaging studies: patients with a low probability should undergo CAG with left ventriculography, while in patients with a high score, transthoracic echocardiography (TTE) should be considered. In the absence of a circumferential ballooning pattern, CAG is recommended. In stable patients with circumferential ballooning patterns, coronary computed tomography angiography (CCTA) is favored to exclude coronary artery disease (CAD). In unstable patients, typical complications of TTS, such as left ventricular outflow tract obstruction (LVOTO), should be determined with TTE and CAG to rule out AMI safely. In patients with normal coronaries on CCTA or CAG and typical ballooning patterns without ‘red flags’ of acute infectious myocarditis, TTS is the most likely diagnosis and can be confirmed after follow-up echocardiography. In case of positive ‘red flags’ of acute infectious myocarditis, cardiac magnetic resonance (CMR) should be performed to confirm the diagnosis [41].
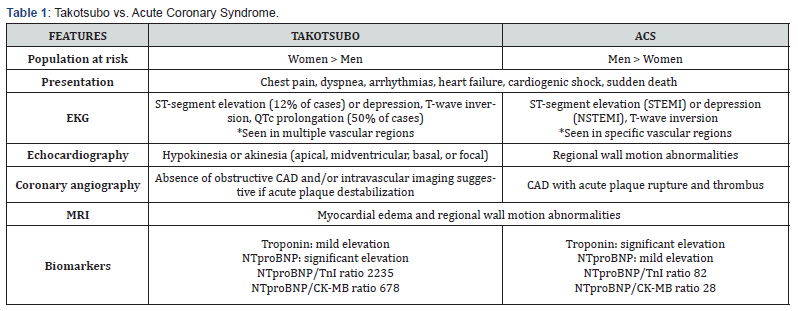
Biomarkers: in a study with a retrospective and prospective double phase, it was found that a cut-off hs-TnT/CKMB ratio of 0.015 distinguished TS from MI with a sensitivity of 85.7% and a specificity of 67.6% (95% CI: 0.71-0.89) in the retrospective phase. In the prospective phase, a ratio of 0.017 differentiated TS from MI, with a sensitivity of 83.3% and a specificity of 78.1% (CI: 0.83-0.94). The hs-TnT/CKMB ratio is a readily available parameter that could be used in conjunction with clinical risk scores, other biomarkers, and ECG findings to discriminate between TS and MI [42]. TS is associated with marked and persistent elevation of NT-proBNP/BNP levels, which correlates with the extent of catecholamine increase and the severity of LV systolic dysfunction [43]. Recently published studies focused on the potential utility of the release and circulation of specific microRNAs (miRNAs) associated with TTS onset [44]. Interleukin (IL)-6 levels appear less elevated, while those of IL-7 are more elevated in TS compared with AMI [45]. In patients with biventricular involvement, it has been shown that plasma concentrations of the stress-responsive cytokine growth differentiation factor-15 increased more rapidly after the onset of TS [46]. A comparison between Takotsubo and Acute Coronary Syndrome is depicted on Table 1.
Treatment Strategies
Currently, no guidelines are established for managing Takotsubo syndrome [47]. Therefore, treatment will be determined by the presence of left ventricular outflow tract obstruction (LVOTO), hemodynamic compromise, and complications. The first step will be to evaluate the patient's hemodynamic status, followed by echocardiography to detect the presence of LVOTO and examination of wall motion abnormality; for example, right ventricular hypokinesis occurs in 33% of the cases [48]. Hemodynamically stable patients are treated with antihypertensive medications such as diuretics, ACE inhibitors, and B-blockers. In individuals with impaired motion of the LV apex, anticoagulant treatment is administered to prevent any thromboembolic event. Intra-aortic balloon pump equipment and cardiac circulatory support are used to treat hemodynamically unstable patients [49].
The presence of a left ventricular outflow tract (LVOTO) is significant for managing Takotsubo syndrome; in previously published studies, LVOT obstruction was evident in 25%-33% of the population studied. [50,51]. It is critical to avoid the use of inotropic drugs, volume depletion, and vasodilator therapy; the therapy for LVOTO accepted is a b-blocker or an a-agonist. There is an exception to using inotropic medications in hypotensive cases due to LV systolic failure that does not have LVOTO. However, it will be short. Lastly, consider the Intra aortic balloon pump as a last resort if LVOTO is refractory.
Complications such as heart failure (HF), thromboembolism, and shock are expected. Therefore, the management has to be personally addressed. There are no specific steps to follow in patients with HF in TTS. Hence HF with reduced ejection fraction management should be followed. Anticoagulation with vitamin K antagonist (warfarin) for three months is indicated when an intraventricular thrombus is detected. Cardiogenic shock increased around 10% of in-hospital mortality during the acute phase of TTS; hypokinesis of the wall is unrelated to the development of hypotensive status or shock [47].
The role of beta-blockers use in pre-admission of the hospital when an adverse cardiovascular event happens has been demonstrated to increase the risk of all-cause complications and cardiac decompensation within the acute phase of TTS and has been linked to upregulation of b-adrenergic receptors with chronic beta-blocker therapy [52].
Disease Prognosis & Potential Complications
Takotsubo syndrome (TTS) was initially considered benign due to its self-limiting clinical course [53]. However, during the acute stage, a substantial number of patients develop severe complications [54]. Some of these, which can affect ionotropic function, include acute heart failure, LV outflow tract obstruction (LVOTO), the presence of LV thrombus, and cardiogenic shock. Also, arrhythmias, heart failure (including pulmonary edema and cardiogenic shock), thromboembolism, cardiac arrest, and rupture can be severe complications in many patients with TTS [55]. Once the diagnosis has been established, conducting continuous telemetry monitoring and repeated echocardiograms is crucial to identify these complications [53]. Moreover, cardiogenic shock can develop in up to 5%-10% of patients with TTS. Its development is associated with physical triggers, young age, and low LV ejection fraction. The mortality rate in TTS with shock is approximately ten times higher. The management can be categorized into cases with and without LVOTO [54].
Recent studies have shown comparable rates of severe in-hospital complications between TTS and acute coronary syndrome [55]. While catecholamine use, cardiogenic shock, resuscitation, and death rates seem equal in both conditions, TTS has been associated with a higher application of invasive or noninvasive ventilation. Up to twenty percent of patients with TTS will develop an in-hospital complication related to hemodynamic and electrical instability during the acute phase of the disease [4]. Not only similarities in hospital complications have been described between TTS and acute coronary syndrome, but also rates of significant adverse cardiac or cerebrovascular event development (which for TTS are reported around 9.9% per patient-year), and death (with a rate of 5.6% per patient-year). Approximately five percent of patients with a Takotsubo cardiomyopathy episode will have a recurrent event [56].
To understand the prognostic factors affecting Takotsubo cardiomyopathy, X. Lu et al. performed a systematic review that analyzed over 60 observational studies. While female sex and emotional triggers were associated with lower short and long-term mortality, male sex, physical triggers, chronic kidney disease, and malignancy led to poorer outcomes [56]. Previous studies indicated that ACEIs and ARBs prescription after discharge was associated with better prognosis [54]. However, the authors reported an uncertain effect. Also, Beta-blockers' role in short- and long-term mortality remains undetermined, which is aligned with previous data [53]. Mood disorders have been found to have an uncertain effect on Takotsubo cardiomyopathy, with different studies reporting divergent results, which may be related to different protocols utilized to identify mood disorders. Left ventricle recovery is essential for long-term prognosis, with early recovery associated with better long-term results and late recovery with less favorable outcomes [56-60].
Conclusion
Takotsubo syndrome, stress-induced cardiomyopathy or broken heart syndrome, is a unique cardiac condition characterized by reversible left ventricular dysfunction. Understanding its epidemiology, risk factors, clinical presentation, diagnosis, treatment, and prognosis is crucial for comprehensive management. Epidemiologically, Takotsubo syndrome predominantly affects postmenopausal women, although it can occur in men and individuals of all ages. The precise incidence and prevalence are not well-defined, but it is increasingly recognized and diagnosed due to improved awareness and diagnostic criteria. Various risk factors have been associated with Takotsubo, including emotional or physical stressors such as bereavement, relationship issues, medical illness, or surgical procedures. Additionally, there is evidence of an association with psychiatric disorders, particularly anxiety and depression. The clinical presentation of Takotsubo often resembles acute myocardial infarction, with symptoms such as chest pain or discomfort, shortness of breath, palpitations, and fatigue. However, the absence of significant coronary artery disease on angiography and the characteristic apical ballooning or other regional wall motion abnormalities seen on echocardiography help differentiate it from myocardial infarction. The diagnosis of Takotsubo relies on clinical presentation, electrocardiographic changes, elevated cardiac biomarkers, and characteristic findings in imaging studies, particularly echocardiography. Additional tests, such as coronary angiography, may be performed to exclude significant coronary artery disease. The management of Takotsubo syndrome involves supportive care, including addressing the underlying triggers or stressors, symptomatic treatment, and close monitoring of cardiac function and hemodynamics. In most cases, the left ventricular dysfunction resolves spontaneously within days to weeks, and the prognosis is generally favorable, with a low risk of recurrence or long-term complications. However, complications like cardiogenic shock or arrhythmias can occur in rare instances, necessitating more intensive management.
In summary, timely recognition, accurate diagnosis, and appropriate supportive care contribute to a favorable prognosis for most individuals affected by this syndrome. In addition to the existing knowledge, further research studies are necessary to understand the underlying pathophysiology of Takotsubo syndrome comprehensively. Despite advances in diagnostic techniques and clinical management, the precise mechanisms behind the development and progression of this condition still need to be understood. Conducting more investigations will provide valuable insights into the intricate interplay of neurohormonal, vascular, and cardiac factors involved in Takotsubo syndrome. A deeper understanding of physiopathology will facilitate the development of targeted therapeutic strategies and enhance patient outcomes. Continued scientific inquiry and collaboration are essential to unraveling the complexities of this intriguing cardiac syndrome.
References
- Templin C, Ghadri JR, Diekmann J, Napp LC, Bataiosu DR, et al. (2015) Clinical Features and Outcomes of Takotsubo (Stress) Cardiomyopathy. N Engl J Med 373(10): 929-938.
- Lyon AR, Bossone E, Schneider B, Sechtem U, Citro R, et al. (2016) Takotsubo syndrome: a Position Statement from the Taskforce on Takotsubo Syndrome of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail 18(1): 8-27.
- Ghadri JR, Wittstein IS, Prasad A, Sharkey S, Dote K, et al. (2018) International Expert Consensus Document on Takotsubo Syndrome (Part I): Clinical Characteristics, Diagnostic Criteria, and Pathophysiology. Eur Heart J 39(22): 2032-2046.
- Lyon AR, Bossone E, Schneider B, Sechtem U, Citro R, et al. (2016) ESC Heart Failure Association Study Group on Takotsubo Syndrome of the European Society of Cardiology. Eur J Heart Fail 18(1): 8-27.
- Templin C, Ghadri JR, Diekmann J, Napp LC, Bataiosu DR, et al. (2015) Clinical Features and Outcomes of Takotsubo (Stress) Cardiomyopathy. N Engl J Med 373(10): 929-938.
- Dote K, Sato H, Tateishi H, Uchida T, Ishihara M (1991) Myocardial Stunning Due to Simultaneous Multivessel Coronary Spasms: A Review of 5 Cases. J Cardiol 21(2): 203-214.
- Wittstein IS, Thiemann DR, Lima JAC, Baughman KL, Schulman SP, et al. (2005) Neurohumoral features of myocardial stunning due to sudden emotional stress. N Engl J Med 352(6): 539-548.
- Paur H, Wright PT, Sikkel MB, Tranter MH, Mansfield C, et al. (2012) High levels of circulating epinephrine trigger apical cardiodepression in a β2-adrenergic receptor/Gi-dependent manner: a new model of Takotsubo cardiomyopathy. Circulation. 126(6): 697-706.
- Akashi YJ, Nakazawa K, Sakakibara M, Miyake F, Musha H, Sasaka K, et al. (2004) 123I-MIBG Myocardial Scintigraphy in Patients with “Takotsubo” Cardiomyopathy. Journal of Nuclear Medicine 45(7): 1121-1127.
- Tsuchihashi K, Ueshima K, Uchida T, Oh-mura N, Kimura K, et al. (2001) Angina Pectoris-Myocardial Infarction Investigations in Japan. Transient left ventricular apical ballooning without coronary artery stenosis: a novel heart syndrome mimicking acute myocardial infarction. Angina Pectoris-Myocardial Infarction Investigations in Japan. J Am Coll Cardiol 38(1): 11-18.
- Roshanzamir S, Showkathali R (2013) Takotsubo cardiomyopathy a short review. Curr Cardiol Rev 9(3): 191-196.
- Frustaci A, Loperfido F, Gentiloni N, Caldarulo M, Morgante E (1991) Catecholamine-induced cardiomyopathy in multiple endocrine neoplasia. A histologic, ultrastructural, and biochemical study. Chest 99(2): 382-385.
- Martin EA, Prasad A, Rihal CS, Lerman LO, Lerman A (2010) Endothelial function and vascular response to mental stress are impaired in patients with apical ballooning syndrome. J Am Coll Cardiol 56(22): 1840-1846.
- Kurisu S, Sato H, Kawagoe T, Ishihara M, Shimatani Y (2002) Tako-tsubo-like left ventricular dysfunction with ST-segment elevation: a novel cardiac syndrome mimicking acute myocardial infarction. Am Heart J 143(3): 448-455.
- Ito K, Sugihara H, Katoh S, Azuma A, Nakagawa M, et al. (2003) Assessment of Takotsubo (ampulla) cardiomyopathy using 99mTc-tetrofosmin myocardial SPECT--comparison with acute coronary syndrome. Ann Nucl Med 17(2): 115-122.
- Ibanez B, Navarro F, Cordoba M, M-Alberca P, Farre J (2005) Tako-tsubo transient left ventricular apical ballooning: is intravascular ultrasound the key to resolve the enigma? Heart 91(1): 102-104.
- Kumar G, Holmes DR Jr, Prasad A (2010) "Familial" apical ballooning syndrome (Takotsubo cardiomyopathy). Int J Cardiol 144(3): 444-445.
- Pison L, De Vusser P, Mullens W (2004) Apical ballooning in relatives. Heart 90(12): e67.
- Sharkey SW, Maron BJ, Nelson P, Parpart M, Maron (2009) MS Adrenergic receptor polymorphisms in patients with stress (tako-tsubo) cardiomyopathy. J Cardiol 53(1): 53-57.
- Handy AD, Prasad A, Olson TM (2009) Investigating genetic variation of adrenergic receptors in familial stress cardiomyopathy (apical ballooning syndrome). J Cardiol 54(3): 516-517.
- Summers MR, Lennon RJ, Prasad A (2010) Pre-morbid psychiatric and cardiovascular diseases in apical ballooning syndrome (tako-tsubo/stress-induced cardiomyopathy): potential pre-disposing factors? J Am Coll Cardiol 55(7): 700-701.
- Nef HM, Möllmann H, Kostin S, Troidl C, Voss S, et al. (2007) Tako-Tsubo cardiomyopathy: intraindividual structural analysis in the acute phase and after functional recovery. Eur Heart J 28(20): 2456-2464.
- Templin C, Diekmann J, Ghadri JR, Napp LC, Bataiosu DR, et al. (2015) Clinical features and outcomes of Takotsubo (stress) cardiomyopathy. N Engl J Med 373(10): 929-938.
- Redfors B, Vedad R, Angeras O, Råmunddal T, Petursson P, et al. (2015) Mortality in Takotsubo syndrome is similar to mortality in myocardial infarction - A report from the SWEDEHEART registry. Int J Cardiol 185: 282-289.
- Kara T, Prasad A, Bybee KA, Lerman A, Barsness GW, et al. (2004) Systematic review: transient left ventricular apical ballooning: a syndrome that mimics ST-segment elevation myocardial infarction. Ann Intern Med 141(11): 858-865.
- Sharkey SW, Windenburg DC, Lesser JR, Maron MS, Hauser RG, et al. (2010) Natural history and expansive clinical profile of stress. 55(4): 333-341.
- Deshmukh A, Kumar G, Pant S, Rihal C, Murugiah K, et al. (2012) Prevalence of Takotsubo cardiomyopathy in the United States. Am Heart J 164(1): 66-71.
- Athanasiadis A, Schwab J, Schneider B, Pistner W, Gottwald U, et al. (2014) Complications in the clinical course of Takotsubo cardiomyopathy. Int J Cardiol 176(1): 199-205.
- Usmani Z, Carson K, Singh K, Sawhney G, Shah R, et al. (2014) Systematic review and meta-analysis of incidence and correlates of recurrence of Takotsubo cardiomyopathy. Int J Cardiol 174(3): 696-701.
- Ghadri JR, Wittstein IS, Prasad A, Sharkey S, Dote K, et al. (2018) International Expert Consensus Document on Takotsubo Syndrome (Part I): Outcome and Management. Eur Heart J 39(22): 2032-2046.
- Nakazawa K, Sakakibara M, Miyake F, Koike H, Sasaka K, et al. (2003) The clinical features of takotsubo cardiomyopathy. QJM: An International Journal of Medicine 96(8): 563-573.
- Akashi YJ, Goldstein DS, Barbaro G, Ueyama T (2008) Takotsubo Cardiomyopathy. Circulation 118(25): 2754-2762.
- Amin HZ, Amin LZ, Pradipta A (2020) Takotsubo Cardiomyopathy: A Brief Review. J Med Life 13(1): 3-7.
- Ghadri JR, Wittstein IS, Prasad A, Sharkey S, Dote k, et al. (2018) International Expert Consensus Document on Takotsubo Syndrome (Part I): Clinical Characteristics, Diagnostic Criteria, and Pathophysiology. Eur Heart J 39(22): 2032-2046.
- O'Gara PT, Kushner FG, Ascheim DD, Donald E Casey Jr, Chung MK, et al. (2013) ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 127(4): e362-e425.
- Ghadri JR, Cammann VL, Templin C (2016) The International Takotsubo Registry: rationale, design, objectives, and first results. Heart Fail Clin 12(4): 597-603.
- Ghadri JR, Cammann VL, Napp LC, Jurisic S, Diekmann J, et al. (2016) International Takotsubo Registry. Differences in the clinical profile and outcomes of typical and atypical takotsubo syndrome: data from the International Takotsubo Registry. JAMA Cardiol 1(3): 335-340.
- Ghadri JR, Cammann VL, Jurisic S, Seifert B, Napp LC, et al. (2017) A novel clinical score (InterTAK Diagnostic Score) to differentiate takotsubo syndrome from acute coronary syndrome: results from the International Takotsubo Registry. European journal of heart failure 19(8): 1036-1042.
- Franke J, Katus HA, Burgdorf C, Schunkert H, Moeller C et al. (2015) Clinical Features and Outcomes of Takotsubo (Stress) Cardiomyopathy. N Engl J Med 373(10): 929–938.
- Bennett J, Ferdinande B, Kayaert P, Wiyono S, Goetschalkx K (2013) Time course of electrocardiographic changes in transient left ventricular ballooning syndrome. Int J Cardiol 169(4): 276-280.
- Ghadri JR, Wittstein IS, Prasad A, Sharkey S, Dote K, et al. (2018) International Expert Consensus Document on Takotsubo Syndrome (Part II): Diagnostic Workup, Outcome, and Management. European heart journal 39(22): 2047-2062.
- Pirlet C, Pierard L, Legrand V, Gach O (2017) Ratio of high-sensitivity troponin to creatine kinase-MB in takotsubo syndrome. International journal of cardiology 243: 300-305.
- Nguyen TH, Neil CJ, Sverdlov AL, Mahadavan G, Chirkov YY, et al. (2011) N-terminal pro-brain natriuretic protein levels in takotsubo cardiomyopathy. Am J Cardiol 108: 1316-1321.
- Kuwabara Y, Ono K, Horie T, Nishi H, Nagao K, et al. (2011) Increased microRNA-1 and microRNA-133a levels in serum of patients with cardiovascular disease indicate myocardial damage. Circ Cardiovasc Genet 4(4): 446-454.
- Elmas E, Haghi D, Lippert C, Kralev S, Lang S, et al. (2012) Platelet and monocyte activity markers and mediators of inflammation in Takotsubo cardiomyopathy. Heart Vessels 27(2): 186-192.
- Stiermaier T, Adams V, Just M, Blazek S, Desch S, et al. (2014) (Growth differentiation factor-15 in Takotsubo cardiomyopathy: diagnostic and prognostic value. Int J Cardiol 173(3): 424-429.
- Di Vece D, Citro R, Cammann VL, Kato K, Gili S, et al. (2019) Outcomes Associated with Cardiogenic Shock in Takotsubo Syndrome: Results from the International Takotsubo Registry. Circulation 139(3): 413-415.
- Singh K, Neil CJ, Nguyen TH, Stansborough J, Chong CR, et al. (2014) Dissociation of early shock in takotsubo cardiomyopathy from either right or left ventricular systolic dysfunction. Heart, Lung & Circulation 23(12): 1141-1148.
- Komamura K, Fukui M, Iwasaku T, Hirotani S, Masuyama T (2014) Takotsubo cardiomyopathy: Pathophysiology, diagnosis and treatment. World Journal of Cardiology 6(7): 602-609.
- El Mahmoud R, Mansencal N, Pilliére R, Leyer F, Abbou N (2008) Prevalence and characteristics of left ventricular outflow tract obstruction in Tako-Tsubo syndrome. American Heart Journal 156(3): 543-548.
- Kawaji T, Shiomi H, Morimoto T, Tazaki J, Imai M, et al. (2015) Clinical impact of left ventricular outflow tract obstruction in takotsubo cardiomyopathy. Circ J 79(4): 839-846.
- Topf A, Mirna M, Dienhart C, Jirak P, Bacher N, et al. (2022) Pretreatment with Betablockers, a Potential Predictor of Adverse Cardiovascular Events in Takotsubo Syndrome. Biomedicines 10(2): 464.
- Medina de Chazal H, Del Buono MG, Keyser-Marcus L, Liangsuo M, Moeller FG (2018) Stress Cardiomyopathy Diagnosis and Treatment: JACC State-of-the-Art Review. J Am Coll Cardiol 72(16): 1955-1971.
- Y-Hassan S, Tornvall P (2018) Epidemiology, pathogenesis, and management of takotsubo syndrome. Clinical Autonomic Research 28: 53-65.
- Fischer T, Vasankari T, Airaksinen KE, Fijalkowski M, Rynkiewicz A, et al. (2015) Clinical Features and Outcomes of Takotsubo Cardiomyopathy. N Engl J Med 373(10): 929-938.
- Li C, Liu Q, Pan S, Dixon RAF, Wang B, et al. (2021) Prognostic factors of Takotsubo cardiomyopathy: a systematic review. ESC Heart Fail 8(5): 3663-3689.
- Ghadri JR, Wittstein IS, Prasad A, Sharkey S, Cammann VL, et al. (2018) International Expert Consensus Document on Takotsubo Syndrome (Part II): Workup, Outcome, and Management. Eur Heart J 39(22): 2047-2062.
- Templin C, Ghadri JR, Diekmann J, Napp C, Dana R, et al. (2015) Clinical Features and Outcomes of Takotsubo (Stress) Cardiomyopathy. N Engl J Med 373(10): 929-938.
- Wittstein IS, Thiemann DR, Lima JA, Baughman KL, Schulman SP, et al. (2005) Neurohumoral features of myocardial stunning due to sudden emotional stress. N Engl J Med 352(6): 539-548.
- Gianni M, Dentali F, Grandi AM, Sumner G, Hiralal R, et al. (2006) Apical ballooning syndrome or Takotsubo cardiomyopathy: a systematic review. Eur Heart J 27(13): 1523-1529.






























