Clinical and Echocardiographic Outcome of Percutaneous Closure of Ventricular Septal Defects
Iyad AL-Ammouri1*, Sahar Karasneh1, Sohayla Y Dababseh2, Fatima A Alkubaisi2, Batool E Al-Masri2, Majd H Al-Khawaldeh2 and Amani Nofal2
1Section of pediatric cardiology, Department of pediatrics, School of Medicine, the University of Jordan, Amman 11942, Jordan
2Department of pediatrics, School of Medicine, the University of Jordan, Amman 11942, Jordan
Submission: February 01, 2023; Published: February 15, 2023/p>
*Corresponding author: Iyad AL-Ammouri, Professor of pediatric cardiology, Section of Pediatric Cardiology, department of pediatrics, School of Medicine, the University of Jordan, Amman 11942, Jordan
How to cite this article: Iyad AL-Ammouri, Sahar Karasneh, Sohayla Y Dababseh, Fatima A Alkubaisi, Batool E Al-Masri, Majd H Al-Khawaldeh and Amani Nofal. Clinical and Echocardiographic Outcome of Percutaneous Closure of Ventricular Septal Defects. J Cardiol & Cardiovasc Ther. 2023; 18(2): 555985. DOI: 10.19080/JOCCT.2023.18.555985
Abstract
Background: Ventricular septal defects (VSD) are the most common congenital cardiac anomalies. Hemodynamically significant defects require closure. Surgery is the gold standard for most defects. Recently, transcatheter closure has provided an acceptable option in selected cases. We aim to evaluate the outcomes of percutaneous closure of VSD and assess the effect of device closure on echocardiographic left-sided chamber parameters.
Methods: A retrospective cross-sectional study included patients who underwent percutaneous VSD closure between 2015 and 2021. Clinical data, angiographic, and echocardiographic images before and after closure were reviewed.
Results: Among 38 patients who had VSD closure during the study period, 34 had isolated defects. The most common defect type was perimembranous (27 patients, 71%). Median age was 6.5 years (0.5-49), and median weight was 19.5 kgs (5-70). Median VSD size was 4 mm (3-10). Most patients were symptomatic (26, 68%). Following closure, all symptomatic patients reported improved symptoms. There were no serious complications during a mean follow up period of 18months (±19). All echocardiographic parameters of left sided volume overload decreased when compared to baseline within the first month of the procedure (left ventricular Z-score dropped from 1.53 ±0.87 to 0.31 ±0.68, p=0.0001, left atrial Z-score dropped from 2.78 ±0.71 to 1.54 ±1.13, p= 0.0001.
Conclusion: Percutaneous closure of VSD in the current era is safe and effective. In addition to improved symptoms of heart failure and exercise intolerance, there is a significant reduction of left atrial and left ventricular size within a short period after closure.
Keywords: Ventricular Septal Defects; Percutaneous Closure; Echocardiography
Introduction
Isolated ventricular septal defects (VSD) account for approximately 40% of all congenital heart lesions [1]. The natural history of VSDs is variable. It has been estimated that spontaneous closure may occur in 12-84% of the defects, with higher rates associated with smaller defects [2-5]. Persistent small defects usually remain asymptomatic and require no treatment unless complicated, while larger defects present early in life with congestive heart failure symptoms and warrant closure [2,5].
Surgery is the traditional modality to close VSDs. However, for the last two decades, advances in percutaneous closure have been described, with several device designs and modifications, resulting in improved success and decreased complication rates [6-9].
At our institution, percutaneous closure of VSD has been done for selected patients since 2015. Indications for closure included symptoms of heart failure or exercise intolerance, left ventricular or left atrial dilatation on echocardiography, and in a few patient, significant residual defect following surgical closure of VSDs.
Our aim in this study is to assess the outcome of percutaneous closure of VSD as a therapeutic modality and to describe the echocardiographic changes of the left ventricular and the left atrial size following percutaneous VSD closure.
Methodology
A Retrospective cross-sectional study included (38) patients who had elective VSD closure at Jordan University Hospital between 2015- 2021. Revision of patient files, echocardiographic and angiographic images were done. Data reviewed included demographic and clinical data, including age at procedure, indication of procedure, type and size of VSD, presence of other congenital anomalies, type of device used to close the defect, approach for closure, complications, and follow up data. Echocardiographic data included parameters of volume overload: Left ventricular end dilastolic dimension (LV), left atrial (LA) dimension as well as left atrial to aortic root dimension ratio (LA/AO), in addition to ejection fraction. The measurements are done in the standard M-Mode echocardiography of long axis parasternal views. Measurements of LV and LA dimensions are converted to Z-scores based on weight and height, while LA/AO ratio is expressed as a numeric value. Echocardiographic data was obtained in three different time points for comparison: At baseline (prior to procedure), first follow up following the VSD closure (within the first six weeks of procedure), and at last follow up. In addition to echocardiographic assessment of left sided volume overload, follow up echocardiography was done to assess device position, the relation of the device to the surrounding structures, any complications such as residual shunts, and the presence of aortic or tricuspid regurgitation.
Categorical data is presented as number (percentage), continuous data is presented as mean (± standard deviation), or median (range). P value of less than 0.05 was considered significant. Statistical analysis was done using online graphpad quickcalcs software (www.graphpad.com/quickcalcs, San Diego, USA).
The study was approved by institutional review board at Jordan University Hospital and by research committee of the University of Jordan.
Procedure
Procedures were performed under either deep sedation or general anesthesia. And via Femoral arterial and venous access. All patients were given 100 IU/kg heparin and prophylactic antibiotics at the start of the procedure. All procedures were guided by transthoracic (TTE), or trans-esophageal echocardiography (TEE) as well as fluoroscopy. Left ventricular angiography was done, and the projection that best profiled the defect was used for sizing and as a reference for fluoroscopic guidance. Defects were crossed either retrogradely from the left ventricle, or antegradely from the right ventricle. Technique of closure of the defect has been described extensively in the literature, and it depends on type and location of the defect, type and design of the occluder device, in addition to the operator experience and preference [6-9]. Device size and type selection were based on echocardiographic and angiographic assessments. Generally, the selected waist size of the device was 2-3 mm larger than the measured VSD diameter.
Results
Thirty-eight patients underwent VSD closure at our hospital between 2015-2021. Females were 24 (63%), median age at intervention was 6.5 years (range 0.5-49), and median weight was 19.5 Kg (5-70). Defects were either perimembranous (27, 71%), or muscular (7, 18%), while two patients had multiple muscular defects, and two patients had both a muscular and perimembranous defect. In addition to the VSD closure, two patients had PDA closure. Four patients were syndromic; three had down syndrome, and one had Williams syndrome. Most patients had symptoms related to the VSD (26, 68%): Eleven patients (29%) had symptoms of heart failure and were on medical therapy before procedure, 10 (26%) children had poor weight gain, 5 patients (13%) had decreased exercise tolerance, and 12 patients had no symptoms. Indications for closure in the asymptomatic patients was left sided dilatation.
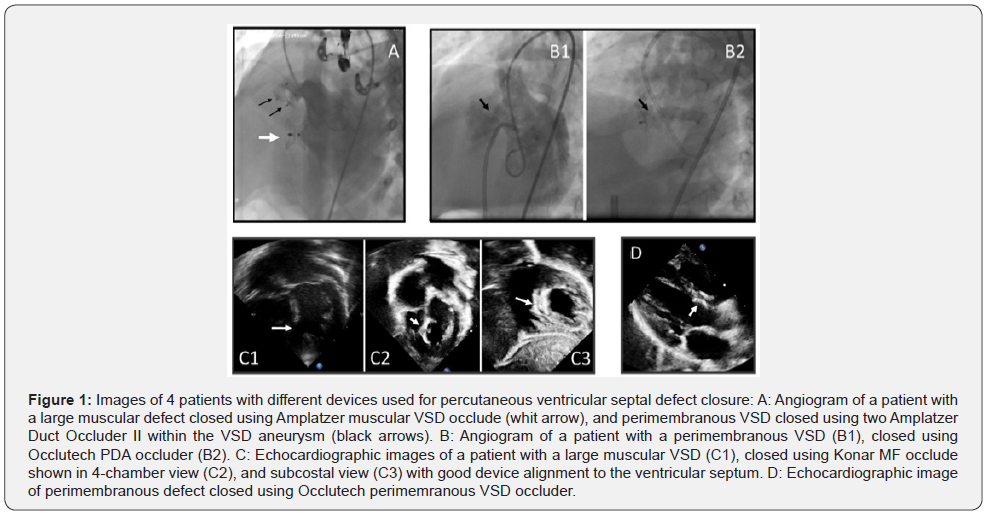
Total of 42 defects were closed in 38 patients (two patients had two defects closed, and one patient had three defects closed. The most commonly used device was Amplatzer duct occluder II (ADOII) (Abbott, CA, USA), in 27 defects (64%). Other devices used included LifeTech muscular device in 4 defects (LifeTech scientific, Shenzhen, China), Occlutech PDA device in 4 defects (Occlutech Holding, Schaffhausen, Switzerland), Amplatzer duct occluder I (ADOI) in 2, LifeTech Konar-MF occluder in 2, Occlutech VSD device in 2, and Amplatzer VSD device in 1patient. Figure 1 illustrates a few images of different devices. Selection of specific device depended on type and size of defect, in addition to device availability at the time of procedure. Median defect size was 4mm (3-11mm).
All procedures were done with both fluoroscopic and echocardiographic guidance. In 19 patients (50%) TEE was used. In the other 50%, TTE was used. There were two incidences of device embolization, both retrieved and the defect closed using a larger device. Approach for crossing the defect was from right ventricle to left ventricle in 16 patients, and from left ventricle to right ventricle in 22 patients. In those with left ventricle to right ventricle approach, 14 patients had closure via arterial catheter (13 ADOII device, and 1 Konar MFO device), while 8 required arteriovenous loop.
All symptomatic patients reported improved symptoms following the procedure, and all patients who were on antifailure medications prior to the procedure were off medications within one month of the closure. Regarding complications of the procedure, one patient had transient epistaxis that was treated conservatively, two patients developed transient intermittent junctional acceleration rhythm which resolved spontaneously within a few days. No patient developed atrioventricular block.
Echocardiographic data
For evaluation of the effect of percutaneous closure of VSD on echocardiographic left ventricular and left atrial size, we included 34 patients with isolated VSD closure. The 4 patients who were not included in the analysis had either a VSD and a PDA (2 patients), or multiple VSDs that were not closed in a single procedure (one had muscular percutaneous VSD closure followed by surgical inlet VSD closure, the other had muscular percutaneous VSD closure followed by perimembranus percutaneous VSD closure 6 months later).
At intial follow up with a mean duration of 4 weeks (±2) following the procedure, 8 (21%) patients had residual shunting (1 moderate, 4 small, and 3 trivial), At the last follow up with mean duration of 18 months (±19), only 6 patients (16%) had residual shunting, (4 trivial and 2 small). There was no aortic valve regurgtitation, and only three patient had trivial tricuspid valve regurgitation.
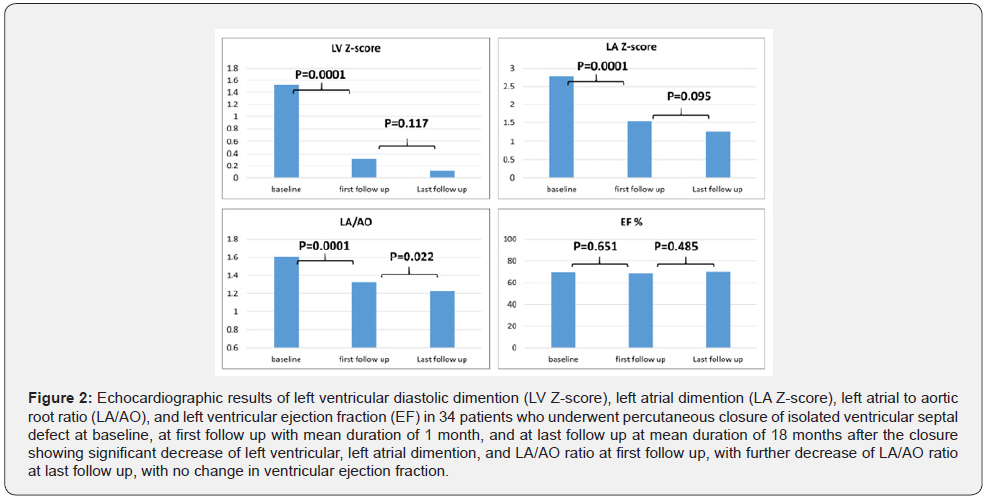
All echocardiographic parameters of left sided volume overload decreased when compared to baseline (Figure 2), the major drop was noted by the first follow up within the first month of the procedure (LV Z-score dropped from 1.53 ±0.87 to 0.31 ±0.68 (p=0.0001), LA Z-score dropped from 2.78 ±0.71 to 1.54 ±1.13 (p= 0.0001), and LA/AO ratio dropped from 1.61 ±0.27 to 1.33 ±0.18 (p=0.0001)). In addition, there was further drop of LA/AO ratio at the last follow up to 1.23 (±0.12) (p=0.022). Other parameters also showed further drop at last follow up with LV Z-score dropping to 0.12 (±0.95), LA Z-score dropping to 1.24 (± 0.8), however without statistical significance (p=0.117 and 0.095, respectively). There was no change in the left ventricular ejection fraction before and after the procedure (69.7 ±5.5 at baseline, 68.8 ±6.5 at first follow up, and 70.2 ±4.5 at last follow up).
Discussion
Left to right shunt in VSDs increases pulmonary blood flow and pulmonary venous return, eventually causing left atrium and left ventricle dilatation. Larger defects typically allow a significant volume overload, following a reduction in pulmonary vascular resistance after birth, and result in early-onset heart failure. Conversely, smaller defects resulting in small shunts generally have a favorable outcome even if persisted. However, systolic dysfunction and a reduction in the compliance of both ventricles due to chronic pressure and volume overload have been demonstrated even in patients with small VSDs who may show left ventricular dilatation on echocardiography and exhibit diminished left ventricular performance during exercise with a subnormal functional capacity with age [10]. Moreover, many long-term complications such as aortic regurgitation, arrhythmia, double chamber right ventricle, and endocarditis have been observed even in small VSDs [4], making their clinical course not entirely benign.
Cardiac structural abnormalities resulting from chronic volume overload associated with VSDs are shown to be partially reversible in patients operated or had percutaneous closure during adulthood [11,12], while early VSD closure is likely to result in complete restoration of the normal cardiac structure. Thus, the reversibility of cardiac structural changes depends to some extent on the degree and the duration of volume overload [13-15].
Thresholds for defect closure nowadays include patients with echocardiographic dilatation of the left atrium, and the left ventricle. Dilated LA and LV diameters adjusted for body surface area (Z-scores) are found to be associated with significant shunting, with Qp: Qs >1.5 [16,17].
A significant reduction in LV Z- score has been demonstrated following percutaneous and surgical closure of VSDs with no significant difference, favoring device closure over surgery in some cases, as device closure has fewer side effects [18]. Our study demonstrated a significant reduction in LV and LA Z-scores, and LA/AO ratio within one month following the procedure. In addition, these parameters seem to continue to decrease gradually after that, indicating restoration of the normal heart size over time [13,18].
Limitations
Our study is limited by its retrospective nature. In addition, it is a single institution experience with a small number of patients who were followed for a relatively short period. To prove the normalization of cardiac chamber parameters and the lack of device-related complications, a larger sample size of patients and a longer follow-up period are required.
Conclusion
Percutaneous ventricular septal defect closure is a highly successful procedure given the wide variety of available devices.
The elimination of the shunt leads to normalization of cardiac size, namely left ventricle and left atrial sizes, in a short period of time, and can be achieved with a very low risk of complications.
Declarations
• Authors received no financial, technical, or other assistance for this work.
• Authors declare no conflict of interest associated with this research.
• All Authors ascertain that they have approved the content of this work.
References
- Hoffman JIE, Kaplan S (2002) The incidence of congenital heart disease. J Am Coll Cardiol 39(12): 1890-1900.
- Natarajan S, Cohen MS (2021) Ventricular Septal Defects. Echocardiogr Pediatr Congenit Hear Dis 247-266.
- Cresti A, Giordano R, Koestenberger M, Spadoni I, Scalese M, et al. (2018) Incidence and natural history of neonatal isolated ventricular septal defects: Do we know everything? A 6-year single-center Italian experience follow-up. Congenit Heart Dis 13(1): 105-112.
- Jortveit J, Leirgul E, Eskedal L, Greve G, Fomina T, et al. (2016) Mortality and complications in 3495 children with isolated ventricular septal defects. Arch Dis Child 101(9): 808-813.
- Viswanathan S, Kumar RK (2017) Should we close small ventricular septal defects? Ann Pediatr Cardiol 10(1): 1-4.
- Brown KN, Adnan G, Kanmanthareddy A (2022) Catheter Management of Ventricular Septal Defect. Stat Pearls.
- Sadiq M, Qureshi AU, Younas M, Arshad S, Hyder SN (2022) Percutaneous closure of ventricular septal defect using LifeTechTM Konar-MF VSD Occluder: initial and short-term multi-institutional results. Cardiol Young 32(5): 755-761.
- Weryński P, Skorek P, Wójcik A, Rudek-Budzyńska A, Dziewulska A, et al. (2021) Recent achievements in transcatheter closure of ventricular septal defects: A systematic review of literature and a meta-analysis. Kardiol Pol 79(2): 161-169.
- Nguyen HL, Tan Phan Q, Huynh Dinh L, Ba Tran H, Won H, et al. (2018) Nit-Occlud Lê VSD coil versus Duct Occluders for percutaneous perimembranous ventricular septal defect closure. Congenit. Heart Dis 13(4): 584-593.
- Maagaard M, Heiberg J, Hjortdal VE (2017) Small, unrepaired ventricular septal defects reveal poor exercise capacity compared with healthy peers: a prospective, cohort study. Int J Cardiol 227: 631-634.
- Amoozgar H, Abdollahi A, Edraki M, Mehdizadegan N, Mohammadi H, et al. (2022) Heart Function and Ventricular Recovery After Percutaneous Closure of Perimembranous Ventricular Septal Defect in Children: A Cross-sectional Study. Iran J Pediatr 32(3): e117730.
- Chikkabyrappa SM, Tretter JT, Doshi AR, Buddhe S, Bhatla P, et al. (2019) The Early “Unnatural” History Following Surgical Repair of Ventricular Septal Defects. Kans J Med 12(4):121-124.
- Aal AA, Hassan HM, Ezzeldin D, El Sayed M (2021) Impact of percutaneous ventricular septal defect closure on left ventricular remodeling and function. Egypt Heart J 73(1): 86.
- Hamdar H, Ghrayeb H, Fakhry B, Chammas E, Chehab G (2022) Treatment of ventricular septal defect in children: Who, when, and how? A 20-years Lebanese multicentric retrospective study. World J Adv Res Rev 14(1): 324-335.
- Bol Raap G, Meijboom FJ, Kappetein AP, Galema TW, Yap SC, et al. (2007) Long-term follow-up and quality of life after closure of ventricular septal defect in adults. Eur J Cardiothorac Surg 32(2): 215-219.
- Gokalp S, Guler Eroglu A, Saltik L, Koca B (2014) Relationships between left heart chamber dilatation on echocardiography and left-to-right ventricle shunting quantified by cardiac catheterization in children with ventricular septal defects. Pediatr Cardiol 35(4): 691-698.
- Gokhroo RK, Anantharaj A, Bisht D, Kishor K, Plakkal N, et al. (2017) A pediatric echocardiographic Z-score nomogram for a developing country: Indian pediatric echocardiography study–The Z-score. Ann Pediatr Cardiol 10(1): 31-38.
- Yang J, Yang L, Yu S, Liu J, Zuo J, et al. (2014) Transcatheter versus surgical closure of perimembranous ventricular septal defects in children: a randomized controlled trial. J Am Coll Cardiol 63(12): 1159-1168.






























