Extracardiac Inappropriate Shocks in Subcutaneous Implantable Cardioverter Defibrillator: Management in Emergency Department
Maurizio Santomauro1*, Mario Petretta2, Livio Imparato3, Carla Riganti4, Gianluigi Iovino1, Antonio Rapacciuolo3, Gaetano Castellano5, Francesco Cacciatore1 and Pasquale Abete1
1Department of Cardiovascular Emergency, Internal Medicine and Geriatric, Federico II University of Naples, Italy
2Department of Translational Medical Sciences, Federico II University of Naples, Italy
3Department of Advanced Biomedical Sciences, Federico II University of Naples, Italy
4General Direction, Medical School, Federico II University of Naples, Italy
5Department of Anesthesiology and Intensive Care, Gemelli Molise Hospital, Campobasso, Italy
Submission: October 15, 2020;Published: November 12, 2020
*Corresponding author: Maurizio Santomauro, Department of Cardiovascular Emergency, Internal Medicine, Geriatric Medical School, Federico II University, Via Sergio Pansini 5- 80131 Naples, Italy
How to cite this article:Santomauro M, Petretta M, Imparato L, Riganti C, Iovino G, et al. Extracardiac Inappropriate Shocks in Subcutaneous Implantable Cardioverter Defibrillator: Management in Emergency Department. J Cardiol & Cardiovasc Ther. 2020; 16(4): 555941. DOI: 10.19080/JOCCT.2020.16.555941
Abstract
Background: The subcutaneous implantable cardioverter defibrillator (S-ICD) is an established treatment for the prevention of sudden cardiac death. In the S-ICD studies, inappropriate shocks (IAS) rate were reported to renge between 5% to 25% and to be mainly due to cardiac and noncardiac oversensing.
Objective: This review highlights data on IAS complications of the S-ICD and is aimed to help in identifying the causes of extracardiac oversensing to facilitate safe and effective emergency management and to reduce the incidence of adverse outcomes. Methods: Literature between January 2010 and March 2020 on IAS and S-ICD was identified by database search. We included studies assessing IAS rates due to extracardiac oversensing. We identified 12 eligible articles including meta-analysis and 28 selected case reports. Results: The total population included 2654 reported patients. Of these, 84 patients received extracardiac IAS. The malfunction of S-ICD due to extracardiac oversensing was related, in particular, to myopotentials (34%) and electromagnetic interferences (29%). The meta-analysis demonstrates an overall high prevalence of extracardiac IAS of 22% (95% CI 8% - 50%); the prevalence was 6% (95% CI 3% - 12%) in patients without the SP (SMART-Pass) filter and 46% (95% CI 12% - 84%) in patients with the SP filter (P=0.047).
Conclusion: Our findings show the high prevalence of extracardiac IAS in patients with S-ICD and demonstrated that the SP is unable to prevent the IAS due to extracardiac reasons. It is important in the Emergency Department for the appropriate management of IAS to identify the various causes.
Keywords: Subcutaneous defibrillator; Inappropriate shock; Extracardiac oversensing; Lead complications; Myopotentials; Electromagnetic interferences
Introduction
Cardiovascular mortality due to ventricular fibrillation (VF) or ventricular tachycardia (VT) represents a significant health problem despite improvements in the prevention and management of underlying cardiovascular disease. Survival of patients with out-of-hospital cardiac arrest worldwide remains poor. In a meta-analysis of Yan et al. [1] the pooled incidence of return of spontaneous circulation was 29.7% (95% CI 27.6– 31.7%), the rate of survival to hospital admission was 22.0% (95% CI 20.7–23.4%), the rate of survival to hospital discharge was 8.8% (95% CI 8.2–9.4%), the pooled 1-month survival rate was 10.7% (95% CI 9.1–13.3%), and the 1-year survival rate was 7.7% (95% CI 5.8–9.5%). The survivors have various therapeutic options such as anti-arrhythmic drugs, radiofrequency or surgical ablation, or implant of implanted cardioverter defibrillator (ICD). Currently, recently introduced S-ICD systems represent an alternative to the implantation of transvenous implanted cardioverter defibrillator (T-ICD) provided that the patient does not require anti-bradycardia pacing or cardiac resynchronization therapy (CRT), recurrent monomorphic VT responsive to antitachycardia pacing (ATP), or pre-existing unipolar pacemaker leads. S-ICD is particularly indicated for primary prevention in younger patients with cardiomyopathies or channelopathies [2]. Furthermore, S-ICD represents the preferred therapy for patients who are at high risk of infection (AHA/ACC/HRS guidelines, Class I recommendation) and for patients without the need of cardiac pacing (Class IIa) [3]. This device reduces VT or VF without direct contact between the implanted system and the vasculature or the endocardium. A can, inserted in the left lateral thoracic wall, and a coil, located anterior to the thoracic cage rib and to the left lateral edge of the sternum [4]. Although initial reports indicated an acceptable rate of IAS when compared to T-ICD [5], novel mechanisms of noise oversensing have recently been reported [6]. After 2016, an automated screening tool (AST) (Boston Scientific) is used to chooses the best sensing configuration and through noise detection and double detection algorithms determines whether oversensing is present [7,8]. The S-ICD sensing algorithm comprises 3 phases: 1) detection phase: this phase filters the input signals to reduce T wave oversensing (TWO); 2) certification phase: the sensed events are classified as certified QRS complexes or as suspected oversensing events. A morphology algorithm based on frequency and slew rate analysis ensures that the signal is cardiac in origin and refuses myopotentials (MP) and electromagnetic interferences (EMI); 3) therapy decision phase: it discriminates between treatable and other high-rate using two programmable zones: conditional and shock zone. In the shock zone, the device analyzes arrhythmia only based on heart rate. In the conditional zone, the device makes supraventricular tachycardia (SVT)–VT discrimination based on static ECG morphology, QRS duration and dynamic ECG morphology. The last function identifies shockable tachyarrhythmias when beat-to-beat morphology varies to indicate that arrhythmia is polymorphic. The SP filter (Boston Scientific) was endorsed on 2016. The SP filter was retroactively installable for A209 systems through an upgrade. The SP filter could be activated following the device setup process and selection of the optimal sensing vector [9,10]. The aim of this algorithm was to reduce IAS without compromising detection of VT. SP reduces the amplitude of lower frequency signals such as T-waves by using a high pass filter. Higher frequency signals such as R-waves, VT and VF remains mostly unchanged. This review summarizes the available data on complications of the S-ICD and aims to help identifying the extracardiac causes of oversensing and the correct management of this device in the ED. We also aimed to evaluate the impact of SP filter on the pooled proportion of extracardiac IAS to the total number of shocks delivered.
Methods
For the purpose of the present investigation, a comprehensive literature on this specific subject search between January 2010 and March 2020 was performed on PubMed, Cochrane central registry, and Google Scholar. The search terms were “subcutaneous implantable cardioverter‐defibrillator and left ventricular assist devices,” “electromagnetic interference and S-ICD,” “Myopotential and S‐ICD,” “inappropriate extracardiac shocks and S-ICD”, “chest compression and S-ICD”, “lead fracture and S-ICD”, “lead dislodgment and S-ICD”, “air entrapment and S-ICD”, “mechanicals complications and S-ICD”. Demographic, extracardiac IAS, programming and actions data were extracted from the reports and authors as needed. The FDA Manufacturer and User Facility Device Experience (MAUDE) database was also queried for reports of S‐ICD and EMI interactions. We included studies assessing IAS rates due to extracardiac oversensing by 3 investigators.
Studies selection and data extraction
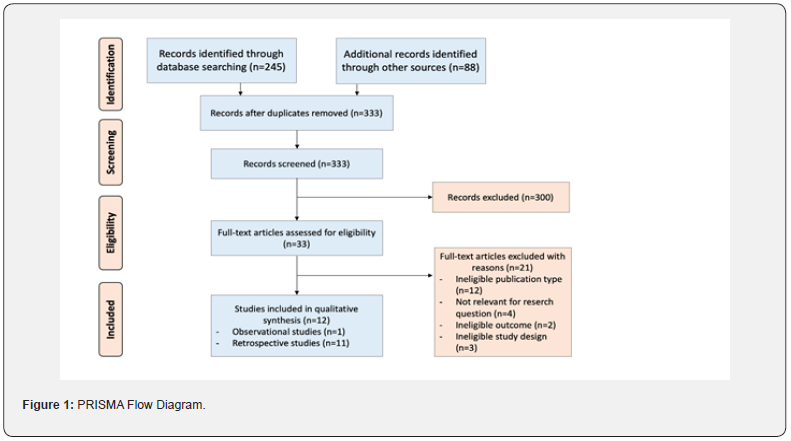
Overall, 333 citations were identified after the removal of duplicates. The references were screened by two independent researchers (MP and GI) and, in case of disagreement, a third researcher (LI) was involved to resolve the differences. The selection process (PRISMA Flow Diagram) is displayed in Figure 1 [11]. Search criteria and methodology were approved by all authors. Titles and abstracts retrieved in the search were reviewed, and observational and comparative studies reporting extracardiac IAS rates in S-ICD were selected. For extracardiac review on causes analysis, articles, abstracts, meta-analysis and editorials were excluded. In the event that there were multiple publications from the same study, the latest study with the most complete data available was selected, and the other publications were not used in order to avoid overlapping cohorts. Because randomized controlled trials (RCT) and non-RCT were included in this meta-analysis, we used the Jadad scale to assess the quality of the RCT, whereas the methodological index for non-randomized studies (MINORS) scale [12] was used to assess non-RCT. If two independent evaluations conflicted, all authors participated in a discussion to resolve the controversy. For selected studies, only data on S-ICD patients were extracted. Extracted data included: number of patients, age, total shocks, number of extracardiac IAS and number of patients with extracardiac IAS. For the metaanalysis, the article type was limited to clinical trials. Twelve full length article were considered eligible for the meta-analysis. Moreover, we identified 28 case reports useful to calculate the percentage of extracardiac IAS in the last 10 years.
Statistical Analyses
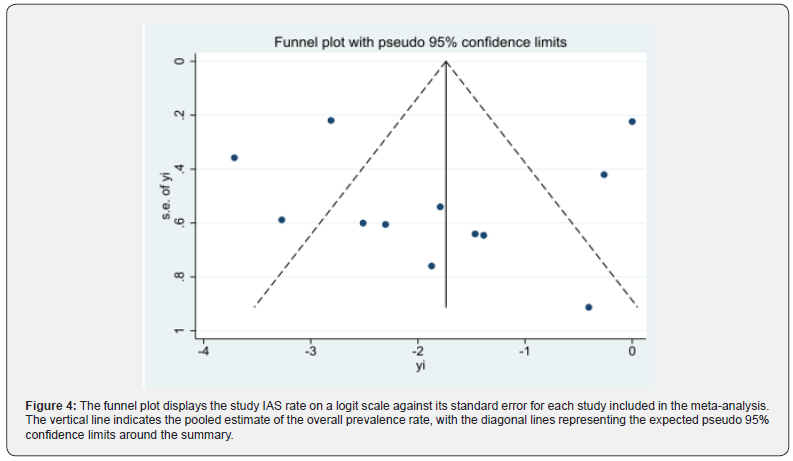
Continuous variables were expressed as mean ± standard deviation and categorical data as percentages. Differences between groups were analyzed by t-test or chi-square test, as appropriate. The main effect size of the meta-analysis was proportional to extracardiac IAS rate of the total number of shocks delivered during the specified follow-up. The user-written Stata metaprop-one package was used to pool proportions and to present weighted sub-group and overall estimates with inversevariance weights. For this purpose the random-effects model the logit transformation was applied, and the result displayed as forest plot. Study specific 95% confidence intervals (CI) were calculated using the Clopper-Pearson exact method. In betweenstudy heterogeneity was evaluated with Cochran’s Q and I square statistics or the likelihood test for random effect vs fixed effect meta-analysis as appropriate. Metaregression analysis was also performed. For this purpose, the extracardiac IAS rate was modeled on the log scale as a linear combination of the regression factors. P-values <0.05 were considered as statistically significant. The presence of publication bias was graphically assessed using a funnel plot, a simple graphical display of a measure of study size against logit of IAS rate. The interpretation of funnel plots is facilitated by the inclusion of diagonal lines representing the 95% confidence limits around the summary treatment effect, showing the expected distribution of studies in the absence of bias. To evaluate potential publication bias, we also performed the Egger weighted regression test and the Begg’s rank test. Finally, percentage of cause of extracardiac IAS in the selected S-ICD studies and case-reports were calculated. Statistical analysis was performed using the Stata software 16.0 (StataCorp 4905 Lakeway Drive College Station, Texas 77845 USA).
Results
After excluding 580 articles for not meeting inclusion/ exclusion criteria, 33 articles remained to be assessed for eligibility (Figure 1). Following assessment of the full-text articles, 21 were excluded for reasons such as: shocks were not specified or rates of extracardiac IAS (rather than just shocks) were not specified. Thus, 12 articles considered eligible for the meta-analysis, and 28 case-reports were selected. The 12 studies included in the metaanalysis are reported in Table 1. The 28 selected case-reports were used for calculating the percentage of extracardiac causes of ICD dysfunction are reported in Table 2. In selected studies and 28 case-reports the malfunction of S-ICD due to oversensing caused by extracardiac signals were: electromagnetic interferences (29%), pectoral or diaphragmatic myopotentials (34%), chest compression (11%), lead damage (1%), air entrapment (23%), electrical interference (1%) and Twiddler syndrome (1%) (Figure 2). The causes of extracardiac IAS and the consequently actions were reported in Table 1 [9-48]. The final population for the metaanalysis included 2627 patients, 1329 without (Group 1) and 1298 with SP filter (Group 2). A total of 84 patients received 167 extracardiac IAS. The random effect meta-analysis of the select studies (9,10,13,14,18,25,27,33,35,42,43,47) demonstrated that in the overall study population the IAS percentage of total shocks delivered due to extracardiac oversensing represent 22% (95% CI 8% - 50%); the percentage was 6% (95% CI 3% - 12%) in patients without the SP filter and 46% (95% CI 12% - 84%) in patients with the SP filter (Figure 3). The funnel plot appears symmetrical (Figure 4), without evidence of bias using both the Egger weighted regression method (P= 0.763) and the Begg rank test (P= 0.304). At meta-regression analysis, the SP filter was significantly associated with a proportion of extracardiac S-ICD dysfunction (P= 0.047).
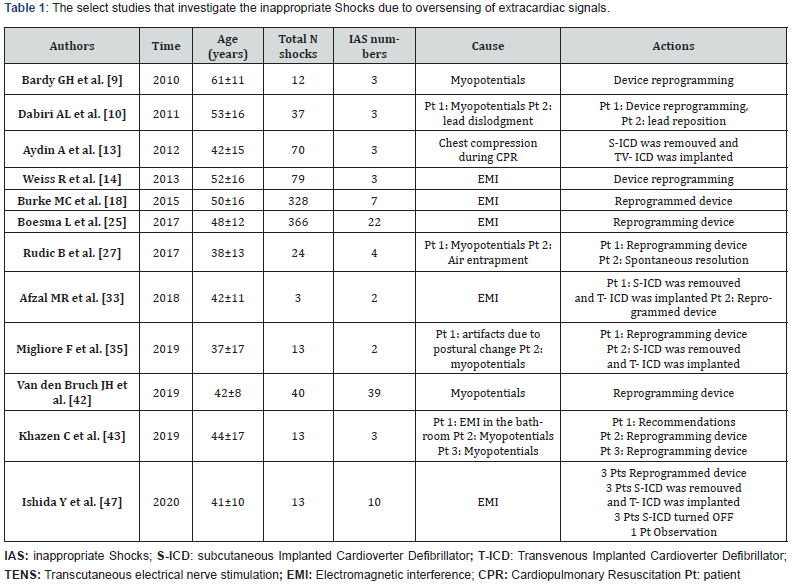



Discussion
Our study first systematically revieved the percentage of causes of extracardiac IAS in full length and case-reports S-ICD studies. IAS derived from the activation of S-ICD therapy without a life-threatening tachyarrhythmia were evaluated in current English literature. During the initial experience with the S-ICD, the IAS rate was high, a prevalence of 4% to 25%, and IAS were associated with increase morbidity [49]. IAS predominantly result from oversensing of cardiac signals due to SVT misinterpreted as ventricular arrhythmias [50]. After addition of the SP filter and additional advancements, contemporary studies have reduced IAS rates from 6.4% to 3.5% annually [7] and the risk of IAS with T-ICD and with S-ICD comparable rates [49]. Theuns et al. [7] showed, with the LATITUDE remote control, that the group SP reduced the risk of the first IAS by 50% (P < .001) and the risk for all IAS by 68% (P < .001) in multivariate analysis adjusted for age and device programming. The incidence of IAS was 4.3% in the SP enabled arm vs 9.7% in the SP disabled arm. In particular, the incidence of extracardiac IAS was 0.5% in the SP enabled arm vs 2.3% in the SP disabled arm (P=0.003). But this study did not specify the causes of extracardiac episodes and the undertaken action. All shock episodes were systematically collected using the remote monitoring system. The EFFORTLESS S-ICD registry collected the data of 472 patients with S-ICD and reported a relatively low incidence of inappropriate therapy (rating of -7%), mostly due to inappropriate cardiac sensing (5.3%) and SVT (1.3%), with a very low incidence of extracardiac inappropriate sensing (0.009%) [51]. But a significant number of case-reports considered that the absence of extracardiac oversensing among patients with S-ICD is underestimated. There are several important reasons that might support such statement. In conclusion, the results of our meta-analysis demonstrate an overall high prevalence of extracardiac IAS of 22%. Interestingly, the summary prevalence was 6% in patients without SP and 46% in those with SP and the difference is statistically significant (P=0.047). The increase in prevalence of extracardiac IAS with SP may be due to recent spread of S-ICD in last years. There is only 10 years of experience with S-ICD. SP is able to reduce IAS from cardiac oversensing [7]. However, extracardiac IAS were already present in the first study of 2010 but for poorly cases suitable, they showed few numbers. Paradoxical effect of the increase in the prevalence of IAS with SP is due to increase in the device in more recent studies that SP can’t avoid. This finding also suggests that, at this time, the SP is unable to eliminate the IAS due to extracardiac reasons. Extracardiac causes of shock are numerous, which effects the management of extracardiac IAS in the ED (Figure 5). In this contest, shock delivery is painful and reduces more battery life. In this report, the most frequent causes secondary to extracardiac signals were EMI (29%) and MP (34%). However, only limited data are available on the impact of EMI on S-ICD function. EMI was not immediately detected on standard device examination but detected on the Marker Channel. Experience with concomitant use of S-ICD and EMI is limited to only few published case reports with discordant results (14,18,25 ,29,30,33,34,43,47,48). Medical equipment which incorporates wireless technology, for example, some infusion pumps, monitoring devices, and ultrasound probes, should also be used at a distance from a S-ICD since these devices may provide a source of EMI. In the case-reports of Gupta et al. [52] and Raman et al. [53] the patient was observed monthly a tour LVAD clinic and no adverse event was seen. In contrast, Afzal et al. [33] and Ishida et al. [47] documented in LVAD patients with S‐ICD, EM Interference in 12 patients. In these cases, EMI may be falsely interpreted as a shockable rhythm and results in 65 J biphasic shock delivery. The fundamental frequencies responsible for the EMI were directly proportional to the pump speed. This is important because there has been some disagreement in the literature regarding the relationship between pump speeds and EMI. Gupta et al. [52] and Raman et al. [53] postulated that the lower operational pump speeds of the LVAD were more likely to be recognized as noise, and less likely to cause sensing interference based on the S‐ICD algorithm. Disabling the shock function of a device before exposure to EMI is therefore advised. Low amplitude of QRS complexes may have contributed to the inability to filter out the external noise. SP filter reduces amplitude of low frequency signals (T waves) but is not useful for high frequency signals like EMI. The EMI filter (non-programmable) of the device was set at 60 Hz. The band pass filter (3-40 Hz) was also enabled. Subcutaneous electrodes and engineered shielding protection have reduced the risk of EMI; however, sources of EMI around the patient should be avoided. If the device has been recently checked and the EMI source site is remote, the likelihood of malfunction is still persistent. For reducing these complications, the Boston Scientific have listed the EMI Compatibility Table for S-ICD [54]. In this document the manufacture recommanded not to use the machines listed since then and is doing well. Offline analysis of the EMI events was invaluable. Educate patients to avoid the device causing the EMI or additional device reprogramming or repositioning is needed to overcome these problems. Although, in different studies and case-reports, we noted that the problem of oversensing related to MP was solved by setting a new configuration in device sensitivity (9,10,15,20,27,28,37,42,43). This is usually managed non-invasively via a change in sensing vector. In the study of Rudic et al. [27] 2 of 62 patients (3.2%) experienced IAS. In 1 patient oversensing of pectoral MP, caused by suboptimal position of the lead, was resolved by placing it toward the right side of the sternum. Reprogramming of the sensing vector resolved the problem. The second patient had 2 IAS in the first 24 hours after implantation and they are related to air entrapment inside the impulse generator pocket. The issue resolved spontaneously without intervention. Van den Bruck et al. [42] assessed 41 patients for MP inducibility. In nearly all patients (90.2%), MP were inducible during isometric chest press (ICP). Secondary vector (70.7%) and alternative vector (75.6%) were most affected during ICP; primary vector was most conditioned during side plank exercise, supporting the body weight on the left arm (51.2%). However, for mechanical extracardiac causes of IAS, the fluoroscopy was clinically useful to demonstrate significant device movement [28]. In case of air entrapment, chest X-rays can often diagnose air entrapment when a large quantity of air sits inside a subcutaneous pocket or lead tunnel. Two kinds of artifacts are commonly seen with air entrapment. Both are related to the entrapped air insulating the sensing electrode. Recently, the twoincision technique is a simplified implant method. But, according to Chinitz et al. [24] and Khazen et al. [43] this new surgical technique predisposes to oversensing and IAS in the early postimplant period, possibly due to introduction of air along the sternal track as the lead is inserted and sheath is removed. This malfunction has been reported and requires only reprogramming the device and may be resolved spontaneously. If this complication does not resolve spontaneously, flushing the sternal track with saline, massaging the skin over the lead and suturing over the lead are suggested to minimize air entrapment [16,17,23,25,27,31,38, 40,41,46]. Unexpectedly, the IAS were due also to oversensing of chest compressions in the setting of a profound postshock bradycardia. Aydin et al. [13] reported 40 consecutive patients of three institutions in Germany. Two episodes in the same patient stopped incorrectly because of under sensing during resuscitation and chest compression after ineffective shocks, respectively. The IAS in the cases reported by Berkowitz et al. [32] and Cmorej et al. [45] were due to oversensing of QRS artefacts that developed during chest compression. With increasing numbers of S-ICD being implanted, the possibility of delivery shock during CPR is likely to represent a challenge in the management of cardiac arrest in S-ICD patients. Magnet deactivation of S-ICD is less reliable and should also be used during CPR maneuvers. In all the cases of malfunctions in emergency situations without a programmer (3200 model, Boston Scientific), the first step is to use a magnet to deactivate repeated shocks. Successful inibition by the magnet is indicated acustically as beeping for 60 seconds. Appropriate placement of the magnet depends on the device S-ICD model [55]. This variability in magnet placement increases the risk of failed deactivation. The magnet should be posizioned off-center so that the curve of the “donut” magnet is over the top or the bottom end of the device. Improper magnet placement may hinder magnet activation and could lead to undesidered delivery of shock theraphy. Magnet application to temporarily deactivate the tachyarrhythmic therapy function is a simple procedure. We believe that consensus statements should consider whether a magnet should be placed over the S-ICD of all patients with IAS. If these conditions cannot be met, consideration should be given to reprogram the device “off” and appropriate precautions taken. Therefore, while the risk of S-ICD activation in the lower extremities is admittedly rare. This case calls into question prior to expert consensus opinion and to management of S-ICD in ED on the lower extremities. Although magnet applications result only in temporary alteration of S-ICD, it should be interrogated at the earliest opportunity to detect any possible programming changes [56].
Limitations
There were limitations to our study which need to be considered. First, the exact proportion of patients with S‐ICD who experience extracardiac IAS is unknown. Also, there is a likely assertation bias because normal functioning devices are unlikely to be reported. In this regard, we only found twenty-two such reports of S‐ICD with confirmed lack of EMI and MP in the literature [9,10,14,15,18-21,25,27,28-30,33-37,42,43,47,48]. It is noteworthy that the largest published patient series of S‐ICD demonstrated that 0.5% to 8% of patients experienced EMI or MP [7,18]. Thus, it is likely that shuch interaction is not a rare event.
Conclusion
Data from reported clinical studies support that the technology of third-generation S-ICD with SP filter has reduced the risk of IAS related to cardiac signals, but do not eliminate the risk of IAS associated with extracardiac signals. Therefore, it is essential in the ED to help improving the management of IAS and to identify the different causes of cardiac and extracardiac IAS. These malfunctions were solved by different approachs: reprogramming device, surgical and education of patient.
Key Teaching Points
• The number of patients with S-ICD presenting in the Emergency Department is increasing.
• Our findings show the high prevalence of extracardiac IAS in patients with S-ICD and demonstrated that, at this time, the SMART-Pass is unable to prevent the IAS due to extracardiac reasons.
• For patient with S-ICD it is crucial to consider also the extracardiac signals as a cause of inappropriate shocks in emergency situation. Shock delivery is painful and utilizes more battery life. These malfunctions were solved by different approachs.
• In the Emergency Department, in case of inappropriate shock without suitable EMBLEM S-ICD 3200 Programmer, the specific Model 6860 or 4520 magnet must be placed above the upper or lower edge of the S-ICD device to inhibit shock delivery. Inhibition of shock delivery by the magnet is signalized acoustically as beeping.
Conflict of Interest
The author has no conflict of interest to disclose.
References
- Yan S, Gan Y, Jiang N, Wang R, Chen Y, et al. (2020)The global survival rate among adult out-of-hospital cardiac arrest patients who received cardiopulmonary resuscitation: a systematic review and meta-analysis Critical Care 24(1):61.
- Al-Khatib SM,Stevenson WG,Ackerman MJ, Bryant WJ, Callans DJ,et al. (2018)2017 AHA/ACC/HRS Guideline for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death.Circulation 138(13):e272-e391.
- Quast ABE, van Dijk VF, Yap SC, Maass AH, Boersma LVA, et al. (2018)Six-year follow-up of the initial Dutch subcutaneous implantable cardioverter-defibrillator cohort: Long-term complications, replacements, and battery longevityJ Cardiovasc Electrophysiol 29(7): 1010-1016.
- Betz JK,Katz DF, Peterson PN, Borne RT, Al-Khatib SM, et al. (2017)Outcomes Among Older Patients Receiving Implantable Cardioverter-Defibrillators for Secondary Prevention From the NCDR ICD Registry. JACC 69(3):265-274.
- Köbe J, Reinke F, Meyer C, Shin DI, Martens E, et al. (2013)Implantation and follow-up of totally subcutaneous versus conventional implantable cardioverter-defibrillators: A multicenter case-control study. Heart Rhythm 10(1):29-36.
- Theuns DA, Burke MC, Allavatam V, et al. (2016)Evaluation of a high pass filter designed to reduce oversensing in the S-ICD. Heart Rhythm13:S10-S11.
- Theuns DA, Brouwer TF, Jones PW, Allavatam V, Donnelley S, et al.(2018)Prospective blinded evaluation of a novel sensing methodology designed to reduce inappropriate shocks by the subcutaneous implantable cardioverter-defibrillator.Heart Rhythm 15(10):1515-1522.
- Brisben AJ, Burke MC, Knight BP, Hahn SJ, Herrmann KL, et al. (2015)New algorithm to reduce inappropriate therapy in the S‐ICD system.J Cardiovasc Electrophysiol26(4):417‐
- Bardy GH, Smith WM, Hood MA, Crozier IG, Melton IC, et al. (2010)An entirely subcutaneous implantable cardioverter-defibrillator. N Engl J Med 363(1):36-44.
- Dabiri AL, Theuns DA, Valk SD, Belle YV, Groot NM, et al. (2011)Clinical experience with a novel subcutaneous implantable defibrillator system in a single center. Clin Res Cardiol 100(9):737-744.
- Moher D, Shamseer L, Clarke M,Ghersi D, Liberati A, et al. (2015)Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement.Syst Rev 1(4):1-9.
- Slim K, Nini E, Forestier D, Kwiatkowski F, Panis Y, et al. (2003)Methodological index for non-randomized studies (minors): development and validation of a new instrument.ANZ J Surg73(9):712-716.
- Aydin A, Hartel F, Schlüter M, Butter C, Köbe J, et al. (2012)Shock efficacy of subcutaneous implantable cardioverter-defibrillator for prevention of sudden cardiac death: initial multicenter experience.CircArrhythmElectrophysiol 5(5):913-919.
- Weiss R, Knight BP, Gold MR, Leon AR, Herre JM,et al. (2013)Safety and efficacy of a totally subcutaneous implantable-cardioverter defibrillator.Circulation 128(9):944-953.
- Alvarez-Acosta L, Romero-Garrido R, Hernandez-Afonzo J (2014)Inappropriate Defibrillator Shock in a Subcutaneous Device Secondary to Repetitive Muscle Contractions.Rev EspCardiol67(6):496-498.
- Zipse MM, Sauer WH, Varosy PD,Aleong RG, Nguyen DT (2014)Inappropriate Shocks due to Subcutaneous Air in a Patient With a Subcutaneous Cardiac defibrillator. Circulation: Arrhythmias and Electrophysiology 7(4):768-770.
- Gamble JHP, Grogono J, Rajappan K, Betts TR, Bashir Y, et al. (2014)Letter by Gamble et al Regarding article” Inappropriate Shocks due to Subcutaneous Air in a Patient With a Subcutaneous Cardiac defibrillator”.Circulation: Arrhythmias and Electrophysiology 7(6):1281.
- Burke MC, Gold MR, Knight BP, Barr CS, Theuns DAMJ, et al. (2015)Safety and Efficacy of the Totally Subcutaneous Implantable Defibrillator: 2-year Results from a Pooled Analysis of the IDE Study and EFFORTLESS Registry. JACC65(16):1605-1615.
- Frommeyer G, Reinke F, Eckardt L, et al. (2015)Inappropriate shock in a subcutaneous ICD due to interference with a street lanternInternational Journal of Cardiol 198:6-8.
- Corzani A, Ziacchi M, Biffi M,Diemberger I, Martignani C, et al. (2015)Inappropriate shock for myopotential over-sensing in patients with subcutaneous ICDIndian Heart J 67(1):56-59.
- Miller MA, Palaniswamy C, Sharma D, Reddy VY (2015)Inappropriate shock from a subcutaneous implantable cardioverter-defibrillator due to transcutaneous electrical nerve stimulationHeart Rhythm 12(7): 1702-1703.
- Kooiman KM, Brouwer TF, Van Halm VP, Knops RE (2015)Subcutaneous implantable cardioverter defibrillator Lead failure due to Twiddler Syndrome.Pacing and Clinical Electrophysiology 38(11):1369-1371.
- Yap SC, Bhagwandier RE, Szili-Torok T,Theuns DAMJ (2015)Air entrapment causing early inappropriate shocks in a patient with a subcutaneous cardioverter-defibrillator Heart Rhythm Case Rep1(3):156-158.
- Chinitz JS, Nadraus P, Darge A,Cacciabaudo J, Altman EJ (2016)Inappropriate Shocks within 24 hours after Implantation of a Subcutaneous defibrillator with a Two-incision TechniqueJournal of Innovation in Cardiac Rhythm Management 7(3):2295-2298.
- Boersma L, Barr C, Knops R,Theuns D, Eckardt L, et al. (2017)EFFORTLESS Investigator Group. Implant and midterm outcomes of the subcutaneous implantable cardioverter-defibrillator registry: the EFFORTLESS study. J Am Coll Cardiol 70(7):830-841.
- Lee S, Souvaliotis N, Metha D, Suri R (2017)Inappropriate shock in a subcutaneous cardiac defibrillator due to residual airClinical Case Reports 5(8):1203-1206.
- Rudic B, Tülümen E, Berlin V,Röger S, Stach K, et al. (2017)Low Prevalence of Inappropriate Shocks in Patients With Inherited Arrhythmia Syndromes With the Subcutaneous Implantable Defibrillator Single Center Experience and Long-Term Follow-Up.J Am Heart Assoc6(10):e006265.
- Chieng D, Stewart B, Paul V(2018)Inappropriate shock from myopotentials due to subcutaneous defibrillator (S-ICD) movement conformed on fluoroscopy with subsequent device pocket revision. Journal of Interventional Cardiac Electrophysiology53(2): 263-265.
- Turner SL, Singh SM(2018)An “UninTENSional” Subcutaneous Implantable Cardioverter-defibrillator. ShockJournal of Innovation in Cardiac Rhythm Management 9(11):3395-3397.
- Ahmed AS, Patel PJ, Bagga S,Gilge JL, Schleeter T, et al.(2018)Troubleshooting electromagnetic interference in a patient with centrifugal flow left ventricular assist device and subcutaneous implantable cardioverter defibrillator. J Cardiovasc Electrophysiol29(3):477-481.
- Taguchi Y, Ishiyuki T, Matsumoto K, Ogino Y, Matsushita H, et al. (2018)An Inappropriate Shock Case Early after Implantation of a Subcutaneous Cardiac defibrillator by Subcutaneous Entrapped Air.Int Heart J 59(2): 417-419.
- Berkowitz EJ, Pleimann BF, Rosenfels LE(2018)Subcutaneous implantable cardioverter defibrillator oversensing and shock delivery due to chest compression during CPR.Pacing Clin Electrophysiol41(12):1687-1690.
- Afzal MR, Ahmed A, Prutkin JM, Saba S, Cha YM, et al.(2018)Multicenter Experience of Concomitant Use of Left Ventricular Assist Devices and Subcutaneous Implantable Cardioverter-Defibrillators.JACC Clinical Electrophysiology4(9):1261‐
- Saini H, Saini A, Leffler J, Eddy S, Ellenbogen KA(2018)Subcutaneous implantable cardioverter defibrillator (S‐ICD) shocks in a patient with a left ventricular assist device. Pacing Clin Electrophysiol41(3):309‐
- Migliore F, Mattesi G, De Franceschi P,Allocca G, Crosato M, et al.(2019)Multicentre experience with the second-generation subcutaneous implantable cardioverter defibrillator and the intermuscular two-incision implantation technique.J Cardiovasc Electrophysiol 30(6):854-864.
- Barnett A, Eckhardt L, Leal MA(2019)Seal plug damage causing inappropriate detection and therapy in a subcutaneous defibrillator system. Heart Rhythm Case Report 5(2):66-69.
- Sasaki T, Nakamura K, Naito S(2019)Subcutaneous Implantable Cardioverter Defibrillator Lead Repositioning for Preventing Inappropriate Shocks Due to Myopotential Oversensing in a Post-Fulminant Myocarditis Patient. Int Heart J 60(2):466-469.
- Yang YC, Aung TT, Bailin SJ, Rhodes TE (2019)Air Entrapment Causing Inappropriate shock From a Subcutaneoous Implantable Cardioverter defibrillator. Cardiol Res 10(2):128-130.
- Camm CF, Rajappan K, Curson M, Tilling L(2019)Twiddler’s syndrome with a subcutaneous implantable cardioverter-defibrillator presenting with an inappropriate shock: a case report. Eur Heart J-Case Report 3(4):1-5.
- Nishinarita R, Kishihara J, Matsuura G, Arakawa Y, Kobayashi S, et al. (2019)Early inappropriate shock in a subcutaneous cardiac defibrillator due to subcutaneous air. J of Arrhythmia35(4):682-684.
- Adduci C, Spadoni L, Palano F, Francia P(2019)Ventricular fibrillation undersensing due to air entrapment in a patient implanted with a subcutaneous cardioverter defibrillator.J Cardiovasc Electrophysiol30(8):1373‐
- van den Bruck JH, Sultan A, Plenge T,Seuthe K, Mödder T, et al. (2019)Incidence of myopotential induction in subcutaneous implantable cardioverter‐defibrillator patients: is the oversensing issue really solved?Heart Rhythm16(10):1523‐
- Khazen C, Magnusson P, Flandorfer J,Schukro C (2019)The subcutaneous implantable cardioverter-defibrillator: A tertiary center experience.Cardiol J 26(5):543-549.
- López‐Gil M, Fontenla A, Delgado JF, Rodríguez-Muñoz D(2019)Subcutaneous implantable cardioverter defibrillators in patients with left ventricular assist devices: case report and comprehensive review. Eur Hear JCase Reports3(2):1‐
- Cmorej P, Smrzova E, Peran D,Bulikova T(2020)CPR Induced Inappropriate Shocks from a Subcutaneous Implantable Cardioverter Defibrillator during Out-of-Hospital Cardiac Arrest. PrehospEmerg Care 24(1):85-89.
- Ghrair F, Dohrmann M, Gautam S (2020)Inappropriate subcutaneous implantable cardioverter defibrillator shock caused by noise resulting from air entrapment in the generator screw set: a rare case report. J Am Coll Card 75(11)(Suppl 1): 2808.
- Ishida Y,PayneJE,Field ME, Gold MR (2020)Electromagnetic interference from left ventricular assist devices in patients with subcutaneous implantable cardioverter‐ JCardiovascElectrophysiol 31(5):1195-1201.
- Utkarsk K, Sohail H(2020)Inappropriate subcutaneous implantable cardioverter-defibrillator shock due to electromagnetic interference. EP Europace22(6): 971.
- Auricchio A, Hudnall JH, Schloss EJ, Sterns LD, Kurita T, et al.(2017)Inappropriate shocks in single-chamber and subcutaneous implantable cardioverter-defibrillators: a systematic review and meta-analysis.Europace19(12):1973-1980.
- Olde Nordkamp LRA, Brouwer TF, Barr C,Theuns DAMJ, Boersma LVA, et al.(2015)Inappropriate shocks in the subcutaneous ICD: incidence, predictors and management.Int J Cardiol195:126‐
- Lambiase PD, Barr C, Theuns DA,Knops D, Neuzil P, et al.(2014)Worldwide experience with a totally subcutaneous implantable defibrillator: early results from the EFFORTLESS S-ICD Registry.Eur Heart J 35(25):1657-1665.
- Gupta A, Subzposh F, Hankins SR,Kutalek SP(2015)Subcutaneous implantable cardioverter-defibrillator implantation in a patient with a left ventricular assist device already in place.Tex Heart Inst J42(2):140-143.
- Raman AS, Shabari FR,Kar B, Loyalka P, Hariharan R(2016)No Electromagnetic Interference Occurred in a Patient with a HeartMate II Left Ventricular Assist System and a Subcutaneous Implantable Cardioverter-Defibrillator. Tex Heart Inst J43(2):183-185.
- Boston Scientific (2017)Electromagnetic (EMI) Compatibility Table for Pacemakers, Transvenous ICDs, S-ICDs and Heart Failure Devices.
- McFaul M, Lombaard S, Arora V, Van Cleve WC, Rooke GA, et al.(2020)Unexpected Shocks from a subcutaneous implantable cardioverter-defibrillator despite attempted reprogramming and magnet use: a case report. AA Practice14(6):e01178.
- Apfelbaum JL, Schulman PL, Mahajan A, Connis RT, Agarkar M (2020)Practice Advisory for the PerioperativeManagement of Patients with Cardiac Implantable Electronic Devices: Pacemakers and Implantable Cardioverter–Defibrillators 2020. Anesthesiology132(2):225-252.






























